Abstract
The activity of neural circuits that underpin particular behaviours are one of the most interesting questions in neurobiology today. This understanding will not only lead to a detailed understanding of learning and memory formation, but also provides a platform for the development of novel therapeutic approaches to a range of neurological disorders that afflict humans. Among the different behavioural paradigms, Pavlovian fear conditioning and its extinction are two of the most extensively used to study acquisition, consolidation and retrieval of fear-related memories. The amygdala, medial prefrontal cortex (mPFC) and hippocampus are three regions with extensive bidirectional connections, and play key roles in fear processing. In this chapter, we summarise our current understanding of the structure and physiological role of these three regions in fear learning and extinction.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
2.1 Introduction
The brain is a complex organ, and disorders of brain function result in a host of disorders, that together make up over 50% of the burden of disease in most societies today. Understanding how neural activity results in thought and behaviour is not only an intrinsically interesting question, but is a crucial step toward finding reliable and specific treatments for neurological disorders. As a result, interest in encoding the neural circuits that underlie specific behaviour across many species has risen immensely. The development of tools such as optogenetics, multi-unit recordings of neurons in behaving animals, and the use of designer drugs that bind to engineered receptors have greatly accelerated this endeavour. One circuit that has evoked intense interest is the neural circuit that triggers fear responses. This circuit is evolutionary preserved and allows animals, including humans, to react rapidly and appropriately to adverse events, and is an essential survival mechanism. Physiological responses during fear include changes in the activity of the limbic system, with activation of the sympathetic nervous system that triggers a fight-or-flight response. As a result, such a response leads to an increase in heart rate, blood pressure and skin conductance, as well as a change in posture and mobility. This circuit and the biochemical mechanisms that underpin its activity have been studied for many years, however, recent findings have started to reveal the precise neural correlates and circuits involved.
2.2 Fear Conditioning and Extinction
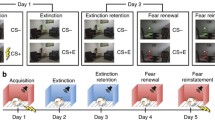
Fear is a normal physiological response triggered by specific events such as a natural threats and disasters or predators, and evokes a transient physiological and behavioural state that returns to baseline after some time. However, repetitive or single traumatic exposure can also lead to abnormal fear processing that result in anxiety related disorders such as post-traumatic stress disorder (PTSD). Due to the complexity of human fear processes, and the limitations in human imaging techniques, the neural circuits that underpin fear have been studied using a classical Pavlovian conditioning procedure called fear conditioning. In this procedure, an emotionally neutral stimulus, the conditioned stimulus (CS), such as a light or tone, is temporally paired with an aversive stimulus, the unconditioned stimulus (US), typically a mild shock. Following a small number of pairings, subjects form an association between the CS and US such that the CS predicts the subsequent US, and subjects begin to respond to the CS with an avoidance response, called the conditioned response (CR). The CR is rapidly acquired, long lasting, and results from the formation and storage of a long-term memory associating the CS with the US. However, subsequent presentations of the CS, not paired with the US, break this association, and lead to a gradual reduction of the CR through a process known as extinction. Since the first studies of Pavlov, it has been appreciated that extinction does not results from an erasure of previous memory associated with the CS but is due, at least in part, to new learning (Pavlov 1927). This idea rests on three key observations in extinction. First, the learnt fear response to the CS can reappear with the passage of time (spontaneous recovery). Secondly, the CR returns when the CS is presented in a context different from the one in which extinction training originally took place (renewal). Third, unexpected delivery of the US following extinction can restore the response to the CS (reinstatement). Both renewal and reinstatement show that the CS retains its ability to drive the CR following extinction. Some of these features of extinction are illustrated in Fig. 2.1. Thus, although the original memory is still present, extinction training results in a new memory trace that inhibits the response to the original CS. In effect, the subject has learnt that a previously aversive situation is no longer dangerous.
Fear learning and fear extinction. In fear conditioning subjects are presented with a neutral stimulus, the conditioned stimulus (CS), such as a tone, that is contingently paired with an aversive stimulus, the unconditioned stimulus (US) typically a foot shock. Following one or several pairings, subjects respond to the initially neutral stimulus with a conditioned response (high fear). Following fear conditioning, subjects are repeatedly presented with the neutral stimuli, but not paired with footshock, and result in a reduction of the fear response (extinction learning). Retrieval of extinction in the same context as extinction learning results in low fear due to extinction of the previously learnt fear. Spontaneous recovery of fear can occur with passage of time (days or years) and subjects again respond with a fearful response. Fear can also be renewed by exposing the animal to the conditioned stimulus outside the extinction context
Fear conditioning and extinction are evolutionarily conserved, and can be demonstrated in all species from insects to humans. Indeed, one of the most famous experiments is the classical fear conditioning trial with the baby called “Albert”. Albert was initially allowed to play with a rat, which he enjoyed, before the experimenter played an unpleasant auditory sound every time Albert touched the rat. After some time, Albert got very distressed just by seeing the rat, which in this case is the conditioned response. Dysfunction in the circuits that mediate fear conditioning and extinction is widely thought to be responsible for a range of anxiety related disorders including phobias and post-traumatic stress disorder. Moreover, treatments for some of these disorders using exposure therapy in which there is controlled repetitive exposure to the fearful stimulus, are based on extinction. Stimulation and lesion studies in animals have identified three key brain regions that contribute to fear learning and extinction: these are the amygdala, medial prefrontal cortex and hippocampus (Marek et al. 2013). Studies in humans using functional magnetic resonance imaging (fMRI), have found that the same three regions are also engaged in humans, suggesting the underlying neural circuits are shared.
2.3 Neural Circuits of Fear and Extinction
Fear responses are not the result of neural activity of single brain structures, but rather result from the orchestrated activity of multiple nuclei, mediated by synaptic connections between them to allow the fear response to occur.
2.4 Anatomy
The amygdala is a temporal lobe structure that is divided into over 20 subnuclei with extensive internuclear connections (Pape and Pare 2010; Sah et al. 2003). These subnuclei are commonly divided into three groups (Price et al. 1987; Sah et al. 2003): a deep basolateral (BLA) group that includes the lateral nucleus, basal nucleus and accessory basal nucleus; a more superficial or cortical-like group that includes the cortical nuclei and nucleus of the lateral olfactory tract; and a centromedial group composed of the medial and central nuclei (CeA). Of these, the BLA and CeA are the most widely studied nuclei of the amygdala, and both structures are highly involved in fear and extinction learning, and are generally thought to form the input and output regions of the amygdala respectively. The BLA is a cortical like structure and contains two types of neurons: glutamatergic principal neurons that form nearly 80% of the total cell population with the remaining being GABAergic interneurons (McDonald 1982, 1992; McDonald and Mascagni 2001; Spampanato et al. 2011). As with cortical regions, interneurons within the BLA are divided into different populations that show distinct electrophysiological properties and expression of particular cytosolic markers (Spampanato et al. 2011). Recent studies have begun to reveal the intrinsic organization and roles of some of these interneuron families, showing that specific types of interneuron make different types of local connections (Jasnow et al. 2013; Rainnie et al. 1991; Woodruff et al. 2006; Woodruff and Sah 2007a, b). Although the BLA does not have a laminar organization, principal neurons are not a single population either, but can be separated into distinct populations by their firing properties (Faber et al. 2001; Washburn and Moises 1992).
The CeA is the dominant output-structure of the amygdala. In contrast to the BLA, the CeA is a striatal-like structure that exclusively contains GABAergic neurons (de Olmos et al. 1985), and is divided into lateral (CeL) and medial (CeM) divisions (Cassell et al. 1986). Such a segregation of these two subregions was made possible by the identification of distinct peptide expression (Cassell et al. 1986) and intrinsic firing properties (Dumont et al. 2002; Lopez de Armentia and Sah 2004; Martina et al. 1999). Moreover, the two subregions of the CeA not only differ in their chemical and electrophysiological properties, but also in their connectivity. In the CeL, neurons receive excitatory inputs from the BLA as well as thalamic and cortical regions (Sah et al. 2003). Stimulation of BLA inputs provide excitation to these neurons (Lopez de Armentia and Sah 2004), and is often accompanied by a disynaptic inhibitory response (Amano et al. 2010; Lopez de Armentia and Sah 2004; Royer et al. 1999). This inhibition comes from two distinct sources: first, a cluster of GABAergic neurons interposed between the BLA and CeL, the intercalated cells (ITC) (Millhouse 1986), that receive excitatory input from the BLA and project to the CeL (Delaney and Sah 2001; Royer et al. 1999; Strobel et al. 2015). Secondly, neurons within the CeL, which are all GABAergic, are extensively interconnected, thereby providing strong local inhibition (Haubensak et al. 2010; Lopez de Armentia and Sah 2004). Recordings from the CeL in vivo have shown that following fear conditioning, some neurons increase their response to the CS, while others are inhibited, suggesting that different cells within the CeA receive different types of input. In contrast to the CeL, much less in understood about neurons in the CeM. However, CeM neurons project to different downstream targets, mediate different physiological responses (Pare and Duvarci 2012), and can be separated on electrophysiological as well as pharmacological grounds (Viviani et al. 2011).
The prefrontal cortex (PFC) is neocortical frontal lobe structure that is involved in a variety of higher cognitive processes such as decision making and attention (Heidbreder and Groenewegen 2003). For emotional learning in humans and primates, two PFC structure are crucially involved, namely the dorsolateral PFC (dlPFC), and the medial PFC (mPFC). However, in rodents, the PFC is much less developed, and the mPFC is the crucial player in fear learning in rodents, where it is cytoarchitectonically divided into four distinct regions from dorsal to ventral: medial precentral cortex, anterior cingulate cortex (ACC), prelimbic (PL) and infralimbic prefrontal cortex (IL) (Heidbreder and Groenewegen 2003).
In primates, the PFC is a regular 6 layered structure, whereas the mPFC of rodents appears to lack the granular cell layer (layer IV). Hence, pyramidal cells are located in layers II/III and layers V/VI (Yang et al. 1996), and in acute brain slices, these neurons show a range of different intrinsic firing properties (Wang et al. 2006) similar to those described for other neocortical regions (Connors and Gutnick 1990). As with the BLA, the mPFC also contains a variety of types of interneuron (Van De Werd et al. 2010), with the expected distribution of interneuronal markers (Markram et al. 2004).
The hippocampus (HPC) has long been identified as the main source for the storage and retrieval of explicit memory. Located in the medial temporal lobe in humans, it can be divided into multiple sub-regions, of which the subiculum and the CA1 region play important roles in integrating fear-related information. The CA1 region can further be separated into the dorsal and ventral portions that play distinct roles. Anatomically, the hippocampal formation is a three layered structure with pyramidal neurons restricted to the cell body layer, and dendritic trees spreading into two layers separating the basal and apical dendrites. Similar to the amygdala and mPFC, a variety of interneurons can be found in the hippocampal formation (Acoli et al. 2008; Freund and Buzsaki 1996). Moreover, the hippocampus is extensively connected with both the mPFC and the amygdala. Until recently, hippocampal efferents to the mPFC, and the reciprocal connections with the amygdala were thought to be exclusively glutamatergic. However, recent studies are suggesting that some GABAergic neurons in the hippocampus may also form long distance connections (McDonald and Mott 2017), but the roles and connections of these projections are largely unknown.
2.5 Functional Roles
2.5.1 The Amygdala
Within the amygdala, anatomical studies indicate that the nuclei are extensively interconnected (Pitkänen et al. 1997), and both CS and US information enters the amygdala at the level of the BLA where it is first processed. These inputs form classical dual component glutamatergic synapses containing alpha-amino-3-hydroxy-5-methyl-4-isoxalepropionic acid (AMPA) and N-Methyl-D-Aspartate (NMDA) receptors (Mahanty and Sah 1996; Weisskopf and LeDoux 1999). Blocking glutamatergic transmission within the BLA by infusion of non-NMDA receptor antagonists blocks fear conditioning, and post-learning infusions block expression of learnt fear (Falls et al. 1992; Kim et al. 1993). These pharmacological manipulations also block fear extinction (Kim et al. 1993), confirming that the BLA is an essential component of the neural circuit that mediates fear conditioning and extinction. In contrast, infusion of selective NMDA receptor antagonists into the amygdala block fear conditioning and extinction learning but have no effect on previously learnt fear (Goosens and Maren 2004; Miserendino et al. 1990). Together, these results have led to the current model in which learning during fear conditioning and extinction requires NMDA-receptor-dependent plasticity of inputs to neurons within the BLA (Mayford et al. 2012). In classical fear conditioning, CS information that is typically either auditory (via auditory cortex and auditory thalamus) or visual (via pulvinar and inferior temporal area), as well as US information (via thalamic inputs from the posterior thalamus) arrives at the BLA (Farb and Ledoux 1999; Lanuza et al. 2008; Sah et al. 2003). This convergent input to neurons within the BLA (Windels et al. 2016), coupled with the associative presentation of CS and US, is thought to result in long term potentiation of inputs carrying CS information to BLA principal neurons (Izquierdo et al. 2016; Pape and Pare 2010). Extinction training also requires NMDA-receptor dependent plasticity of glutamatergic input to BLA principal neurons (Falls et al. 1992), however, how this plasticity is initiated during repetitive CS presentation is not known. Single unit recordings during fear conditioning and extinction suggest that, following fear learning, the CS activates a population of principal neurons that have been called “fear” neurons (Herry et al. 2008). These “fear neurons” in turn, project directly to the central amygdala (CeA), and downstream projections from the CeA initiate the physiological responses seen in conditioned fear (Ehrlich et al. 2009; Pape and Pare 2010; Pare and Duvarci 2012). In extinction, fear neurons loose their CS-evoked activity, and a new set of neurons, called “extinction” neurons are instead driven by the CS (Herry et al. 2008). The activity of these neurons effectively inhibits the fear response.
Projections from the BLA enter the central amygdala at the level of the CeL, and single unit studies have shown that following fear conditioning, the CS drives a population of neurons called ‘ON neurons’. These cells locally inhibit a different population of tonically active ‘OFF neurons’(Ciocchi et al. 2010; Haubensak et al. 2010). These ‘OFF’ cells are GABAergic, and project to the CeM and the overall impact is disinhibition of neurons in the CeM by the CS (Ciocchi et al. 2010; Haubensak et al. 2010). Thus, following fear conditioning, the CS evokes activity of CeM neurons to trigger a fear response (Ciocchi et al. 2010; Haubensak et al. 2010). Following extinction training, there is a reduction in the activity in ‘fear neurons’ in the BLA, and ‘extinction neurons’ become active (Herry et al. 2008). As described above, the ITC neurons form a set of GABAergic neurons that provide feed-forward inhibition to the CeA. Synaptic input from the BLA to ITC neurons also show NMDA receptor-dependent plasticity (Royer and Paré 2002), and it has been proposed that following extinction, plasticity of these inputs results in an increase in disynaptic inhibition to the CeA, effectively reducing the activity of ON neurons, thus inhibiting the fear response (Amano et al. 2010). An attractive possibility is that BLA extinction neurons are selectively engaged in driving this feed-forward inhibition of the CeL. Together, these finding show that within the amygdala, distinct circuits mediate fear expression and extinction, and the population of neurons engaged by fear learning and extinction form distinct sets that are driven by distinct inputs.
2.5.2 Medial Prefrontal Cortex
In addition to the direct projections that carry CS information to the amygdala, “top-down” information from the prefrontal cortex also modulates amygdala activity. The first data that suggested a role for the mPFC in fear learning came from experiments in which this structure was ablated, resulting in a deficit in extinction memory (Morgan et al. 1993). It was therefore suggested that the mPFC is required for consolidation of extinction. Subsequent stimulation and inactivation studies of the mPFC have established that this region is involved in both fear conditioning and extinction (Burgos-Robles et al. 2007; Corcoran and Quirk 2007; Laurent and Westbrook 2008; Sotres-Bayon and Quirk 2010), with the infralimbic (IL) and prelimbic mPFC (PL) having distinct roles (Burgos-Robles et al. 2007). While the amygdala is engaged during acquisition and expression of learnt fear, the PL plays a key role in consolidation and recall of fear memory (Maren and Quirk 2004; Quirk and Mueller 2008). Thus, inactivation of the PL after fear acquisition results in reduced fear responses (Corcoran and Quirk 2007). The PL in turn sends direct glutamatergic projections to the BLA, and injection of anterograde tracer into the PL labels terminals largely limited to the basal nucleus of the BLA (McDonald 1998; McDonald et al. 1996). As described above, activity of BLA neurons is required for fear expression, and this excitatory input from the PL to the BLA is thought to modulate this activity and fear expression. However, the relationship between direct CS sensory input to the BLA and that mediated via the PL, and how these interact, is not clear.
In contrast to the PL, the IL does not appear to have a significant role in either fear or extinction learning, but is required for consolidation, and perhaps expression of extinction memory. Recent experiments using optogenetics to either enhance or silence neural activity has provided direct proof for the role of the IL in fear extinction (Do-Monte et al. 2015). Moreover, in support of chemical inactivation studies, electrophysiological recordings demonstrate that following extinction training, neurons in the IL show enhanced responses to the CS (Milad and Quirk 2002). Interestingly, infusion of NMDA receptor antagonists into the IL, either before or immediately after extinction training, impair extinction learning (Burgos-Robles et al. 2007), again suggesting that in addition to the BLA, synaptic plasticity in the IL may also be also required in consolidation of extinction learning. Memory consolidation is well known to require gene transcription and protein synthesis (Lubin et al. 2011), and supporting the role of mPFC in the consolidation of memory for fear extinction, evidence exists for the necessity of protein synthesis and gene transcription within the mPFC during the establishment of long-lasting fear extinction memories (Mamiya et al. 1993; Santini et al. 2004).
While the role of the IL in extinction memory is well established, the neural circuits between the IL and the amygdala that mediate this action remain controversial. Injection of anterograde tracers into the IL show extensive labelling in the lateral amygdala as well as intermediate capsule, a region between the BLA and CeA (McDonald 1998; McDonald et al. 1996). Several lines of evidence using neuronal tracing, specific lesioning and neuronal activity markers have shown that ITC neurons are active during extinction, leading to a model in which IL activity in extinction drives ITC neurons thereby inhibiting the output of the CeA (Amano et al. 2010; Freedman et al. 2000; Likhtik et al. 2008; McDonald et al. 1996; Pinto and Sesack 2008). However, recent studies suggest that afferents from the IL do not, in fact, innervate ITC neurons directly but rather target BLA neurons, which in turn target ITC neurons (Pinard et al. 2012; Strobel et al. 2015). Thus, how IL activity in extinction inhibits amygdala outputs that mediate fear responses remains unclear.
As described above, the mPFC sends projections to the amygdala that are involved in fear and extinction. In return, the mPFC receives afferents from the amygdala as well as a number of cortical and subcortical regions (Conde et al. 1995). Thus, amygdala and mPFC activity during fear learning and extinction are likely mediated by reciprocal synaptic connections between them (Quirk and Mueller 2008; Sotres-Bayon and Quirk 2010). In support of this proposal, single unit recordings in vivo show that within the BLA, both ‘fear neurons’ and ‘extinction neurons’ have connections with the mPFC, with ‘fear neurons’ only sending projections to the mPFC while ‘extinction neurons’ appear to be reciprocally connected to the mPFC (Herry et al. 2008). Moreover, inactivation of the BLA can reduce the response of PL neurons to the CS (Sotres-Bayon et al. 2012). Together, these results suggest complex interactions between the amygdala and PL in processing a conditioned stimulus.
2.5.3 Hippocampus
The hippocampal formation forms a major part of the medial temporal lobe system, and has been linked with emotional regulation since the first studies of Papez (1937). In agreement with this, the hippocampus has extensive connections with both the mPFC and the amygdala (McDonald and Mott 2016). In fear conditioning, subjects are placed in a particular environment where the CS is contingently paired with the US. In this paradigm, subjects learn to associate both the cue (tone) and the context (the environment) with an aversive event. Following fear conditioning, subjects show defensive behaviours (e.g., freezing) to the context in which learning took place (contextual fear memory), but also to the cue (the tone—cued conditioning) in contexts different from those where learning took place. The hippocampus is well known to be involved in processing information regarding space, and lesions of the dorsal hippocampus impair contextual fear memory but have little effect on cued conditioning to an auditory stimulus (Maren and Holt 2000). Thus, it is likely that the conditioned response to the context and the cue use distinct neural circuits with the hippocampus playing a major role in defining contextual cues.
Unlike cued fear conditioning, fear extinction is highly context dependent as the fear memory trace can be retrieved outside the fear extinction context (renewal). This context dependency in extinction is dependent on the ventral hippocampus (Hobin et al. 2006; Ji and Maren 2007). As described above, the IL plays a key role in fear extinction, and in agreement with the role of the hippocampus in fear extinction, there are extensive projections from the hippocampus to the mPFC (Parent et al. 2010). These arise mainly in the CA1 region and subiculum (Hoover and Vertes 2007), and innervate both pyramidal neurons and interneurons in the PL and IL (Parent et al. 2010). In the PL, inactivation studies show that hippocampal input can inhibit activity of pyramidal neurons (Sotres-Bayon et al. 2012). However, the effect of hippocampal input on the IL is not known, and how hippocampal activity modulates extinction via the IL is currently not understood. These circuits that mediate fear learning and extinction are summarised in Fig. 2.2.
Schematic of the neural circuitry involved in fear and its extinction. Under control conditions, sensory information which will be the conditioned stimulus (CS) reaches the amygdala at the level of the basolateral amygdala (BLA). This incoming input innervate excitatory principal neurons (triangles) and local inhibitory interneurons (circles). The BLA in turn projects to the central amygdala that contains inhibitory neurons with extensive local connections. Input arrives in the lateral division of the central amygdala (CeL), that has inhibitory connections with the medial division (CeM). Neurons in the central amygdala show different levels of tonic activity. During fear conditioning, CS inputs as well as the aversive unconditioned inputs (US) converge on principal neurons in the BLA, and as a result CS inputs are potentiated. Following consolidation, subsequent presentation of the CS enhances the activity of one set of principal neurons in the BLA that are now called “fear neurons”. As result input to the central amygdala is larger and drives the activity of a set of neurons called “on neurons”, these neurons in turn locally inhibit neurons in the CeL labelled “off neurons”. These “off neurons” are thought to project to the CeM and disinhibition of the neurons in the CeM mediates the physiological response during fear expression. The prelimbic preFig. 2.2 (continued) frontal cortex (PLPFC) and the hippocampus (HPC) are also involved in fear expression, however, their exact roles in driving fear neurons are not well understood. Following extinction, CS drive of “fear neurons” in the BLA is weakened while activity of a different set of neurons, now called “extinction neurons” is enhanced most likely due to synaptic plasticity of inputs to these cells. It is likely that extinction neurons, in turn drive a set of inhibitory neurons, the intercalated cell masses (ITCs) that then inhibit response of neurons in the central amygdala thereby reducing the fear response. In extinction, the infralimbic prefrontal cortex (ILPFC) and the HPC are also engaged, both of which send projections to the amygdala, and are important for extinction expression
2.6 Conclusions
Fear conditioning and extinction are two well-preserved learning paradigms seen in all mammalian species and involve the storage, consolidation and retrieval of a memory trace. It is widely believed that unravelling the mechanisms that underlie these learnt responses will provide a detailed understanding of learning and memory formation in the mammalian brain. The functional similarity between fear and anxiety disorders, and the fact that extinction recapitulates treatment strategies for these disorders, suggests that understanding the mechanisms that underpin these behaviours will lead to the development of treatments for human anxiety-related disorders. The neural circuits that mediate these two behaviours and the synaptic, biochemical and epigenetic changes that accompany them are beginning to be understood; however, there are clearly many gaps in our understanding. The development of new techniques to interrogate neural circuits in awake behaving animals holds much promise for the future.
References
Acoli, GA et. al., Petilla terminology: nomenclature of features of GABAergic interneurons of the cerebral cortex. Nat. Rev. Neuroscience. 2008; 9:557-568.
Amano T, Unal CT, Pare D. Synaptic correlates of fear extinction in the amygdala. Nat Neurosci. 2010;13:489–94.
Burgos-Robles A, Vidal-Gonzalez I, Santini E, Quirk GJ. Consolidation of fear extinction requires NMDA receptor-dependent bursting in the ventromedial prefrontal cortex. Neuron. 2007;53:871–80.
Cassell MD, Gray TS, Kiss JZ. Neuronal architecture in the rat central nucleus of the amygdala: a cytological, hodological, and immunocytochemical study. J Comp Neurol. 1986;246:478–99.
Ciocchi S, Herry C, Grenier F, Wolff SB, Letzkus JJ, Vlachos I, Ehrlich I, Sprengel R, Deisseroth K, Stadler MB, et al. Encoding of conditioned fear in central amygdala inhibitory circuits. Nature. 2010;468:277–82.
Conde F, Maire-Lepoivre E, Audinat E, Crepel F. Afferent connections of the medial frontal cortex of the rat. II. Cortical and subcortical afferents. J Comp Neurol. 1995;352:567–93.
Connors BW, Gutnick MJ. Intrinsic firing patterns of diverse neocortical neurons. Trends Neurosci. 1990;13:99–103.
Corcoran KA, Quirk GJ. Activity in prelimbic cortex is necessary for the expression of learned, but not innate, fears. J Neurosci. 2007;27:840–4.
de Olmos J, Hardy H, Heimer L. Amygdala. In: Paxinos G, editor. The rat nervous system. Sydney: Academic; 1985. p. 317–223.
Delaney AJ, Sah P. Pathway-specific targeting of GABA(A) receptor subtypes to somatic and dendritic synapses in the central amygdala. J Neurophysiol. 2001;86:717–23.
Do-Monte FH, Manzano-Nieves G, Quinones-Laracuente K, Ramos-Medina L, Quirk GJ. Revisiting the role of infralimbic cortex in fear extinction with optogenetics. J Neurosci. 2015;35:3607–15.
Dumont EC, Martina M, Samson RD, Drolet G, Paré D. Physiological properties of central amygdala neurons: species differences. Eur J Neurosci. 2002;15:544–52.
Ehrlich I, Humeau Y, Grenier F, Ciocchi S, Herry C, Luthi A. Amygdala inhibitory circuits and the control of fear memory. Neuron. 2009;62:757–71.
Faber ESL, Callister RJ, Sah P. Morphological and electrophysiological properties of principal neurons in the rat lateral amygdala in vitro. J Neurophysiol. 2001;85:714–23.
Falls WA, Miserendino MJ, Davis M. Extinction of fear-potentiated startle: blockade by infusion of an NMDA antagonist into the amygdala. J Neurosci. 1992;12:854–63.
Farb CR, Ledoux JE. Afferents from rat temporal cortex synapse on lateral amygdala neurons that express NMDA and AMPA receptors. Synapse. 1999;33:218–29.
Freedman LJ, Insel TR, Smith Y. Subcortical projections of area 25 (subgenual cortex) of the macaque monkey. J Comp Neurol. 2000;421:172–88.
Freund TF, Buzsaki G. Interneurons of the hippocampus. Hippocampus. 1996;6:347–470.
Goosens KA, Maren S. NMDA receptors are essential for the acquisition, but not expression, of conditional fear and associative spike firing in the lateral amygdala. Eur J Neurosci. 2004;20:537–48.
Haubensak W, Kunwar PS, Cai H, Ciocchi S, Wall NR, Ponnusamy R, Biag J, Dong HW, Deisseroth K, Callaway EM, et al. Genetic dissection of an amygdala microcircuit that gates conditioned fear. Nature. 2010;468:270–6.
Heidbreder CA, Groenewegen HJ. The medial prefrontal cortex in the rat: evidence for a dorso-ventral distinction based upon functional and anatomical characteristics. Neurosci Biobehav Rev. 2003;27:555–79.
Herry C, Ciocchi S, Senn V, Demmou L, Muller C, Luthi A. Switching on and off fear by distinct neuronal circuits. Nature. 2008;454:600–6.
Hobin JA, Ji J, Maren S. Ventral hippocampal muscimol disrupts context-specific fear memory retrieval after extinction in rats. Hippocampus. 2006;16:174–82.
Hoover WB, Vertes RP. Anatomical analysis of afferent projections to the medial prefrontal cortex in the rat. Brain Struct Funct. 2007;212:149–79.
Izquierdo I, Furini CR, Myskiw JC. Fear Memory. Physiol Rev. 2016;96:695–750.
Jasnow AM, Ehrlich DE, Choi DC, Dabrowska J, Bowers ME, McCullough KM, Rainnie DG, Ressler KJ. Thy1-expressing neurons in the basolateral amygdala may mediate fear inhibition. J Neurosci. 2013;33:10396–404.
Ji J, Maren S. Hippocampal involvement in contextual modulation of fear extinction. Hippocampus. 2007;17:749–58.
Kim M, Campeau S, Falls WA, Davis M. Infusion of the non-NMDA receptor antagonist CNQX into the amygdala blocks the expression of fear-potentiated startle. Behav Neural Biol. 1993;59:5–8.
Lanuza E, Moncho-Bogani J, Ledoux JE. Unconditioned stimulus pathways to the amygdala: effects of lesions of the posterior intralaminar thalamus on foot-shock-induced c-Fos expression in the subdivisions of the lateral amygdala. Neuroscience. 2008;155:959–68.
Laurent V, Westbrook RF. Distinct contributions of the basolateral amygdala and the medial prefrontal cortex to learning and relearning extinction of context conditioned fear. Learn Mem. 2008;15:657–66.
Likhtik E, Popa D, Apergis-Schoute J, Fidacaro GA, Pare D. Amygdala intercalated neurons are required for expression of fear extinction. Nature. 2008;454:642–5.
Lopez de Armentia M, Sah P. Firing properties and connectivity of neurons in the rat lateral central nucleus of the amygdala. J Neurophysiol. 2004;92:1285–94.
Lubin FD, Gupta S, Parrish RR, Grissom NM, Davis RL. Epigenetic mechanisms: critical contributors to long-term memory formation. Neuroscientist. 2011;17:616–32.
Mahanty NK, Sah P. The physiology of excitatory synapses in the lateral and basolateral amygdala. Soc Neurosci. Abstracts 22; 1996.
Mamiya N, Goldenring JR, Tsunoda Y, Modlin IM, Yasui K, Usuda N, Ishikawa T, Natsume A, Hidaka H. Inhibition of acid secretion in gastric parietal cells by the Ca2+/calmodulin-dependent protein kinase II inhibitor KN-93. Biochem Biophys Res Commun. 1993;195:608–15.
Marek, R. Strobel, C., Bredy, TW., Pankaj Sah, P. The amygdala and medial prefrontal cortex: partners in the fear circuit. The Journal of Physiology. 2013; 591(10):2381–2391
Maren S, Holt W. The hippocampus and contextual memory retrieval in Pavlovian conditioning. Behav Brain Res. 2000;110:97–108.
Maren S, Quirk GJ. Neuronal signalling of fear memory. Nat Rev Neurosci. 2004;5:844–52.
Markram H, Toledo-Rodriguez M, Wang Y, Gupta A, Silberberg G, Wu C. Interneurons of the neocortical inhibitory system. Nat Rev Neurosci. 2004;5:793–807.
Martina M, Royer S, Pare D. Physiological properties of central medial and central lateral amygdala neurons. J Neurophysiol. 1999;82:1843–54.
Mayford M, Siegelbaum SA, Kandel ER. Synapses and memory storage. Cold Spring Harb Perspect Biol. 2012;4(6):a005751.
McDonald AJ. Neurons of the lateral and basolateral amygdaloid nuclei: a golgi study in the rat. J Comp Neurol. 1982;212:293–312.
McDonald AJ. Projection neurons of the basolateral amygdala: a correlative Golgi and retrograde tract tracing study. Brain Res Bull. 1992;28:179–85.
McDonald AJ. Cortical pathways to the mammalian amygdala. Prog Brain Res. 1998;55:257–332.
McDonald AJ, Mascagni F. Colocalization of calcium-binding proteins and GABA in neurons of the rat basolateral amygdala. Neuroscience. 2001;105:681–93.
McDonald AJ, Mott DD. Functional neuroanatomy of amygdalohippocampal interconnections and their role in learning and memory. J Neurosci Res. 2017;95(3):797–820.
McDonald AJ, Mascagni F, Guo L. Projections of the medial and lateral prefrontal cortices to the amygdala: a Phaseolus vulgaris leucoagglutinin study in the rat. Neuroscience. 1996;71:55–75.
Milad MR, Quirk GJ. Neurons in medial prefrontal cortex signal memory for fear extinction. Nature. 2002;420:70–4.
Millhouse OE. The intercalated cells of the amygdala. J Comp Neurol. 1986;247:246–71.
Miserendino MJD, Sananes CB, Melia KR, Davis M. Blocking of acquisition but not expression of conditioned fear-potentiated startle by NMDA antagonists in the amygdala. Nature. 1990;345:716–8.
Morgan MA, Romanski LM, LeDoux JE. Extinction of emotional learning: contribution of medial prefrontal cortex. Neurosci Lett. 1993;163:109–13.
Pape HC, Pare D. Plastic synaptic networks of the amygdala for the acquisition, expression, and extinction of conditioned fear. Physiol Rev. 2010;90:419–63.
Papez JW. A proposed mechanism of emotion. Arch Neurol Psychiatry. 1937;38:725–43.
Pare D, Duvarci S. Amygdala microcircuits mediating fear expression and extinction. Curr Opin Neurobiol. 2012;22:717–23.
Parent MA, Wang L, Su J, Netoff T, Yuan LL. Identification of the hippocampal input to medial prefrontal cortex in vitro. Cereb Cortex. 2010;20:393–403.
Pavlov IP. Conditioned reflexes. New York: Dover; 1927.
Pinard CR, Mascagni F, McDonald AJ. Medial prefrontal cortical innervation of the intercalated nuclear region of the amygdala. Neuroscience. 2012;205:112–24.
Pinto A, Sesack SR. Ultrastructural analysis of prefrontal cortical inputs to the rat amygdala: spatial relationships to presumed dopamine axons and D1 and D2 receptors. Brain Struct Funct. 2008;213:159–75.
Pitkänen A, Savander V, LeDoux JE. Organization of intra-amygdaloid circuitries in the rat: an emerging framework for understanding functions of the amygdala. Trends Neurosci. 1997;20:517–23.
Price JL, Russchen FT, Amaral DG. The limbic region. II: the amygdaloid complex. In: Bjorklund A, Hökfelt T, Swanson LW, editors. Handbook of chemical neuroanatomy, vol. 5, Integrated systems of the CNS, part I. Amsterdam: Elsevier Science; 1987.
Quirk GJ, Mueller D. Neural mechanisms of extinction learning and retrieval. Neuropsychopharmacology. 2008;33:56–72.
Rainnie DG, Asprodini EK, Schinnick-Gallagher P. Inhibitory transmission in the basolateral amygdala. J Neurophysiol. 1991;66:999–1009.
Royer S, Paré D. Bidirectional synaptic plasticity in intercalated amygdala neurons and the extinction of conditioned fear responses. Neuroscience. 2002;115:455–62.
Royer S, Martina M, Paré D. An inhibitory interface gates impulse traffic between the input and output stations of the amygdala. J Neurosci. 1999;19:10575–83.
Sah P, Faber ES, Lopez De Armentia M, Power J. The amygdaloid complex: anatomy and physiology. Physiol Rev. 2003;83:803–34.
Santini E, Ge H, Ren K, Pena de Ortiz S, Quirk GJ. Consolidation of fear extinction requires protein synthesis in the medial prefrontal cortex. J Neurosci. 2004;24:5704–10.
Sotres-Bayon F, Quirk GJ. Prefrontal control of fear: more than just extinction. Curr Opin Neurobiol. 2010;20:231–5.
Sotres-Bayon F, Sierra-Mercado D, Pardilla-Delgado E, Quirk GJ. Gating of fear in prelimbic cortex by hippocampal and amygdala inputs. Neuron. 2012;76:804–12.
Spampanato J, Polepalli J, Sah P. Interneurons in the basolateral amygdala. Neuropharmacology. 2011;60:765–73.
Strobel C, Marek R, Gooch HM, Sullivan RK, Sah P. Prefrontal and auditory input to intercalated neurons of the amygdala. Cell Rep. 2015. https://doi.org/10.1016/j.celrep.2015.02.008. [Epub ahead of print].
Van De Werd HJ, Rajkowska G, Evers P, Uylings HB. Cytoarchitectonic and chemoarchitectonic characterization of the prefrontal cortical areas in the mouse. Brain Struct Funct. 2010;214:339–53.
Viviani D, Charlet A, van den Burg E, Robinet C, Hurni N, Abatis M, Magara F, Stoop R. Oxytocin selectively gates fear responses through distinct outputs from the central amygdala. Science. 2011;333:104–7.
Wang Y, Markram H, Goodman PH, Berger TK, Ma J, Goldman-Rakic PS. Heterogeneity in the pyramidal network of the medial prefrontal cortex. Nat Neurosci. 2006;9:534–42.
Washburn MS, Moises HC. Electrophysiological and morphological properties of rat basolateral amygdaloid neurons in vitro. J Neurosci. 1992;12:4066–79.
Weisskopf MG, LeDoux JE. Distinct populations of NMDA receptors at subcortical and cortical inputs to principal cells of the lateral amygdala. J Neurophysiol. 1999;81:930–4.
Windels F, Yan S, Stratton PG, Sullivan R, Crane JW, Sah P. Auditory Tones and Foot-Shock Recapitulate Spontaneous Sub-Threshold Activity in Basolateral Amygdala Principal Neurons and Interneurons. PLoS One. 2016;11:e0155192.
Woodruff AR, Sah P. Inhibition and synchronization of basal amygdala principal neuron spiking by parvalbumin-positive interneurons. J Neurophysiol. 2007a;98:2956–61.
Woodruff AR, Sah P. Networks of parvalbumin-positive interneurons in the basolateral amygdala. J Neurosci. 2007b;27:553–63.
Woodruff AR, Monyer H, Sah P. GABAergic excitation in the basolateral amygdala. J Neurosci. 2006;26:11881–7.
Yang CR, Seamans JK, Gorelova N. Electrophysiological and morphological properties of layers V-VI principal pyramidal cells in rat prefrontal cortex in vitro. J Neurosci. 1996;16:1904–21.
Acknowledgments
This work for funded by grants from the National Health and Medical Research Council of Australia and the Centre for Integrative Brain Function from the Australian Research Council (CE140100007).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Marek, R., Sah, P. (2018). Neural Circuits Mediating Fear Learning and Extinction. In: Cheung-Hoi Yu, A., Li, L. (eds) Systems Neuroscience. Advances in Neurobiology, vol 21. Springer, Cham. https://doi.org/10.1007/978-3-319-94593-4_2
Download citation
DOI: https://doi.org/10.1007/978-3-319-94593-4_2
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-94591-0
Online ISBN: 978-3-319-94593-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)