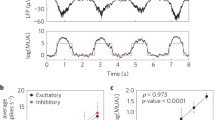
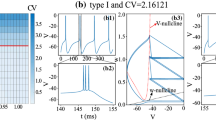
Abstract
Rhythmic bursting is the most striking behavior of cultured cortical networks and may start in the second week after plating. In this study, we focus on the intervals between spontaneously occurring bursts, and compare experimentally recorded values with model simulations. In the models, we use standard neurons and synapses, with physiologically plausible parameters taken from literature. All networks had a random recurrent architecture with sparsely connected neurons. The number of neurons varied between 500 and 5,000. We find that network models with homogeneous synaptic strengths produce asynchronous spiking or stable regular bursts. The latter, however, are in a range not seen in recordings. By increasing the synaptic strength in a (randomly chosen) subset of neurons, our simulations show interburst intervals (IBIs) that agree better with in vitro experiments. In this regime, called weakly synchronized, the models produce irregular network bursts, which are initiated by neurons with relatively stronger synapses. In some noise-driven networks, a subthreshold, deterministic, input is applied to neurons with strong synapses, to mimic pacemaker network drive. We show that models with such “intrinsically active neurons” (pacemaker-driven models) tend to generate IBIs that are determined by the frequency of the fastest pacemaker and do not resemble experimental data. Alternatively, noise-driven models yield realistic IBIs. Generally, we found that large-scale noise-driven neuronal network models required synaptic strengths with a bimodal distribution to reproduce the experimentally observed IBI range. Our results imply that the results obtained from small network models cannot simply be extrapolated to models of more realistic size. Synaptic strengths in large-scale neuronal network simulations need readjustment to a bimodal distribution, whereas small networks do not require such changes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Amit DJ, Brunel N (1997) Dynamics of a recurrent network of spiking neurons before and following learning. Netw Comput Neural Syst 8: 373–404
Baker RE, Corner MA, Pelt J (2006) Spontaneous neuronal discharge patterns in developing organotypic mega-co-cultures of neonatal rat cerebral cortex. Brain Res 1101: 29–35
Bi GQ, Poo MM (1998) Synaptic modifications in cultured hippocampal neurons: dependence on spike timing, synaptic strength, and postsynaptic cell type. J Neurosci 18: 10464–10472
Coles S (2001) An introduction to statistical modelling of extreme values. Springer-Verlag, London
Destexhe A, Badoual M, Piwkowska Z, Bal T, Rudolph M (2004) A novel method for characterizing synaptic noise in cortical neurons. Neurocomputing 58–60: 191–196
Eckmann JP, Jacobi S, Marom S, Moses E, Zbinden C (2008) Leader neurons in population bursts of 2D living neural networks. New J Phys 10: 015011
Eytan D, Marom S (2006) Dynamics and effective topology underlying synchronization in networks of cortical neurons. J Neurosci 26: 8465–8476
French DA, Gruenstein EI (2006) An integrate-and-fire model for synchronized bursting in a network of cultured cortical neurons. J Comput Neurosci 21: 227–241
Gibson JR, Connors BW (2003) Chemical and electrical synapses in neocortex. In: Arbib MA (ed) Handbook of brain theory and neural networks. 2 edn. MIT Press, Cambridge, pp 725–729
Giugliano M, Darbon P, Arsiero M, Lüscher HR, Streit J (2004) Single-neuron discharge properties and network activity in dissociated cultures of neocortex. J Neurophysiol 92: 977–996
Gritsun TA, le Feber J, Stegenga J, Rutten WLC (2010) Network bursts in cortical cultures are best simulated using pacemaker neurons and adaptive synapses. Biol Cybern 102: 1–18
Hansel D, Sompolinsky H (1996) Chaos and synchrony in a model of a hypercolumn in visual cortex. J Comput Neurosci 3: 7–34
Hubbard JI, Stenhouse D, Eccles RM (1967) Origin of synaptic noise. Science 157: 330–331
Ichikawa M, Muramoto K, Kobayashi K, Kawahara M, Kuroda Y (1993) Formation and maturation of synapses in primary cultures of rat cerebral cortical cells: An electron microscopic study. Neurosci Res 16: 95–103
Izhikevich EM (2003) Simple model of spiking neurons. IEEE Trans Neural Netw 14: 1569–1572
Kitano K, Fukai T (2007) Variability v.s. synchronicity of neuronal activity in local cortical network models with different wiring topologies. J Comput Neurosci 23: 237–250
Latham PE, Richmond BJ, Nirenberg S, Nelson PG (2000) Intrinsic dynamics in neuronal networks. II. Experiment. J Neurophysiol 83: 828–835
le Feber J, Van Pelt J, Rutten WLC (2009) Latency-related development of functional connections in cultured cortical networks. Biophys J 96: 3443–3450
le Feber J, Rutten WLC, Stegenga J, Wolters PS, Ramakers GJA, Pelt J (2007) Conditional firing probabilities in cultured neuronal networks: a stable underlying structure in widely varying spontaneous activity patterns. J Neural Eng 4: 54–67
Maeda E, Robinson HPC, Kawana A (1995) The mechanisms of generation and propagation of synchronized bursting in developing networks of cortical neurons. J Neurosci 15: 6834–6845
Mainen ZF, Sejnowski TJ (1995) Reliability of spike timing in neocortical neurons. Science 268: 1503–1506
Markram H, Wang Y, Tsodyks M (1998) Differential signaling via the same axon of neocortical pyramidal neurons. Proc Natl Acad Sci USA 95: 5323–5328
Muller TH, Swandulla D, Zeilhofer HU (1997) Synaptic connectivity in cultured hypothalamic neuronal networks. J Neurophysiol 77: 3218–3225
Nesse WH, Borisyuk A, Bressloff PC (2008) Fluctuation-driven rhythmogenesis in an excitatory neuronal network with slow adaptation. J Comput Neurosci 25: 317–333
Rodriguez-Molina VM, Aertsen A, Heck DH (2007) Spike timing and reliability in cortical pyramidal neurons: effects of EPSC kinetics, input synchronization and background noise on spike timing. PLoS One 2: e319
Romijn HJ, Van Huizen F, Wolters PS (1984) Towards an improved serum-free, chemically defined medium for long-term culturing of cerebral cortex tissue. Neurosci Biobehav Rev 8: 301–334
Scott DW (1979) On optimal and data-based histograms. Biometrika 66: 605–610
Segev R, Shapira Y, Benveniste M, Ben-Jacob E (2001) Observations and modeling of synchronized bursting in two-dimensional neural networks. Phys Rev 64: 011920
Segev R, Benveniste M, Hulata E, Cohen N, Palevski A, Kapon E, Shapira Y, Ben-Jacob E (2002) Long term behavior of lithographically prepared in vitro neuronal networks. Phys Rev Lett 88: 1181021–1181024
Song S, Miller KD, Abbott LF (2000) Competitive Hebbian learning through spike-timing-dependent synaptic plasticity. Nat Neurosci 3: 919–926
Stegenga J, le Feber J, Marani E, Rutten WLC (2008) Analysis of cultured neuronal networks using intra-burst firing characteristics. IEEE Trans Biomed Eng 55(4): 1382–1390
Stevens CF, Zador AM (1998) Input synchrony and the irregular firing of cortical neurons. Nat Neurosci 1: 210–217
Strata F, Atzori M, Molnar M, Ugolini G, Tempia F, Cherubini E (1997) A pacemaker current in dye-coupled hilar interneurons contributes to the generation of giant GABAergic potentials in developing hippocampus. J Neurosci 17: 1435–1446
Teich MC, Saleh BEA, Perina J (1984) Role of primary excitation statistics in the generation of antibunched and sub-poisson light. J Opt Soc Am B 1: 366–389
Toledo-Rodriguez M, Gupta A, Wang Y, Wu CZ, Markram H (2003) Neocortex: basic neuron types. In: Arbib MA (ed) Handbook of brain theory and neural networks. 2 edn. MIT Press, Cambridge, pp 719–725
Tsodyks M, Pawelzik K, Markram H (1998) Neural networks with dynamic synapses. Neural Comput 10: 821–835
Tsodyks M, Uziel A, Markram H (2000) Synchrony generation in recurrent networks with frequency-dependent synapses. J Neurosci 20: RC50
Van Pelt J, Wolters PS, Corner MA, Rutten WLC, Ramakers GJA (2004) Long-term characterization of firing dynamics of spontaneous bursts in cultured neural networks. IEEE Trans Biomed Eng 51: 2051–2062
Vladimirski BB, Tabak J, O’Donovan MJ, Rinzel J (2008) Episodic activity in a heterogeneous excitatory network, from spiking neurons to mean field. J Comput Neurosci 25: 39–63
Wagenaar DA, Pine J, Potter SM (2006) An extremely rich repertoire of bursting patterns during the development of cortical cultures. BMC Neurosci 7:11
Wiedemann UA, Luthi A (2003) Timing of network synchronization by refractory mechanisms. J Neurophysiol 90: 3902–3911
Zhang LI, Tao HW, Holt CE, Harris WA, Poo MM (1998) A critical window for cooperation and competition among developing retinotectal synapses. Nature 395: 37–44
Acknowledgements
The work presented in this article was funded by the EU Marie-Curie NEURoVERS-it project (MRTN-CT-2005-019247).
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Gritsun, T., le Feber, J., Stegenga, J. et al. Experimental analysis and computational modeling of interburst intervals in spontaneous activity of cortical neuronal culture. Biol Cybern 105, 197–210 (2011). https://doi.org/10.1007/s00422-011-0457-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00422-011-0457-3