Abstract
The concept of the “extended amygdala”, developed and explored by Lennart Heimer, Jose de Olmos, George Alheid, and their collaborators, has had an enormous impact on the field of neuroscience and on our own work. Measuring fear-potentiated startle test using conditioned stimuli that vary in length we suggest that the central nucleus of the amygdala (CeA) and the lateral division of the bed nucleus of the stria terminalis (BNSTL) are involved in short-term versus long-term fear responses we call phasic versus sustained fear, respectively. Outputs from the basolateral amygdala (BLA) activate the medial division of the CeA (CeAM) to very rapidly elicit phasic fear responses via CeAM projections to the hypothalamus and brainstem. The BLA also projects to the BNSTL, which together with other BNSTL inputs from the lateral CeA (CeAL) initiate a slower developing, but sustained fear response, akin to anxiety. We hypothesize this occurs because the CeAL releases the peptide corticotropin releasing hormone (CRF) into the BNSTL which facilitates the release of glutamate from BLA terminals. This activates the BNSTL which projects to hypothalamic and brainstem areas similar to those innervated by the CeAM that mediate the specific signs of fear and anxiety. The generality of this idea is illustrated by selective studies looking at context conditioning, social defeat, drug withdrawal and stress induced reinstatement.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Based on early observations by Johnston (1923), the concept of the “extended amygdala” was developed and explored in great detail by Lennart Heimer, Jose de Olmos, George Alheid, and their many collaborators to the great benefit of behavioral researchers such as ourselves. Among their many significant findings, and of particular relevance to our research, they showed that the central (CeA) and medial (MeA) nuclei of the amygdala and the bed nucleus of the stria terminalis (BNST) were connected by columns of cells located throughout the stria terminalis, the fiber tract that connects these amygdala nuclei with the BNST, and also in a ventrally located sublenticular part of the basal forebrain (Alheid and Heimer, 1988; Alheid et al. 1998). They showed also that the CeA projected primarily to the lateral division of the BNST (BNSTL) and that the MeA projected primarily to the medial division of the BNST (BNSTM). This was followed by a series of observations that the CeA and BNSTL shared many common attributes in terms of inputs, outputs, cell types, and neurochemical makeup, especially with respect to the high levels of several peptides found in both structures (Alheid et al. 1995). Based on the many similarities between these two components of the extended amygdala, and also on the known involvement of the CeA in conditioned fear (Kapp et al. 1979), our laboratory began also to evaluate the role of the BNSTL. This work was thoroughly reviewed in Walker et al. (2003) and will only be summarized briefly here.
CeA versus BNST involvement in fear responses: initial findings
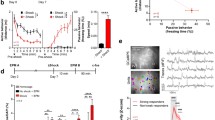
Similar to earlier work in which freezing was used as a fear measure (Iwata et al. 1986; LeDoux et al. 1988), we found that CeA but not BNSTL lesions also blocked fear-potentiated startle, in this case to a 3.2 s light that had previously been paired with footshock (Hitchcock and Davis 1986, 1991). We later found that startle also was increased after several minutes of exposure to bright light, even without prior pairings with shock (Walker and Davis 1997a). This “light-enhanced startle” effect could be reduced by anxiolytic compounds (de Jongh et al. 2002; Walker and Davis 2002; de Jongh et al. 2003) suggesting it represented an unconditioned anxiogenic effect of bright light, consistent with previous evidence that rats and mice will avoid brightly lit areas if possible and will show signs of anxiety if avoidance is not possible (DeFries et al. 1966; File and Hyde 1978; Crawley 1981). In contrast to fear-potentiated startle to short-duration conditioned fear stimuli (CSs), this light-enhanced startle effect was not blocked by local infusion of the AMPA receptor antagonist NBQX into the CeA but was blocked by local infusions into the BNSTL.
We also found that the BNSTL, but not the CeA, was involved in the startle increases produced by intraventricular infusions of the anxiogenic peptide corticotropin releasing factor (CRF). Thus, excitotoxic BNSTL lesions, or local infusions of a CRF antagonist into the BNSTL, but not into the CeA, completely blocked CRF enhanced startle (Davis et al. 1997). In addition, CRF infusions directly into the BNSTL (Lee and Davis 1997), but not into the CeA or any other part of the amygdala (Liang et al. 1992), also facilitated startle amplitude. The observed parallels between CRF- and light-enhanced startle suggested that CRF receptors in the BNSTL might contribute to light-enhanced startle. Using systemic administration of a proprietary non-peptide CRF-1 receptor antagonist, we have now found that CRF-R1 blockade does indeed disrupt light-enhanced startle, but has no effect on fear-potentiated startle at the doses tested (Fig. 1, and also see de Jongh et al. 2003).
What is perhaps most valuable about these specific paradigms (i.e., fear-potentiated and light-enhanced startle) is that, procedurally, they are so very similar. Each uses increased startle as a measure of fear, and light as the stimulus that triggers that fear. Indeed, the visual stimulus used in most of our fear-potentiated startle experiments is physically identical to that used in the light-enhanced startle procedure, differing only in its duration and conditioning history. These similarities are useful in that they greatly constrain the range of possible interpretations that might account for the observed differences in terms of CeA versus BNSTL involvement, and also to CRF receptor blockade.
In our initial paper describing the differential involvement of the CeA versus BNSTL in fear-potentiated and light-enhanced startle, we suggested that there were two possibilities that might account for the double dissociation between the involvement of these two structures in these two effects (Walker and Davis 1997a). One was that the CeA plays a special role in mediating conditioned fear responses, whereas the BNSTL plays a special role in unconditioned responses. The other was that the CeA plays a special role in mediating short-duration fear responses, whereas the BNSTL plays a special role in longer-duration responses. Based on recent data from our laboratory and elsewhere, we now believe that the second hypothesis is correct. We specifically suggest that the medial division of the CeA (CeAM) and its projections to brainstem areas which mediate many of the behaviors that are influenced by fear, is critical for short-duration fear responses whereas the lateral division of the CeA (CeAL) and it’s CRF-containing projections to the BNSTL are critical for sustained fear responses. We have also suggested that sustained fear in this model may be more akin to anxiety, as that term is commonly used, than is short-duration fear and as such may be particularly relevant clinically. These are the data we will concentrate on in this review.
Differential involvement of the CeA versus BNST in short- versus long-duration startle increases
The most direct test of these two alternatives (conditioned versus unconditioned or short- versus long-duration) would be to evaluate the effect of a BNSTL manipulation on fear responses elicited by a stimulus that is both conditioned and also long in duration. If blockade of BNSTL function disrupted the influence of such a stimulus, these results would be consistent with the short- versus long-duration hypothesis and inconsistent with the conditioned versus unconditioned hypothesis. We have now developed procedures that combine these two variables and have, in fact, found that the BNSTL is only involved in startle increases produced by long-duration (i.e., minutes as opposed to seconds) CSs.
In one such procedure, rats are placed into our standard startle box and presented with silence for the first 8 min followed by a continuous 8 min low-frequency-filtered noise. During this auditory stimulus, seven 0.5 s, 0.4 mA footshocks are presented using random interstimulus intervals. This procedure is done twice within a session for three total sessions spread across three consecutive days. In more recent versions we have presented seven clicker stimuli with durations ranging from 3 s to 8 min together with co-terminating footshock, also on each of three consecutive days. With both versions, the rats learn that when the CS comes on they are at risk for shock, but they do not know exactly when that shock might occur. Prior to training and also on the test day, startle amplitude is measured for 8 min before presentation of the CS and for 8 min during presentation of the CS in a context distinctively different from that used in training. For each animal, a percent change score from the pre-conditioning to the post-conditioning test is calculated. It should be emphasized that this is very different than our typical procedure in which the CS onset to shock interval is constant (typically 3.2 s) during training, and the CS onset to startle interval during testing is the same. Under those conditions, rats quickly learn to predict when the shock will occur as indicated by the fact that fear-potentiated startle is greatest when the CS-startle interval used in testing matches the CS-shock interval used in training (Davis et al. 1989).
Using this new procedure we typically see that there is relatively little change in startle amplitude from the pre- to the post-conditioning test during the pre-CS period (i.e., very little generalized context conditioning) but a large and consistent increase from the pre- to post-conditioning test when the CS is present. Using this design, we evaluated the effect of pre-test intra-BNSTL and intra-CeA NBQX infusions. As indicated in Fig. 2, intra-BNSTL infusions decreased the late (i.e., sustained) component of the fear-potentiated startle effect (min 5–8), but not the early component (min 1–4), whereas intra-CeA NBQX infusions had no effect using these parameters. These results are consistent with the hypothesis that the BNSTL plays a selective role in long-duration fear responses. That is, once the auditory cue had been on for 4 min the BNSTL, but not the CeA, became critical for this sustained fear response. In fact, during the first 4 min, inactivation of the BNSTL actually increased the magnitude of fear potentiated startle, consistent with recent data showing that the BNSTL may tonically inhibit fear-potentiated startle (Meloni et al. 2006). In fact, in the rat, the BNST sends a very strong projection to the CeAM (Fig. 3), which in one way or the other may tonically inhibit the expression of fear-potentiated startle.
Intra-BNSTL infusions decreased the late (i.e., sustained) component of the fear-potentiated startle effect (min 5–8), but not the early component (min 1–n4), whereas intra-CeA NBQX infusions had no effect using these parameters. These results are consistent with the hypothesis that the BNSTL plays a selective role in long-duration fear responses
Biotinylated dextran amine, an anterograde neuronal tracer, infused into the BNST (a) reveals projections to many of the same hypothalamic and brainstem areas involved in specific signs of fear that are also innervated by the CeAM, (c–f), and also to the CeAM itself (b), suggesting that the BNST may modulate CeAM-mediated fear
On the other hand, it was somewhat surprising that inactivation of the CeA did not disrupt fear-potentiated startle during the first 4 min of the auditory CS, given previous findings that electrolytic (Hitchcock and Davis 1987) or chemical (Campeau and Davis 1995) CeA lesions completely block fear-potentiated startle to 3.2 s auditory CSs, and that intra-CeA NBQX infusions block fear-potentiated startle to 3.2 s visual CSs (Walker and Davis 1997b). Further experiments are underway, using a larger group of rats, to more precisely determine when short-duration fear turns into sustained fear (as defined by susceptibility to these types of manipulations), using either NBQX into the CeA or a CRF-1 receptor antagonist given systemically (see below). We predict that fear-potentiated startle at very early parts of the 8 min CS will be blocked by inactivation of the CeA and spared by systemic administration of a CRF antagonist.
It should be noted also that the two paradigms differ in that the shock is very predictable in the phasic fear paradigm, but much less so in the sustained fear paradigm. Thus, it is possible that this aspect of the procedure alone renders sustained fear CeA-independent. To a large degree, fear duration and US predictability are inexorably intertwined. One is almost invariably a confound for the other. That is, for a highly predictable US (e.g., one which always occurs 3.2 s after CS onset), fear can be very brief, lasting only for the moment in which shock is anticipated (and this appears to be exactly what well-trained rats do Davis et al. 1989). For a less predictable shock (e.g., one which may occur at any time during an 8 min CS), fear must be maintained for the duration of the CS. It is possible however to disentangle these variables. Very recently, we found that animals trained with the paradigm that supports good sustained fear (i.e., variable duration CSs up to 8 min with coterminating footshock) also show high levels of fear-potentiated startle to a 3.7 s CS (Walker, Miles and Davis, in preparation). Because the conditioning procedure is the same, so is US predictability (in this case, it is very unpredictable). Thus, differences in the effect of any pretest manipulation would have to be attributed to differences in response duration rather than predictability. Although we have not yet done this with BNST inactivation, we very recently found that a CRF1 receptor antagonist that completely blocks sustained fear, has no effect on short-duration fear, irrespective of the conditioning procedure (i.e., irrespective of predictability—Walker, Miles and Davis, in preparation). Results from Waddell et al. (2006a), in which BNST lesions disrupted conditioned suppression to a 10 but not 1-min min clicker CS—both with very predictable co-terminating footshocks (discussed in next section)—would also appear more consistent with the conclusion that response duration rather than predictability is the key variable.
Figure 2 also shows data from animals with cannula placements in the basolateral amygdala (BLA). These data show that BLA inactivation disrupts fear-potentiated startle throughout the 8 min CS, consistent with its blockade of fear-potentiated startle both to a 3.2 s tone (Campeau and Davis 1995) and sustained fear (Fig. 2) in the light-enhanced startle paradigm (Walker and Davis 1997b). We believe that the BLA’s involvement in short-duration fear is mediated by projections to the CeAM, which in turn projects to the startle pathway, and that it’s involvement in sustained fear is mediated by projections to the BNSTL, which also projects to the startle pathway.
In summary, these data indicate that the BNSTL does indeed play an important role in the expression of conditioned fear responses, provided that those responses are maintained for a relatively long time. Based on the finding that an unconditioned visual stimulus begins to increase startle approximately 60 s after light onset (Davis et al. 1989) and the fact that inactivation of the BNST does not disrupt fear-potentiated startle to a 3.2 s visual CS (Walker and Davis 1997a), we suspect the transition from CeAM to BNSTL involvement may begin between 4 and 60 s after CS onset, although it may be several minutes before the response is fully dependent on the BNSTL.
Differential involvement of the CeA versus BNST in short- versus long-duration fear responses: corroborating evidence from other laboratories and paradigms
Another stimulus that is both conditioned and long in duration is context. Sullivan et al. (2004) have now reported that post-training electrolytic BNSTL lesions disrupt freezing as well as corticosterone responses to a context CS but do not affect these same responses to a brief auditory CS, whereas CeA lesions disrupt freezing to both. On the basis of those findings, they suggested that the BNSTL plays a special role in context-elicited fear. However, in light of our most recent findings, we wonder if a more parsimonious interpretation may be that BNSTL lesions disrupt context-elicited freezing simply because context CSs are invariably long in duration, and so too are the responses they evoke. Recent work by Waddell et al. (2006b) are especially relevant in that regard. They found that pre-training BNSTL lesions blocked conditioned suppression to a 10 min but not to a 1 min clicker CS. Interestingly, they also found that BNSTL lesions disrupted the context-dependent reinstatement of conditioned suppression (i.e., in previously extinguished animals) by footshock. Overall, a differential involvement of the BNST in long- versus short-duration fear responses has now been demonstrated across three different response measures—startle, freezing, and conditioned suppression. As such, the pattern does not appear to be idiosyncratic to a particular response measure.
What is the role of CRF receptors?
The same hypothesis that accounts for the differential involvement of the CeA and BNSTL in fear-potentiated versus light-enhanced startle might also account for the differential involvement of CRF receptors (as shown previously in Fig. 1). That is, CRF receptors might contribute to long- but not short-duration fear responses, irrespective of whether the fear or anxiety response is conditioned or unconditioned. In fact, prior studies have shown that the non-specific CRF antagonist, α-helical CRF9–41, does indeed disrupt sustained freezing responses to context CSs (Kalin and Takahashi 1990; Deak et al. 1999). We recently evaluated the same CRF-1 receptor antagonist used in Fig. 1 on fear-potentiated startle to the 8 min auditory CS. As shown in Fig. 4 (black bars), systemic administration of the CRF-1 receptor antagonist dose-dependently blocked fear-potentiated startle to the long-duration CS. In contrast to the effect of intra-BNSTL AMPA receptor blockade, however, the effect was apparent even during the first few minutes (the data for the entire 8 min CS period are combined in Fig. 3).
As previously noted, the same doses did not block fear-potentiated startle to a 3.7 s visual CS. To ensure that that dissociation was not indicative of a less interesting modality-specific effect, in a different group of rats we assessed the CRF-1 receptor antagonist’s ability to block fear-potentiated startle to a 3.7 s auditory CS. This too was unaffected (Fig. 4 gray bars). In fact, fear-potentiated startle in the intermediate dose group was actually greater, though not significantly so, than fear-potentiated startle in the vehicle group. Given the involvement of CRF in BNST functions, and evidence presented earlier that BNST inactivation actually facilitates fear-potentiated startle to short-duration threat cues (e.g., Meloni et al. 2006, and see also Fig. 2), one wonders if this nominal increase reflects random between-group variability or something more meaningful. In any case, the results clearly suggest that CRF-1 receptors contribute to long- (minutes) but not short- (seconds) duration fear responses in these paradigms, even though the time course does not exactly mirror that of AMPA receptor blockade in the BNSTL. However, as mentioned above, we expect that if we used a large number of rats and sampled startle every 10 s or so during the 8 min CS, the CRF-1 receptor antagonist would not block fear-potentiated startle during the earliest part of the 8 min CS but only after the CS had been on for some time. Alternatively, it is possible that fear-potentiated startle during the first few minutes of the 8 min CS is dependent on CRF but not AMPA receptors in the BNSTL or that the systemically administered CRF receptor antagonist acts elsewhere to disrupt fear-potentiated startle during the first few minutes of CS exposure. A possible site of action for the early effect is the BLA, where CRF receptors are found in even greater abundance than in the BNSTL (DeSouza et al. 1985; Chen et al. 2000). Perhaps then, CRF receptors in the BLA contribute to an intermediate phase of the fear response (e.g., during the first several minutes of the long-duration CS), whereas those in the BNSTL contribute to the more delayed component of this response. These ideas are currently being tested.
Circumstantial evidence for independent roles of the CeAM versus CeAL
The CeA can be divided into several subnuclei which include, most notably, the medial and lateral (CeAM and CeAL) subdivisions, respectively. Although both areas project to the BNSTL (Sun et al. 1991; Petrovich and Swanson 1997; Bourgeais et al. 2001; Dong et al. 2001), they are otherwise very different. First, the CeAM has many projections to brainstem nuclei and other areas that mediate behaviors influenced by fear (c.f., Davis 2000; Davis and Whalen 2001), including areas that mediate or modulate the acoustic startle response (Shammah-Lagnado et al. 1987; Rosen et al. 1991; Fendt et al. 1994; Meloni and Davis 1999; Shi et al. 2002). In contrast, CeAL projections to these areas are much more limited (Schwaber et al. 1982; Veening et al. 1984; Gray and Magnusson 1987, 1992). The CeAL projects instead to the substantia innominata, to the CeAM, and quite prominently to the BNSTL (Sun et al. 1991; Petrovich and Swanson 1997; Bourgeais et al. 2001; Dong et al. 2001).
The CeAM and CeAL are also very different in terms of their neurotransmitter content. Whereas CeAL neurons stain for a variety of neuropeptide transmitters, these same peptides are largely absent from CeAM neurons (Wray and Hoffman 1983; Veening et al. 1984; Moga and Gray 1985; Cassell et al. 1986; Gray and Magnusson 1987; Shimada et al. 1989; Otake et al. 1995; Day et al. 1999). One peptide found in great abundance in the CeAL is CRF. In fact, CeAL neurons are a major source of BNSTL CRF. This was demonstrated by Sakanaka et al. (1986) who found that electrolytic CeA but not BLA lesions dramatically reduced BNSTL CRF-immunoreactivity—nearly depleting it entirely from dorsal BNSTL. Many neurons within the BNSTL are themselves CRF-positive (Cummings et al. 1983; Cintra et al. 1987; Gray and Magnusson 1987; Shimada et al. 1989; Phelix and Paul 1990; Gray and Magnuson 1992; Makino et al. 1994a, b; Watts and Sanchez-Watts 1995; Day et al. 1999; Veinante et al. 2003), and CRF-positive neurons in both areas invariably express GABA (Veinante et al. 1997; Day et al. 1999). As high-frequency stimulation is known to favor peptide release (e.g., Lundberg et al. 1986; Bartfai et al. 1988; Whim 1989; Bourque 1991; Ip 1994), the findings suggest that the influence of these neurons (i.e., either inhibitory or excitatory) on downstream structures may vary as a function of the pattern of afferent activity. More specifically, sustained high-frequency activation may favor the release of CRF.
It is also of some interest that CRF-positive neurons in the CeAL and dorsal BNSTL express glucocorticoid receptors (Cintra et al. 1987; Honkaniemi et al. 1992; Lechner and Valentino 1999). The regulation of these neurons by stress hormones would be compatible with the hypothesis that the CeAL and BNSTL participate in long-duration responses to sustained threats that do not need to be as temporally precise as those to more immediate or predictable threats (e.g., fixed CS-US interval).
If CeAL neurons are involved in BNSTL-dependent effects, then it is necessary to account for the failure of intra-CeA NBQX infusions to disrupt light-enhanced startle in Walker and Davis (1997b) or the late stage of fear-potentiated startle to the 8 min CS in Fig. 2. One possibility is that the CeAM neurons thought to mediate short-duration fear responses via direct projections to the brainstem are AMPA-responsive, whereas the CeAL neurons that may mediate longer duration fear responses indirectly by way of projections to the BNSTL are not. Perhaps those neurons are driven instead by activation of other receptor types such as glucocorticoid and/or calcitonin gene related peptide (CGRP) receptors. Indeed, previous studies have shown that chronic corticosterone administration upregulates CRF mRNA in CeAL and BNSTL neurons (Swanson and Simmons 1989; Makino et al. 1994a,b; Watts and Sanchez-Watts 1995; Shepard et al. 2000; Liu et al. 2004), and interacts synergistically with CRF to increase startle amplitude (Lee et al. 1994). Calcitonin gene related peptide (CGRP) is also an interesting candidate, especially given its preferential distribution (Haring et al. 1991; Honkaniemi et al. 1992; Harrigan et al. 1994; Dobolyi et al. 2005) and that of its receptors (Kruger et al. 1988) within the lateral versus medial CeA. These receptors, when activated, produce various symptoms associated with fear and anxiety such as heart rate and blood pressure increases (Nguyen et al. 1986; Brown and Gray 1988), antinociception (Xu et al. 2003), and freezing (Kocorowski and Helmstetter 2001). CGRP-positive terminals directly innervate stress-responsive (Honkaniemi et al. 1992) CRF-containing neurons within the CeAL (Harrigan et al. 1994). Perhaps then, glutamate selectively activates CeAM neurons that mediate short-duration fear responses whereas CGRP, corticosterone and/or other peptide receptors selectively influence CeAL neurons which mediate more sustained fear responses. Having shown that intra-CeA NBQX infusions selectively influence the former, it will now be interesting to evaluate the effect of some of these other ligands on short- and long-duration startle increases.
Another difference is that the CeAM receives input from almost all other nuclei within the amygdala whereas the rat CeAL receives virtually no amygdala input at all (Jolkkonen and Pitkanen 1998). Instead, prominent inputs to the CeAL include those from insular and entorhinal cortices (Yasui et al. 1991; Sun et al. 1994; McDonald et al. 1997), and also from the paraventricular nucleus of the thalamus (PVT—Berendse and Groenewegen 1991; Turner and Herkenham 1991; Moga et al. 1995; Li and Kirouac 2008; Vertes and Hoover 2008)—areas that project very lightly to CeAM.
Projections from the PVT are especially interesting insofar as the PVT is one of the most stress-responsive areas in the brain, based on the induction of c-fos with a variety of stressors (Chastrette et al. 1991; Beck and Fibiger 1995; Cullinan et al. 1995; Duncan et al. 1996; Bhatnagar and Dallman 1998; Bhatnagar and Dallman 1999; Bubser and Deutch 1999). In fact, Bhatnagar and Dalman (1998) have suggested that the PVT to amygdala to paraventricular nucleus of the hypothalamus projection is a key regulator of the hypothalamic-pituitary-adrenal response to stress. Moreover, the PVT innervates regions in the extended amygdala, including the CeAL that contain corticotropin-releasing factor (CRF) neurons, many of which receive apparent contacts from PVT fibers (Li and Kirouac 2008). Interestingly, the PVT also appears to be involved in circadian rhythms and the BNST shows periodicity in clock gene expression that is highly similar to, and dependent upon, the suprachiasmatic nucleus (Amir et al. 2004).
Cortical inputs to CeAL are also interesting in that they raise the intriguing possibility that these inputs might mediate, at least in more cognitive species, the autonomic accompaniments of something more aptly described as ‘worry’. In fact, the insular cortex has now been found in several fMRI studies to become active when human subjects are told to anticipate shock (Phelps et al. 2001), or learn to expect other aversive stimuli in the course of conditioning procedures (Buchel et al. 1998; Ploghaus et al. 1999), as has the BNST (and see also Kalin 2005 for threat-induced BNST activation in nonhuman primates; Straube et al. 2007). The insular cortex also projects heavily to BNSTL as well as to the posterior part of the BLA (BLAP) (e.g., Yasui et al. 1991; McDonald et al. 1999), which itself projects to the BNSTL.
Functional evidence for an involvement of a CRF-containing CeAL→BNSTL projection in long-lasting anxiety and stress effects
Jasnow et al. (2004) found that social defeat behavior in Syrian Hamsters was reduced by pre-defeat unilateral electrolytic CeA lesions and also by pre-test unilateral intra-BNSTL D-Phe CRF12–41 infusions. The combined manipulation, on opposite sides of the brain, produced an even greater effect. On the basis of these results, they concluded that “stress activates CRF-containing neurons in the CeA, which then releases CRF within the BNST.” Similarly, Erb et al. (2001) showed that neither unilateral intra-CeA TTX infusions nor unilateral intra-BNSTL infusions of the CRF receptor antagonist D-Phe CRF12–41 disrupted the shock-induced reinstatement of extinguished cocaine-seeking behavior, but that the combination of both treatments, again on opposite sides of the brain, did, leading them to conclude that a “CRF-containing pathway from CeA to BNST is involved in mediating the effects of CRF…on the reinstatement of cocaine seeking.”
Although the results of both studies are consistent with the conclusions that were drawn, they are not definitive insofar as only a partial implementation of the crossed-lesion design was used. That is, neither study compared the effect of contralateral versus ipsilateral CRF antagonist infusions (e.g., left CeA lesion + CRF antagonist into right BNST versus CeA lesion + CRF antagonist into left BNST). Assuming that a serial CeA-to-BNST circuit is critical for the behavior in question, one would predict that the effect of ipsilateral CeA + BNST treatments would be less than that of contralateral CeA + BNST treatments, and equal in magnitude to that obtained by unilaterally manipulating either structure alone. Also, intra-CeA TTX infusions in the Erb et al. (2001) study and electrolytic CeA lesions in the Jasnow et al. (2004) study would have interrupted communication between the BLA and BNSTL because fibers from BLA project through the CeA on their way to the BNSTL (Davis and Whalen 2001; Dong et al. 2001). Thus, the observed behavioral effects in these studies might have been attributable to an interruption of this pathway instead. Thus, it will be important to determine if fiber-sparing inactivation of the CeA will reproduce these intriguing findings.
Role of the CeA and BNST in drug withdrawal
There is now considerable evidence that some of the anxiogenic effects of drug withdrawal involve activation of the both the CeA and BNST, and may involve CRF-containing projections from the CeAL to the BNST. This seems to be the case for the affective components of anxiety (e.g., avoidance of the place where drug withdrawal occurred) but not the somatic symptoms (e.g., classic signs of drug withdrawal such as wet dog shakes, teeth chattering, ptosis, postural abnormalities).
In the CeAL, naloxone-precipitated morphine withdrawal induced CRE-mediated transcription in approximately half of the neurons that are immuno-positive for CRF (Shaw-Lutchman et al. 2002), and significantly increases the number of c-Fos-positive neurons in the CeA and BNST (Gracy et al. 2001; Frenois et al. 2002; Jin et al. 2004; Frenois et al. 2005). Although c-Fos was not found in CRF-positive immunoreactive neurons in the amygdala (Hamlin et al. 2004), the number of CRF-immunoreactive neurons was significantly lower in withdrawn versus non-withdrawn rats, leading Veinante et al. (2003) to suggest that opiate withdrawal might actually have led to a massive activation and subsequent depletion of CRF from those cells. In fact, Olive et al. (2002) have shown that ethanol withdrawal does produce a large (~100%) increase in extracellular CRF in the BNST. As indicated above, naloxone-precipitated morphine withdrawal increases c-fos in the CeA as well as the BNST. Consistent with a serial circuit for withdrawal-associated effects, c-fos activation in the BNST is disrupted by excitotoxic CeA lesions, whereas c-fos activation in the CeA is not disrupted by excitotoxic BNST lesions. Moreover, lesions in either area disrupted the affective component of withdrawal, namely conditioned place aversions to the location in which withdrawal occurred (Nakagawa et al. 2005).
Although intraventricular or intra-CeA α-helical CRF9–41 infusions, as well as systemic injections of the CRF-R1 antagonist CP-154,526 (Iredale et al. 2000; Lu et al. 2000; McNally and Akil 2002) block many of the somatic signs of morphine withdrawal (e.g., wet dog shakes, teeth chattering, ptosis, postural abnormalities), α-helical CRF9–41 infusions directly into the BNST do not (McNally and Akil 2002). Also, the CRF antagonist D-Phe-CRF12–41 infused into the CeA but not the BNST, has been found to reduce ethanol consumption in alcohol-dependent animals (Funk et al. 2006). Thus, CRF in the BNST does not seem to be involved in the somatic components of opiate withdrawal.
On the other hand, there is considerable evidence to show that CRF (Heinrichs et al. 1995; Contarino and Papaleo 2005; Stinus et al. 2005) and the BNST, and more specifically norepinephrine acting on β-adrenergic receptors in the BNST, are involved in the affective state associated with drug withdrawal. The BNST has one of the highest concentrations of norepinephrine in the brain, and opiate withdrawal activates BNST-projecting cells in the A1 and A2 noradrenergic cell groups of the caudal medulla. Lesions of the ventral noradrenergic bundle, which interrupt the projection of these cell groups to the BNST, reduce opiate-withdrawal-induced place aversion, as do injections of β-adrenergic-receptor antagonists into the BNST itself, even though these same manipulations have little effect on the somatic symptoms of withdrawal (Aston-Jones et al. 1999; Delfs et al. 2000; and see also Cecchi et al. 2007).
Interestingly, noradrenergic bundle lesions did not reduce conditioned place aversions when shock was used as a US (Delfs et al. 2000), suggesting the effect was specific either to drug withdrawal or to addicted animals. On the other hand, lesions of the BNST did block conditioned place aversions to somatic pain produced by intra-peritoneal acetic acid injections or formalin injections in the paw, but had no effect on acetic acid-induced nociceptive writhing and actually increased formalin-induced nociceptive behaviors (Deyama et al. 2007). Taken together, these data suggest the BNST is primarly involved in the aversive affective symptoms of drug withdrawal. In fact, the reduction of the affective aspects of withdrawal using manipulations of the BNST, in the face of little or no effect on the somatic symptoms of withdrawal, may make the decrease in affective symptoms even more impressive.
BLA→BNSTL projections
The preceding section focused on the possible involvement of CeAL to BNSTL projections in sustained anxiety and aversive states. It is important to note also that the BNSTL. receives substantial inputs from the BLA—particularly from caudal part of the BLA (BLAp) —(Weller and Smith 1982; McDonald 1991; Dong et al. 2001). This may explain why NBQX infusions into the caudal rather than rostral BLA (Fig. 5) blocked light-enhanced startle in Walker and Davis (1997b). We have also found more recently that infusions of an NBQX/muscimol cocktail into the BLA completely block CRF-enhanced startle (Fig. 6). Although excitotoxic BLA lesions only attenuated CRF-enhanced startle in Lee and Davis (1997), the more caudal elements of the BLA were spared in that study. Moreover, electrolytic lesions of the CeA, through which BLA fibers pass on their way to the BNSTL (Davis and Whalen 2001; Dong et al. 2001) also block CRF-enhanced startle (Liang et al. 1992). Together, these results suggest that BLAP inputs to the BNSTL may be involved in CRF- as well as light-enhanced startle, and perhaps in sustained fear responses more generally.
NBQX infusions into the caudal rather than rostral Bla blocked light-enhanced startle in Walker and Davis (1997b)
One model that could account for these results posits that the activation of CRF receptors in the BNSTL potentiates either the release of glutamate from BLA terminals (i.e., pre-synaptic receptors), or the response of BNSTL neurons (i.e., post-synaptic receptors) to glutamate released these same BLAP or other neurons (Fig. 7). Findings consistent with the first possibility have been presented in abstract form by Forray et al. (2005). Using in vivo microdialysis, they found that intra-BNSTL infusions of high-K+ evoke a substantial rise in extracellular BNSTL glutamate in rats that had undergone a chronic stress procedure, and that this response was completely prevented by co-infusion of the CRF-1 receptor antagonist NBI 27914. Taken as a whole, these findings are consistent with a neural model in which (a) CeAL neurons release CRF into the BNSTL, (b) CRF binds to presynaptic receptors on BLAP terminals (c) CRF receptor activation increases the release of glutamate from these terminals, and (d) glutamatergic input to BNSTL neurons drives sustained threat responses (Fig. 8). We are currently testing this model using lesions that are selective for the CeAL versus the CeAM, microdialysis, and local drug infusions. More generally, we believe that the CeAL and the BNSTL, compared to the CeAM play a critical role in sustained responses to long-duration threats akin to anxiety. In some cases these effects are mediated by CRF and norepinephrine in either the CeAL, the BNSTL or both.
Schematic model of how we believe the extended amygdala produces sustained fear. CeAL neurons release CRF which binds to receptors located on the terminals of glutamatergic BLAP neurons. Activation of these CRF receptors increases glutamate release and, therefore, excitatory drive onto BNSTL neurons. These BNSTL neurons project to other areas, mostly in the brainstem, that mediate behaviors influenced by fear
Although we have limited this brief review to findings that we believe to be most directly relevant to the neural substrates of short- versus long-duration fear responses, many other studies have found evidence for a differential involvement of various components of the extended amygdala in a variety of other effects. Most notably these involve different patterns of morphological changes in the extended amygdala in different types of stress which have different effects on anxiety (Vyas et al. 2002, 2003), the critical role of the BNST as a relay between the hippocampus’s inhibitory modulation of the paraventricular nucleus of the hypothalamus (Cullinan et al. 1993) and the importance that norepinephrine plays in this circuitry (Forray and Gysling 2004), the role of the BNST in stress-induced anorexia (Ciccocioppo et al. 2003; Ciccocioppo et al. 2004) and the “learned helplessness” model of depression (Maier et al. 1993; Hammack et al. 2004), differential modulation in the amygdala versus BNST of stress-induced anxiety by norepinephrine and galanin (Morilak et al. 2003), differential effects of repeated urocortin infusions into the amygdala versus BNST on the development of anxiety and vulnerability to lactate-induced ‘panic’ (Sajdyk et al. 1999; Sajdyk and Gehlert 2000; Rainnie et al. 2004; Lee et al. 2008) and the role of the BNST versus the amygdala in predator odor induced anxiety (Fendt et al. 2003). As a whole, these studies point to the widespread influence of the concept of an “extended amygdala” and, therefore, to the significance of Lennart Heimers’ work—a body of research which has already proven influential in many different areas of behavioral neuroscience, including our own, and which we are certain will have an enduring impact on the field as a whole. We believe this would please Dr. Heimer very much.
References
Alheid G, De Olmos JS, Beltramino CA (1995) Amygdala and Extended Amygdala. In: Paxinos G (ed) The rat nervous system. Academic Press, New York, pp 495–578
Alheid GF, Heimer L (1988) New perspectives in basal forebrain organization of special relevance for neuropsychiatric disorders: the striatopallidal, amygdaloid, and corticopetal components of substantia innominata. Neuroscience 27:1–39. doi:10.1016/0306-4522(88)90217-5
Alheid GF, Beltramino CA, De Olmos JS, Forbes MS, Swanson DJ, Heimer L (1998) The neuronal organization of the supracapsular part of the stria terminalis in the rat: the dorsal component of the extended amygdala. Neuroscience 84:967–996. doi:10.1016/S0306-4522(97)00560-5
Amir S, Lamont EW, Robinson B, Stewart J (2004) A circadian rhythm in the expression of PERIOD2 protein reveals a novel SCN-controlled oscillator in the oval nucleus of the bed nucleus of the stria terminalis. J Neurosci 24:781–790. doi:10.1523/JNEUROSCI.4488-03.2004
Aston-Jones G, Delfs JM, Druhan J, Zhu Y (1999) The bed nucleus of the stria terminalis. A target site for noradrenergic actions in opiate withdrawal. Ann NY Acad Sci 877:486–498. doi:10.1111/j.1749-6632.1999.tb09284.x
Bartfai T, Iverfeldt K, Fisone G, Serfozo P (1988) Regulation of the release of coexisting neurotransmitters. Annu Rev Pharmacol Toxicol 28:285–310. doi:10.1146/annurev.pa.28.040188.001441
Beck CHM, Fibiger HC (1995) Conditioned fear-induced changes in behavior and in the expression of the immediate early gene c-fos: with and without diazepam pretreatment. J Neurosci 15:709–720
Berendse HW, Groenewegen HJ (1991) Restricted cortical termination fields of the midline and intralaminar thalamic nuclei in the rat. Neuroscience 42:73–102. doi:10.1016/0306-4522(91)90151-D
Bhatnagar S, Dallman M (1998) Neuroanatomical basis for facilitation of hypothalamic-pituitary-adrenal responses to a novel stressor after chronic stress. Neuroscience 84:1025–1039. doi:10.1016/S0306-4522(97)00577-0
Bhatnagar S, Dallman MF (1999) The paraventricular nucleus of the thalamus alters rhythms in core temperature and energy balance in a state-dependent manner. Brain Res 851:66–75. doi:10.1016/S0006-8993(99)02108-3
Bourgeais L, Gauriau C, Bernard JF (2001) Projections from the nociceptive area of the central nucleus of the amygdala to the forebrain: a PHA-L study in the rat. Eur J Neurosci 14:229–255. doi:10.1046/j.0953-816x.2001.01640.x
Bourque CW (1991) Activity-dependent modulation of nerve terminal excitation in a mammalian peptidergic system. Trends Neurosci 14:28–30. doi:10.1016/0166-2236(91)90180-3
Brown MR, Gray TS (1988) Peptide injections into the amygdala of conscious rats: Effects on blood pressure, heart rate and plasma catecholamines. Regul Pept 21:95–106. doi:10.1016/0167-0115(88)90094-8
Bubser M, Deutch AY (1999) Stress induces Fos expression in neurons of the thalamic paraventricular nucleus that innervate limbic forebrain sites. Synapse 32:13–22. doi :10.1002/(SICI)1098-2396(199904)32:1<13::AID-SYN2>3.0.CO;2-R
Buchel C, Morris J, Dolan RJ, Friston KJ (1998) Brain systems mediating aversive conditioning: an event-related fMRI study. Neuron 20:947–957. doi:10.1016/S0896-6273(00)80476-6
Campeau S, Davis M (1995) Involvement of the central nucleus and basolateral complex of the amygdala in fear conditioning measured with fear-potentiated startle in rats trained concurrently with auditory and visual conditioned stimuli. J Neurosci 15:2301–2311
Cassell MD, Gray TS, Kiss JZ (1986) Neuronal architecture in the rat central nucleus of the amygdala: a cytological, hodological, and immunocytochemical study. J Comp Neurol 246:478–499. doi:10.1002/cne.902460406
Cecchi M, Capriles N, Watson SJ, Akil H (2007) Beta1 adrenergic receptors in the bed nucleus of stria terminalis mediate differential responses to opiate withdrawal. Neuropsychopharmacology 32:589–599. doi:10.1038/sj.npp.1301140
Chastrette N, Pfaff DW, Gibbs RB (1991) Effects of daytime and nighttime stress on Fos-like immunoreactivity in the paraventricular nucleus of the hypothalamus, the habenula, and the posterior paraventricular nucleus of the thalamus. Brain Res 563:339–344. doi:10.1016/0006-8993(91)91559-J
Chen Y, Brunson KL, Muller MB, Cariaga W, Baram TZ (2000) Immunocytochemical distribution of corticotropin-releasing hormone receptor type-1 (CRF(1))-like immunoreactivity in the mouse brain: light microscopy analysis using an antibody directed against the C-terminus. J Comp Neurol 420:305–323. doi :10.1002/(SICI)1096-9861(20000508)420:3<305::AID-CNE3>3.0.CO;2-8
Ciccocioppo R, Cippitelli A, Economidou D, Fedeli A, Massi M (2004) Nociceptin/orphanin FQ acts as a functional antagonist of corticotropin-releasing factor to inhibit its anorectic effect. Physiol Behav 82:63–68. doi:10.1016/j.physbeh.2004.04.035
Ciccocioppo R, Fedeli A, Economidou D, Policani F, Weiss F, Massi M (2003) The bed nucleus is a neuroanatomical substrate for the anorectic effect of corticotropin-releasing factor and for its reversal by nociceptin/orphanin FQ. J Neurosci 23:9445–9451
Cintra A, Fuxe K, Harfstrand A, Agnati LF, Wikstrom AC, Okret S et al (1987) Presence of glucocorticoid receptor immunoreactivity in corticotrophin releasing factor and in growth hormone releasing factor immunoreactive neurons of the rat di- and telencephalon. Neurosci Lett 77:25–30. doi:10.1016/0304-3940(87)90601-X
Contarino A, Papaleo F (2005) The corticotropin-releasing factor receptor-1 pathway mediates the negative affective states of opiate withdrawal. Proc Natl Acad Sci USA 102:18649–18654. doi:10.1073/pnas.0506999102
Crawley JN (1981) Neuropharmacologic specificity of a simple animal model for the behavioral actions of benzodiazepines. Pharmacol Biochem Behav 15:695–699. doi:10.1016/0091-3057(81)90007-1
Cullinan WE, Herman JP, Watson SJ (1993) Ventral subicular interaction with the hypothalamic paraventricular nucleus: Evidence for a relay in the bed nucleus of the stria terminalis. J Comp Neurol 332:1–20. doi:10.1002/cne.903320102
Cullinan WE, Herman JP, Battaglia DF, Akil H, Watson SJ (1995) Pattern and time course of immediate early gene expression in rat brain following acute stress. Neuroscience 64:477–505. doi:10.1016/0306-4522(94)00355-9
Cummings S, Elde R, Ells J, Lindall A (1983) Corticotropin-releasing factor immunoreactivity is widely distributed within the central nervous system of the rat: an immunohistochemical study. J Neurosci 3:1355–1368
Davis M (2000) The role of the amygdala in conditioned and unconditioned fear and anxiety. In: Aggleton JP (ed) The amygdala, vol 2. Oxford University Press, Oxford, pp 213–287
Davis M, Whalen P (2001) The amygdala: Vigilance and emotion. Mol Psychiatry 6:13–34. doi:10.1038/sj.mp.4000812
Davis M, Schlesinger LS, Sorenson CA (1989) Temporal specificity of fear-conditioning: effects of different conditioned stimulus-unconditioned stimulus intervals on the fear-potentiated startle effect. J Exp Psychol Anim Behav Process 15:295–310. doi:10.1037/0097-7403.15.4.295
Davis M, Walker DL, Lee Y (1997) Roles of the amygdala and bed nucleus of the stria terminalis in fear and anxiety measured with the acoustic startle reflex: Possible relevance to PTSD. Ann NY Acad Sci 821:305–331. doi:10.1111/j.1749-6632.1997.tb48289.x
Day HE, Curran EJ, Watson SJ Jr, Akil H (1999) Distinct neurochemical populations in the rat central nucleus of the amygdala and bed nucleus of the stria terminalis: evidence for their selective activation by interleukin-1Beta. J Comp Neurol 413:113–128. doi :10.1002/(SICI)1096-9861(19991011)413:1<113::AID-CNE8>3.0.CO;2-B
de Jongh R, Groenink L, van der Gugten J, Olivier B (2002) Pharmacological validation of the light-enhanced startle paradigm as a putative animal model of anxiety. Psychopharmacology (Berl) 159:176–180. doi:10.1007/s002130100914
de Jongh R, Groenink L, van der Gugten J, Olivier B (2003) Light-enhanced and fear-potentiated startle: temporal characteristics and effects of alpha-helical corticotropin-releasing hormone. Biol Psychiatry 54:1041–1048. doi:10.1016/S0006-3223(03)00468-2
Deak T, Nguyen KT, Ehrlich AL, Watkins LR, Spencer RL, Maier SF et al (1999) The impact of the nonpeptide corticotropin-releasing hormone antagonist antalarmin on behavioral and endocrine responses to stress. Endocrinology 140:79–86. doi:10.1210/en.140.1.79
DeFries JC, Hegmann JP, Weir MW (1966) Open-field behavior in mice: evidence for a major gene effect mediated by the visual system. Science 154:1577. doi:10.1126/science.154.3756.1577
Delfs JM, Zhu Y, Druhan JP, Aston-Jones G (2000) Noradrenaline in the ventral forebrain is critical for opiate withdrawal-induced aversion. Nature 403:430–434. doi:10.1038/35000212
DeSouza EB, Insel TR, Perrin MH, Rivier J, Vale WW, Kuhar MJ (1985) Corticotropin-releasing factor receptors are widely distributed within the rat central nervous system: an autoradiographic study. J Neurosci 5:3189–3203
Deyama S, Nakagawa T, Kaneko S, Uehara T, Minami M (2007) Involvement of the bed nucleus of the stria terminalis in the negative affective component of visceral and somatic pain in rats. Behav Brain Res 176:367–371. doi:10.1016/j.bbr.2006.10.021
Dobolyi A, Irwin S, Makara G, Usdin TB, Palkovits M (2005) Calcitonin gene-related peptide-containing pathways in the rat forebrain. J Comp Neurol 489:92–119. doi:10.1002/cne.20618
Dong HW, Petrovich GD, Swanson LW (2001) Topography of projections from amygdala to bed nuclei of the stria terminalis. Brain Res Brain Res Rev 38:192–246. doi:10.1016/S0165-0173(01)00079-0
Duncan GE, Knapp DJ, Breese GR (1996) Neuroanatomical characterization of Fos induction in rat behavioral models of anxiety. Brain Res 713:79–91. doi:10.1016/0006-8993(95)01486-1
Erb S, Salmaso N, Rodaros D, Stewart J (2001) A role for the CRF-containing pathway from central nucleus of the amygdala to bed nucleus of the stria terminalis in the stress-induced reinstatement of cocaine seeking in rats. Psychopharmacology (Berl) 158:360–365. doi:10.1007/s002130000642
Fendt M, Koch M, Schnitzler H-U (1994) Lesions of the central grey block the sensitization of the acoustic startle response in rats. Brain Res 661:163–173. doi:10.1016/0006-8993(94)91193-2
Fendt M, Endres T, Apfelbach R (2003) Temporary inactivation of the bed nucleus of the stria terminalis but not of the amygdala blocks freezing induced by trimethylthiazoline, a component of fox feces. J Neurosci 23:23–28
File SE, Hyde JRG (1978) Can social interaction be used to measure anxiety. Br J Pharmacol 62:19–24
Forray MI, Gysling K (2004) Role of noradrenergic projections to the bed nucleus of the stria terminalis in the regulation of the hypothalamic-pituitary-adrenal axis. Brain Res Brain Res Rev 47:145–160. doi:10.1016/j.brainresrev.2004.07.011
Forray MI, Gonzales M, Hadwed N, Gonzalez MP (2005) Chronic immobilization stress increases glutamatergic transmission in the rat bed nucleus of the stria terminalis. in vivo microdialysis studies. Soc Neuosci Abstr Program No. 187.3
Frenois F, Cador M, Caille S, Stinus L, Le Moine C (2002) Neural correlates of the motivational and somatic components of naloxone-precipitated morphine withdrawal. Eur J Neurosci 16:1377–1389. doi:10.1046/j.1460-9568.2002.02187.x
Frenois F, Stinus L, Di Blasi F, Cador M, Le Moine C (2005) A specific limbic circuit underlies opiate withdrawal memories. J Neurosci 25:1366–1374. doi:10.1523/JNEUROSCI.3090-04.2005
Funk CK, O’Dell LE, Crawford EF, Koob GF (2006) Corticotropin-releasing factor within the central nucleus of the amygdala mediates enhanced ethanol self-administration in withdrawn, ethanol-dependent rats. J Neurosci 26:11324–11332. doi:10.1523/JNEUROSCI.3096-06.2006
Gracy KN, Dankiewicz LA, Koob GF (2001) Opiate withdrawal-induced fos immunoreactivity in the rat extended amygdala parallels the development of conditioned place aversion. Neuropsychopharmacology 24:152–160. doi:10.1016/S0893-133X(00)00186-X
Gray TS, Magnusson DJ (1987) Neuropeptide neuronal efferents from the bed nucleus of the stria terminalis and central amygdaloid nucleus to the dorsal vagal complex in the rat. J Comp Neurol 262:365–374. doi:10.1002/cne.902620304
Gray TS, Magnuson DJ (1992) Peptide immunoreactive neurons in the amygdala and the bed nucleus of the stria terminalis project to the midbrain central gray in the rat. Peptides 13:451–460. doi:10.1016/0196-9781(92)90074-D
Hamlin AS, Buller KM, Day TA, Osborne PB (2004) Effect of naloxone-precipitated morphine withdrawal on c-fos expression in rat corticotropin-releasing hormone neurons in the paraventricular hypothalamus and extended amygdala. Neurosci Lett 362:39–43. doi:10.1016/j.neulet.2004.02.033
Hammack SE, Richey KJ, Watkins LR, Maier SF (2004) Chemical lesion of the bed nucleus of the stria terminalis blocks the behavioral consequences of uncontrollable stress. Behav Neurosci 118:443–448. doi:10.1037/0735-7044.118.2.443
Haring C, Humpel C, Skofitsch G, Krobath J, Javorsky F, Saria A (1991) Calcitonin gene-related peptide in the amygdaloid complex of the rat: immunohistochemical and quantitative distribution, and drug effects on calcium dependent, potassium-evoked in vitro release. Synapse 8:261–269. doi:10.1002/syn.890080404
Harrigan EA, Magnuson DJ, Thunstedt GM, Gray TS (1994) Corticotropin releasing factor neurons are innervated by calcitonin gene-related peptide terminals in the rat central amygdaloid nucleus. Brain Res Bull 33:529–534. doi:10.1016/0361-9230(94)90079-5
Heinrichs SC, Menzaghi F, Schulteis G, Koob GF, Stinus L (1995) Suppression of corticotropin-releasing factor in the amygdala attenuates aversive consequences of morphine withdrawal. Behav Pharmacol 6:74–80. doi:10.1097/00008877-199501000-00011
Hitchcock JM, Davis M (1986) Lesions of the amygdala, but not of the cerebellum or red nucleus, block conditioned fear as measured with the potentiated startle paradigm. Behav Neurosci 100:11–22. doi:10.1037/0735-7044.100.1.11
Hitchcock JM, Davis M (1987) Fear-potentiated startle using an auditory conditioned stimulus: effect of lesions of the amygdala. Physiol Behav 39:403–408. doi:10.1016/0031-9384(87)90242-3
Hitchcock JM, Davis M (1991) The efferent pathway of the amygdala involved in conditioned fear as measured with the fear-potentiated startle paradigm. Behav Neurosci 105:826–842. doi:10.1037/0735-7044.105.6.826
Honkaniemi J, Pelto-Huikko M, Rechardt L, Isola J, Lammi A, Fuxe K et al (1992) Colocalization of peptide and glucocorticoid receptor immunoreactivities in rat central amygdaloid nucleus. Neuroendocrinology 55:451–459. doi:10.1159/000126156
Ip NY (1994) Pattern of presynaptic nerve activity can determine the type of neurotransmitter regulating a postsynaptic event. Nature 311:472–474. doi:10.1038/311472a0
Iredale PA, Alvaro JD, Lee Y, Terwilliger R, Chen YL, Duman RS (2000) Role of corticotropin-releasing factor receptor–1 in opiate withdrawal. J Neurochem 74:199–208. doi:10.1046/j.1471-4159.2000.0740199.x
Iwata J, LeDoux JE, Meeley MP, Arneric S, Reis DJ (1986) Intrinsic neurons in the amygdala field projected to by the medial geniculate body mediate emotional responses conditioned to acoustic stimuli. Brain Res 383:195–214. doi:10.1016/0006-8993(86)90020-X
Jasnow AM, Davis M, Huhman KL (2004) Involvement of central amygdalar and bed nucleus of the stria terminalis corticotropin-releasing factor in behavioral responses to social defeat. Behav Neurosci 118:1052–1061. doi:10.1037/0735-7044.118.5.1052
Jin C, Araki H, Nagata M, Suemaru K, Shibata K, Kawasaki H et al (2004) Withdrawal-induced c-Fos expression in the rat centromedial amygdala 24 h following a single morphine exposure. Psychopharmacology (Berl) 175:428–435
Johnston JB (1923) Further contribution to the study of the evolution of the forebrain. J Comp Neurol 35:337–481. doi:10.1002/cne.900350502
Jolkkonen E, Pitkanen A (1998) Intrinsic connections of the rat amygdaloid complex: projections originating in the central nucleus. J Comp Neurol 395:53–72. doi :10.1002/(SICI)1096-9861(19980525)395:1<53::AID-CNE5>3.0.CO;2-G
Kalin NH (2005) Brain regions associated with the expression and contextual regulation of anxiety in primates. Biol Psychiatry 58:796–804. doi:10.1016/j.biopsych.2005.05.021
Kalin NH, Takahashi LK (1990) Fear-motivated behavior induced by prior shock experience is mediated by corticotropin-releasing hormone systems. Brain Res 509:80–84. doi:10.1016/0006-8993(90)90311-X
Kapp BS, Frysinger RC, Gallagher M, Haselton JR (1979) Amygdala central nucleus lesions: effect on heart rate conditioning in the rabbit. Physiol Behav 23:1109–1117. doi:10.1016/0031-9384(79)90304-4
Kocorowski LH, Helmstetter FJ (2001) Calcitonin gene-related peptide released within the amygdala is involved in Pavlovian auditory fear conditioning. Neurobiol Learn Mem 75:149–163. doi:10.1006/nlme.2000.3963
Kruger L, Mantyh PW, Sternini C, Brecha NC, Mantyh CR (1988) Calcitonin gene-related peptide (CGRP) in the rat central nervous system: patterns of immunoreactivity and receptor binding sites. Brain Res 463:223–244. doi:10.1016/0006-8993(88)90395-2
Lechner SM, Valentino RJ (1999) Glucocorticoid receptor-immunoreactivity in corticotrophin-releasing factor afferents to the locus coeruleus. Brain Res 816:17–28. doi:10.1016/S0006-8993(98)00900-7
LeDoux JE, Iwata J, Cicchetti P, Reis DJ (1988) Different projections of the central amygdaloid nucleus mediate autonomic and behavioral correlates of conditioned fear. J Neurosci 8:2517–2529
Lee Y, Davis M (1997) Role of the hippocampus, bed nucleus of the stria terminalis and amygdala in the excitatory effect of corticotropin releasing hormone on the acoustic startle reflex. J Neurosci 17:6434–6446
Lee Y, Schulkin J, Davis M (1994) Effect of corticosterone on the enhancement of the acoustic startle reflex by corticotropin releasing factor (CRF). Brain Res 666:93–98. doi:10.1016/0006-8993(94)90286-0
Lee Y, Fitz S, Johnson PL, Shekhar A (2008) Repeated Stimulation of CRF Receptors in the BNST of Rats Selectively Induces Social but not Panic-Like Anxiety. Neuropsychopharmacology (Advance online publication)
Li S, Kirouac GJ (2008) Projections from the paraventricular nucleus of the thalamus to the forebrain, with special emphasis on the extended amygdala. J Comp Neurol 506:263–287. doi:10.1002/cne.21502
Liang KC, Melia KR, Campeau S, Falls WA, Miserendino MJD, Davis M (1992) Lesions of the central nucleus of the amygdala, but not of the paraventricular nucleus of the hypothalamus, block the excitatory effects of corticotropin releasing factor on the acoustic startle reflex. J Neurosci 12:2313–2320
Liu IY, Lyons WE, Mamounas LA, Thompson RF (2004) Brain-derived neurotrophic factor plays a critical role in contextual fear conditioning. J Neurosci 24:7958–7963. doi:10.1523/JNEUROSCI.1948-04.2004
Lu L, Liu D, Ceng X, Ma L (2000) Differential roles of corticotropin-releasing factor receptor subtypes 1 and 2 in opiate withdrawal and in relapse to opiate dependence. Eur J Neurosci 12:4398–4404. doi:10.1046/j.1460-9568.2000.01310.x
Lundberg JM, Rudehill A, Sollevi A (1986) Frequency- and reserpine-dependent chemical coding of sympathetic transmission: differential release of noradrenaline and neuropeptide Y from pig spleen. Neurosci Lett 63:96–100. doi:10.1016/0304-3940(86)90020-0
Maier SF, Grahn RE, Kalman BA, Sutton LC, Wiertelak EP, Watkins LR (1993) The role of the amygdala and dorsal raphe nucleus in mediating the behavioral consequences of inescapable shock. Behav Neurosci 107:377–388. doi:10.1037/0735-7044.107.2.377
Makino S, Gold PW, Schulkin J (1994)a Corticosterone effects on corticotropin-releasing hormone mRNA in the central nucleus of the amygdala and the parvocellular region of the paraventricular nucleus of the hypothalamus. Brain Res 640:105–112. doi:10.1016/0006-8993(94)91862-7
Makino S, Gold PW, Schulkin J (1994)b Effects of corticosterone on CRH mRNA and content in the bed nucleus of the stria terminalis; comparison with the effects in the central nucleus of the amygdala and the paraventricular nucleus of the hypothalamus. Brain Res 657:141–149. doi:10.1016/0006-8993(94)90961-X
McDonald AJ, Shamman-Lagnado SJ, Shi CJ, Davis M (1999) Cortical afferents to the extended amygdala. In: McGinty JF (ed) Annals of the New York Academy of Sciences. Annals of the New York Academy of Sciences, New York, pp 309–338
McDonald J (1991) Topographical organization of amygdaloid projections to the caudatoputamen, nucleus accumbens, and related striatal-like areas of the rat brain. Neuroscience 44:15–33. doi:10.1016/0306-4522(91)90248-M
McDonald RJ, Murphy RA, Guarraci FA, Gortler JR, White NM, Baker AG (1997) Systematic comparison of the effects of hippocampal and fornix-fimbria lesions on acquisition of three configural discriminations. Hippocampus 7:371–388. doi :10.1002/(SICI)1098-1063(1997)7:4<371::AID-HIPO3>3.0.CO;2-M
McNally GP, Akil H (2002) Role of corticotropin-releasing hormone in the amygdala and bed nucleus of the stria terminalis in the behavioral, pain modulatory, and endocrine consequences of opiate withdrawal. Neuroscience 112:605–617. doi:10.1016/S0306-4522(02)00105-7
Meloni EG, Davis M (1999) Muscimol in the deep layers of the superior colliculus/mesencephalic reticular formation blocks expression but not acquisition of fear-potentiated startle in rats. Behav Neurosci 113:1152–1160. doi:10.1037/0735-7044.113.6.1152
Meloni EG, Jackson A, Gerety LP, Cohen BM, Carlezon WA Jr (2006) Role of the bed nucleus of the stria terminalis (BST) in the expression of conditioned fear. Ann N Y Acad Sci 1071:538–541. doi:10.1196/annals.1364.059
Moga MM, Gray TS (1985) Evidence for corticotropin-releasing factor, neurotensin, and somatostatin in the neural pathway from the central nucleus of the amygdala to the parabrachial nucleus. J Comp Neurol 241:275–284. doi:10.1002/cne.902410304
Moga MM, Weis RP, Moore RY (1995) Efferent projections of the paraventricular thalamic nucleus in the rat. J Comp Neurol 359:221–238. doi:10.1002/cne.903590204
Morilak DA, Cecchi M, Khoshbouei H (2003) Interactions of norepinephrine and galanin in the central amygdala and lateral bed nucleus of the stria terminalis modulate the behavioral response to acute stress. Life Sci 73:715–726. doi:10.1016/S0024-3205(03)00392-8
Nakagawa T, Yamamoto R, Fujio M, Suzuki Y, Minami M, Satoh M et al (2005) Involvement of the bed nucleus of the stria terminalis activated by the central nucleus of the amygdala in the negative affective component of morphine withdrawal in rats. Neuroscience 134:9–19. doi:10.1016/j.neuroscience.2005.03.029
Nguyen KQ, Sills MA, Jacobowitz DM (1986) Cardiovascular effects produced by microinjection of calcitonin gene-related peptide into the rat central amygdaloid nucleus. Peptides 7:337–339. doi:10.1016/0196-9781(86)90233-0
Olive MF, Koenig HN, Nannini MA, Hodge CW (2002) Elevated extracellular CRF levels in the bed nucleus of the stria terminalis during ethanol withdrawal and reduction by subsequent ethanol intake. Pharmacol Biochem Behav 72:213–220. doi:10.1016/S0091-3057(01)00748-1
Otake K, Ruggiero DA, Nakamura Y (1995) Adrenergic innervation of forebrain neurons that project to the paraventricular thalamic nucleus in the rat. Brain Res 697:17–26. doi:10.1016/0006-8993(95)00749-G
Petrovich GD, Swanson LW (1997) Projections from the lateral part of the central amygdalar nucleus to the postulated fear conditioning circuit. Brain Res 763:247–254. doi:10.1016/S0006-8993(96)01361-3
Phelix CF, Paul WK (1990) Demonstration of distinct corticotropin releasing factor—containing neuron populatins in the bed nucleus of the stria terminalis. A light and electron microscoic immunocytochemical study in the rat. Histochemistry 94:345–364. doi:10.1007/BF00266441
Phelps EA, O’Connor KJ, Gatenby JC, Gore JC, Grillon C, Davis M (2001) Activation of the left amygdala to a cognitive representation of fear. Nat Neurosci 4:437–441. doi:10.1038/86110
Ploghaus A, Tracey I, Gati JS, Clare S, Menon RS, Matthews PM et al (1999) Dissociating pain from its anticipation in the human brain. Science 284:1979–1981. doi:10.1126/science.284.5422.1979
Rainnie DG, Bergeron R, Sajdyk TJ, Patil M, Gehlert DR, Shekhar A (2004) Corticotrophin releasing factor-induced synaptic plasticity in the amygdala translates stress into emotional disorders. J Neurosci 24:3471–3479. doi:10.1523/JNEUROSCI.5740-03.2004
Rosen JB, Hitchcock JM, Sananes CB, Miserendino MJD, Davis M (1991) A direct projection from the central nucleus of the amygdala to the acoustic startle pathway: anterograde and retrograde tracing studies. Behav Neurosci 105:817–825. doi:10.1037/0735-7044.105.6.817
Sajdyk TJ, Gehlert DR (2000) Astressin, a corticotropin releasing factor antagonist, reverses the anxiogenic effects of urocortin when administered into the basolateral amygdala. Brain Res 877:226–234. doi:10.1016/S0006-8993(00)02638-X
Sajdyk TJ, Schober DA, Gehlert DR, Shekhar A (1999) Role of corticotropin-releasing factor and urocortin within the basolateral amygdala of rats in anxiety and panic responses. Behav Brain Res 100:207–215. doi:10.1016/S0166-4328(98)00132-6
Sakanaka M, Shibasaki T, Lederis K (1986) Distribution and efferent projections of corticotropin-releasing factor-like immunoreactivity in the rat amygdaloid complex. Brain Res 382:213–238. doi:10.1016/0006-8993(86)91332-6
Schwaber JS, Kapp BS, Higgins GA, Rapp PR (1982) Amygdaloid and basal forebrain direct connections with the nucleus of the solitary tract and the dorsal motor nucleus. J Neurosci 2:1424–1438
Shammah-Lagnado SJ, Negrao N, Silva BA, Ricardo JA (1987) Afferent connections of the nuclei reticularis pontis oralis and caudalis: A horseradish peroxidase study in the rat. Neuroscience 20:961–989. doi:10.1016/0306-4522(87)90256-9
Shaw-Lutchman TZ, Barrot M, Wallace T, Gilden L, Zachariou V, Impey S et al (2002) Regional and cellular mapping of cAMP response element-mediated transcription during naltrexone-precipitated morphine withdrawal. J Neurosci 22:3663–3672
Shepard JD, Barron KW, Myers DA (2000) Corticosterone delivery to the amygdala increases corticotropin-releasing factor mRNA in the central amygdaloid nucleus and anxiety-like behavior. Brain Res 861:288–295. doi:10.1016/S0006-8993(00)02019-9
Shi C-J, Zhou X-L, Davis M (2002) A GABAergic projection from the central extended amygdala to the deep mesencephalic nucleus in rats. Society for Neuroscience Abstract 28:Abstract 284.214
Shimada S, Inagaki S, Kubota Y, Ogawa N, Shibasaki T, Takagi H (1989) Coexistence of peptides (corticotropin releasing factor/neurotensin and substance P/somatostatin) in the bed nucleus of the stria terminalis and central amygdaloid nucleus of the rat. Neuroscience 30:377–383. doi:10.1016/0306-4522(89)90259-5
Stinus L, Cador M, Zorrilla EP, Koob GF (2005) Buprenorphine and a CRF1 antagonist block the acquisition of opiate withdrawal-induced conditioned place aversion in rats. Neuropsychopharmacology 30:90–98. doi:10.1038/sj.npp.1300487
Straube T, Mentzel HJ, Miltner WH (2007) Waiting for spiders: brain activation during anticipatory anxiety in spider phobics. Neuroimage 37:1427–1436. doi:10.1016/j.neuroimage.2007.06.023
Sullivan GM, Apergis J, Bush DE, Johnson LR, Hou M, Ledoux JE (2004) Lesions in the bed nucleus of the stria terminalis disrupt corticosterone and freezing responses elicited by a contextual but not by a specific cue-conditioned fear stimulus. Neuroscience 128:7–14. doi:10.1016/j.neuroscience.2004.06.015
Sun N, Roberts L, Cassell D (1991) Rat central amygdaloid nucleus projections to the bed nucleus of the stria terminalis. Brain Res Bull 27:651–662. doi:10.1016/0361-9230(91)90041-H
Sun N, Yi H, Cassell MD (1994) Evidence for a GABAergic interface between cortical afferents and brainstem projection neurons in the rat central extended amygdala. J Comp Neurol 340:43–64. doi:10.1002/cne.903400105
Swanson LW, Simmons DM (1989) Differential steroid hormone and neural influences on peptide mRNA levels in CRH cells of the paraventricular nucleus: a hybridization histochemical study in the rat. J Comp Neurol 285:413–435. doi:10.1002/cne.902850402
Turner BH, Herkenham M (1991) Thalamoamygdaloid projections in the rat: a test of the amygdala’s role in sensory processing. J Comp Neurol 313:295–325. doi:10.1002/cne.903130208
Veening JG, Swanson LW, Sawchenko PE (1984) The organization of projections from the central nucleus of the amygdala to brain stem sites involved in central autonomic regulation: a combined retrograde transport-immunohistochemical study. Brain Res 303:337–357. doi:10.1016/0006-8993(84)91220-4
Veinante P, Stoeckel ME, Freund-Mercier MJ (1997) GABA- and peptide-immunoreactivities co-localize in the rat central extended amygdala. Neuroreport 8:2985–2989. doi:10.1097/00001756-199709080-00035
Veinante P, Stoeckel ME, Lasbennes F, Freund-Mercier MJ (2003) c-Fos and peptide immunoreactivities in the central extended amygdala of morphine-dependent rats after naloxone-precipitated withdrawal. Eur J Neurosci 18:1295–1305. doi:10.1046/j.1460-9568.2003.02837.x
Vertes RP, Hoover WB (2008) Projections of the paraventricular and paratenial nuclei of the dorsal midline thalamus in the rat. J Comp Neurol 508:212–237. doi:10.1002/cne.21679
Vyas A, Bernal S, Chattarji S (2003) Effects of chronic stress on dendritic arborization in the central and extended amygdala. Brain Res 965:290–294. doi:10.1016/S0006-8993(02)04162-8
Vyas A, Mitra R, Shankaranarayana Rao BS, Chattarji S (2002) Chronic stress induces contrasting patterns of dendritic remodeling in hippocampal and amygdaloid neurons. J Neurosci 22:6810–6818
Waddell J, Morris RW, Bouton ME (2006)a Effects of bed nucleus of the stria terminalis lesions on conditioned anxiety: aversive conditioning with long-duration conditional stimuli and reinstatement of extinguished fear. Behav Neurosci 120:324–336. doi:10.1037/0735-7044.120.2.324
Waddell J, Morris RW, Bouton ME (2006)b Effects of bed nucleus of the stria terminalis lesions on conditioned anxiety: aversive conditioning with long-duration conditional stimuli and reinstatement of extinguished fear. Behav Neurosci 120:324–336. doi:10.1037/0735-7044.120.2.324
Walker DL, Davis M (1997)a Anxiogenic effects of high illumination levels assessed with the acoustic startle paradigm. Biol Psychiatry 42:461–471. doi:10.1016/S0006-3223(96)00441-6
Walker DL, Davis M (1997)b Double dissociation between the involvement of the bed nucleus of the stria terminalis and the central nucleus of the amygdala in light-enhanced versus fear-potentiated startle. J Neurosci 17:9375–9383
Walker DL, Davis M (2002) Light enhanced startle: Further pharmacological and behavioral evaluation. Psychopharmacology (Berl) 159:304–310. doi:10.1007/s002130100913
Walker DL, Toufexis DJ, Davis M (2003) Role of the bed nucleus of the stria terminalis versus the amygdala in fear, stress, and anxiety. Eur J Pharmacol 463:199–216. doi:10.1016/S0014-2999(03)01282-2
Watts AG, Sanchez-Watts G (1995) Region-specific regulation of neuropeptide mRNAs in rat limbic forebrain neurones by aldosterone and corticosterone. J Physiol 484(Pt 3):721–736
Weller KL, Smith DA (1982) Afferent connections to the bed nucleus of the stria terminalis. Brain Res 232:255–270. doi:10.1016/0006-8993(82)90272-4
Whim MD (1989) Frequency-dependent release of peptide cotransmitters from identified cholinergic motor neurons in Aplysia. Proc Natl Acad Sci USA 86:9034–9038. doi:10.1073/pnas.86.22.9034
Wray S, Hoffman GE (1983) Organization and interrelationship of neuropeptides in the central amygdaloid nucleus of the rat. Peptides 4:525–541. doi:10.1016/0196-9781(83)90059-1
Xu W, Lundeberg T, Wang YT, Li Y, Yu LC (2003) Antinociceptive effect of calcitonin gene-related peptide in the central nucleus of amygdala: activating opioid receptors through amygdala-periaqueductal gray pathway. Neuroscience 118:1015–1022. doi:10.1016/S0306-4522(03)00069-1
Yasui Y, Saper C, Cechetto D (1991) Calcitonin gene-related peptide (CGRP) immunoreactive projections from the thalamus to the striatum and amygdala in the rat. J Comp Neurol 308:293–310. doi:10.1002/cne.903080212
Acknowledgments
This research was supported by National Institute of Mental Health grants MH069056, MH47840, MH57250 and MH59906, and the Science and Technology Center (The Center for Behavioral Neuroscience of the National Science Foundation under Agreement No. IBN–9876754) and the Yerkes Base Grant. “Principles of laboratory animal care (NIH publication No. 86–23, revised 1985) were followed and all procedures were approved by the Emory IACUC.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Walker, D.L., Davis, M. Role of the extended amygdala in short-duration versus sustained fear: a tribute to Dr. Lennart Heimer. Brain Struct Funct 213, 29–42 (2008). https://doi.org/10.1007/s00429-008-0183-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-008-0183-3