Abstract
Introduction
Non-adherence to anti-hormonal therapy is a major problem in gynecologic oncology. Reasons reported are side effects and lack of support. The aim of our study was an analysis of influence of experiences of patients with endocrine therapy and communication and information on this topic and their influence on adherence.
Methods
We developed a structured questionnaire which was tested in a pilot version and then programmed as online questionnaire and presented to patient members of self-help and breast cancer organizations.
Results
Patients only had received scarce information on endocrine therapy. Only 12.8 % stated that their questions were answered in detail, 43.2 % got no answers or only non-detailed answers. 76 % had side effects limiting functions of daily life. 60 % of physicians did not react on these side effects. There is a significant correlation between number and intensity of side effects and non-adherence or disruption of therapy (p = 0.029 and p < 0.01, respectively). Women who reported having received detailed answers to their questions also reported better adherence (p = 0.014).
Conclusion
In order to improve adherence, detailed information on side effects and answers in case of symptoms are most important. Physicians should not rely on presenting written information but should mainly engage in direct communication.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Anti-hormonal therapy is one of the decisive elements for adjuvant as well as palliative therapy for patients with breast cancer. In comparison with chemotherapy, it is considered by most physicians to have less side effects. Side effects of anti-hormonal therapy have a strong influence on quality of life, and physical as well as mental and social functioning. Discontinuation and non-adherence in the adjuvant setting may result in more relapses and reduced disease-free or overall survival. In the palliative setting, progress of disease and change to chemotherapy have to be considered. In the literature, a rate of non-adherence and discontinuation of 30–60 %of women in adjuvant therapy is reported (Partridge et al. 2003; Thurlimann et al. 2005; Howell et al. 2005; Lash et al. 2006; van Herk-Sukel et al. 2010; Huiart et al. 2011; Güth et al. 2012). Most often quoted reasons are side effects and reduced quality of life (Bell et al. 2013).
In 2012, a French working group reported their data on young women’s reasons of discontinuation and non-adherence. During the first period, a lack of understandable information and insufficient social support were the main reasons. Only later on, side effects of treatment and less fear of relapse turned to be the important reasons. Lack of social support and failures in communication were persisting reasons (Cluze et al. 2012 ). The decisive role of communication has also been shown by (Liu et al. 2013).
In Germany, most women with breast cancer are treated in certified breast centers that have to prove a high standard of treatment and psychosocial support. Thus, communication with patients should be on a high level, and women should receive all the information they need for shared decision making.
In the present study, we aim at collecting first data on the relationship between communication and non-adherence in Germany. In order to find out whether women in Germany face the same difficulties as those in France, we decided to collaborate with different German self-help groups and do a survey on their members.
Results of this survey are of high importance for physicians and may be of relevance for societies engaged in the national processes which aim at improving the quality of cancer care.
Methods
In a first step, a questionnaire was developed for the survey. The focus was entirely on the perspective of the patient. From the data from the literature, a first draft of the questionnaire was developed from a scientific point of view. This draft was passed to three leaders of patient advocacy for a first review. The revised questionnaire was tested in a group of patients attending a seminar on breast cancer in 2012. Twenty-three women participated in this anonymous pretest. This pretest was analyzed according to whether participants were able to answer the questions. Furthermore, participants were asked to give feedback concerning questions they thought difficult to answer.
Apart from a few changes in terminology, the questionnaire was well suited and the final version was consented to by the scientists and patients advocates. It is made up of the following sections (see also supplemental material):
-
1.
Demographic data (six questions).
-
2.
Data concerning breast cancer, former and current therapy (13 questions).
-
3.
Information concerning anti-hormonal therapy and decision making on anti-hormonal therapy (12 questions) given by the physician in charge looked for and/or found by other sources.
-
4.
Anti-hormonal therapy (type, side effects, and consequences drawn) (12 questions).
-
5.
General support (one question).
We used closed questions, providing lists of possible answers (f.ex. Which types of treatment did you get so far? (several answers possible): operation, chemotherapy, radiotherapy, antibody, endocrine therapy; including the possibility to choose “no answer”). In cases where a rating of the participants was necessary, we prespecified answers using four-point Likert scales (f.ex. If an anti-hormonal therapy been proposed to you have the side effects been discussed with you? It was not discussed—It has been briefly discussed—It has been discussed—It has been thoroughly explained. Or: How has support been by…your family: very well–well-sufficient–low–none).
Besides the pretest with patients advocates, the questionnaire has not been tested for reliability or validity.
Inclusion criteria were patients with endocrine-responsive breast cancer (according to their knowledge). Exclusion criteria were male patients.
This questionnaire was prepared as a programmed online questionnaire, which was distributed by the use of a hyperlink to the members of the following four self-help groups and associations for breast cancer in Germany: Frauenselbsthilfe nach Krebs (Women’s Self-help after Cancer) BRCA-Netzwerk (BRCA-Network), KOMBRA (annual training program for patients with breast cancer and professionals on communication on breast cancer), and Brustkrebs Deutschland (Breast Cancer Germany). The access to the questionnaire was open from January to March 2013.
According to the regulations of the Ethics committee of the university hospital Frankfurt/Main for anonymous surveys, no ethics evaluation was necessary.
Results
Demographic and treatment data
A total of 523 patients took part in the survey, opening the questionnaire and answering at least one question. 281 participants answered all questions. These were included in the statistical analysis. More than half of the participants were in the age group 40–59, the median age being 51 years. The youngest participant was 20 and the eldest 76. Three quarters were married and 70 % had children. Nearly half had a certificate of secondary education and about half a university entrance diploma or a university degree. The demographic and oncological data are presented in Table 1.
Information concerning anti-hormonal therapy and decision making on anti-hormonal therapy
Anti-hormonal therapy was recommended to 92.9 % of participants. Of all participants, 89 % ever had taken or actually took an endocrine therapy.
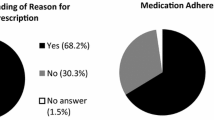
Table 2 summarizes how detailed patients thought the information was that they received from their physicians regarding this therapy. While 63.0 % of participants rated the information they got as comprehensible, 28.1 % did not think so. Only 12.8 % stated that their questions were answered in a detailed manner, 36.3 % in a satisfactory manner, while 43.2 % got no answers or only non-detailed answers. Additional written information was given to 24.9 %.
The decision for or against anti-hormonal therapy was made alone by 8.9 % of participants. In 45.6 % of cases, both physician and patient decided together, and in 37.4 %, it was the physician alone who made the decision.
Anti-hormonal therapy (type, side effects, and consequences drawn)
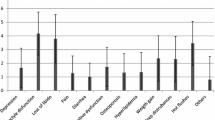
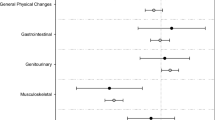
Side effects were experienced by 85.8 % of participants. From those, 25.6 % had 1–4 side effects, 49.5 % had 5–8 side effects, and 10.7 % had 9–13 side effects. The types of side effects are shown in Table 3.
Moderate side effects from which patients did not feel any limitations in daily life were experienced by 10.3; 40.9 % had stronger side effects experiencing some limitations in daily life, and 35.2 % had strong side effects experiencing heavy limitations in daily life.
In 59.8 % of patients, no consequences resulted from the discussion of the side effects with the physician. In 4.3 %, dosage or frequency of drug was changed. In 6.8 %, any supportive therapy was prescribed and in 14.6 %, the type of anti-hormonal therapy was changed. A total of 14.6 % of participants interrupted or stopped taking anti-hormonal therapy.
General support
Support from family and friends is presented in Table 4. Nearly 80 % rated the quality of support by the family as very well or well, only 7.5 % as low. Also support by husband was mostly rated high (60 % very well and well and only 5 % low).
Adherence
Whereas age, partnership, and education do not correlate with non-adherence, having someone with breast cancer in the family is associated with significantly better adherence. In contrast, carriers of BRCA 1 or 2 genes do not show better adherence. Information regarding effects has no impact, whereas patients who got information on side effects have a significantly better adherence. The number of side effects and even more so the strength of side effects are associated with non-adherence and disruption of therapy (p = 0.029 and p < 0.01, respectively). Besides the strength of side effects, newly occurring depression is a feature of highest significance (p < 0.001). Patients who got information on possibilities of supportive therapy in the case of side effects tend to have a better adherence than those who did not. While comprehensiveness of information in general has no influence, getting detailed answers to questions leads to significantly better adherence (p = 0.014). Support by friends also is of high importance (p = 0.036), while support by family members and husbands did not show significant correlations.
Discussion
In our survey, 523 patients took part and 281 questionnaires were suitable for the statistical analysis. We are not able to determine how many potential participants we reached with our survey. The FSH has 1,131 members but it is unknown, how many have access to the Internet. The BRCA network has 190 members. KOMBRA and Breast Cancer Germany not only address patients but also professionals. This uncertainty of the percentage of participants in relation to patients addressed is one of the main limitations of our study. The second main limitation also stems from the setting of our study as online survey which was distributed by cooperating organizations. From this selection bias, younger and better informed women most probably are overrepresented in our survey. This argument is supported by the distribution of educational degrees, which is shifted to higher degrees in comparison with the general population in Germany. As younger women with higher education are more likely to ask questions and insist on answers, one may conclude that they should have better opportunities to get information from their physicians. On the other hand, it may be that they are more demanding, which makes them rate the answers as being insufficient more often. Nevertheless, our findings are in line with the French data that were also derived from younger women [17]. The third limitation is the missing psychometric evaluation concerning reliability and validity. As 40.6 % of participants did not use anti-hormonal therapy at the time of participation in the study, they reported on side effects by hindsight. This might have introduced recall bias.
Yet, despite these limitations, the key message of our results is that in Germany, women with breast cancer report having received only scarce information on anti-hormonal treatment. In particular, they report side effects being seldom discussed and possibilities of supportive therapy in the case of side effects being even less often discussed. Additionally, they experience their questions being rarely answered in detail.
Anti-hormonal therapy has been interrupted or stopped by 14.6 % of the participants. In comparison with the literature, this rate of non-adherence is low. On the other hand, this rate is in line with others from anonymous population-based surveys. One reason for the low level of non-adherence may be that we only gathered self-reported data.
In the literature, an abundance of partly contradicting data exists concerning the risk factors of non-adherence. In their review of the literature, Murphy and colleagues found a rate of 31–73 % of non-adherence and discontinuation measured at the end of 5 years of treatment. Extremes of age (older or younger), increasing out-of-pocket costs, follow-up care with a general practitioner (vs. Oncologist), switching from one form of therapy to another, and treatment side effects are negatively associated with adherence and/or persistence. Taking more medication at baseline, referral to an oncologist, and earlier age at diagnosis were positively associated with adherence and/or persistence (Murphy et al. 2012). While some authors discuss older age as a risk factor (Wigertz et al. 2012; Nekhlyudov et al. 2011), Huiart et al. (2011) did not find higher rates of adherence in young women in their survey. Hershman et al. (2010) even reported women younger than 40 years to have the highest risk of discontinuation. According to these conflicting data, our data do not show any correlation of non-adherence with age at all.
In the literature, adherence is positively associated with being married, large tumor size, receipt of chemotherapy or radiotherapy, and negatively associated with smoking. No significant associations with obesity or physical activity have been reported. Data on education or income are contradictory (Wigertz et al. 2012; Hershman et al. 2010; Land et al. 2011; Güth et al. 2008). Our data do not show any correlation with education and tumor size. On the other side, number and strength of side effects are decisive for non-adherence and disruption of therapy. This is in line with the literature (van de Water et al. 2012).
Early discontinuation and non-adherence to anti-hormonal therapy are associated with increased mortality (survival at 10 years, 73.6 versus 80.7 % for women who discontinue and 77.8 versus 81.7 % in case of non-adherence or adherence) [23]. Regarding these numbers, increasing adherence will be able to significantly improve the survival of women with breast cancer. Different approaches have been suggested, one of which will be to actively address and treat side effects. Chlebowski and Geller (2006) made up a list of suggestions which focus on the accessibility and convenience of treatment, improving the knowledge of patients and increasing shared decision making.
In the recently published PACT study, 4,844 patients in Germany getting an aromatase inhibitor were randomized to standard care or standard care and additional educational material. This study did not show a difference between both arms regarding compliance and persistence (Hadjy et al. 2013). In our survey, written material had no influence and shared decision making only a weak one on adherence. Even if information on supportive therapy in case of side effects is given, this does not improve adherence. In contrast, the decisive step is giving detailed answers to all questions. Thus, in order to improve adherence, physicians and breast cancer centers should not rely on presenting written information but should mainly engage in giving answers. This fact is also of high importance for the development of health systems as there might be a strong impact on survival data from investments in physician–patient communication.
References
Bell RJ, Fradkin P, Schwarz M, Davis SR (2013) Understanding discontinuation of oral adjuvant endocrine therapy by women with hormone receptor-positive invasive breast cancer nearly 4 years from diagnosis. Menopause 20(1):15–21
Chlebowski RT, Geller ML (2006) Adherence to endocrine therapy for breast cancer. Oncology 71(1–2):1–9
Cluze C, Rey D, Huiart L, BenDiane MK, Bouhnik AD, Berenger C, Carrieri MP, Giorgi R (2012) Adjuvant endocrine therapy with tamoxifen in young women with breast cancer: determinants of interruptions vary over time. Ann Oncol 23:882–890
Güth U, Huang DJ, Schötzau A, Zanetti-Dällenbach R, Holzgreve W, Bitzer J, Wight E (2008) Target and reality of adjuvant endocrine therapy in postmenopausal patients with invasive breast cancer. Br J Cancer 99(3):428–433
Güth U, Myrick ME, Kilic N, Eppenberger-Castori S, Schmid SM (2012) Compliance and persistence of endocrine adjuvant breast cancer therapy. Breast Cancer Res Treat 131(2):491–499
Hadjy P, Blettner M, Harbeck N, Jackisch C, Lück HJ, Windemuth-Kieselbach C, Zaun S, Kreienberg R (2013) The patient’s anastrozole compliance to therapy (PACT) program: a randomized, in-practice study in the impact of a standardized information program on persistence and compliance to adjuvant endocrine therapy in postmenopausal women with early breast cancer. Ann Oncol 24:505–512
Hershman DL, Kushi LH, Shao T, Buono D, Kershenbaum A, Tsai WY, Fehrenbacher L, Gomez SL, Miles S, Neugut AI (2010) Early discontinuation and nonadherence to adjuvant hormonal therapy in a cohort of 8,769 early-stage breast cancer patients. J Clin Oncol 28(27):4120–4128
Howell A, Cuzick J, Baum M, Buzdar A, Dowsett M, Forbes JF, Hoctin-Boes G, Houghton J, Locker GY, Tobias JS (2005) ATAC Trialists’ group: results of the ATAC (Arimidex, Tamoxifen, Alone or in Combination) trial after completion of 5 years’ adjuvanttreatment for breast cancer. Lancet 365:60–62
Huiart L, Dell’Aniello S, Suissa S (2011) Use of tamoxifen and aromatase inhibitors in a large population-based cohort of women with breast cancer. Br J Cancer 104(10):1558–1563
Land SR, Cronin WM, Wickerham DL, Costantino JP, Christian NJ, Klein WM, Ganz PA (2011) Cigarette smoking, obesity, physical activity, and alcohol use as predictors of chemoprevention adherence in the National Surgical Adjuvant Breast and Bowel Project P-1 Breast Cancer Prevention Trial. Cancer Prev Res (Phila) 4(9):1393–1400
Lash TL, Fox MP, Westrup JL, Fink AK, Silliman RA (2006) Adherence to tamoxifen over the five-year course. Breast Cancer Res Treat 99:215–220
Liu Y, Malin JL, Diamant AL, Thind A, Maly RC (2013) Adherence to adjuvant hormone therapy in low-income women with breast cancer: the role of provider-patient communication. Breast Cancer Res Treat 137:829–836
Murphy CC, Bartholomew LK, Carpentier MY, Bluethmann SM, Vernon SW (2012) Adherence to adjuvant hormonal therapy among breast cancer survivors in clinical practice: a systematic review. Breast Cancer Res Treat 134(2):459–478
Nekhlyudov L, Li L, Ross-Degnan D, Wagner AK (2011) Five-year patterns of adjuvant hormonal therapy use, persistence, and adherence among insured women with early-stage breast cancer. Breast Cancer Res Treat 130(2):681–689
Partridge AH, Wang PS, Winer EP, Avorn J (2003) Nonadherence to adjuvant tamoxifen therapy in women with primary breast cancer. J Clin Oncol 21:602–606
Thurlimann B, Coates AS, Mouridsen H, Mauriac L, Forbes JF, Paridaens R, Castiglione-Gertsch M, Gelber RD, Rabaglio M, Smith I, Wardley A, Price KN, Goldhirsch A (2005) Breast international group (BIG) 1–98 collaborative group: a comparison of letrozole and tamoxifen in postmenopausal women with early breast cancer. N Engl J Med 353:2747–2757
van de Water W, Bastiaannet E, Hille ET, Meershoek-Klein Kranenbarg EM, Putter H, Seynaeve CM, Paridaens R, de Craen AJ, Westendorp RG, Liefers GJ, van de Velde CJ (2012) Age-specific nonpersistence of endocrine therapy in postmenopausal patients diagnosed with hormone receptor-positive breast cancer: a TEAM study analysis. Oncologist 17(1):55–63
van Herk-Sukel MP, van de Poll-Franse LV, Voogd AC, Nieuwenhuijzen GA, Coebergh JW, Herings RM (2010) Half of breast cancer patients discontinue tamoxifen and any endocrine treatment before the end of the recommended treatment period of 5 years: a population-based analysis. Breast Cancer Res Treat 122(3):843–851
Wigertz A, Ahlgren J, Holmqvist M, Fornander T, Adolfsson J, Lindman H, Bergkvist L, Lambe M (2012) Adherence and discontinuation of adjuvant hormonal therapy in breast cancer patients: a population-based study. Breast Cancer Res Treat 133(1):367–373
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wuensch, P., Hahne, A., Haidinger, R. et al. Discontinuation and non-adherence to endocrine therapy in breast cancer patients: is lack of communication the decisive factor?. J Cancer Res Clin Oncol 141, 55–60 (2015). https://doi.org/10.1007/s00432-014-1779-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-014-1779-z