Abstract
The purpose of the present study was based on assessments of the antiparasitic activities of synthesized titanium dioxide nanoparticles (TiO2 NPs) utilizing leaf aqueous extract of Catharanthus roseus against the adults of hematophagous fly, Hippobosca maculata Leach (Diptera: Hippoboscidae), and sheep-biting louse, Bovicola ovis Schrank (Phthiraptera: Trichodectidae). The synthesized TiO2 NPs were analyzed by X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FTIR), scanning electron microscopy (SEM), and atomic force microscopy (AFM). The formation of the TiO2 NPs synthesized from the XRD spectrum compared with the standard confirmed spectrum of titanium particles formed in the present experiments were in the form of nanocrystals, as evidenced by the peaks at 2θ values of 27.43°, 36.03°, and 54.32°. The FTIR spectra of TiO2 NPs exhibited prominent peaks at 714 (Ti–O–O bond), 1,076 (C–N stretch aliphatic amines), 1,172 (C–O stretching vibrations in alcoholic groups), 1,642 (N–H bend bond), and 3,426 (O–H stretching due to alcoholic group). SEM analysis of the synthesized TiO2 NPs clearly showed the clustered and irregular shapes, mostly aggregated and having the size of 25–110 nm. By Bragg’s law and Scherrer’s constant, it is proved that the mean size of synthesized TiO2 NPs was 65 nm. The AFM obviously depicts the formation of the rutile and anatase forms in the TiO2 NPs and also, the surface morphology of the particles is uneven due to the presence of some of the aggregates and individual particles. Adulticidal parasitic activity was observed in varying concentrations of aqueous leaf extract of C. roseus, TiO2 solution, and synthesized TiO2 NPs for 24 h. The maximum parasitic activity was observed in aqueous crude leaf extracts of C. roseus against the adults of H. maculata and B. ovis with LD50 values of 36.17 and 30.35 mg/L, and r 2 values of 0.948 and 0.908, respectively. The highest efficacy was reported in 5 mM TiO2 solution against H. maculata and B. ovis (LD50 = 33.40 and 34.74 mg/L; r 2 = 0.786 and 0.873), respectively, and the maximum activity was observed in the synthesized TiO2 NPs against H. maculata and B. ovis with LD50 values of LD50 = 7.09 and 6.56 mg/L, and r 2 values of 0.880 and 0.913, respectively. This method is considered as an innovative alternative approach to control the hematophagous fly and sheep-biting louse.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The widespread development of tick and fly resistance and high cost of the conventional ectoparasitic drugs have limited the control of veterinary parasites and hence led to evaluation of medicinal plants as an alternative source to control parasites and the tick. The production losses largely result from meat quantity loss caused by tick worry, and the death of cattle from tick fever and the reduction in meat quantity associated with tick infestation were estimated (Sing et al. 1983). Hippobosca maculata is a serious pest of equines in India and is also cosmopolitan in distribution (Parashar et al. 1991). These flies are adapted to a more or less continuous existence on the bodies of their host. Blood sucking flies cause damage to livestock through both direct damage, and as vectors of viral and bacterial infections, the hematophagous flies, Hippoboscidae, are well known to infest sheep and cattle in different parts of the world (Soulsby 1982). Paramphistomosis has been a neglected trematode infectious disease in ruminants but has recently emerged as an important cause of productivity loss. Economic loss caused by paramphistome infections has not been estimated but may be greater than those caused by many other parasites (Hanna et al. 1988). H. maculata is responsible for losses in milk, meat, and leather production and for the death of a number of animals, which results in economic losses associated with cattle production, and a recent survey on acaricide resistance conducted through questionnaire reported a large-scale acaricide resistance in India (FAO 2004). Bovicola ovis is a scurf feeder which spends its entire life on sheep. It thermoregulates by moving up and down the wool fibers to remain within its narrow thermal tolerance range.
An important area of research in nanotechnology is the synthesis of nanoparticles of different chemical compositions, sizes, and controlled monodispersity. Currently, there is an ever-growing need to develop environmentally benign nanoparticle synthesis processes. Nanoparticles play an indispensable role in drug delivery, diagnostics, imaging, sensing, gene delivery, artificial implants, and tissue engineering (Morones et al. 2005). The plant-mediated biosynthesis of nanoparticles is advantageous over chemical and physical methods because it is a cost-effective and environment-friendly method, where it is not necessary to use high pressure, energy, temperature, and toxic chemicals (Goodsell 2004).
Current applications to livestock for flies control primarily use products containing organophosphates or synthetic pyrethroids. However, some flies species have been becoming resistant to these insecticides as a result of repeated exposure (Foil et al. 2004), and these chemicals may have negative effects on non-target organisms including humans. Parasite control, in general, is based on the use of chemical insecticides. Unfortunately, repeated use of chemicals has many negative side effects, including the possibility of causing the development of chemical resistance in some ticks population, and food and environmental contamination if these products are improperly used. Public concerns about the environmental impact and safety of chemical applications are driving research into alternative, sustainable methods for parasites control, including biological control (Chandler et al. 2000). There is a development of experimental processes for the synthesis of nanoparticles of different sizes, shapes, and controlled dispersity. With the development of new chemical or physical methods, the concern for environmental contaminations are also heightened as the chemical procedures involved in the synthesis of nanomaterials generate a large amount of hazardous by-products. Thus, there is a need for “green chemistry” that includes a clean, nontoxic, and environment-friendly method of nanoparticle synthesis (Mukherjee et al. 2001).
In recent years, nanoparticle composites have become important owing to their small size and large surface area and because they exhibit unique properties not seen in bulk materials. As a result, nanoparticles (NPs) have useful applications in photovoltaic cells, optical and biological sensors, conductive materials, and coating formulations (Templeton et al. 2000), and the use of plants for nanoparticle synthesis can be advantageous over other biological processes because it eliminates the elaborate process of maintaining cell cultures and can also be suitably scaled up for large-scale nanoparticle synthesis (Shankar et al. 2004). Nanoparticles have attracted considerable attention because of their various applications. Jha et al. (2009) reported that the synthesized TiO2 NPs using Lactobacillus sp. and Sachharomyces cerevisae possessed anti-bacterial and anti-fungal properties. Use of plant extract for the synthesis of nanoparticles could be advantageous over other environmentally benign biological processes because it eliminates the elaborate process of maintaining cell cultures. Recently, green TiO2 nanoparticles have been synthesized using natural products like Nyctanthes arbor-tristis extract (Sundrarajan and Gowri 2011).
Catharanthus roseus L. (Vinca rosea) belonging to family Apocynaceae is known with various names in India and all over the world. Hot water decoction of the leaves or the whole plant is used for treatment of diabetes in several countries (Novello and Sprague 1957). C. roseus reported as antineoplastic agents to treat leukemia, Hodgkin’s disease, malignant lymphomas, neuroblastoma, rhabdomyosarcoma, Wilms’ tumor, and other types of cancers (Fischhof et al. 1996). Nayak (2006) reported that the dried or wet leaf extracts of C. roseus were applied as a paste on wounds in some rural communities. The fresh juice from the leaves of C. roseus made into a tea has been used by Ayurvedic physicians in India and other countries for external use to treat skin problems, dermatitis, eczema, and acne (El-Sayed and Cordell 1981).
Materials and methods
Materials
The leaves of C. roseus were collected from Melvisharam (12°56′23″N, 79°14′23″E), Tamil Nadu, India. Taxonomic identification was done by Dr. C. Hema from the Department of Botany, Arignar Anna Government Arts College for Women, Walajapet, Vellore, India. The voucher specimen was numbered and kept in our research laboratory for further reference. Titanium dioxide (TiO2 purity 99.0%) was purchased from Himedia, Mumbai, India.
Parasite collection
The newly attached adults H. maculata Leach (Diptera: Hippoboscidae) were collected from the softer skin inside the thigh, flanks, abdomen, brisket, and forelegs of naturally infested cattle and sheep. H. maculata adults have a short, straight capitulum, and brown to cream colored body. The adult of B. ovis Schrank (Phthiraptera: Trichodectidae) were collected from the body of goats. The sheep body louse is a pale yellow insect, brown transverse stripe on the abdomen, and a broad, red-brown head. The parasites were identified by Department of Veterinary Parasitology, Madras Veterinary College, Tamil Nadu Veterinary and Animal Sciences University, Chennai, Tamil Nadu, India.
Synthesis of titanium dioxide nanoparticles
The fresh leaf of C. roseus broth solution was prepared by taking 10 g of thoroughly washed and finely cut leaves in a 250 mL Erlenmeyer flask along with 100 mL of sterilized double distilled water and then boiling the mixture for 10 min before finally decanting it. The extract was filtered with Whatman filter paper no. 1 and stored at −15°C and could be used within 1 week. The filtrate was treated with 20 ml of aqueous leaf extract added in 80 ml of 5 mM (39.94 mg of TiO2 powder in 100 mL Milli-Q water) solution in an Erlenmeyer flask under stirring at 50°C. After 4 h of continuous stirring, the formed light green color change indicated the formation of TiO2 NPs.
Filter paper impregnated bioassay test
The adults of H. maculata and B. ovis were collected from various parts of both sheep and goats. The aqueous leaf extract of C. roseus, TiO2 solution, and synthesized titanium dioxide nanoparticles (TiO2 NPs) were used in filter paper impregnated tests in sealed glass jars. Different concentrations of aqueous extract and TiO2 solution ranging from aliquot of 6.12, 12.5, 25, 50, and 100 mg/L and synthesized TiO2 NPs of 5, 10, 15, 20, and 25 mg/L were prepared and a series of filter paper envelopes (Whatman filter paper no. 1, ~216 cm2) with micropores treated with each concentration of samples. Each envelope was treated with 3 mL solution uniformly distributed with a pipette on internal surfaces. Five envelopes were impregnated with each tested solution. The control papers were impregnated with distilled water only. H. maculata and B. ovis (20–50 individuals) were collected directly from infested animals. Each test was performed by placing 20 adults of H. maculata and B. ovis into the series of filter paper envelopes (Whatman filter paper no.1, ∼216 cm2) with micropores treated with each concentration. The numbers of dead adults were counted for 24 h. The death of the parasites was confirmed when there was cessation of motility or waggling of the appendages upon touching with a needle. The dose–response data were subjected to probit analysis to determine the LD50 values for the 24 h exposure under constant climatic conditions (25°C, 12/12 h L/D; Nyamador et al. 2010).
Dose–response bioassay
The crude leaf extract of C. roseus, TiO2 solution (5 mM), and synthesized TiO2 NPs were subjected to dose–response bioassay for parasitic activity against the adults of H. maculata and B. ovis. Different concentrations ranging from 6.12 to 100 mg/L (for aqueous plant extracts, TiO2) and 5.0 to 25 mg/L (for synthesized TiO2 NPs) were prepared for parasitic activity. The numbers of dead parasites were counted after 24 h of exposure, and the percentage of mortality was reported from the average of five replicates. However, at the end of 24 h, the selected test samples turned out to be equal in their toxic potential.
Data analysis
The average parasite mortality data were subjected to probit analysis for calculating LC50, and other statistics at 95% fiducial limits of upper confidence limit and lower confidence limit were calculated by using the software developed by Reddy et al. (1992). Results with p < 0.05 were considered to be statistically significant.
Characterization of TiO2 nanoparticles
The resulting pellet was dissolved in de-ionized water and filtered through Millipore filter (0.45 μm). The synthesized nanoparticles were identified by X-ray diffraction (XRD) spectroscopy (PerkinElmer Spectrum One instrument, PW1830 instrument operating at a voltage of 40 kV and a current of 30 mA with CuKα radiation). Fourier transform infrared (FTIR) spectra of the samples were measured using PerkinElmer Spectrum One instrument in the diffuse reflectance mode at a resolution of 4 cm−1 in KBr pellets. Powder samples for the FTIR were prepared similar to powder diffraction measurements. The FTIR spectra of synthesized TiO2 NPs taken were analyzed, which discussed for the possible functional groups for the formation of nanoparticles. For the scanning electron microscopic studies, 25 μL of sample was sputter-coated on copper stub, and the images of nanoparticles were studied using scanning electron microscopy (SEM; JEOL, ModelJFC-1600). Topography was studied using atomic force microscopy (AFM; PARKS scanning probe microscope) working in the non-contact mode. AFM images have been processed using XEI software given by PARKS system (Horcas et al. 2007).
Results
In the filter paper impregnated bioassay method, the aqueous leaf extracts, TiO2 solution (5 mM), and synthesized TiO2 NPs of C. roseus were noted; however, the synthesized TiO2 NPs showed 100% mortality against the adults of H. maculata and B. ovis at the concentration of 5,10,15,20, and 25 mg/L. The mortality of aqueous leaf extracts of C. roseus against the adults of H. maculata and B. ovis showed the values of LD50 = 36.17 and 30.35 mg/L, and r 2 = 0.948 and 0.908, respectively. Values of the efficacy of 5 mM TiO2 solution against H. maculata and B. ovis showed the LD50 values of 33.40 and 34.74 mg/L, and r 2 values of 0.786 and 0.873, respectively. The synthesized TiO2 NPs by C. roseus extract against the adults of H. maculata and B. ovis reported the LD50 values of 7.09 and 6.56 mg/L, and r 2 values of 0.880 and 0.913, respectively (Table 1). In the present study, the parasitic activity results showed the highest mortality in synthesized TiO2 NPs than the aqueous leaf extract of C. roseus. Each test included a control group (distilled water) with five replicates for each individual concentration. All the tested components that showed lethal effect and mortality were positively dose dependent. The results showed that the optimal hours for measuring the percent mortality adults of H. maculata and B. ovis synthesized TiO2 NPs were 13%, 22%, 38%, 82%, and 100%, and 11%, 29%, 44%, 63%, and 99% at 1, 6, 12, 18, and 24 h, respectively. The comparative study of larvicidal and adulticidal activity of different nanoparticles are shown in Table 2.
Characterization of TiO2 NPs
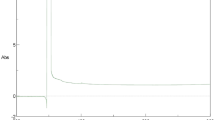
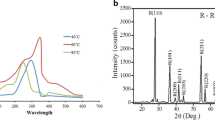
The formation of TiO2 NPs synthesized using C. roseus leaf extract was supported by X-ray diffraction measurements. XRD analysis showed three distinct diffraction peaks at 27.43°, 36.03°, and 54.32° which indexed the planes 110, 101, and 211, respectively, of the cubic face centered TiO2 (JCPDS no. 21–1272). The average grain size formed in the biosynthesis was determined using Scherrer’s formula, d = 0.89λ/βcosθ was estimated as 100 nm for the higher intense peak (Fig. 1). The sharp peaks and absence of unidentified peaks confirmed the crystallinity and higher purity of prepared nanoparticles.
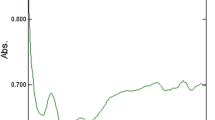
FTIR spectroscopy analyses were carried out to identify the biomolecules responsible for capping of the bioreduced TiO2 NPs synthesized using plant extract. Figure 2 shows the synthesized TiO2 NPs using C. roseus leaf aqueous extract where the absorption peaks were located at 714 (Ti–O–O bond), 1,076 (C–N stretch aliphatic amines), 1,172 (C–O stretching vibrations in alcoholic groups), 1,642 (N–H bend bond), and 3,426 (O–H stretching due to alcoholic group). SEM analyses of the synthesized TiO2 NPs clearly showed clustered and irregular shapes, mostly aggregated and having an average size of 25 to 110 nm with interparticle distance. It is clear in Fig. 3a, b the clustered structures with sizes of 65 nm. The line profile image was drawn by the XEI software and the horizontal line at 6 μm on a 2D AFM image. The height of the particles existing across the line can be evaluated by placing two cursors. The AFM was performed in order to know the topological map of the surface of the synthesized nanoparticles. The surface area of the nanoparticles has increased dramatically showing with the increase in the peaks Fig. 4a, b. The AFM obviously depicts the formation of the rutile and anatase forms in the TiO2 NPs, and also, the surface morphology of the particles is uneven due to the presence of some of the aggregates and individual particles.
Discussion
The adulticidal activity of aqueous leaf extracts and synthesized TiO2 NPs of C. roseus showed maximum activity observed in aqueous extract of C. roseus and the synthesized TiO2 NPs against H. maculata and B. ovis. The nanoparticle synthesis is important as the instability or aggregation of nanoparticles would lead to a decrease in their biological activities (Kvitek et al. 2008). The larvicidal aqueous crude leaf extracts and synthesized silver nanoparticles (Ag NPs) of Mimosa pudica showed the highest mortality in synthesized Ag NPs against the larvae of Rhipicephalus (Boophilus) microplus (8.98 mg/L, r 2 = 0.479; Marimuthu et al. 2011). In Nelumbo nucifera leaf, synthesized Ag NPs maximum efficacy was observed in crude methanol, aqueous, and synthesized Ag NPs against the larvae of Anopheles subpictus (LC50 = 8.89, 11.82, and 0.69 ppm; LC90 = 28.65, 36.06, and 2.15 ppm) and against the larvae of Culex quinquefasciatus (LC50 = 9.51, 13.65, and 1.10 ppm; LC90 = 28.13, 35.83, and 3.59 ppm), respectively (Santhoshkumar et al. 2011). The synthesized zinc oxide nanoparticles against R. microplus and Pediculus humanus capitis, and the larvae of A. subpictus and C. quinquefasciatus showed LC50 values of 29.14, 11.80, 11.14, and 12.39 mg/L, respectively (Kirthi et al. 2011). Jayaseelan et al. (2011b) reported the aqueous extract and synthesized Ag NPs of Musa paradisiaca in Haemaphysalis bispinosa with LC50 values of 28.96 and 1.87 mg/L and in H. maculata 31.02 and 2.02 mg/L, respectively. A cardiac glycosidal (cardenolide) extract isolated from Calotropis procera was tested for their effects against the larvae and adult stages of Hyalomma dromedarii (Al-Rajhy et al. 2003). Melaleuca alternifolia oil and neem seed oil exhibited acaricidal effects against nymphal Ixodes ricinus and adults of Hyalomma anatolicum excavatum ticks, respectively (Abdel-Shafy and Zayed 2002; Iori et al. 2005).Organophosphates are still very effective against B. ovis, but control is hampered by inadequate application via plunge or shower dipping (Levot 1995). Heath et al. (1995) reported that the approach would be through the effect exhibited by azadirachtin, the most active ingredient from Azadirachta indica, which decreases the number of the sheep-biting louse B. ovis by 85% to 100% on treated sheep.
The physicochemical parameters including morphology, crystallinity, agglomeration state, and surface active groups play significant role in toxicity of the metal oxide NPs (Handy et al. 2008). The XRD peaks at 2θ = 25.25° (101) and 48.0° confirm the characteristic facets for anatase form of TiO2 (Liu et al. 2005). In a related pervious study, it has been observed that the crystal structure of nano-TiO2 contributed to cytotoxicity, with anatase TiO2 showing more toxicity than rutile TiO2 (Sayes et al. 2006). A couple of distinct Bragg reflections corresponding to (110), (101), and (211) sets of lattice planes were manifested in the X-ray diffraction patterns.
FTIR spectra of TiO2 NPs exhibited prominent peaks at 3,426, 1,642, 1,172, 1,076, and 714 cm−1. The 3,427 cm−1 absorbance bands are known to be associated with the stretching O–H due alcoholic group. In particular, the 1,642 cm−1 indicates the presence of H bend bond for 1° for proteins. The band observed at 1,172 cm−1 is due to C–O stretching vibrations in alcoholic groups in carbohydrates. The bands in alcoholic groups are due to C–O stretching vibrations in carbohydrate observed at 1,076 cm−1 got oxidized to unsaturated and 714 cm−1 peak at due to Ti–O–O bond (Sadiq et al. 2011; http://orgchem.colorado.edu/hndbksupport/specttutor/irchart.html).
SEM analysis showed the particle size between 25 and 110 nm as well as the cubic structure of the nanoparticles (Khandelwal et al. 2010). The size of the TiO2 nanoparticles was measured by AFM in contact mode with silicon cantilevers with force constant of 0.02–0.77 N m−1 and tip height of 10–15 mm. For imaging by AFM, the sample was suspended in acetone and spins coated on a silicon wafer. The acetone vaporized leaving the particles behind (Mahapatraa et al. 2008).
Mouchet et al. (2008) reported that a high mortality rate (85%) was noted at the highest double-walled carbon nanotube concentration (500 mg L−1) against the larvae of Xenopus laevis. Baun et al. (2008) indicated the toxicity of C60, carbon nanotubes, and zinc oxide to an aquatic invertebrate, Daphnia magna. Although an attempt to develop essential oil for pesticides and insecticides has been made in a variety of water-soluble formulations such as nanoemulsion incorporated with β-cypermethrin (Wang et al. 2007) and essential oil-loaded microcapsules for pest control (Moretti et al. 2002). Recent studies demonstrated that TiO2 NPs were synthesized by two microalgae species Scenedesmus sp. and Chlorella sp. (Sadiq et al. 2011). The photocatalytic inactivation of Escherichia coli and Pichia pastoris was studied with combustion synthesized TiO2 (Sontakke et al. 2010). Prasad et al. (2009) have reported photocatalytic inactivation of Bacillus anthracis, which was studied by using titania nanomaterials.
The present green synthesis shows that the environmentally benign and renewable source of C. roseus is used as an effective reducing agent for the synthesis of TiO2 NPs. This biological reduction of metal would be boon for the development of clean, nontoxic, and environmentally acceptable green approach to produce metal nanoparticles, involving organisms even ranging to higher plants. The formed TiO2 NPs are highly stable and have significant parasitic activity.
References
Abdel-Shafy S, Zayed AA (2002) In vitro acaricidal effect of plant extract of neem seed oil (Azadirachta indica) on egg, immature, and adult stages of Hyalomma anatolicum excavatum (Ixodoidea: Ixodidae). Vet Parasitol 106:89–96
Al-Rajhy DH, Alahmed AM, Hussein HI, Kheir SM (2003) Acaricidal effects of cardiac glycosides, azadirachtin and neem oil against the camel tick, Hyalomma dromedarii (Acari: Ixodidae). Pest Manag Sci 59(11):1250–1254
Anjali CH, SudheerKhan S, Goshen KM, Magdassi S, Mukherjee A, Chandrasekaran N (2010) Formulation of water-dispersible nanopermethrin for larvicidal applications. Ecotoxicol Environ Saf 73:1932–1936
Baun A, Hartmann NB, Grieger K, Kusk KO (2008) Ecotoxicity of engineered nanoparticles to aquatic invertebrates: a brief review and recommendations for future toxicity testing. Ecotoxicology 17:387–396
Chandler D, Davidson G, Pell JK, Ball BV, Shaw K, Sunderland KD (2000) Fungal biocontrol of acari. Biocontrol Sci Tech 10:357–384
El-Sayed A, Cordell GA (1981) Catharanthamine, a new antitumor bisindole alkaloid from C. roseus. J Nat Prod 44:289–293
FAO (2004) Resistance management and integrated parasite control in ruminants—guidelines, module 1—Ticks: acaricide resistance: diagnosis, management and prevention. Food and Agriculture Organization, Animal Production and Health Division, Rome, p 53
Fischhof PK, Moslinger-Gehmayr R, Herrmann WM, Friedmann A, Russmann DL (1996) Theraupetic efficacy of vincamine in dementia. Neuropsychobiology 34:29–35
Foil LD, Coleman P, Eisler M, Fragoso-Sanchez H, Garcia-Vazquez Z, Guerrero FD, Jonsson N, Langstaff IG, Li AY, Machila N, Miller RJ, Morton J, Pruett JH, Torr S (2004) Factors that influence the prevalence of acaricide resistance and tick-borne diseases. Vet Parasitol 125:163–181
Goodsell DS (2004) Bionanotechnology: lessons from nature. Wiley, Hoboken
Handy RD, Kammer FV, Lead JR, Hassello M, Owen R, Crane M (2008) The toxicology and chemistry of the manufactured NPs. Ecotoxicology 17:287–314
Hanna REB, Williamson DS, Mattison RG, Nizami WA (1988) Seasonal reproduction in Paramphistomum epiclitum and Gastrothylax crumenifer, rumen paramphistomes of the Indian water buffalo, and comparison with the biliary paramphistome Gigantocotyle explanatum. Int J Parasitol 18:513–521
Heath ACG, Lampkin N, Jowett JH (1995) Evaluation of non-conventional treatments for control of the biting louse (Bovicola ovis) on sheep. Med Vet Entomol 4:407–412
Horcas I, Fernandez R, Gomez-Rodriguez JM, Colchero J, Gomez-Herrero J, Baro AM (2007) WSXM: software for scanning probe microscopy and a tool for nanotechnology. Re Sci Instrum 78:013705, http://orgchem.colorado.edu/hndbksupport/specttutor/irchart.html
Iori A, Grazioli D, Gentile E, Marano G, Salvatore G (2005) Acaricidal properties of the essential oil of Melaleuca alternifolia Cheel (tea tree oil) against nymphs of Ixodes ricinus. Vet Parasitol 129:173–176
Jayaseelan C, Rahuman AA, Rajakumar G, Vishnu Kirthi A, Santhoshkumar T, Marimuthu S, Bagavan A, Kamaraj C, Zahir AA, Elango G (2011a) Synthesis of pediculocidal and larvicidal silver nanoparticles by leaf extract from heartleaf moonseed plant. Tinospora cordifolia Miers. Parasitol Res. doi:10.1007/s00436-010-2242-y
Jayaseelan C, Rahuman AA, Rajakumar G, Santhoshkumar T, Kirthi AV, Marimuthu S, Bagavan A, Kamaraj C, Zahir AA, Elango G, Velayutham K, Rao KV, Karthik L, Raveendran S (2011b) Efficacy of plant-mediated synthesized silver nanoparticles against hematophagous parasites. Parasitol Res 11:2473–2476
Jha AK, Prasad K, Kulkarni AR (2009) Synthesis of TiO2 nanoparticles using microorganisms. Colloids Surf B Biointerfaces 71(2):226–229
Khandelwal N, Abhijeet S, Devendra J, Upadhyay MK, Verma HN (2010) Green synthesis of silver nanoparticles using Argimone mexicana leaf extract and evaluation of their antimicrobial activities. Dig J Nanomat Biostruct 5:483–489
Kirthi AV, Rahuman AA, Rajakumar G, Marimuthu S, Santhoshkumar T, Jayaseelan C, Velayutham K (2011) Acaricidal, pediculocidal and larvicidal activity of synthesized ZnO nanoparticles using wet chemical route against blood feeding parasites. Parasitol Res 109:461–72
Kvitek L, Panacek A, Soukupova J, Kolar M, Vecerova R (2008) Effect of surfactant and polymers on stability and antibacterial activity of silver nanoparticles(NPs). J Phys Chem 112:5825–5834
Levot GW (1995) Resistance and the control of sheep ectoparasitic. Int J Parasitol 25(11):1355–1362
Liu Z, Hong L, Guo B (2005) Physicochemical and electrochemical characterization of anatase titanium dioxide NPs. J Power Sour 143:231–235
Mahapatraa O, Bhagatb M, Gopalakrishnana C, Arunachalamb KD (2008) Ultrafine dispersed CuO nanoparticles and their antibacterial activity. J Exp Nanosci 3(3):185–193
Marimuthu S, Rahuman AA, Rajakumar G, Santhoshkumar T, Kirthi AV, Jayaseelan C, Bagavan A, Zahir AA, Elango G, Kamaraj C (2011) Evaluation of green synthesized silver nanoparticles against parasites. Parasitol Res 10:2212–2224
Moretti MDL, Sanna-Passino G, Demontis S, Bazzoni E (2002) Essential oil formulations useful as a new tool for insect pest control. AAPS Pharm Sci Tech 13:1–11
Morones JR, Elechiguerra JL, Camacho A, Holt K, Kouri JB, Ramfrez JT, Yacaman MJ (2005) The bactericidal effect of silver nanoparticles. Nanotechnology 16:2346–2353
Mouchet F, Landois P, Sarremejean E, Bernard G, Puech P, Pinelli E, Flahaut E, Gauthier L (2008) Characterisation and in vivo ecotoxicity evaluation of double-wall carbon nanotubes in larvae of the amphibian Xenopus laevis. Aquat Toxicol 87(2):127–137
Mukherjee P, Ahmad A, Mandal D, Senapati S, Sainkar SR, Khan MI, Ramani R, Parischa R, Ajayakumar PV, Alam M, Sastry M, Kumar R (2001) Bioreduction of AuCl(4)(−) ions by the fungus. Verticillium sp. and surface trapping of the gold nanoparticles formed. Angew Chem Int Ed Engl 40(19):3585–3588
Nayak S (2006) Influence of ethanol extract of Vinca rosea on wound healing in diabetic rats online. J Biol Sci 6(2):51–55
Novello FC, Sprague JM (1957) Alkaloids of Vinca rosea Linn. J Am Chem Sot 79:20–28
Nyamador WS, Ketoh GK, Amevoin K, Nuto Y, Koumaglo HK, Glitho IA (2010) Variation in the susceptibility of two Callosobruchus species to essential oils. J Stored Prod Res 46:48–51
Parashar BD, Gupta GP, Rao KM (1991) Control of the haematophagous fly H. maculata a serious pest of equines by deltamethrin. Med Vet Entomol 5:363–367
Prasad GK, Agarwal GS, Singh B, Rai GP, Vijayaraghavan R (2009) Photocatalytic inactivation of Bacillus anthracis by titania nanomaterials. J Hazard Mater 165:506–510
Rajakumar G, Rahuman AA (2011) Acaricidal activity of aqueous extract and synthesized silver nanoparticles from Manilkara zapota against R (B) microplus. Res Vet Sci. doi:10.1016
Ramyadevi J, Jeyasubramanian K, Marikani A, Rajakumar G, Rahuman AA, Santhoshkumar T, Kirthi AV, Jayaseelan C, Marimuthu S (2011) Copper nanoparticles synthesized by polyols process used to control hematophagous parasites. Parasitol Res. doi:10.1007/s00436-011-2387-3
Reddy PJ, Krishna D, Murthy US, Jamil K (1992) A microcomputer FORTRAN program for rapid determination of lethal concentration of biocides in mosquito control. CABIOS 8:209–213
Sadiq IM, Dalai S, Chandrasekaran N, Mukherjee A (2011) Ecotoxicity study of titania (TiO2) NPs on two micro algae species: Scenedesmus sp. and Chlorella sp. Ecotoxicol Environ Saf 10:10–16
Sakulku U, Nuchuchua O, Uawongyart N, Puttipipatkhachorn S, Soottitantawat A, Ruktanonchai U (2009) Characterization and mosquito repellent activity of citronella oil nanoemulsion. Int J Pharm 372:105–111
Santhoshkumar T, Rahuman AA, Rajakumar G, Marimuthu S, Bagavan A, Jayaseelan C, Zahir AA, Elango G, Kamaraj C (2011) Synthesis of silver nanoparticles using Nelumbo nucifera leaf extract and its larvicidal activity against malaria and filariasis vectors. Parasitol Res 10:2115–2124
Sayes CM, Wahi R, Kurian PA, Lie Y, West L, Ausman KD, Warheit DB, Colvin VL (2006) Correlating nanoscale titania structure with toxicity a cytotoxicity and inflammatory response study with human dermal fibroblasts and human lung epithelial cells. Toxicol Sci 92:174–185
Shankar SS, Rai A, Ahmad A, Sastry M (2004) Rapid synthesis of Au, Ag, and bimetallic Au core-Ag shell nanoparticles using Neem (Azadirachta indica) leaf broth. J Colloid Interface Sci 275:496–502
Sing NC, Johnston LAY, Leatch G (1983) The economics of cattle tick control in the dry tropical Australia. Aust Vet J 60:37–39
Sontakke S, Modak J, Madras G (2010) Photocatalytic inactivation of E. coli and Pichia pastoris with combustion synthesized titanium dioxide. Chem Eng J. doi:10.1016
Soulsby EJL (1982) Helminths, arthropods and protozoa of domesticated animals. Bailliere Tindall, London
Sundrarajan M, Gowri S (2011) Green synthesis of titanium dioxide nanoparticles by Nyctanthes arbor-tristis leaves extract. Chalcogenide Letters 447–451
Templeton AC, Wuelfing WP, Murray RW (2000) Monolayer protected cluster molecules. Acc Chem Res 33:27
Wang L, Li X, Zhang G, Dong J, Eastoe J (2007) Oil-in-water nanoemulsion for pesticide formulations. J Colloid Interface Sci 314:230–235
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Velayutham, K., Rahuman, A.A., Rajakumar, G. et al. Evaluation of Catharanthus roseus leaf extract-mediated biosynthesis of titanium dioxide nanoparticles against Hippobosca maculata and Bovicola ovis . Parasitol Res 111, 2329–2337 (2012). https://doi.org/10.1007/s00436-011-2676-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-011-2676-x