Abstract
Background
Endocrine therapy is a mainstay of prostate cancer therapy. Given that few data exist on patient physician communication with regard to this field of therapy and adherence, we conducted a survey of patient members of a German support organization.
Patients and methods
We developed a structured questionnaire that was tested in a pilot version and then programmed as an online questionnaire.
Results
The questionnaire was completed by 694 patients. While 58 % of participants rated the information they received as comprehensive, 42 % did not. Fifty-one percentage stated that they were informed of side effects in detail, and 35 % received information on supportive treatments available in the event of side effects. Patients with higher education more often reported receiving information on side effects (p = 0.036) as well as alternatives for treatment (p = 0.001). Only 13 % stated that their questions were answered in detail, with 43 % receiving no answers or only non-detailed answers. Additional information was sought by 82 %, mostly from the Internet (67 %) and patient support groups (66 %). Seventy-six percentage experienced side effects that imposed limitations on their daily activities. Of those patients with side effects, 60 % reported that their physicians did not react to their complaints. There is a significant association between side effects in general and depression in particular and non-adherence (p < 0.01 and p = 0.002, respectively). In contrast, better information on side effects is associated with better adherence (p < 0001).
Conclusion
In order to improve adherence, detailed information on side effects and comprehensive supportive care is most important. Physicians should not rely on written information but should rather mainly engage in direct communication.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Endocrine therapy is one of the mainstays of prostate cancer therapy. It is considered by most physicians to have fewer side effects than chemotherapy. Yet these side effects may nonetheless have a significant influence on quality of life, and physical as well as mental and social functioning. With more and more men diagnosed with prostate cancer at an early stage, long-term endocrine treatments and the number of patients receiving these therapies will increase. In spite of recent advances with chemotherapy, endocrine therapy offers better risk–benefit ratios for hormonally sensitive tumors. Moreover, recent new developments for endocrine therapy offer new treatment options.
Side effects from endocrine therapy include symptoms that affect quality of life (sexual dysfunction, hot flashes, gynecomastia and sarcopenia), but they may also cause such secondary diseases as osteoporosis, insulin resistance, hyperglycemia, metabolic syndrome and cardiovascular diseases (Harle et al. 2006; Cluze et al. 2012). These side effects are tolerated in cases of prolongation of overall survival in the adjuvant setting (Kumar et al. 2006; Widmarl et al. 2009; Nair et al. 2002; Payne and Mason 2011; Bourke et al. 2013) or in the control of disease.
Different strategies exist to minimize side effects and optimize benefit: choice of anti-hormonal drugs, late instead of early start and intermittent instead of continuous treatment in palliative care (Mottet et al. 2012; German Interdisciplinary Guideline Prostate Cancer).
In a previous survey of young breast cancer patients, a lack of understandable information and insufficient social support were the main reasons for discontinuation and non-adherence to endocrine therapy during the first period of treatment. Only later on did the side effects of treatment and to a lesser degree fear of relapse turn out to be the most important reasons. Lack of social support and failures in communication were persisting reasons (Studer et al. 2011). The decisive role of communication has also been shown by Liu and colleagues (Liu et al. 2013).
To our knowledge, no publications presenting data on communication between patient and physician on the subject of side effects exist.
In the present study, we aimed at collecting first data on information provided to patients concerning endocrine therapy and its side effects, as well as the relationship between communication and non-adherence. We conducted our survey in collaboration with the German prostate cancer support organization Bundesverband Prostatakrebs Selbsthilfe (BPS).
Methods
In a first step, a questionnaire was developed for the survey. A first draft of the questionnaire was developed from our questionnaire on breast cancer. This draft was passed to the leaders of the patient support organization for a first review. The revised questionnaire was tested with a group of patients from one regional group in spring 2013. Twenty men participated in this anonymous pretest. The pretest was analyzed according to whether participants were able to answer the questions. Furthermore, the leader of this group gave feedback concerning questions he thought were ambiguous or difficult to answer. The questionnaire has not been tested for reliability or validity.
The final version of the questionnaire was approved by the scientists and leaders of the patient support organization. It is made up of the following seven sections (see also supplemental material):
-
1.
Current or former endocrine therapy or proposal of endocrine therapy.
-
2.
Demographic data (5 questions).
-
3.
Data concerning prostate cancer, former and current therapy (9 questions).
-
4.
Endocrine therapy (type, side effects) (5 questions).
-
5.
Information concerning endocrine therapy the patient was given or sought on his own (6 questions).
-
6.
Decision making on endocrine therapy, possibility of discussing the therapy with someone and support by others (3 questions).
-
7.
Thoughts of cessation of therapy (1 question).
-
8.
Use of complementary and alternative therapy.
We used closed questions, providing lists of possible answers (e.g., Which therapies have you received so far? (several answers possible): radiotherapy, operation, chemotherapy, anti-hormonal therapy, naturopathy).
In cases in which a rating was necessary, we provided answers using 4-point Likert scale (e.g., Do you feel supported by your family and friends? Not at all—a little bit—good—very good).
Inclusion criteria were patients with prostate cancer who had either received or were then receiving endocrine therapy or to whom endocrine therapy had been proposed by their physician. Exclusion criteria were all patients who did not answer with “yes” to one of these three criteria.
The questionnaire was prepared as a programmed online questionnaire distributed by hyperlink to leaders of the regional organizations of the BPS. These leaders were asked to pass the hyperlink on to members of their groups. As we received several requests for a print version for members lacking Internet access, we also provided a PDF of the questionnaire to the group leaders, which could be sent back to the BPS office. Access to the questionnaire was open from September to November 2013.
Statistical procedures were performed with SPSS, version 19, calculating frequencies, mean values, standard deviations and analyzing correlations using a bivariate analysis.
Results
Demographic and treatment data
In total, 694 patients took part in the survey and all but 14 fulfilled the inclusion criteria and were included in the statistical analysis even if each question was only answered by half of the participants or less. Given that the questions left unanswered differed among participants, we have decided against excluding any participant. All percentages refer to the number of participants who answered questions from that section. The demographic and oncological data are presented in Table 1.
The question on continuous or intermittent androgen blockade was answered by 249 participants, of whom 126 (50.6 %) receive an intermittent blockade and 123 (49.4 %) a continuous blockade.
Information concerning endocrine therapy and decision making on endocrine therapy
Table 2 and 3 summarize data on patients’ estimate of the information they received from their physicians concerning endocrine therapy. Of all participants, 323 (47.5 %) answered questions in this section. While 58 % of participants rated the information they received as comprehensive, 42 % did not rate it as such. About half of participants indicated that all of their questions had been answered by the physician, while slightly fewer reported receiving information on alternative treatment options. Only 51 % reported being informed of side effects in detail, and only 35 % received information on supportive treatments available in the event of side effects. Additional written information was given to 28 %.
Patients with higher education more often reported receiving information on side effects (p = 0.036) as well as alternatives for treatment (p = 0.001).
Additional information was gathered by 262 patients (81.6 %, 321 answered these questions). The most important sources of information were the Internet (216 or 67 %) and patient support groups (212, 66 %), followed by lectures (156, 49 %), a second opinion (145, 45 %) and books (132, 41 %). Other patients outside the patient support groups were identified by 109 (34 %) and general practitioners by 95 (30 %). Only a few reported having asked a non-medical practitioner (27, 8 %). There is no association between age or education and the search for additional information. Yet, patients with higher education more often used the Internet (p = 0.001).
The decision for or against anti-hormonal therapy was made by 17.3 % of patients unilaterally. In 57.0 % of cases, physician and patient decided together, and in 16.7 % of cases, the physician alone made the decision. This is independent of age and education.
Anti-hormonal therapy (type, side effects and consequences drawn)
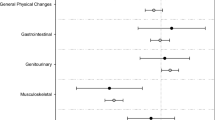
Side effects were experienced by 249 of the 331 participants who answered the question (75 %). The types of side effects and values of patients’ rating are shown in Fig. 1. Other complaints mostly referred to muscle pain or gynecomastia. The question “To what extent does anti-hormonal therapy influence your life?” was answered by 61 (19 %) as not at all, while 119 (37 %) reported few constraints and 143 (44 %) reported stronger to strong constraints, with 38 (12 %) rating them as very strong. Age, marital status and education did not have any influence on the number or severity of side effects.
Of 264 patients who discussed side effects with their physician, 27 (10.4 %) received another type of anti-hormonal therapy, for 45 (17.3 %) intermittent blockade was introduced and, in 10 cases (3.8 %), treatment was stopped. A supportive therapy was introduced for 4 patients (13.1 %).
General support
About 70 % (226 of 322) of the participants stated that they had somebody to talk to about their problems and anxieties. This was the physician for 168 (74 %), the partner or another family member for 143 and 142 patients, respectively (63 % each), other patients for 134 (59 %) and friends for 101 (44.7 %).
Adherence
Of 320 patients who answered the question, 85 (26.5 %) had disrupted the endocrine therapy or planned to do so. Of these, 23 (27 %) based this decision on side effects, 3 (3.5 %) on deficits in care and only 1 on a lack of information. Fifty-one (60 %) had other reasons, mostly because the PSA was stable or no disease had been shown in diagnostic procedures and a break had been consented to, and some because of an increase in PSA rising that was interpreted as castration resistance. Higher education is associated with less non-adherence, while more and more severe side effects are associated with more non-adherence, and better information on side effects is associated with better adherence (all p < 0001). Developing a depression is correlated with non-adherence (p = 0.002).
Discussion
The key message of our results is that in Germany, men with prostate cancer to a high degree report not having received comprehensive information on endocrine therapy, including information on side effects and supportive treatments. Information provision is better in cases of higher education (university), with these patients taking a more active role in decision making.
There are some limitations to our study. First of all, we are not able to determine how many potential participants we reached with our survey. The BPS has 240 support groups, but it is unknown how many were informed by group leaders and had access to the Internet or obtained the PDF of the questionnaire. The second limitation is a probable selection bias incurred by addressing members of a self-help organization. Patients experiencing side effects may be overrepresented. In comparison with German society as a whole, participants with higher education were also overrepresented. The third limitation is the missing evaluation of the questionnaire for reliability and validity. Furthermore, regarding adherence, we did not use any means to verify participants’ answers, which may result in lower non-adherence being reported. Additionally, the quality of information was only evaluated as reported by the patients. Given that no records of the communication were available and we were not able to address their physicians, it may be that more information was provided than was recalled by the participants. Yet this nonetheless does not compromise the essential point that a high proportion of patients with prostate cancer do not feel adequately informed with regard to the endocrine therapy they have been prescribed. The last major limitation is the fact that most of the questions were answered only by about half or even fewer of the participants.
Most participants experienced side effects that had a strong impact on their quality of life. Yet even if discussed with the physician, most side effects were not treated by supportive means or a change of therapy. While this is one of the main reasons for women to stop endocrine therapy (Studer et al. 2011; Gueth et al. 2012; Liu et al. 2013; Bell et al. 2013), there is only a small rate of non-adherence reported by the patients in our survey. This small rate is in accordance with data we derived from a survey of breast cancer patients. In this survey as well, a substantial number of breast cancer patients reported not having received information on side effects and more so on treatment options in the event of side effects. Even when turning to the physicians for help with side effects, the majority of women reported that no action was taken (Wuensch et al. 2015). As developing depression is significantly correlated with non-adherence, this side effect demands more attention from the physicians.
Most patients in our survey reported looking for information from other sources, the Internet and self-help groups being named most often. Accordingly, the BPS in Germany has installed a highly professional counseling system via telephone free for any patient.
The Internet is a source of information of growing importance. Yet for many patients, the abundance of information, the lack of trustworthiness and the question of relevance are obstacles to using the Internet. Accordingly, the BPS provides some information on its website that complies with most criteria for evidence-based patient information (see, e.g., Steckelberg et al. 2005; http://www.discern.org.uk/discern_instrument.php; https://www.healthonnet.org/HONcode/German). As written information the patient version of the national cancer guideline on prostate cancer provides reliable information approved by experts (http://www.krebsgesellschaft.de/download/2009-pl-pca.pdf; http://www.krebsgesellschaft.de/download/patientenleitlinie_prostatakrebs_2_2013.pdf). Yet it comprises more than 150 pages (in two parts without screening), which may be too complicated for men with lower education or health literacy. Lectures, which are already are a main source of additional information, may help those for whom reading and selecting relevant information may be a challenging task. For these lectures, physicians might be trained in order to present scientific data in a manner accessible to lay people.
On the other hand, in spite of all these additional sources of information, the physician is the most important source of information for the patient. Accordingly, there are two main tasks to be solved by the medical community:
-
Evaluation and compilation of data on side effects of endocrine therapy in men and supportive treatments.
-
Training of communication skills for medical students and physicians.
Finally, an adequate reimbursement for time spent in communication would appear essential to improving quality of care.
The German national guideline on prostate cancer recommends adequately informing each patient of the side effects of endocrine therapy. It lists the most significant side effects and provides some recommendations on supportive therapy (erectile dysfunction, hot flushes, gynecomastia). Yet for many side effects such as metabolic disorders, loss of muscle mass or pain, fatigue and cognitive dysfunction, only general recommendations such as physical activity are listed. While there is an abundance of literature relating to the side effects of endocrine therapy and their management in cases of breast cancer, only few publications on the topic exist for prostate cancer. Considering the significance of these side effects, more extensive information for physicians on how to prevent and treat the side effects of the treatments they prescribe, together with improved communication skills, could help to enhance quality of life for many patients.
References
Bell RJ, Fradkin P, Schwarz M, Davis SR (2013) Understanding discontinuation of oral adjuvant endocrine therapy by women with hormone receptor-positive invasive breast cancer nearly 4 years from diagnosis. Menopause 20(1):15–21
Bourke L, Kirkbridge P, Hooper R, Rosario AJ, Chico TJA, Rosario DJ (2013) Endocrine therapy in prostate cancer: time fr reappraisal of risks, benefits and cost-effectiveness? Brit J Cancer 108(9):13
Cluze C, Rey D, Huiart L, BenDiane MK, Bouhnik AD, Berenger C, Carrieri MP, Giorgi R (2012) Adjuvant endocrine therapy with tamoxifen in young women with breast cancer: determinants of interruptions vary over time. Ann Oncol 23:882–890
Gueth U, Myrick ME, Kilic N, Eppenberger-Castori S, Schmid SM (2012) Compliance and persistence of endocrine adjuvant breast cancer therapy. Breast Cancer Res Treat 131(2):491–499
Harle LK, Maggio M, Shahani S, Braga-Basaria M, Basaria S (2006) Endocrine complications of androgen-deprivation therapy in men with prostate cancer. Clin Adv Hematol Oncol 4(9):687–696
https://www.healthonnet.org/HONcode/German/. Assessed 27 July 2014
http://www.krebsgesellschaft.de/download/2009-pl-pca.pdf. Assessed 27 July 2014
http://www.krebsgesellschaft.de/download/patientenleitlinie_prostatakrebs_2_2013.pdf. Assessed 27 July 2014
http://www.discern.org.uk/discern_instrument.php. Assessed 27 July 2014
Interdisziplinäre Leitlinie der Qualität S3 zur Früherkennung, Diagnose und Therapie der verschiedenen Stadien des Prostatakarzinoms; Langversion 3.1–2. Aktualisierung, Oktober 2014; AWMF-RegisterNummer 043/022OL
Kumar S, Shelley M, Harrison C, Coles B, Wilt TJ, Mason MD (2006) Neo-adjuvant and adjuvant hormone therapy for localised and locally advanced prostate cancer. Cochrane Database Syst Rev 4:CD006019
Liu Y, Malin JL, Diamant AL, Thind A, Maly RC (2013) Adherence to adjuvant hormone therapy in low-income women with breast cancer: the role of provider-patient communication. Breast Cancer Res Treat 137:829–836
Mottet N, Peneau M, Mzeron JJ, Molinie V, Richaud P (2012) Addition of radiotherapy to long-term androgen deprivation in locally advanced prostate cancer: an open randomised phase 3 trial. Eur Urol 62(2):213–219
Nair B, Wilt T, MacDonald R, Rutks I (2002) Early versus deferred androgen suppression in the treatment of advanced prostatic cancer. Cochrane Database Syst Rev 1:CD003506
Payne H, Mason M (2011) Androgen deprivation therapy as adjuvant/neoadjuvant to radiotherapy for high-risk localised and locally advanced prostate cancer: recent developments. Br J Cancer 105(11):1628–1634
Steckelberg A, Berger B, Köpke S, Heesen C, Muehlhauser I (2005) Kriterien für evidenzbasierte Patienteninformationen. Z ärztl Fortb Qualitätssich 99:343–351
Studer U, Whelan P, Albrecht W, Casselman J, Reijke TMD, Knoenageö H, Madersbacher S, Isorna S, Sundaram SK, Collette L (2011) Long term results of immediate versus deferred deprivation in patients with no local treatment for T0-4 N0-2 M0 prostate cancer (EORTC 30891). J Urol 185(4S):e144
Widmarl A, Klepp O, Solberg A, Damber JE, Angelsen A, Fransson P, Lund JA, Tasdemir I, Hoyer M, Wiklund F, Fossa SD (2009) Endocrine treatment with or without radiotherapy, in locally advances prostate cancer (SPCG-7/SFUO-3): an open randomised phase III trial. Lancet 373(9660):301–308
Wuensch P, Hahne A, Haidinger R, Meißler K, Tenter B, Stoll C, Senf B, Huebner J (2015) Discontinuation and non-adherence to endocrine therapy in breast cancer patients: is lack of communication the decisive factor? J Cancer Res Clin Oncol 141(1):55–60
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they do have no conflicts of interest.
Ethical approval
All procedures performed were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. According to the regulations of the ethics committee of the University Hospital Frankfurt/Main for anonymous surveys, no ethics evaluation was necessary. As it was an anonymous survey, no written informed consent was obtained from the participants who voluntarily took part.
Rights and permissions
About this article
Cite this article
Jung, B., Stoll, C., Feick, G. et al. Prostate cancer patients’ report on communication about endocrine therapy and its association with adherence. J Cancer Res Clin Oncol 142, 465–470 (2016). https://doi.org/10.1007/s00432-015-2059-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-015-2059-2