Abstract
Background
Pancreatoduodenectomy in Germany is performed by a broad range of hospitals. A diversity of operative techniques is employed as no guidelines exist for intra- and perioperative management. We carried out a national survey to determine the de facto German standards for pancreatoduodenectomy, assess quality assurance measures, and identify relevant issues for further investigation.
Methods
A questionnaire evaluating major outcome variables, case load, preferred surgical procedures, and perioperative management during pancreatoduodenectomy was developed and sent to 211 German hospitals performing >12 pancreatoduodenectomies per year (requirement for certification as a pancreas center). Statistical analysis was carried out using the Fisher Exact, Mann–Whitney U, and Spearman tests.
Results
The final response rate was 86 % (182/211). The preferred technique and de facto German standard for pancreatoduodenectomy was pylorus-preserving pancreatoduodenectomy with pancreatojejunostomy carried out via duct-to-mucosa anastomosis with interrupted sutures using PDS 4.0. The minority of German pancreas centers were certified (18–48 %). The certification rate increased with higher capacity levels and case load (P < 0.05); however, significant correlations between the fistula rate and hospital case load, hospital capacity level, or hospital certification status were not seen.
Conclusion
This study revealed a distinct variety of management strategies for pancreatic surgery and available evidence-based data was not necessarily translated into clinical practice. The limited certification rate represented a shortcoming of quality assurance. The data emphasize the need for further trials to answer the questions whether hospital certifications and omission of drains improve outcome after pancreatoduodenectomy and for the establishment of guidelines for pancreatoduodenectomy.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pancreatoduodenectomy (PD) for tumors of the pancreatic head is performed in many German hospitals varying considerably in size and capacity. A diversity of operative approaches and surgical materials exist for the procedure while no clear gold standard has yet been defined. The same is true for the non-surgical aspects of patient care in PD, as there are no general guidelines for intraoperative and perioperative management. This is especially remarkable in light of the procedure’s significant complications and persisting mortality rates even in specialized high-volume centers.
The pylorus-preserving pancreatoduodenectomy by Traverso–Longmire (PPPD) and the classic (Kausch–)Whipple pancreatoduodenectomy (CWPD) are the usual techniques for the management of pancreatic head tumors with PPPD being preferred by the majority of surgeons [1, 2]. For either operation, the two common techniques are pancreatogastrostomy (PG) and pancreatojejunostomy (PJ). There is no consensus as to which is the superior method; previous meta-analyses did not show significant benefits for one method over the other while a retrospective study did report a lower incidence of intra-abdominal complications for pancreatogastrostomy vs. pancreatojejunostomy [3–5]. In that study, however, no differences were found for the rates of pancreatic and biliary fistulas, rate of delayed gastric emptying, and mortality. In addition to the general differences between reconstruction approaches, each method for itself can be performed in different fashions as several different suture techniques and materials exist. Evidence-based data regarding which combination of methods promises the best results are not available. Thus, the absence of clearly defined guidelines becomes comprehensible as the literature does not allow for distinct conclusions as to which reconstruction method should be favored and in which form it should be performed.
In addition to the variations of the surgical procedures, perioperative management also offers multiple approaches. The time point of return to solid food, amount and type of infusions, and time point of drain removal are some of the variables that contribute to the considerable differences in institution-specific protocols. Next to these standard care approaches, fast track programs have become more and more common and several studies report favorable results for patients undergoing major surgery, including pancreatic surgery [6–8]. At present, however, neither the literature nor clinical practices divulge a standard for perioperative management during pancreatoduodenectomy.
Pancreatic fistulas represent a notable complication after pancreatoduodenectomy. Two powerful study groups, the International Study Group on Pancreatic Fistula and the International Study Group of Pancreatic Surgery, have provided the necessary framework for this process by publishing a clear definition for pancreatic fistulas and a system for recording and reporting clinical data during pancreatoenterostomy [9, 10]. Nonetheless, inconsistent assessment of postoperative pancreatic fistulas prevail due to lacking or varying standards in surgical departments, such as missing or inconsistent measurement of quantity and quality of drain fluids.
Clinical experience is an important outcome factor in pancreatic surgery and recent studies correlate reduced morbidity and mortality to higher caseloads [11–15]. Many hospitals strive to improve patient care and prove their qualification by obtaining certifications. In times of rising financial awareness, certifications also take on a role as patient acquisition and case load increasing tools. Attesting a variety of medical standards often summarized as pathways, certifications allow for control of own operational sequences [16]. Over the past years, an abundance of certifications has sprung into existence in Germany and in times of growing competition, obtaining such certifications has become vital. Certifications in the field of pancreatic cancer are especially sensible, as the corresponding surgery is challenging and fraught with risk.
Despite an existing guideline for the treatment of pancreatic cancer in Germany [17], there exist no standards for the many operative and perioperative aspects of pancreatoduodenectomy. While this is the case, an important tool of quality assurance is already in place and is likely to have already improved treatment quality: the certification process for pancreatic centers. We developed a questionnaire to determine which treatment algorithms for pancreatoduodenectomy are currently employed in Germany and are the de facto standard for the procedure. Further, we examine the role of certifications on patient treatment. We believe this information will be both interesting and useful as it may be used to further improve the quality of pancreatic surgery.
Methods
Questionnaire
A questionnaire was developed consisting of eight main question blocks with a combined total of 38 parameters to evaluate the general aspects of the hospitals and their strategy for carrying out pancreatoduodenectomy (Fig. 1). The questionnaire was created using the present literature and our own clinical experience [10, 18–20].
Contacted hospitals
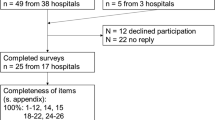
An online database (white list/www.weisse-liste.de) was used to identify German hospitals that were subsequently contacted and sent the questionnaire. Search items were as follows: OPS-Code 5–52 (pancreatic operations), OPS-Code 5–524 (partial resection of the pancreas), and OPS-Code 5–525 (total pancreatectomy). Of the hospitals identified by these terms, 211 reported a case load of >12 pancreatoduodenectomies per year, one of the requirements for certification as a pancreas center. These hospitals were sent the questionnaire in December 2011. The questionnaires were addressed to the head of the surgical department asking for objectively recorded figures. After 8 weeks, an identical questionnaire was sent to all hospitals that did not reply. The contacted hospitals were comprised of 18 % (38/211) university hospitals, 22 % (46/211) maximum care hospitals, 39 % (83/211) tertiary hospitals, and 21 % (44/211) basic care hospitals.
Statistics
Data analysis was performed using SPSS Statistics 20 (IBM). The Fisher exact test was utilized for contingency tables. The Mann–Whitney U test and Spearman Test were used for non-parametric testing. Incompletely or faultily filled-out questionnaires were excluded from analysis for the unanswered questions. All tests were two-sided and considered significant at P < 0.05.
Results
Response rate
The response rate after the first mailing was 77.3 % (163/211) which increased to 86.3 % (182/211) after the second mailing. The composition of contacted and responding hospitals was similar; most non-responding hospitals were basic care providers (Fig. 2).
Questionnaire results
The following paragraphs provide summaries of the collected data with the corresponding details being presented in tables.
Pathways, certification, and case load
While most centers had implemented a pathway for pancreatoduodenectomy, only a minority were certified pancreas centers (29 %) (Table 1). The rate of certification increased with higher capacity levels: 20 % of basic care, 18 % of tertiary care, 37 % of maximum care providers, and 48 % of university hospitals held pancreas center certifications. Further, the number of pancreatoduodenectomies per year also increased with hospital capacity level and was not evenly distributed (P < 0.05, Fig. 3). Case load was correlated to certification status: certified hospitals had significantly higher caseloads than non-certified hospitals (P < 0.01).
Operative approach
PPPD was the favored standard approach for pancreatoduodenectomy over CWPD, while 69 % reported the occasional use of CWPD. Pancreatojejunostomy with a duct-to-mucosa anastomosis was the preferred reconstruction technique, mostly carried out as a single button suture using PDS 4.0. Absorbable threads were favored by the vast majority. Use of a pancreatic stent was not common; those that were employed were equally divided into internally and externally draining ones. Fibrin glue was used by less than 10 % of hospitals routinely. Easy flow drains were the most commonly employed abdominal drains (Table 2).
Postoperative management
Monitoring of pancreas enzyme levels in drain fluids was carried out in the majority of hospitals, as well as the insertion of a gastric tube. Eighty-one percent of the hospitals returned their patients to solid food during the first three postoperative days. Use of somatostatin (octreotide) differed considerably. The time point of drain removal was only standardized in 55 % of the hospitals. Including hospitals with non-standardized removal plans, intra-abdominal drains were mostly removed between POD (postoperative day) 3 and 5. A standard postoperative CT scan was highly uncommon with only one hospital reporting this practice (Table 3).
Complications—rate of postoperative pancreatic fistula
The majority of hospitals reported low fistula rates: 68 % of hospitals reported their grade A fistula rate to be below 10 %, 95 % reported grade B fistula rates of below 10 and 87.6 reported grade C fistula rates of below 5 %. There were no significant correlations between the fistula rate and hospital case load, hospital capacity level, or hospital certification status (Table 4).
Discussion
There exists no gold standard for the surgical and perioperative management of pancreatic head resections. The current literature describes several procedures and management strategies that are reported to improve patient outcome but there is no consensus for a specific pathway. Because of these uncertainties in surgical treatment and the lack of standardization, we performed a national survey to evaluate the currently favored treatment approaches in order to (1) disseminate knowledge of the de facto standard, (2) assess quality assurance measures in pancreatic surgery, and (3) identify relevant issues for further investigation.
In Germany, in 2011 and 2012, research has been conducted on surgical treatment concepts [19] and the correlation of case load to outcome in pancreatic surgery [20]. Alsfasser et al. [20] have demonstrated an overall mortality in all German hospitals between 2.58 and 3.98 % after pancreatic surgery and showed a decreased mortality with increasing volume. Our survey was intentionally kept short and simple in order to achieve a high response rate and obtain representative results. Previously investigated aspects [19, 20] have only partially been re-visited in our questionnaire. In retrospect, the missing mortality data has been considered as a disadvantage of this study. However, the high response rate of 86 % in our study reflects the ongoing interest of German surgeons regarding pancreatic surgery and the topicality of the subject. The presented data supply an accurate picture of the current surgical routine and reveal correlations.
The preferred technique and de facto German standard for pancreatoduodenectomy was PPPD with pancreatojejunostomy carried out via duct-to-mucosa anastomosis with interrupted sutures using PDS 4.0. PPPD being favored corresponds to other studies reporting that PPPD has prevailed over CWPD in German hospitals [1, 21]. Interestingly, there is no clear consensus in the literature to warrant this strong preference for PPPD; some studies have stated PPPD to be the faster procedure with less blood loss [1, 21], while a recent review and meta-analysis conclude that there is no evidence of relevant differences in mortality, morbidity, and survival between PPPD and CWPD [22, 23].
A similar debate is ongoing concerning the most suitable reconstruction technique after pancreatoduodenectomy; a significantly higher rate of postoperative pancreatic fistula (POPF) and increased mortality has been reported for PJ vs. PG while a prospective randomized controlled trial comparing the two methods did not detect significant differences [5, 24, 25]. The latter findings have been confirmed by Wellner et al. who stated similar complication rates in a comparison of both techniques [26]. Then again, a meta-analysis reported PG to be superior to PJ due to lower overall morbidity (but with similar results regarding POPF and mortality) [5]. Thus, there can presently be no certainty as to which is the more suitable technique—yet German surgeons intensely preferred pancreatojejunostomy over pancreatogastrostomy. Further light will be shed on this issue by the RECOnstruction after partial PANCreatoduodenectomy (RECOPANC) trial, a prospective randomized controlled multicenter trial comparing PG and PJ [27]. RECOPANC involves 14 academic centers and is planned to conclude in 2016.
In addition to the above, other technical options that a surgeon faces during pancreatoduodenectomy must also be navigated without the help of guidelines for the same reason of insufficient data or no consensus in the literature. This applies to the questions of duct to mucosa vs. invagination [28–31], whether to use trans-anastomotic pancreatic stents [32–35] and whether to administer somatostatin postoperatively [36–40].
Further, evidence-based data concerning continuous vs. interrupted suture techniques is scarce. In the present study, surgeons predominantly performed interrupted sutures, while a retrospective study [41] for duct-to-mucosa anastomoses reported reduced POPF rates using continuous sutures compared to an interrupted technique.
Similarly, the removal of intra-abdominal drains was conducted predominantly after POD 3 and was only standardized in 55 % of hospitals. At present, drain management after pancreatic surgery is a topic of intense interest. The literature provides data that intra-abdominal drains can be safely removed on POD 3 after standard pancreatoduodenectomy and that indeed prolonged drain insertion increases the risk of postoperative complications [42, 43]. Most recently, a single center study showed that the use of drains during pancreatic surgery was associated with longer hospital stay, increased morbidity, increased fistula- and readmission rates and did not alter the reintervention- or mortality rates. The authors concluded that routine prophylactic drainage after pancreatic resection could be safely abandoned [44]. The present study has shown that the available data is not inevitably transferred to daily clinical practice. On the other hand, the available data is insufficient and obviously does not convince the majority of surgeons to change draining habits. This matter will only be likely resolved by randomized controlled multicenter studies assessing these issues.
Postoperative pancreatic fistulas still represent the main postoperative complication after PD. The present data did not reveal significant correlations between POPF and hospital caseload, hospital capacity level, or hospital certification status. However, these results should be treated with caution: it is remarkable that rates above 10 % of clinically relevant fistulas (grade B + C) were only reported by a combined total of 7.1 % of the responding hospitals. The majority of the current literature reports POPF rates between 5 and 15 %, most of these data stemming from specialized centers and experienced surgeons with above-average outcomes. An explanation for the low POPF rates reported in the survey is that the POPF rate may not always represent (the requested) objectively documented current clinical data but be subjectively influenced estimates of the reporting surgeons.
Several studies have discussed the correlations of a surgeon’s experience, hospital volume, and patient outcome. In 2002, Birkmeyer et al. reported a significant correlation of increased hospital volume to reduced mortality [12]. One year later, their group emphasized the role of the surgeon, concluding that even within high-volume hospitals, the more frequently operating surgeons have better results [45]. Nowadays, it is accepted that high-volume surgeons in high-volume centers produce the best outcomes [13–15, 46, 47]. Surprisingly, data of our survey did not show significant correlations between fistula rate and hospital case load and hospital capacity level or hospital certification status. A weakness of this study is the missing question/data of the surgeons’ caseloads that would have completely assessed the known volume–outcome relations.
In Germany, two organizations award pancreas center certifications: The Deutsche Gesellschaft für Allgemein- und Viszeralchirurgie (DGAV; German General and Visceral Surgery Association) and the Deutsche Krebsgesellschaft (DKG; German Cancer Association) (www.dgav.de, www.krebsgesellschaft.de). The general requirements are a minimum of pancreatic cancer cases and pancreatic resections per year, defined morbidity and mortality rates, and implementation of pathways incorporating structured and interdisciplinary therapies along with an interdisciplinary tumor board. However, pancreatoduodenectomy is performed in German hospitals of all capacity levels, ranging from university hospitals to basic care hospitals, with caseloads from 1–300 pancreatoduodenectomies per year. In the present study, the caseload reported varied considerably from ∼12 PD per year to well over 100 PD annually. In addition, high-volume centers displayed a significantly higher certification rate with those centers performing >100 PD per year, all being certified. In summary, the present data show a positive correlation between caseload, capacity level, and certification/pathway rate. The certification of hospitals by accredited expert panels helps to optimize internal processes and reduces clinical vulnerabilities and subsequently morbidity and mortality. The process of certification requires the development of pathways and clinical pathways are established to be associated with improved documentation, reduced in-hospital mortality, reduced in-hospital complications, reduced length of hospital stay, and reduced hospital costs [48–50].
Conclusion
Despite existing studies and clinical recommendations for pancreatic surgery, the present study revealed a huge variety of perioperative and operative treatments due to the absence of strict guidelines. Only a minority of German pancreatic centers were officially certified, representing a shortcoming of quality assurance. The data emphasize the need for further trials to answer the questions whether (a) hospital certifications, (b) omission of drains after pancreatoduodenectomy, and (c) continuous sutures for the anastomosis improved the surgical outcome of patients after pancreatoduodenectomy. These results may be translated into guidelines for the management of pancreatoduodenectomy—guidelines that must then be stringently applied to improve the outcome after pancreatoduodenectomy
Abbreviations
- CWPD:
-
Classic (Kausch-)Whipple pancreatoduodenectomy
- DKG:
-
Deutsche Krebsgesellschaft, German Cancer Association
- DGAV:
-
Deutsche Gesellschaft für Allgemein- und Viszeralchirurgie, German Association for General and Visceral Surgery
- PD:
-
Pancreatoduodenectomy
- PG:
-
Pancreatogastrostomy
- PJ:
-
Pancreatojejunostomy
- POD:
-
Postoperative day
- POPF:
-
Postoperative pancreatic fistula
- PPPD:
-
Pylorus-preserving pancreatoduodenectomy by Traverso–Longmire
References
Glanemann M, Bahra M, Neuhaus P (2008) Pylorus-preserving pancreatic head resection: a new standard for tumors. Chirurg 79(12):1107–1114
Smigielski J, Piskorz L, Kutwin L, Brocki M (2012) Comparison of early results of surgical treatment in patients with pancreatic cancer. Pol Przegl Chir 84(1):1–5
McKay A, Mackenzie S, Sutherland FR, Bathe OF, Doig C, Dort J, Vollmer CM Jr, Dixon E (2006) Meta-analysis of pancreaticojejunostomy versus pancreaticogastrostomy reconstruction after pancreaticoduodenectomy. Br J Surg 93(8):929–936
Wente MN, Shrikhande SV, Muller MW, Diener MK, Seiler CM, Friess H, Buchler MW (2007) Pancreaticojejunostomy versus pancreaticogastrostomy: systematic review and meta-analysis. Am J Surg 193(2):171–183
Schlitt HJ, Schmidt U, Simunec D, Jager M, Aselmann H, Neipp M, Piso P (2002) Morbidity and mortality associated with pancreatogastrostomy and pancreatojejunostomy following partial pancreatoduodenectomy. Br J Surg 89(10):1245–1251
di Sebastiano P, Festa L, De Bonis A, Ciuffreda A, Valvano MR, Andriulli A, di Mola FF (2011) A modified fast-track program for pancreatic surgery: a prospective single-center experience. Langenbecks Arch Surg 396(3):345–351
van Bree SH, Vlug MS, Bemelman WA, Hollmann MW, Ubbink DT, Zwinderman AH, de Jonge WJ, Snoek SA, Bolhuis K, van der Zanden E et al (2011) Faster recovery of gastrointestinal transit after laparoscopy and fast-track care in patients undergoing colonic surgery. Gastroenterology 141(3):872–880, e871-874
Hall TC, Dennison AR, Bilku DK, Metcalfe MS, Garcea G (2012) Enhanced recovery programmes in hepatobiliary and pancreatic surgery: a systematic review. Ann R Coll Surg Engl 94(5):318–326
Bassi C, Dervenis C, Butturini G, Fingerhut A, Yeo C, Izbicki J, Neoptolemos J, Sarr M, Traverso W, Buchler M (2005) Postoperative pancreatic fistula: an international study group (ISGPF) definition. Surgery 138(1):8–13
Shukla PJ, Barreto SG, Fingerhut A, Bassi C, Buchler MW, Dervenis C, Gouma D, Izbicki JR, Neoptolemos J, Padbury R et al (2010) Toward improving uniformity and standardization in the reporting of pancreatic anastomoses: a new classification system by the International Study Group of Pancreatic Surgery (ISGPS). Surgery 147(1):144–153
Balzano G, Zerbi A, Capretti G, Rocchetti S, Capitanio V, Di Carlo V (2008) Effect of hospital volume on outcome of pancreaticoduodenectomy in Italy. Br J Surg 95(3):357–362
Birkmeyer JD, Siewers AE, Finlayson EV, Stukel TA, Lucas FL, Batista I, Welch HG, Wennberg DE (2002) Hospital volume and surgical mortality in the United States. N Engl J Med 346(15):1128–1137
de Wilde RF, Besselink MG, van der Tweel I, de Hingh IH, van Eijck CH, Dejong CH, Porte RJ, Gouma DJ, Busch OR, Molenaar IQ (2012) Impact of nationwide centralization of pancreaticoduodenectomy on hospital mortality. Br J Surg 99(3):404–410
Topal B, Van de Sande S, Fieuws S, Penninckx F (2007) Effect of centralization of pancreaticoduodenectomy on nationwide hospital mortality and length of stay. Br J Surg 94(11):1377–1381
van Heek NT, Kuhlmann KF, Scholten RJ, de Castro SM, Busch OR, van Gulik TM, Obertop H, Gouma DJ (2005) Hospital volume and mortality after pancreatic resection: a systematic review and an evaluation of intervention in the Netherlands. Ann Surg 242(6):781–788, discussion 788–790
Post S (2010) Quality management—certification: curse or blessing? Zentralbl Chir 135(1):1–2
Adler TS G, Bischoff SC, Brambs H-J, Feuerbach S, Grabenbauer G, Hahn S, Heinemann V, Hohenberger W, Langrehr JM, Lutz MP, Micke O, Neuhaus H, Neuhaus P, Oettle H, Schlag PM, Schmid R, Schmiegel W, Schlottmann K, Werner J, Wiedenmann B, Kopp I (2007) S3-Leitlinie, Exokrines Pankreaskarzinom 2007. Z Gastroenterol 45(6):487–523
Shen HN, Lu CL (2011) Incidence, resource use, and outcome of acute pancreatitis with/without intensive care: a nationwide population-based study in Taiwan. Pancreas 40(1):10–15
Sargent M, Boeck S, Heinemann V, Jauch KW, Seufferlein T, Bruns CJ (2011) Surgical treatment concepts for patients with pancreatic cancer in Germany—results from a national survey conducted among members of the "Chirurgische Arbeitsgemeinschaft Onkologie" (CAO) and the "Arbeitsgemeinschaft Internistische Onkologie" (AIO) of the Germany Cancer Society (DKG). Langenbecks Arch Surg 396(2):223–229
Alsfasser G, Kittner J, Eisold S, Klar E (2012) Volume-outcome relationship in pancreatic surgery: the situation in Germany. Surgery 152(3 Suppl 1):S50–S55
Karanicolas PJ, Davies E, Kunz R, Briel M, Koka HP, Payne DM, Smith SE, Hsu HP, Lin PW, Bloechle C et al (2007) The pylorus: take it or leave it? Systematic review and meta-analysis of pylorus-preserving versus standard Whipple pancreaticoduodenectomy for pancreatic or periampullary cancer. Ann Surg Oncol 14(6):1825–1834
Diener MK, Heukaufer C, Schwarzer G, Seiler CM, Antes G, Buchler MW, Knaebel HP (2008) Pancreaticoduodenectomy (classic Whipple) versus pylorus-preserving pancreaticoduodenectomy (pp Whipple) for surgical treatment of periampullary and pancreatic carcinoma. Cochrane Database Syst Rev 2, CD006053
Diener MK, Fitzmaurice C, Schwarzer G, Seiler CM, Antes G, Knaebel HP, Buchler MW (2011) Pylorus-preserving pancreaticoduodenectomy (pp Whipple) versus pancreaticoduodenectomy (classic Whipple) for surgical treatment of periampullary and pancreatic carcinoma. Cochrane Database Syst Rev 5, CD006053
Oussoultzoglou E, Bachellier P, Bigourdan JM, Weber JC, Nakano H, Jaeck D (2004) Pancreaticogastrostomy decreased relaparotomy caused by pancreatic fistula after pancreaticoduodenectomy compared with pancreaticojejunostomy. Arch Surg 139(3):327–335
Yeo CJ, Cameron JL, Maher MM, Sauter PK, Zahurak ML, Talamini MA, Lillemoe KD, Pitt HA (1995) A prospective randomized trial of pancreaticogastrostomy versus pancreaticojejunostomy after pancreaticoduodenectomy. Ann Surg 222(4):580–588, discussion 588–592
Wellner UF, Sick O, Olschewski M, Adam U, Hopt UT, Keck T (2012) Randomized controlled single-center trial comparing pancreatogastrostomy versus pancreaticojejunostomy after partial pancreatoduodenectomy. J Gastrointest Surg 16(9):1686–1695
Wellner UF, Brett S, Bruckner T, Limprecht R, Rossion I, Seiler C, Sick O, Wegener I, Hopt UT, Keck T (2012) Pancreatogastrostomy versus pancreatojejunostomy for RECOnstruction after partial PANCreatoduodenectomy (RECOPANC): study protocol of a randomized controlled trial UTN U1111-1117-9588. Trials 13:45
Greene BS, Loubeau JM, Peoples JB, Elliott DW (1991) Are pancreatoenteric anastomoses improved by duct-to-mucosa sutures? Am J Surg 161(1):45–49, discussion 49–50
Matsumoto Y, Fujii H, Miura K, Inoue S, Sekikawa T, Aoyama H, Ohnishi N, Sakai K, Suda K (1992) Successful pancreatojejunal anastomosis for pancreatoduodenectomy. Surg Gynecol Obstet 175(6):555–562
Berger AC, Howard TJ, Kennedy EP, Sauter PK, Bower-Cherry M, Dutkevitch S, Hyslop T, Schmidt CM, Rosato EL, Lavu H et al (2009) Does type of pancreaticojejunostomy after pancreaticoduodenectomy decrease rate of pancreatic fistula? A randomized, prospective, dual-institution trial. J Am Coll Surg 208(5):738–747, discussion 747–739
Kim JH, Yoo BM, Kim WH (2009) Which method should we select for pancreatic anastomosis after pancreaticoduodenectomy? World J Surg 33(2):326–332
Pessaux P, Sauvanet A, Mariette C, Paye F, Muscari F, Cunha AS, Sastre B, Arnaud JP (2011) External pancreatic duct stent decreases pancreatic fistula rate after pancreaticoduodenectomy: prospective multicenter randomized trial. Ann Surg 253(5):879–885
Zhou Y, Yang C, Wang S, Chen J, Li B (2011) Does external pancreatic duct stent decrease pancreatic fistula rate after pancreatic resection?: a meta-analysis. Pancreatology 11(3):362–370
Frozanpor F, Lundell L, Segersvard R, Arnelo U (2012) The effect of prophylactic transpapillary pancreatic stent insertion on clinically significant leak rate following distal pancreatectomy: results of a prospective controlled clinical trial. Ann Surg 255(6):1032–1036
Xiong JJ, Altaf K, Mukherjee R, Huang W, Hu WM, Li A, Ke NW, Liu XB (2012) Systematic review and meta-analysis of outcomes after intraoperative pancreatic duct stent placement during pancreaticoduodenectomy. Br J Surg 99(8):1050–1061
Gans SL, van Westreenen HL, Kiewiet JJ, Rauws EA, Gouma DJ, Boermeester MA (2012) Systematic review and meta-analysis of somatostatin analogues for the treatment of pancreatic fistula. Br J Surg 99(6):754–760
Kollmar O, Moussavian MR, Richter S, de Roi P, Maurer CA, Schilling MK (2008) Prophylactic octreotide and delayed gastric emptying after pancreaticoduodenectomy: results of a prospective randomized double-blinded placebo-controlled trial. Eur J Surg Oncol 34(8):868–875
Alghamdi AA, Jawas AM, Hart RS (2007) Use of octreotide for the prevention of pancreatic fistula after elective pancreatic surgery: a systematic review and meta-analysis. Can J Surg 50(6):459–466
Graham JA, Johnson LB, Haddad N, Al-Kawas F, Carroll J, Jha R, Wong J, Maglaris D, Mertens S, Fishbein T (2011) A prospective study of prophylactic long-acting octreotide in high-risk patients undergoing pancreaticoduodenectomy. Am J Surg 201(4):481–485
Vanounou T, Pratt WB, Callery MP, Vollmer CM Jr (2007) Selective administration of prophylactic octreotide during pancreaticoduodenectomy: a clinical and cost-benefit analysis in low- and high-risk glands. J Am Coll Surg 205(4):546–557
Tsuji M, Kimura H, Konishi K, Yabushita K, Maeda K, Kuroda Y (1998) Management of continuous anastomosis of pancreatic duct and jejunal mucosa after pancreaticoduodenectomy: historical study of 300 patients. Surgery 123(6):617–621
Bassi C, Molinari E, Malleo G, Crippa S, Butturini G, Salvia R, Talamini G, Pederzoli P (2010) Early versus late drain removal after standard pancreatic resections: results of a prospective randomized trial. Ann Surg 252(2):207–214
Kawai M, Tani M, Terasawa H, Ina S, Hirono S, Nishioka R, Miyazawa M, Uchiyama K, Yamaue H (2006) Early removal of prophylactic drains reduces the risk of intra-abdominal infections in patients with pancreatic head resection: prospective study for 104 consecutive patients. Ann Surg 244(1):1–7
Correa-Gallego C, Brennan MF, D’Angelica M, Fong Y, Dematteo RP, Kingham TP, Jarnagin WR, Allen PJ (2013) Operative drainage following pancreatic resection: analysis of 1122 patients resected over 5 years at a single institution. Ann Surg. In press.
Birkmeyer JD, Stukel TA, Siewers AE, Goodney PP, Wennberg DE, Lucas FL (2003) Surgeon volume and operative mortality in the United States. N Engl J Med 349(22):2117–2127
Kim CG, Jo S, Kim JS (2012) Impact of surgical volume on nationwide hospital mortality after pancreaticoduodenectomy. World J Gastroenterol 18(31):4175–4181
Parks RW, Bettschart V, Frame S, Stockton DL, Brewster DH, Garden OJ (2004) Benefits of specialisation in the management of pancreatic cancer: results of a Scottish population-based study. Br J Cancer 91(3):459–465
Rotter T, Kinsman L, James E, Machotta A, Gothe H, Willis J, Snow P, Kugler J (2010) Clinical pathways: effects on professional practice, patient outcomes, length of stay and hospital costs. Cochrane Database Syst Rev 3, CD006632
Muller MK, Dedes KJ, Dindo D, Steiner S, Hahnloser D, Clavien PA (2009) Impact of clinical pathways in surgery. Langenbecks Arch Surg 394(1):31–39
Munitiz V, Martinez-de-Haro LF, Ortiz A, Ruiz-de-Angulo D, Pastor P, Parrilla P (2010) Effectiveness of a written clinical pathway for enhanced recovery after transthoracic (Ivor Lewis) oesophagectomy. Br J Surg 97(5):714–718
Acknowledgments
The authors thank Mrs. Ulrike Kein for her outstanding work in the mailing and documentation of the questionnaire. The project was funded by an unrestricted grant of the Foerderverein Peter Geiger, Beilstein, Germany.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Christina Haane, Wolf Arif Mardin, and Christina Schleicher contributed equally to this study.
Rights and permissions
About this article
Cite this article
Haane, C., Mardin, W.A., Schmitz, B. et al. Pancreatoduodenectomy—current status of surgical and perioperative techniques in Germany. Langenbecks Arch Surg 398, 1097–1105 (2013). https://doi.org/10.1007/s00423-013-1130-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-013-1130-1