Abstract
Seedling recruitment is a critical driver of population dynamics and community assembly, yet we know little about functional traits that define different recruitment strategies. For the first time, we examined whether trait relatedness across germination and seedling stages allows the identification of general recruitment strategies which share core functional attributes and also correspond to recruitment outcomes in applied settings. We measured six seed and eight seedling traits (lab- and field-collected, respectively) for 47 varieties of dryland grasses and used principal component analysis (PCA) and cluster analysis to identify major dimensions of trait variation and to isolate trait-based recruitment groups, respectively. PCA highlighted some links between seed and seedling traits, suggesting that relative growth rate and root elongation rate are simultaneously but independently associated with seed mass and initial root mass (first axis), and with leaf dry matter content, specific leaf area, coleoptile tissue density and germination rate (second axis). Third and fourth axes captured separate tradeoffs between hydrothermal time and base water potential for germination, and between specific root length and root mass ratio, respectively. Cluster analysis separated six recruitment types along dimensions of germination and growth rates, but classifications did not correspond to patterns of germination, emergence or recruitment in the field under either of two watering treatments. Thus, while we have begun to identify major threads of functional variation across seed and seedling stages, our understanding of how this variation influences demographic processes—particularly germination and emergence—remains a key gap in functional ecology.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Functional traits are used to describe ecological variation among plant species and provide a mechanistic understanding of species distributions, community assembly, and how these influence ecosystem function and services (Bello et al. 2010; Garnier and Navas 2012; McGill et al. 2006; Westoby and Wright 2006). This line of work has been instrumental in identifying trait tradeoffs that limit independent trait variation and uncovering potential ecological strategies favored under different environmental conditions (e.g., Bernard-Verdier et al. 2012; Cornwell and Ackerly 2009; Wright and Westoby 1999). While there has been sustained headway in describing key dimensions of trait variation in plants, efforts to integrate this understanding in a manner that facilitates practical advances in ecosystem conservation and restoration have been slow to develop but represent one of the most critical areas of plant functional trait research (Funk et al. 2008; Laughlin 2014).
One key gap limiting application of trait-based frameworks is our lack of understanding of traits driving variation in recruitment outcomes (Garnier and Navas 2012). The vast majority of our current understanding of functional trait spectra and strategies is centered around plant tissue economics and the influence of these traits on resource capture, conservation and growth (e.g., Lambers and Poorter 1992; Reich 2014). Along a tissue economics spectrum, species constructing thinner or less dense leaves or roots create more absorptive surface area per unit of biomass, resulting in more rapid return on tissue investments and higher relative growth rates (RGRs) (Reich 2014; Shipley 2006). At the opposite end, species constructing thicker or denser leaves and roots tend to have slower growth but greater tissue longevity and stress tolerance, which could promote survival (Poorter and Bongers 2006; Ryser 1996). Species may also vary in the plastic response of these attributes to changing environmental conditions, which could be key in differentiating performance among coexisting species (Valladares et al. 2007). Given the potential for early seedling survival to influence recruitment rates (Gómez-Aparicio 2008), these seedling functional traits could provide important insight into demographic patterns. However, there is also growing evidence that a bulk of plant mortality can occur prior to vegetative stages, during germination and emergence (James et al. 2011; Leishman and Westoby 1994; Sharitz and McCormick 1973). In order to understand the implications of trait variation for populations, communities and ecosystems, it is critical to expand our understanding beyond plant growth and resource capture to include key traits influencing transition probabilities across early life stages.
A wide array of traits may influence germination and emergence processes, but few studies have attempted to explore and integrate trait variation within and across these key life stage transitions (but see Gardarin et al. 2011; Thompson et al. 1996). Seed mass is most broadly studied among seed traits. Though it is typically compared to seedling function and survival (Moles and Westoby 2006), evidence suggests that light and temperature stratification requirements for germination may be negatively and positively associated with seed mass, respectively (Milberg et al. 2000; Pearson et al. 2002). Phenological patterns of germination and seminal root growth could also influence recruitment probabilities and are tightly tied to requirements for minimum soil temperature, minimum water potential, and hydrothermal time accumulation (Bradford 2002; Gummerson 1986; Harris and Wilson 1970). Furthermore, the size, shape, and density of coleoptiles or cotyledons could influence survival of emerging seedlings under harsh abiotic conditions (Ganade and Westoby 1999; Gardarin et al. 2010). A major challenge in identifying general recruitment strategies is that traits and ecological dimensions related to germination and emergence may be unrelated to those influencing seedling resource capture (Grime et al. 1997; Leishman and Westoby 1992). However, early work by Grime et al. (1981) and Shipley et al. (1989) documents at least one cohesive spectrum linking large-seeded species to both slow germination rates and slow seedling growth rates across several life forms. Ultimately, if germination and emergence are key processes influencing recruitment outcomes, it is essential to identify and incorporate related traits into a broader understanding of ecological strategies and variation among plants.
Whether trait variation across multiple seed and seedling stages can be used to identify general ecological strategies which ultimately predict recruitment outcomes is unknown. It has been suggested that functional groups of species sharing key trait attributes may have common responses to environmental stressors and ecosystem effects (Boutin and Keddy 1993; Lavorel et al. 1997). These trait-based groups, or functional types, should provide a simpler, generalizable alternative to species-based models and management while capturing community responses better than groups based simply on life form or life history. However, it is unclear whether functional types could effectively predict demographic responses, which are difficult and time-consuming to measure.
The first two objectives of this study were to look for major axes of variation and tradeoffs among seed and seedling functional traits of dryland grasses, and to use these trait data to identify recruitment types (i.e., functionally similar species or varieties identified via cluster analysis) among candidate restoration grasses. We then asked whether these recruitment types captured variation in probabilities of germination, emergence or seedling survival under two water levels, providing a foundation for a general framework to predict recruitment outcomes.
Materials and methods
Study system and experimental approach
Trait and life stage transition data were collected for 47 grass varieties (i.e., ecotypes or cultivars) broadly distributed across dryland systems of western North America (Table 1). Study species represent key native and non-native perennials used in restoration efforts in this region, two annual grasses that commonly invade following disturbance (Bromus tectorum L. and Bromus japonicas L.), and two introduced annual grasses that have shown little ability to invade these systems [Eremopyron triticeum (Gaertn.) Nevski and Lolium multiflorum (Lam.)]. Seeds were obtained from private seed companies, US Department of Agriculture (USDA) Plant Materials Centers, and personal collection.
Seedling traits and life stage transitions were measured at a field site in the Northern Great Basin Experimental Range near Burns, Oregon (43°27′N, 119°42′W, 1400 m elevation, 283 mm average annual precipitation). We arranged 300 tilled 1-m2 plots in a 15 × 20 grid and randomly assigned each variety to six plots. Plots were hand sown (400 seeds per plot) to monitor probabilities of emergence and seedling survival. Plots also contained one germination bag (50 seeds per bag, fully buried, 2- to 5-cm depth) and two open-top seedling bags (all bags buried with open top flush to soil surface and extended to 10-cm depth to allow unrestrained seedling growth within the bag) made of nylon mesh and filled with field soil. Germination and open-topped seedling bags were removed from the site at different times to quantify germination probabilities or seedling traits, respectively (described below).
Seeds were sown during November 2011. Because precipitation for the study year (130 mm) was less than 50 % of average, all plots were supplemented with 65 mm of water over six April watering events (approximately 11 mm per event), prior to emergence to simulate the effect of a typical spring storm period on soil moisture (low-water treatment). Row coverings (Agribon AG-19) were temporarily placed over all plots during watering to reduce evaporative loss. To capture life stage transition rates under two moisture scenarios and to quantify differences in trait plasticity among grass seedlings, half of the plots per variety received an additional 65-mm watering treatment distributed as 12 events (ranging 2.5–10 mm depending on soil moisture status) between March and May 2012 (high-water treatment).
Trait measurement
Seedling traits were sampled during spring 2012. Once seedlings in all open-top bags of a given variety and treatment had established (i.e., developed a fully expanded true leaf), seedlings were thinned to one seedling of uniform size per bag. One set of open-top bags was harvested immediately, and a second set was harvested 3 weeks later. Fresh leaves and roots were scanned (WinRHIZO; Regent Instruments, Sainte-Foy, Canada) for total area and length, respectively, and leaves were kept moist and weighed for fresh mass within 24 h. Roots, leaves, and shoots were then dried at 60 °C for at least 3 days and weighed for dry mass. Traits collected from initial and final harvests were used to calculate RGR and root elongation rate (RER) (Hunt et al. 2002). All other seedling traits in the analysis, except initial root mass (RMi), were collected from final harvest seedlings. Traits collected under the high-water treatment were used only to estimate RGR plasticity, following the phenotypic plasticity index described by Valladares et al. (2006) (Table 2). We sampled three individuals per variety, treatment, and harvest to calculate seedling trait means.
Coleoptile tissue density (CTD) was calculated as the ratio of dry to fresh mass of newly emerged coleoptiles (<1 cm tall) grown in five regularly watered pots per variety outside of the Eastern Oregon Agricultural Research Center greenhouse in Burns in spring 2012 (Ganade and Westoby 1999). Seed mass was the average seed weight from three lots of 50 seeds, oven-dried for 2 days at 60 °C.
Hydrothermal germination parameters, including base temperature, base water potential, and hydrothermal time for germination, are useful indices describing temperature, moisture, and temporal requirements for germination, respectively (Allen et al. 2000; Bradford 2002). To estimate these parameters, germination trials were performed for each variety at three sub-optimal temperatures (6, 12, and 18 °C) and three water potentials (−0.033, −0.5, −1.0 MPa) in a factorial design following experimental methodology and equipment described by Hardegree et al. (2003). Six replicate germination vials (30 seeds vial−1) per treatment and variety were filled with polyethylene glycol 8000 water potential solutions and distributed randomly among temperature chambers such that no two replicates were assigned the same chamber. Germinated seeds (radicle >2 mm) were counted at least every other day and pooled across replicates to create cumulative germination curves scaled by the maximum percent germination observed for a given seedlot (Hardegree et al. 2013). Interpolation was used to calculate time to germination for every fifth percentile of the seed population up to and including the 25th percentile. Inverses of these values (per day germination rates) were then used to estimate hydrothermal time, base water potential, and base temperature closely following modeling methods of Gummerson (1986) and based on the hydrothermal time equation:
where Ɵ HT is hydrothermal time, T b is base temperature for germination, Ψ b(G) is base water potential for germination for a given germination percentile G, t(G) is the time to germination for G, and T and Ψ are environmental temperature and water potential, respectively.
We also calculated average annual rate sum, which integrates potential germination response relative to predicted field conditions and can be used as a relative index of potential germination performance among different seedlots (Hardegree et al. 2013). Rate sum estimates the number of times a field-planted seedlot would reach a given germination percentile in a year, on average, if the seed population was planted at the beginning of the year and replaced by an identical seed population every time the germination percentile was reached. Rate sums thus translate hydrothermal parameters into a more ecologically relevant index of relative germination rate given a realistic treatment of actual field-variable conditions of seedbed microclimate (Hardegree et al. 2013). We estimated average annual rate sum by predicting and summing hourly 25 % germination rates for each year of a 44-year simulation of seedbed temperature and water potential for a field site in Ada County, Idaho (Hardegree et al. 2013). Hourly germination rates [t(25)−1] were predicted by applying estimated Ɵ HT, Ψ b(25) and T b parameters to the hydrothermal time (Eq. 1), where T and Ψ were temperatures and water potentials, respectively, for each hour of the simulation. Average annual rate sums ranged from around five for slower germinating seedlots to over 28 for the most rapidly germinating seedlots. All trait data are described in Table 2, and trait means and SDs are provided in Electronic Supplementary Material, Appendix S1.
Life stage transition probabilities
To account for differences in initial seedlot viability (see germination probability below), tetrazolium (TZ) testing was performed when prior viability tests (germination or TZ) were unavailable or indicated less than 90 % germinability (Appendix S1). TZ testing followed the Association of Official Seed Analysts and was conducted at the Oregon State University Seed Laboratory (Corvallis, OR). Conditional life stage transition probabilities, including germination, emergence, seedling survival and cumulative survival (Table 2) (Fenner and Thompson 2005), were calculated from three germination bags and seeded plots per grass variety and treatment. Germination probability was the average proportion of germinated seeds (radicle >2 mm) in germination bags divided by seedlot viability. Emergence and seedling mortality were monitored in 1-m2 plots, in which emerged seedlings were marked and monitored biweekly from April to dormancy onset in late June. Emergence was the average proportion of germinated seeds per plot (estimated no. germinated seeds = germination probability × 400 sown seeds per plot) resulting in coleoptiles (i.e., non-autotrophic shoots) that penetrated the surface. Seedling survival was the proportion of emerged seedlings surviving (i.e., remaining intact and flexible) to the end of the first growing season. Cumulative survival was the proportion of sown seeds resulting in survived seedlings over the first growing season (i.e., first-year recruitment success). Life stage transition data are summarized in Table 2 and provided in Appendix S1.
Analyses
Mean trait values were captured in a trait matrix with 47 grass varieties and 14 traits. Missing data comprised less than 3 % of the entire trait matrix, and in each of these cases, data were substituted with averages from conspecific varieties or columns in order to retain ecologically relevant grass varieties in the analysis (substitutions noted in Appendix S1). Rate sum and leaf dry matter content (LDMC) values were log transformed to reduce outlier influence, and all traits were relativized by standard deviates to give equal weights to traits with different units (McCune and Grace 2002). Data were checked for linearity, and proportional life stage transition rates were logit transformed to meet distributional assumptions.
Pearson correlation coefficients were calculated between all traits to aid the interpretation of trait relationships across seed and seedling stages. Principal components analysis (PCA) was then performed on the full trait matrix with Varimax rotation to reduce trait variation down to a few key axes describing relationships and tradeoffs among seed and seedling traits. In order to explicitly test whether varieties were similarly related to one another in seed and seedling trait spaces, we split the full trait matrix into a seedling trait matrix and seed trait matrix with eight and six trait columns, respectively (Table 2). CTD was classified as a seed trait because of its functional relevance in the pre-emergence period. A Mantel test was then performed on two Euclidean distance matrices (47 varieties × 47 varieties, in trait space) from seed and seedling traits, using a randomization test of statistical significance (1000 runs). Mantel tests estimate the correlation between two distance matrices, providing a standardized Mantel statistic (r), and a significance test of the null hypothesis of no relationship between matrices.
In order to identify and describe emergent functional types during recruitment (sensu Lavorel et al. 1997), hierarchical agglomerative cluster analysis was performed on the trait matrix using Euclidean distances and Ward’s linkage method. We minimized weighting of highly correlated traits in cluster separation by retaining just one trait when pairs of highly correlated traits occurred (r > 0.70) (Fry et al. 2014). We thus performed cluster analysis with 11 of the 14 traits, retaining seed mass, specific leaf area (SLA) and RGR while excluding respective correlates RMi, LDMC and RER. The appropriate number of clusters was determined from visual assessment of cluster separation in the dendrogram and in the PCA ordination space to identify possible cutoff points. As an objective verification of cluster cutoff selection, clusters were tested for trait differences using one-way ANOVA. We also examined the dendrogram and PCA ordination space for late-merging or outlying (i.e., relative to the cluster center) varieties, respectively. Psathyrostachys juncea var. Vinall was distant from all clusters and was removed from cluster analysis with no additional effects on cluster membership (dendrograms in Appendix S2). We also used discriminant analysis with leave-one-out cross-validation to validate cluster membership and found 43 of 46 original cluster assignments to be supported (Appendix S2) (Fry et al. 2014). Once clusters were established, we conducted one-way ANOVAs (α = 0.05) with Tukey’s honest significant difference for post hoc multiple comparisons to test for cluster differences in life stage transition probabilities under both high and low water treatments. PCA, mantel tests, cluster analysis and discriminant analysis were performed in R under the packages pysch, ecodist, cluster, and MASS, respectively (R v. i386 3.1.1 R Core Team 2014).
Results
A Mantel test weakly rejected the null hypothesis of no relationship between seed and seedling trait distance matrices (standardized Mantel r = 0.183, P = 0.035). Out of 91 possible correlations between 14 traits, there was some evidence of correlation (α = 0.10) in 30 cases: between seedling traits (n = 13), between seed traits (n = 6), and between seed and seedling traits (n = 11) (Table 3). Most strong correlations within and among stages were captured in trait spectrums identified by PCA and are described below, though some bivariate trait associations were not captured by ordination. Among these bivariate associations, varieties with lower RGR tended to have more positive RGR plasticity in response to water. Additionally, varieties with higher rate sums tended to have lower base temperatures, more negative base water potentials and lower hydrothermal times for germination (Table 3).
Four axes were retained from PCA (RC1-RC4), cumulatively explaining 65 % of the total variance among grass traits (Table 4; Fig. 1a, b). Each axis represented a separate set of traits which loaded highly onto the axis, indicating that these traits were highly correlated with one another across grasses. The first axis (RC1) had positive loadings of RGR and RER and negative loadings of RMi and seed mass, representing a spectrum from small seed/root mass and rapid growth to large seed/root mass and slow growth. The second axis (RC2) represented a separate growth spectrum connecting seedling growth rate to leaf and coleoptile traits and germination rate, with positive loadings of RGR, RER, SLA, and rate sum and negative loadings of LDMC and CTD. The third axis (RC3) captured variation in germination requirements, with opposite loadings of hydrothermal time and base water potential. Finally, the fourth axis (RC4) described a tradeoff in root investment strategy with opposite loadings of SRL and RMR. Notably, while rate sum and CTD shared weak correlations with key traits on RC2 (Table 3), less than 50 % of the variation in these traits was ultimately captured by the components (h 2-values; Table 4). Similarly, despite some bivariate associations with other traits (Table 3), base temperature for germination and RGR plasticity were not strongly represented by any single axis.
Trait loadings (vectors) and species scores (points) along principal component analysis axes a 1 and 2, and b 1 and 3. The magnitude and direction of each trait vector along a given axis represents the correlation between the trait and that axis. Symbol letters indicate trait-based recruitment types identified by cluster analysis (Table 5; clusters A–F). RGR Relative growth rate, RER root elongation rate, LDMC leaf dry matter content, SLA specific leaf area, CTD coleoptile tissue density, SRL specific root length, RM i initial root mass, RMR RM ratio
Cluster analysis separated grasses into six trait-based recruitment types (clusters A-F; assignments given in Table 1) sharing similar collections of trait attributes. Examination of the dendrogram (Appendix S2) suggested that “natural groups” (sensu McCune and Grace 2002) occurred at cutoffs of either two or six clusters, when long cluster stems signified substantial between-cluster differences. Six relatively homogeneous clusters were more meaningful for the study’s objectives. Further cluster separation (i.e., to seven clusters) yielded groups with just two conspecific or congeneric varieties, while further merging (i.e., to five clusters) yielded large groups with minimal distances between subsequent mergers. Selection of six clusters was further verified by the occurrence of significant trait differences between all clusters. Given the small sample size of some clusters (n = 3), these estimates of significance were relatively conservative.
Clusters were broadly separated by seedling growth rate (rapid,clusters A–D; slow, clusters E, F) and germination rate (i.e., rate sum; rapid, clusters A, B; slow, clusters C–F), with other traits underlying these differences (Table 5). Cluster A (n = 3) represented annual grasses with the most rapid growth and germination traits accompanied by low CTD and LDMC, high SLA, small initial roots with rapid RER, and the lowest hydrothermal times and base temperatures for germination. Cluster B (n = 21) represented the most rapidly germinating and growing perennials, with average leaf and root attributes but above-average rate sums with more negative base water potentials for germination. Cluster C (n = 5) grasses shared some leaf and root attributes with cluster B, but exhibited below-average rate sums and were further separated by the tendency to grow faster in drier conditions (negative RGR plasticity). Cluster D (n = 3) also represented varieties with rapid growth and slower germination, primarily separated from other clusters by very high hydrothermal time estimates. Clusters E (n = 7) and F (n = 7) were both separated by slow growth and germination rates, but represented very different strategies. Cluster E represented small-seeded grasses with lower SLA and above-average RMR, whose seeds and plants were water sensitive with positive RGR plasticity and the least negative base water potentials for germination. Cluster F represented the largest-seeded grasses with large initial roots but slow RGR and RER, low CTD and slower predicted germination rates with higher base temperatures. While cluster A consisted of all invasive annual grasses (three of four in the study), native and non-native perennials were spread throughout other clusters (Table 1). Dendrogram structure, trait means and SEs for each cluster, and significant trait differences between clusters are given in Table 5.
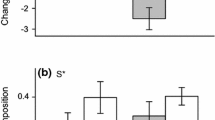
No significant differences were detected among clusters with respect to germination probability under either water treatment, and weak but insignificant differences were detected among species with respect to emergence (Table 6). Clusters differed significantly with respect to seedling survival under the low water treatment and cumulative survival under the high water treatment (Table 6). However, multiple comparisons revealed that significant differences occurred between just two clusters or were non-detectable, respectively (Fig. 2a, b).
Life stage transition probabilities of individual grass varieties (closed circles) within six trait-based recruitment types identified by cluster analysis (Table 5, clusters A–F) under (a–d) low water and (e–h) high-water treatments. Means (open circles) and SEs for each cluster are shown. Different letters indicate significant differences between groups. Although cumulative survival probability differed significantly between clusters in the high-water treatment (Table 6), multiple comparisons were non-significant
Discussion
In this exploratory analysis of trait variation across life stages, we characterized new links and divisions between seedling growth and leaf function, root function and germination. First, our results affirmed the seed mass—growth rate tradeoff previously found among woody and herbaceous life forms (e.g., Baraloto et al. 2005; Houghton et al. 2013; Wright and Westoby 1999) and linked this spectrum to root traits. Trait associations along RC1 suggested that larger seed mass enabled massive root growth upon germination, which could promote early seedling survival among slow-growing grasses. However, smaller seeded grasses with less massive initial roots tended to compensate with more rapid root elongation and seedling growth rates through establishment. Hallett et al. (2011) identified a similar positive link between seed mass and early root length among Mediterranean-climate forbs; while small-seeded species did not appear to achieve higher root growth rates overall in the first 2 months, rates were much more variable. Together, this evidence suggests that small-seeded species have the potential for rapid elongation of small initial root systems, but that this is dependent on microsite and external resource availability. Because seed mass is easily measured and has implications for seedling survival and growth, it has become a key trait in ecological strategies (Moles and Westoby 2006; Westoby 1998). However, germination traits did not link strongly to seed mass or this spectrum, indicating that seed mass alone is unlikely to capture functional differences in recruitment response.
Interestingly, we also identified a separate growth-related spectrum (RC2) in which rapidly growing seedlings not only displayed higher SLA and lower LDMC [as predicted by the leaf economics spectrum (Poorter et al. 2009; Shipley 2006)], but tended to have lower tissue density from the time of germination (low CTD) and more rapid germination. In conjunction with previous studies suggesting a link between rapid seedling growth and germination in macrophytes (Shipley et al. 1989), wetland plants (Shipley and Parent 1991) and terrestrial plants (Grime et al. 1981), our results indicate that it may be possible to make general predictions about the speed of germination based on easily measurable growth-related traits like SLA. However, while rate sum was weakly correlated with SLA and RER (Table 3), PCA axes together explained less than 50 % of the variation in rate sum across species (Table 4), suggesting substantial variation in the speed of germination external to this leaf tissue spectrum (illustrated through cluster analysis, described below). Similarly, the separate association of seedling growth rate with traits on RC1 and RC2 highlights that RGR has multiple underlying mechanisms which may not be captured by one aspect of seed or leaf function alone. Ultimately, these two axes support previous work identifying seed mass and leaf physiology as two independent axes which define plant strategies globally (Diaz et al. 2004; Laughlin et al. 2010; Westoby 1998), while suggesting that these spectra may also extend to aspects of root and germination function.
Importantly, some seed and belowground traits with potential implications for recruitment showed little correspondence to aboveground function. Hydrothermal time and base water potential for germination were highly correlated (as found in Allen et al. 2000) and fell out separately on RC3, with the general lack of correspondence to seedling traits (Table 3) suggesting that neither the driest conditions in which a species can germinate, nor the accumulation of moisture and temperature required to germinate, were strongly related to general strategies of seedling development in this environment. No other studies to our knowledge have examined these trait links directly, although some studies have suggested that these attributes may tie into certain strategies, such as salt tolerance or invasiveness (Allen et al. 2000; Pérez-Fernández et al. 2000). While theory predicts weaker associations between seed and seedling function across species (Grime et al. 1997; Leishman and Westoby 1992), the separation of the fourth axis tradeoff between root allocation (RMR) and root tissue efficiency (SRL) from root elongation and seedling growth was more surprising. RMR and SRL influence resource acquisition and are often linked to some aspect of whole plant growth (reviewed by Reich 2014). However, our results suggest that while RER may be tied to whole plant growth across species, root morphological traits are not always linearly associated with their aboveground counterparts [e.g., SLA, as found in Tjoelker et al. (2005)] or with growth. Given the equivocality of above- and belowground relationships across relatively few studies (reviewed by Freschet et al. 2010), there must be a greater effort to measure root traits across species and environments to understand how they contribute to concepts of ecological strategy.
The identification of four separate trait axes indicates that while some aspects of seed and seedling function are related, ecological strategy during recruitment is ultimately a product of location along several functional spectra which may influence germination, emergence, and seedling survival. A major benefit of cluster analysis was that it allowed us to isolate general recruitment types representing possible combinations of attributes from many dimensions of trait variation. We separated six groups of grasses with core-defining attributes across seed and seedling stages, but found substantial challenges in their ability to predict recruitment responses.
Although there are few studies to which identified recruitment types can be compared, the major veins of cluster separation—growth rate and germination rate—were similar to those which typically isolate adult strategies. For example, growth rate represents a key spectrum in plant ecology (Reich 2014) and has been used to distinguish stress-tolerant species (slower growing) from ruderal and competitive species (typically faster-growing annuals and perennials, respectively) under the competitor–stress tolerant–ruderal (CSR) scheme (Grime 1977). Incorporating both seedling growth rate and predicted germination rate, we found that four of six types maintained either rapid (groups A, B) or slow (groups E, F) developmental rates throughout recruitment. Under the CSR scheme, we might thus apply the classification of these groups as ruderal annuals (group A), competitive perennials (group B) and stress-tolerant perennials (groups E, F) to recruitment stages. In contrast to the concept of stress tolerance in slow-growing plants which survive harsh conditions, slow germinators required relatively favorable cues to germinate (e.g., higher temperatures for group F or wetter conditions for group E). While this may result in fewer germinants from the seed pool in a given year, these conservative attributes may minimize the chance of encountering fatal environmental hazards such as freezing or extreme drought during the earliest vulnerable stages (Donohue et al. 2010). In contrast to the parallel relationship between germination rate and seedling growth rate observed among other grasses, the two remaining groups, C and D, exhibited slower germination traits, but grew rapidly once emerged. Collectively, these groups highlighted the range of recruitment niches which may be filled given the multi-dimensional nature of trait variation across seed and seedling stages (Table 5).
Grass membership within clusters also has important implications for the applied value of recruitment types. Three of four invasive annual grasses fell into group A, exhibiting the most rapid germination and RERs—a rapid development syndrome often associated with invasive species (Pyšek and Richardson 2007). While no perennials reached these rapid germination rates (as found in Grime et al. 1981; Shipley et al. 1989; Shipley and Parent 1991), germination rates of group B were highest, suggesting that this group may be most directly competitive with invasive annual recruitment. Importantly, varieties of the same species were sometimes classified as different recruitment types. For example, while four varieties of Elymus elymoides displayed similar leaf and RGR attributes, substantial variation in seed size, initial root mass, plasticity and germination attributes resulted in assignment to three different clusters (Table 1), demonstrating the extent of recruitment-related trait variation which can occur between conspecific populations. Indeed, significant directional selection on morphology and phenology was observed in restoration-sown E. elymoides populations after just 2 years (Kulpa and Leger 2013), highlighting that both seed source and growing condition may be important considerations in trait measurement and subsequent species classification into recruitment types (Cochrane et al. 2015). This also emphasizes the critical importance of considering traits in addition to species identity in restoration approaches and ecological models, and makes a case for the identification of key traits which could eventually be used to separate recruitment strategies related to patterns of recruitment.
Although identified recruitment types were linked to a suite of seed and seedling attributes, they had a limited ability to predict life stage transition probabilities under either water scenario (Table 6; Fig. 2). Based on the functional characteristics of recruitment types, we might have expected higher germination probabilities in groups A and B (given high rate sum and earlier germination), higher survival in groups C and E under low and high water conditions, respectively (given RGR plasticity), and lower germination probabilities but higher survival in groups D and F. While seedling survival differed between a couple clusters, as a whole, survival probabilities exceeded 0.60 across all but four grass varieties, regardless of watering treatment. Given that water limitation was apparently removed as a survival barrier in this experiment, we had limited ability to test the potential of trait-based recruitment strategies to predict seedling performance. However, the traits and strategies included here could be relevant to seedling survival under other conditions—particularly if drought or competition became a significant limiting factor. For example, Pillay and Ward (2014) found that seedling RGR and SLA were indicative of seedling competitive effects and responses across savanna tree species, respectively, which could favor functional types similar to group A annual grasses. In contrast, germination and emergence limitations were not overcome across grass varieties, suggesting a stronger or more complex range of environmental limitations acting on these processes in this environment. We observed within-cluster variation in germination and emergence probabilities as high five- and 12-fold, respectively, with little evidence of distinct cluster differences in these probabilities. Thus, while we demonstrate that germination and emergence processes can be of substantial importance to understanding recruitment dynamics, substantial challenges remain in the application of functional traits to unraveling these processes.
In a recent study, we examined direct links between a subset of the present traits and germination and emergence probabilities, and found that some of the variation in these processes could be explained by seed mass and CTD, but that much more remained to be explained (Larson et al. 2015). While traits related to germination and especially emergence are relatively understudied in functional ecology, the complexity and variability of environmental controls on these processes could also pose a significant challenge. For example, although we moved beyond testing the influence of single traits on germination probabilities to identifying recruitment types sharing a suite of germination attributes reflecting temperature and moisture requirements, germination variability remained high within clusters.
Generally, plant functional types have been used to examine longer term responses of communities over time [e.g., shifts in abundance as a result of grazing (Louault et al. 2005), fragmentation (Kolb and Diekmann 2005) or fire (Müller et al. 2007)]. In comparison, predicting recruitment probabilities in a given year depends on capturing responses to much finer-scale spatiotemporal variability in environmental filters (e.g., Gómez-Aparicio 2008; Young et al. 2015). Thus, while very high rate sums of group A annuals indicate rapid germination responses which may allow higher germination totals in a given year, observed germination probabilities were among the lowest across grasses under both water treatments (Fig. 2). Lack of germination in trial bags pulled as early as January-March (authors’ unpublished data) suggested that dormancy or seed death may have been sparked by unanticipated seed responses to drought or freezing prior to germination (Clauss and Venable 2000; Elberse and Breman 1990). Given the substantial and complex influence of environmental context (water, temperature, soil biota, environmental hazards) on early life stage transition rates, using recruitment types to predict demographic patterns in a given year is likely to be impossible without considering environmental context at a finer scale and exploring a wider range of relevant traits, such as dormancy, seed-persistence metrics, germination phenology and shoot and radicle physiology (Baskin and Baskin 1988; Dalling et al. 2011; Long et al. 2015). Moving forward, a key task will be to extend empirical studies to consider additional traits and environments over longer time periods to identify whether general dimensions of trait variation are able to capture fine- or broad-scale recruitment patterns.
Given the multi-dimensional nature of recruitment trait variation observed across seed and seedling stages (at least four axes among traits here), recruitment types (i.e., clusters) which capture sets of related attributes among species may provide an advantageous tool in plant functional ecology. However, the lack of correspondence between recruitment types and life stage transition rates identified here suggests that the functional traits underlying recruitment types (e.g., germination and growth rates) were insufficient to capture responses to the particular filters in this environment. In order to link these demographic patterns to functional traits, we must develop a better understanding of how coordinated sets traits interact with the environment during separate recruitment stages. For example, incorporating dormancy and seed longevity traits (which may be unrelated to hydrothermal time requirements) into germination functional types may better predict seed responses in different environments and years. These germination types may exist independent of seedling strategies, emergence and survival. The ability to link traits and demography may ultimately be limited by the feasibility of including ever-finer scales of environmental and trait information; however, efforts to describe recruitment patterns across species and environmental gradients are beginning to emerge (Fraaije et al. 2015; Gómez-Aparicio 2008), and traits and functional types which could differentiate performance during recruitment remain virtually untested across species in trait-based ecology. There is substantial opportunity to incorporate these ideas into empirical studies to explore general relationships between traits, the environment, and recruitment dynamics.
References
Allen PS, Meyer SE, Khan MA (2000) Hydrothermal time as a tool in comparative germination studies. Seed Biology: Advances and Applications. Wallingford, UK, CABI 401–410
Baraloto C, Forget P-M, Goldberg DE (2005) Seed mass, seedling size and neotropical tree seedling establishment. J Ecol 93:1156–1166
Baskin CC, Baskin JM (1988) Germination ecophysiology of herbaceous plant species in a temperate region. Am J Bot 75:286–305. doi:10.2307/2443896
Bello Fd et al (2010) Towards an assessment of multiple ecosystem processes and services via functional traits. Biodivers Conserv 19:2873–2893. doi:10.1007/s10531-010-9850-9
Bernard-Verdier M, Navas M-L, Vellend M, Violle C, Fayolle A, Garnier E (2012) Community assembly along a soil depth gradient: contrasting patterns of plant trait convergence and divergence in a Mediterranean rangeland. J Ecol 100:1422–1433. doi:10.1111/1365-2745.12003
Boutin C, Keddy PA (1993) A functional classification of wetland plants. J Veg Sci 4:591–600
Bradford KJ (2002) Applications of hydrothermal time to quantifying and modeling seed germination and dormancy. Weed Sci 50:248–260. doi:10.1614/0043-1745(2002)050[0248:aohttq]2.0.co;2
Clauss MJ, Venable DL (2000) Seed germination in desert annuals: an empirical test of adaptive bet hedging. Am Nat 155:168–186. doi:10.1086/303314
Cochrane A, Yates CJ, Hoyle GL, Nicotra AB (2015) Will among-population variation in seed traits improve the chance of species persistence under climate change? Glob Ecol Biogeogr 24:12–24. doi:10.1111/geb.12234
Cornwell WK, Ackerly DD (2009) Community assembly and shifts in plant trait distributions across an environmental gradient in coastal California. Ecol Monogr 79:109–126. doi:10.1890/07-1134.1
Dalling JW, Davis AS, Schutte BJ, Elizabeth Arnold A (2011) Seed survival in soil: interacting effects of predation, dormancy and the soil microbial community. J Ecol 99:89–95
Diaz S et al (2004) The plant traits that drive ecosystems: evidence from three continents. J Veg Sci 15:295–304
Donohue K, Rubio de Casas R, Burghardt L, Kovach K, Willis CG (2010) Germination, postgermination adaptation, and species ecological ranges. Annu Rev Ecol Evol Syst 41:293–319. doi:10.1146/annurev-ecolsys-102209-144715
Elberse WT, Breman H (1990) Germination and establishment of sahelian rangeland species. II. Effects of water availability. Oecologia 85:32–40. doi:10.2307/4219473
Fenner M, Thompson K (2005) The ecology of seeds. Cambridge University Press
Fraaije RGA, ter Braak CJF, Verduyn B, Breeman LBS, Verhoeven JTA, Soons MB (2015) Early plant recruitment stages set the template for the development of vegetation patterns along a hydrological gradient. Funct Ecology:n/a-n/a. doi:10.1111/1365-2435.12441
Freschet GT, Cornelissen JHC, Van Logtestijn RSP, Aerts R (2010) Evidence of the ‘plant economics spectrum’ in a subarctic flora. J Ecol 98:362–373. doi:10.1111/j.1365-2745.2009.01615.x
Fry EL, Power SA, Manning P (2014) Trait-based classification and manipulation of plant functional groups for biodiversity–ecosystem function experiments. J Veg Sci 25:248–261
Funk JL, Cleland EE, Suding KN, Zavaleta ES (2008) Restoration through reassembly: plant traits and invasion resistance. Trends Ecol Evol 23:695–703. doi:10.1016/j.tree.2008.07.013
Ganade G, Westoby M (1999) Seed mass and the evolution of early-seedling etiolation. Am Nat 154:469–480
Gardarin A, Dürr C, Colbach N (2010) Effects of seed depth and soil aggregates on the emergence of weeds with contrasting seed traits. Weed Res 50:91–101
Gardarin A, Dürr C, Colbach N (2011) Prediction of germination rates of weed species: relationships between germination speed parameters and species traits. Ecol Model 222:626–636. doi:10.1016/j.ecolmodel.2010.10.005
Garnier E, Navas M-L (2012) A trait-based approach to comparative functional plant ecology: concepts, methods and applications for agroecology. A review. Agron Sustain Dev 32:365–399. doi:10.1007/s13593-011-0036-y
Gómez-Aparicio L (2008) Spatial patterns of recruitment in Mediterranean plant species: linking the fate of seeds, seedlings and saplings in heterogeneous landscapes at different scales. J Ecol 96:1128–1140. doi:10.1111/j.1365-2745.2008.01431.x
Grime JP (1977) Evidence for the existence of three primary strategies in plants and its relevance to ecological and evolutionary theory. Am Nat 111:1169–1194
Grime JP, Mason G, Curtis AV, Rodman J, Band SR (1981) A comparative study of germination characteristics in a local flora. J Ecol 69:1017–1059. doi:10.2307/2259651
Grime JP et al (1997) Integrated screening validates primary axes of specialisation in plants. Oikos 79:259–281. doi:10.2307/3546011
Gummerson RJ (1986) The effect of constant temperatures and osmotic potentials on the germination of sugar beet. J Exp Bot 37:729–741
Hallett L, Standish R, Hobbs R (2011) Seed mass and summer drought survival in a Mediterranean-climate ecosystem. Plant Ecol 212:1479–1489. doi:10.1007/s11258-011-9922-2
Hardegree SP, Flerchinger GN, Van Vactor SS (2003) Hydrothermal germination response and the development of probabilistic germination profiles. Ecol Model 167:305–322. doi:10.1016/s0304-3800(03)00192-3
Hardegree SP et al (2013) Hydrothermal assessment of temporal variability in seedbed microclimate. Rangel Ecol Manage 66:127–135
Harris GA, Wilson AM (1970) Competition for moisture among seedlings of annual and perennial grasses as influenced by root elongation at low temperature. Ecology 51:530–534. doi:10.2307/1935392
Houghton J, Thompson K, Rees M (2013) Does seed mass drive the differences in relative growth rate between growth forms? Proc R Soc B-Biol Sci. doi:10.1098/Rspb.2013.0921
Hunt R, Causton DR, Shipley B, Askew AP (2002) A modern tool for classical plant growth analysis. Ann Bot 90:485–488
James JJ, Svejcar TJ, Rinella MJ (2011) Demographic processes limiting seedling recruitment in arid grassland restoration. J Appl Ecol 48:961–969
Kolb A, Diekmann M (2005) Effects of life-history traits on responses of plant species to forest fragmentation. Conserv Biol 19:929–938
Kulpa SM, Leger EA (2013) Strong natural selection during plant restoration favors an unexpected suite of plant traits. Evol Appl 6:510–523
Lambers H, Poorter H (1992) Inherent variation in growth rate between higher plants: a search for physiological causes and ecological consequences. Adv Ecol Res 34:187–261
Larson JE, Sheley RL, Hardegree SP, Doescher PS, James JJ (2015) Seed and seedling traits affecting critical life stage transitions and recruitment outcomes in dryland grasses. J Appl Ecol 52:199–209. doi:10.1111/1365-2664.12350
Laughlin DC (2014) Applying trait-based models to achieve functional targets for theory-driven ecological restoration. Ecol Lett 17:771–784
Laughlin DC, Leppert JJ, Moore MM, Sieg CH (2010) A multi-trait test of the leaf-height-seed plant strategy scheme with 133 species from a pine forest flora. Funct Ecol 24:493–501. doi:10.1111/j.1365-2435.2009.01672.x
Lavorel S, McIntyre S, Landsberg J, Forbes T (1997) Plant functional classifications: from general groups to specific groups based on response to disturbance. Trends Ecol Evol 12:474–478
Leishman MR, Westoby M (1992) Classifying plants into groups on the basis of associations of individual traits-evidence from Australian semi-arid woodlands. J Ecol 80:417–424. doi:10.2307/2260687
Leishman MR, Westoby M (1994) The role of seed size in seedling establishment in dry soil conditions—experimental evidence from semi-arid species. J Ecol 82:249–258. doi:10.2307/2261293
Long RL et al (2015) The ecophysiology of seed persistence: a mechanistic view of the journey to germination or demise. Biol Rev 90:31–59. doi:10.1111/brv.12095
Louault F, Pillar V, Aufrere J, Garnier E, Soussana JF (2005) Plant traits and functional types in response to reduced disturbance in a semi-natural grassland. J Veg Sci 16:151–160
McCune B, Grace JB (2002) Analysis of ecological communities. MjM software design, Gleneden Beach, OR
McGill BJ, Enquist BJ, Weiher E, Westoby M (2006) Rebuilding community ecology from functional traits. Trends Ecol Evol 21:178–185. doi:10.1016/j.tree.2006.02.002
Milberg P, Andersson L, Thompson K (2000) Large-seeded spices are less dependent on light for germination than small-seeded ones. Seed Sci Res 10:99–104
Moles AT, Westoby M (2006) Seed size and plant strategy across the whole life cycle. Oikos 113:91–105
Müller S, Overbeck G, Pfadenhauer J, Pillar V (2007) Plant functional types of woody species related to fire disturbance in forest–grassland ecotones. Plant Ecol 189:1–14. doi:10.1007/s11258-006-9162-z
Pearson TRH, Burslem DFRP, Mullins CE, Dalling JW (2002) Germination ecology of Neotropical pioneers: interacting effects of environmental conditions and seed size. Ecology 83:2798–2807
Pérez-Fernández MA, Lamont BB, Marwick AL, Lamont WG (2000) Germination of seven exotic weeds and seven native speciesin south-western Australia under steady and fluctuating water supply. Acta Oecol 21:323–336. doi:10.1016/s1146-609x(00)01084-5
Pillay T, Ward D (2014) Competitive effect and response of savanna tree seedlings: comparison of survival, growth and associated functional traits. J Veg Sci 25:226–234. doi:10.1111/jvs.12075
Poorter L, Bongers F (2006) Leaf traits are good predictors of plant performance across 53 rain forest species. Ecology 87:1733–1743
Poorter H, Niinemets Ü, Poorter L, Wright IJ, Villar R (2009) Causes and consequences of variation in leaf mass per area (LMA): a meta-analysis. New Phytol 182:565–588
Pyšek P, Richardson DM (2007) Traits associated with invasiveness in alien plants: where do we stand? In: Nentwig N (ed) Biological invasions. Springer, Berlin, pp 97–125
Reich PB (2014) The world-wide ‘fast–slow’ plant economics spectrum: a traits manifesto. J Ecol 102:275–301
Ryser P (1996) The importance of tissue density for growth and life span of leaves and roots: a comparison of five ecologically contrasting grasses. Funct Ecol 10:717–723. doi:10.2307/2390506
Sharitz RR, McCormick JF (1973) Population dynamics of two competing annual plant species. Ecology 54:723–740. doi:10.2307/1935669
Shipley B (2006) Net assimilation rate, specific leaf area and leaf mass ratio: which is most closely correlated with relative growth rate? A meta-analysis. Funct Ecol 20:565–574. doi:10.1111/j.1365-2435.2006.01135.x
Shipley B, Parent M (1991) Germination responses of 64 wetland species in relation to seed size, minimum time to reproduction and seedling relative growth rate. Funct Ecol 5:111–118. doi:10.2307/2389561
Shipley B, Keddy PA, Moore DRJ, Lemky K (1989) Regeneration and establishment strategies of emergent macrophytes. J Ecol 77:1093–1110
Thompson K, Hillier SH, Grime JP, Bossard CC, Band SR (1996) A functional analysis of a limestone grassland community. J Veg Sci 7:371–380
Tjoelker MG, Craine JM, Wedin D, Reich PB, Tilman D (2005) Linking leaf and root trait syndromes among 39 grassland and savannah species. New Phytol 167:493–508
Valladares F, Sanchez-Gomez D, Zavala MA (2006) Quantitative estimation of phenotypic plasticity: bridging the gap between the evolutionary concept and its ecological applications. J Ecol 94:1103–1116
Valladares F, Gianoli E, Gómez JM (2007) Ecological limits to plant phenotypic plasticity. New Phytol 176:749–763. doi:10.1111/j.1469-8137.2007.02275.x
Westoby M (1998) A leaf-height-seed (LHS) plant ecology strategy scheme. Plant Soil 199:213–227. doi:10.1023/a:1004327224729
Westoby M, Wright IJ (2006) Land-plant ecology on the basis of functional traits. Trends Ecol Evol 21:261–268. doi:10.1016/j.tree.2006.02.004
Wright IJ, Westoby M (1999) Differences in seedling growth behaviour among species: trait correlations across species, and trait shifts along nutrient compared to rainfall gradients. J Ecol 87:85–97. doi:10.1046/j.1365-2745.1999.00330.x
Young TP, Zefferman EP, Vaughn KJ, Fick S (2015) Initial success of native grasses is contingent on multiple interactions among exotic grass competition, temporal priority, rainfall and site effects. AoB Plants. doi:10.1093/aobpla/plu081
Acknowledgments
The authors thank M. Bernard-Verdier, E. Schupp, and two anonymous reviewers for comments which greatly improved earlier versions of this manuscript. We also thank L. Ziegenhagen, B. Bingham, L. Smith, A. Boehm and L. Kosberg for assistance with data collection and E. Sabry, A. Muldoon, B. McCune, T. Jones and USDA Plant Materials personnel for providing resources and helpful feedback on key aspects of the project.
Author contribution statement
J. E. L., J. J. J., R. L. S., P. S. D. and S. P. H. contributed to the conception and design of the experiments. J. E. L., J. J. J. and S. P. H. performed the experiments. J. E. L. analyzed the data. J. E. L. and J. J. J. wrote the manuscript; other authors provided editorial advice.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Funding
This study was funded as part of the USDA-Agricultural Research Service Area-wide Ecologically-based Invasive Plant Management of Annual Grasses program.
Additional information
Communicated by John M. Stark.
Highlighted student research statement
Utilizing germination, belowground and leaf traits of 47 grasses, we show that trait variation during recruitment is multidimensional in its influences on seed and seedling function. Furthermore, we detected an unexpectedly high degree of intraspecific variation in the nature of trait interactions. However, weak links to recruitment highlight a key knowledge gap in trait-based ecology. This study provides a framework for further explorations of trait interactions and influence during critical life history phases of seed germination and seedling recruitment.
Electronic supplementary material
Below is the link to the electronic supplementary material.
442_2015_3430_MOESM1_ESM.docx
Species data (means and SDs), including seedlot viability, life stage transition probabilities, and seed and seedling trait data. (DOCX 74 kb)
442_2015_3430_MOESM2_ESM.docx
Cluster analysis dendrograms and results of discriminant analysis leave-one-out cross-validation of cluster assignments. (DOCX 75 kb)
Rights and permissions
About this article
Cite this article
Larson, J.E., Sheley, R.L., Hardegree, S.P. et al. Do key dimensions of seed and seedling functional trait variation capture variation in recruitment probability?. Oecologia 181, 39–53 (2016). https://doi.org/10.1007/s00442-015-3430-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-015-3430-3