Abstract
Purpose
We compared the modulation of force steadiness by different types of electrical nerve stimulation in young (n = 13, 25 ± 4 years) and older (n = 12, 78 ± 5 years) adults.
Methods
The protocol involved four types of isometric contractions with the wrist-extensor muscles at 10% of the maximal force. Three of the contractions involved electrical nerve stimulation that comprised two forms of neuromuscular electrical stimulation (NMES) to evoke muscle contractions and a voluntary contraction with superimposed transcutaneous electrical nerve stimulation (TENS) at an intensity less than motor threshold.
Results
The coefficient of variation (CV) for force during voluntary wrist extension was less (P = 0.03) for young (1.82 ± 0.43%) than older adults (2.80 ± 1.08%). The CV for force did not differ between age groups during the three types of electrical nerve stimulation but was reduced relative to the value observed during voluntary wrist extension for older adults. In contrast, the CV for force increased during the voluntary contraction with superimposed TENS for young adults but not for older adults. Moreover, there were significant negative correlations in older adults between the CV for force during the voluntary contraction and its decrease with electrical nerve stimulation.
Conclusion
Differences in the CV for force between the evoked and voluntary contractions for the two age groups suggest that the variance in common synaptic input to motor neurons during steady voluntary contractions with the wrist extensors is greater for older adults than young adults.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
When an individual performs a submaximal isometric contraction to match a constant target force, the applied force invariably fluctuates about the prescribed target. The force fluctuations are usually greater for older adults (Galganski et al. 1993; Laidlaw et al. 2000; Tracy and Enoka 2002; Carville et al. 2006) and individuals with neurological disorders (Bilodeau et al. 2000; Hyngstrom et al. 2014; Arpin et al. 2016) compared with healthy young adults but can be reduced with practice and training (Laidlaw et al. 1999; Bilodeau et al. 2000; Salonikidis et al. 2009; Kobayashi et al. 2014). Critically, the normalized amplitude of the force fluctuations (coefficient of variation for force) during voluntary isometric contractions are strongly associated with the variance in the common synaptic input received by the motor neurons of concurrently active motor units (Negro et al. 2009; Farina and Negro 2015; Farina et al. 2016; Feeney et al. 2017; Thompson et al. 2018). Moreover, the coefficient of variation force during steady contractions with the wrist muscles is more strongly associated with the variance in the estimated common synaptic input to motor neurons for older adults than for young adults (Feeney et al. 2018).
The synaptic inputs responsible for the common modulation of motor unit discharge times are present only during voluntary contractions, as demonstrated by Jones et al. (2002) when they compared the standard deviation of force fluctuations over a range of target forces during voluntary contractions with those evoked by neuromuscular electrical stimulation (NMES). They found that the standard deviation of the force fluctuations increased with target force during brief voluntary contractions but essentially remained constant across target forces when the contractions were evoked by eliciting action potentials in intramuscular axons with NMES. Consequently, differences in the amplitude of the force fluctuations across conditions relative to the value observed during NMES-evoked contractions indicate modulation of the variance in the common synaptic input to motor neurons.
Our study extended the approach of Jones et al. (2002) by modulating electrical nerve stimulation parameters to activate different proportions of intramuscular motor and sensory axons. In general, the specific intramuscular axons engaged by electrical nerve stimulation depend on the pulse duration, frequency of the stimulus trains, and intensity of the applied current (Baldwin et al. 2006; Dean et al. 2007; Bergquist et al. 2011; Chipchase et al. 2011; Lagerquist et al. 2010). Conventional NMES protocols evoke contractions by activating motor axons with pulse durations of 0.2 to 0.5 ms, frequencies in the range of 40 to 80 Hz, and currents of less than 100 mA (Maffiuletti 2010; Vanderthommen and Duchateau 2007; Veldman et al. 2016). In contrast, longer pulse durations and greater frequencies, such as 1 ms and 100 Hz, have been shown to elicit action potentials in both motor and sensory axons and thereby involve a central contribution to the evoked force in some individuals, at least with the application of relatively low currents (Collins et al. 2002; Lagerquist et al. 2009; Bergquist et al. 2011; Clair-Auger et al. 2012; Wegrzyk et al. 2015). Moreover, the sensory volley activated by electrical nerve stimulation applied over the muscle is distributed widely throughout the central nervous system (Chipchase et al. 2011; Schmidt et al. 2011; Thompson et al. 2011; Gueugneau et al. 2017) and may influence the variance in the common modulation of synaptic input to motor neurons.
In contrast to NMES, conventional transcutaneous electrical nerve stimulation (TENS) involves narrow-pulsewidths (0.1 to 0.25 ms), either low (≤ 10 Hz) or high (≥ 50 Hz) frequencies, and the application of weak currents that activate sensory axons exclusively, without evoking a muscle contraction by directly activating intramuscular motor axons (Sluka and Walsh 2003). Stronger TENS currents that also do not evoke a muscle contraction, as used in our study, increase the types and numbers of afferent fibers activated by the electrical stimulation (Moran et al. 2011; Walker et al. 2014; Gomes-Osman et al. 2017). The application of TENS at a current below motor threshold has been found to reduce the coefficient of variation for force during a weak, steady contraction with the plantar flexors of young men (Kouzaki et al. 2012), which suggests that the sensory volley elicited by TENS reduced the variance of the common synaptic input received by the motor neurons engaged in the steady isometric contractions.
The purpose of our study was to compare the modulation of force steadiness by electrical nerve stimulation in young and older adults and thereby infer the relative influence of the engaged pathways on the variance in the common synaptic input received by the motor neurons innervating the wrist-extensor muscles. The protocol involved three types of electrical nerve stimulation applied to the wrist-extensor muscles during which force steadiness was quantified as the coefficient of variation for force and compared with the value observed during a voluntary isometric contraction. The target force for the evoked and voluntary contractions was 10% of the value achieved during a maximal voluntary contraction (MVC). The three types of electrical nerve stimulation manipulated the amount of sensory feedback that might influence the amplitude of the fluctuations in the force generated by the wrist extensors. We hypothesized that the coefficient of variation for force would be least during the contractions evoked by conventional NMES for both groups of subjects but that the modulation of force steadiness by the other two types of electrical nerve stimulation relative to the values observed during the voluntary contraction would differ for young and older adults.
Methods
Thirteen young (25 ± 4 years; five men) and 12 older (78 ± 5 years; seven men) adults met the inclusion criteria and provided informed consent to participate in our study. The experimental procedures were approved by the Institutional Review Board at the University of Colorado Boulder (Protocol #17–0064) and were in accordance with the Declaration of Helsinki. All participants were free of neuromuscular disease, reported no orthopedic problems that impacted the arms, hands, or fingers, and were not taking medications known to influence neuromuscular or cognitive function. All subjects participated in one experimental session that comprised three parts (muscle strength, force steadiness, and electrical nerve stimulation) and lasted approximately 1 h.
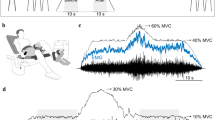
Muscle strength
The protocol began with an assessment of the strength of the muscles that contributed to wrist extension of the right arm, which was quantified as the peak force reached during an MVC. Subjects were seated comfortably with the right arm abducted by ~ 0.79 rad from the trunk, the elbow flexed to 1.57 rad with the pronated forearm resting on a metal stand, and the wrist secured on a padded brace (Fig. 1). Each MVC trial involved an increase in force from rest to maximum over 3 s and then sustaining it for 3 s. In addition to verbal encouragement during each MVC, visual feedback of the force was provided on a monitor (43.2 cm) located at eye level ~ 1.5 m in front of the subject. The average peak force from two trials that were within 5% of each other was designated as the MVC force. Participants also performed an MVC at the end of the protocol to provide a measure of performance fatigability.
The force exerted during wrist extension (both the voluntary and evoked contractions) was measured with a JR3 six-axis force transducer attached to a rigid restraint (900-N range, 176 N/V; Woodland, CA, USA). The subject was positioned so that the back of the right hand, just proximal to the knuckles, could push up against the rigid restraint that was attached to the JR3 transducer during wrist extension. The force signals were digitized with a Power 1401 (Cambridge Electronic Design, CED; Cambridge, UK) at 1000 samples/s and stored on a computer.
Force steadiness
After the assessment of MVC force, participants performed submaximal isometric contractions with the wrist-extensor muscles to match the 10% MVC target force for 30 s. Each subject was asked to match the target line and then maintain a steady contraction. The force exerted by the subject was displayed as a horizontal line with an increase in force indicated by an upward displacement of the line. The visual target was presented as a horizontal line with low-gain feedback at a visual angle of 0.75 ° (Baweja et al. 2010, 2012; Park et al. 2017b). Subjects performed a 10-s practice trial before beginning the assessment of force steadiness.
Electrical nerve stimulation
The final part of the protocol involved three electrical nerve stimulation conditions, which were performed in a randomized order. The current was delivered through conductive stimulating electrode pads that were attached over the wrist extensors (PALS Platinum Neurostimulation Electrodes, 5 cm2; model 895,220, Axelgaard Manufacturing Co., Ltd.; Lystrup, Denmark). A constant-current stimulator (DS7A Digitimer; Welwyn Garden City, UK) was used to provide a monophasic, rectangular current to the stimulating electrode pads with the cathode placed near the proximal end of the wrist extensors and the anode placed ~ 2 cm distal to the cathode (Fig. 1). The placement varied slightly between subjects but was generally over the muscle belly and ~ 1 inch from the carpal bones. Two types of evoked contractions were investigated: wide-pulse NMES (1 ms) at 100 Hz and narrow-pulse NMES (0.2 ms) at 50 Hz. The currents were set to evoke contractions that matched the 10% MVC target force. Subjects were asked to relax and allow the NMES to evoke a contraction for 30 s during which no visual feedback was provided. Participants completed a single trial for each type of NMES in a randomized order.
Subsequently, participants performed a 30-s voluntary contraction to match a 10% MVC target force with the wrist extensors while concurrently receiving TENS applied with a clinical device (LG-TEC Elite, LGMedSupply, LLC; Cherry Hill, NJ, USA). The TENS current was set to 1 mA less than the intensity required to produce a visible contraction in the forearm muscles, which produced a tingling sensation with no observable increase in force. The stimulus parameters for TENS were 0.2 ms pulses at 50 Hz. Representative records for a single participant are shown in Fig. 2.
Representative force trace for an older adult during the 30-s wrist-extension tasks with the right arm. Once the target was matched (10% MVC force), the subject was asked to maintain a steady contraction, which, for this person, was associated with a slight decline in the applied force. Critically, there were no obvious corrective actions during the steady contraction. Inset indicates a 10-s interval of the steadiest segment of the trial. The CV for force during this evoked (wide-pulse NMES) contraction was 1.33%
Data analysis
The magnitude of the force fluctuations about the target force was quantified as the coefficient of variation for force (ratio of standard deviation relative to the mean) to provide a measure of force steadiness (Galganski et al. 1993). A moving 10-s window (100-ms increments) was used to identify the steadiest 10 s (lowest coefficient of variation for force) of each 30-s trial. The force recorded during the NMES trials was detrended with a best-fit 2nd order polynomial function prior to quantifying the force fluctuations, similar to the approach used by Jones et al. (2002).
The power in the 0–0.5 Hz and 0.5–1 Hz bandwidths of the force signal was calculated with Welch’s periodogram. Power in each of the two bandwidths was determined from the detrended force signals over the steadiest 10-s window (Moon et al. 2014). The resulting resolution was 0.06 Hz. The integral of the power in the 0–0.5 Hz and 0.5–1.0 Hz bandwidths were divided by the total power from 0 to 1 Hz to calculate the percentage of power in each bandwidth. These low-frequency bandwidths—when quantified with sufficient resolution—are strongly correlated with force variability (Moon et al. 2014; Lodha and Christou 2017; Park et al. 2017a).
Statistics
After confirming the normality of the data with the Shapiro–Wilk test, a 2 × 4 (age-by-condition) analysis of variance (ANOVA) was used to compare the influence of condition (voluntary contraction, two types of NMES, and superimposed TENS) and age on force steadiness. Mauchly’s sphericity test was applied to confirm that the assumption of equal variances was met to implement a repeated-measures ANOVA to compare young (P = 0.124) and older (P = 0.124) adults. Although the interaction effect was not statistically significant, previous work on force steadiness has demonstrated the utility of a within-group analysis (Feeney et al. 2018). Within age groups, a repeated-measures ANOVA examined the influence of each type of stimulation on the coefficient of variation for force. Paired t tests with Bonferroni corrections of the P values were applied post-hoc to compare the coefficient of variation for force between groups.
Effect size for the ANOVA was estimated with η2, whereas Cohen’s d was used for post-hoc pairwise comparisons. Effect sizes of 0.1 were considered small and those ≥ 0.5 were deemed large. All correlations were examined with Pearson’s product-moment correlation. An alpha level of P < 0.05 was used to identify significant differences for all tests, except the paired t tests, which were adjusted according to the Bonferroni criteria. Data are presented in the text as mean ± SD. Statistical analyses were performed using R (CRAN, R version 3.4.1) based on the advice of experts in biostatistics.
Results
Our report comprises the results on force steadiness—coefficient of variation for force—during voluntary and evoked contractions performed by 13 young and 12 older adults. All participants were right-handed, as verified by the laterality quotient (0.9 ± 0.2) of the Edinburgh Handedness Inventory (Oldfield 1971). There were no statistically significant differences between young and older adults for any of the descriptive characteristics (Table 1).
As a foundation for interpreting the influence of the pathways engaged by electrical nerve stimulation on force steadiness, we measured the coefficient of variation for force when the two groups of participants performed the voluntary wrist-extension action at 10% MVC force. Young adults (1.82 ± 0.43%) were steadier than older adults (2.80 ± 1.08%) (P = 0.002, Cohen’s d = 0.9). The 2 × 4 ANOVA (two age groups by four contraction conditions) indicated that there was a significant main effect for contraction condition (P = 0.0035, η2 = 0.09) but not for age (P = 0.062, η2 = 0.013) or the interaction (P = 0.07, = 0.07). Moreover, post-hoc analysis with paired t tests did not detect any condition comparison that met the adjusted P value (< 0.0083).
Within each age group, a repeated-measures ANOVA identified a significant effect for contraction condition in older adults (P = 0.0006, η2 = 0.18) but not young adults (P = 0.068, η2 = 0.11), which indicated that the coefficient of variation for force differed significantly between evoked and voluntary contractions for older adults.
The current required to evoke a contraction in the wrist extensors to match the target force was not statistically different between groups for both wide-pulse, 100-Hz NMES (young: 6.3 ± 4.7 mA; older: 8.5 ± 4.2 mA; P = 0.24) and narrow-pulse, 50-Hz NMES (young: 19.0 ± 6.9 mA; older: 25.0 ± 6.7 mA; P = 0.06). However, the TENS current was significantly less (P = 0.04, η2 = 0.20) for young (9.8 ± 1.4 mA) than older adults (11.0 ± 1.3 mA). The average force evoked by both types of NMES during the 10-s contractions did not increase for any participant, but instead it either remained constant or decreased slightly (Fig. 2). In addition, the average force was not altered by the application of TENS.
The three types of electrical nerve stimulation elicited different results in the young and older adults (Table 2). The coefficient of variation for force (mean ± SD, [95% confidence interval]) for the young adults was not statistically different during either NMES condition (wide-pulse, 100 Hz: 1.93 ± 0.68%, [1.52–2.34]; narrow-pulse, 50 Hz: 1.83 ± 0.59%, [1.47–2.19]) compared with the voluntary contraction with the wrist extensors (1.80 ± 0.43%, [1.56–2.08]). In contrast, the coefficient of variation for force was less in older adults during both NMES-evoked conditions (wide-pulse, 100 Hz: 2.01 ± 0.67%. [1.60–2.44]; narrow-pulse, 50 Hz: 1.69 ± 0.62%, [1.30–2.09]) than during voluntary wrist extension (2.8 ± 1.1%, [2.00–3.36]). Conversely, the application of TENS during a voluntary contraction increased the coefficient of variation for force in young adults (2.41 ± 1.02%, [1.79–3.03]) relative to the value observed during voluntary wrist extension, but there was no statistically significant difference in the amplitude of the force fluctuations during the voluntary contractions with and without TENS for older adults (2.29 ± 0.76%, [1.80–2.77]).
As indicated in Fig. 3, however, there was substantial variability across subjects in the influence of electrical nerve stimulation on the coefficient of variation for force relative the value observed during voluntary wrist extension. Nonetheless, there were statistically significant effects within each group when the change in the amplitude of the force fluctuations was compared with the value observed during the voluntary contraction. Older adults exhibited statistically significant negative correlations (P < 0.05) for all three types of electrical nerve stimulation (two types of NMES and TENS) between the change in the coefficient of variation for force and the value observed during voluntary wrist extension (Fig. 3d–f; Table 2). The negative correlations indicate that older adults with greater force fluctuations during the voluntary contraction experienced greater decreases in the force fluctuations during all three types of electrical nerve stimulation.
Percent change in the coefficient of variation for force during the three electrical nerve stimulation conditions relative to the value during voluntary wrist extension for young (a–c) and older (d–f) adults. a, d Wide-pulse 100-Hz NMES. b, e Narrow-pulse 50-Hz NMES. c, f Voluntary contraction with the wrist extensors during superimposed TENS
In contrast, there was only one statistically significant correlation for young adults. As with the older adults, young adults with greater force fluctuations during voluntary wrist extension experienced greater decreases in the force fluctuations during wide-pulse 100-Hz NMES (Fig. 3a).
Approximately 93% of the power in the force signal for all conditions was located below 10 Hz. There was no significant effect of contraction condition (P = 0.35) or age (P = 0.06) on the power in the low-frequency bandwidth (0–0.5 Hz). Although the difference was not statistically significant, 72.2 ± 13.3% of the power in the force signal was between 0 and 0.5 Hz for young adults and 82.2 ± 9.8% for older adults (P = 0.06). Similarly, there were no statistically significant differences in the percentage of power in the low-frequency bandwidth between narrow − 50 Hz NMES (79.9 ± 13.4%), wide-100 Hz NMES (78.3 ± 16.7%) and TENS (80.2 ± 12.2%).
MVC force during wrist extension at the end of the protocol was 97 ± 27 N for young adults and 114 ± 47 N for older adults, neither of which was statistically different (young: P = 0.43; older: P = 0.61) from that measured at the beginning of the experiment (Table 1). Thus, the protocol did not induce any performance fatigability as indicated by the assessment of MVC force.
Discussion
The main finding of our study was that the influence of electrical nerve stimulation on the normalized amplitude of the force fluctuations differed for young and older adults. The coefficient of variation for the force was minimal during the contractions evoked by conventional NMES (narrow-pulse, 50 Hz) (Jones et al. 2002) and increases in the force fluctuations during the other three conditions were assumed to indicate an increase in the variance of the common synaptic input received by the involved motor neurons (Farina and Negro 2015; Farina et al. 2016; Negro et al. 2016; Feeney et al. 2018; Thompson et al. 2018).
Consistent with our hypothesis, the coefficient of variation for force was least during narrow-pulse, 50-Hz NMES for both age groups. However, the coefficient of variation for force during wide-pulse, 100-Hz NMES was not statistically different from that during narrow-pulse, 50-Hz NMES for either age group. In contrast to the NMES-evoked contractions, superimposed TENS during a voluntary contraction increased the coefficient of variation for force in young adults, but not older adults. Critically, both types of NMES improved force steadiness—reduced the coefficient of variation for force—in older adults. Although the coefficient of variation for force was not significantly different during voluntary contractions with and without TENS in older adults, the changes in the coefficient of variation for force during TENS were significantly and negatively correlated with the value observed during the voluntary contraction. The negative correlation indicates that those older adults who were least steady during the voluntary contraction exhibited the greatest reduction in the coefficient of variation for force when TENS was superimposed on the voluntary contraction. A similar negative correlation was observed for young adults, but only during wide-pulse, 100-Hz NMES.
Neuromuscular electrical stimulation
Our study was partially motivated by the findings of Jones et al. (2002) that the standard deviation for force during voluntary contractions scales linearly with target force (20–70% MVC) for young adults, whereas the standard deviation for force is constant during contractions evoked by NMES. Jones and colleagues used an NMES protocol (pulse width: 300 µs; frequency: 25–30 Hz) that was similar to our narrow-pulse, 50-Hz condition. Their results indicate that most of the force fluctuations during voluntary isometric contractions are attributable to differences in central factors that influence motor unit activity, whereas the force fluctuations associated with peripheral signal transduction are relatively constant.
The application of NMES generates action potentials in intramuscular motor axons (Hultman et al. 1983) that are widely distributed throughout a muscle (Yu et al. 2016; Buckmire et al. 2018). Due to the capacity of an action potential to be propagated in both directions from the location at which a current has been applied to an axon (Magladery and McDougal 1950; Schieppati 1987; Puksa et al. 2003), surface stimulation is able to activate both superficial and deep regions of a muscle (Adams et al. 1993; Okuma et al. 2013). Presumably, action potentials elicited in an intramuscular branch of a motor axon can be propagated antidromically to invade other branches of the motor axon and thereby generate action potentials in muscle fibers that are located at a greater distance away from the site of stimulation. The resulting distribution of action potentials produces an asynchronous activation of muscle fibers and low-amplitude fluctuations in the evoked force (Collins et al. 2002; Lagerquist et al. 2009). Although the average evoked force increases with the intensity of the applied current, the amplitude of the force fluctuations remains relatively constant (Jones et al. 2002). Our current findings indicate that neither the normalized amplitude nor the spectral content of the force fluctuations (coefficient of variation for force) was consistently influenced by the stimulus parameters that evoke the same average force; namely between wide, high-frequency and narrow, low-frequency NMES.
Another finding was that the coefficient of variation for force during the NMES-evoked contractions was statistically reduced from that observed during voluntary contractions in older adults. This finding indicates that the force fluctuations exhibited by young adults during voluntary contractions were equivalent in amplitude to those produced by the asynchronous activation of intramuscular motor axons by both types of NMES. In contrast, the amplitude of the force fluctuations exhibited by older adults during voluntary contractions, which are likely attributable to the variance in the common synaptic input to motor neurons (Negro et al. 2016; Feeney et al. 2018; Thompson et al. 2018), was greater than those produced by the NMES-evoked asynchronous activation of motor axons.
Moreover, the relative difference in force fluctuations between the NMES-evoked and voluntary contractions varied within groups for three of the four conditions; the exception was narrow-pulse NMES for young adults. In addition, there were significant negative correlations between the amplitude of the force fluctuations during the voluntary contraction and the size of the reduction in the fluctuations during the NMES conditions for older adults. The negative correlations indicated that individuals with greater force fluctuations during the steady voluntary contractions experienced greater decreases in the force fluctuations during the evoked contractions, suggesting that NMES might be an effective intervention to improve force control in those older adults who exhibit impairments in force control, such as during tasks requiring manual dexterity. This finding also indicates variability across participants in the extent to which the variance in the common synaptic input to motor neurons (during voluntary contractions) exceeded that of asynchronous activation of motor axons (during evoked contractions).
Transcutaneous electrical nerve stimulation
The concurrent application of TENS during the voluntary contraction significantly increased the coefficient of variation for force in young adults, but not in older adults. The current applied during TENS was just below motor threshold, which elicits action potentials in a wide range of sensory axons (Radhakrishnan and Sluka 2005; Moran et al. 2011; Pantaleao et al. 2011; Walker et al. 2014; De Nunzio et al. 2018). A number of participants reported a tingling sensation located beneath the stimulation electrodes, which suggests that the influence of the current was quite localized. The increase in the coefficient of variation for force exhibited by young adults suggests that the additional sensory feedback elicited with submotor TENS increased the variance in the common synaptic input to motor neurons. Similarly, Jones et al. (2002) found that the superimposition of NMES on a voluntary contraction performed by young adults increased their standard deviation for force.
The absence of a significant difference in the coefficient of variation for force during voluntary contractions with and without concurrent TENS in older adults suggests that the influence of the additional sensory volley may have been saturated in this group. Although aging is usually accompanied by a reduction in the numbers of sensory receptors, a decline in the responsiveness of the receptors, and an increase in the level of presynaptic inhibition (Proske and Gandevia 2012; Baudry 2016), the absence of a significant difference in the coefficient of variation for force suggests that TENS did not elicit a consistent increase in the variance of the common input received by the motor neurons.
Although the initial power calculations based on comparable studies enrolled fewer subjects than we did in our study, it is possible that an increase in sample size might have increased the number of significant differences between groups. However, a post-hoc power analysis of the difference in CV for force between groups suggested that our study had adequate statistical power (83.5%). Nonetheless, there was a statistically significant negative correlation for older adults between the reduction in the force fluctuations when TENS was applied during the voluntary contraction compared with the value observed during the voluntary contraction alone. This finding suggests that those older adults with worse force steadiness during the voluntary contraction (greater force fluctuations) presumably experienced a greater reduction in the variance in the common synaptic input to motor neurons due to the additional sensory feedback, which was opposite to the main effect observed in young adults. The concurrent application of TENS, therefore, attenuated at least one major source of variance in the common synaptic input to motor neurons, but only in those older adults who were least steady during the submaximal, isometric contraction with the wrist extensors.
Limitations
There were some limitations in our study. Although the force fluctuations during steady isometric contractions are strongly associated with the variability in the common modulation of motor unit activity in an agonist muscle (Negro et al. 2009; Farina and Negro 2015; Farina et al. 2016; Feeney et al. 2016, 2018; Thompson et al. 2018), other factors—such as activity in synergistic and antagonistic muscles—can contribute to the amplitude of the force fluctuations, and ought be considered with our interpretation that differences in the amplitude of the force fluctuations across conditions is attributable to changes in the variance of the common synaptic input to motor neurons. Similarly, the differential influence of the three types of electrical nerve stimulation on the force fluctuations in the two age groups suggest the involvement of different pathways, but the specific afferent fibers that contribute to the effects in each cohort remain to be identified. Moreover, the effects may differ for other muscle groups, even though many motor neuron pools appear to receive a dominant slow-varying common synaptic input (Negro et al. 2016).
Conclusion
The force fluctuations exhibited by young adults during a steady, voluntary contraction with the wrist extensors were equivalent in amplitude to those observed during NMES-evoked contractions. In contrast, the force fluctuations of older adults were greater during the voluntary contraction than during the NMES-evoked contractions. Nonetheless, the amplitude of the force fluctuations during the NMES-evoked contractions was similar for young and older adults. The superimposition of TENS during a voluntary contraction, which provided additional sensory feedback, increased the force fluctuations in young adults, but not in older adults. In addition, the effects elicited by three types of electrical nerve stimulation were negatively correlated with the amplitude of the force fluctuations (coefficient of variation for force) during the voluntary contraction, except for one condition in young adults. The differences between the two groups of participants indicated the relative influence of the variance in the common synaptic input to motor neurons in adding to the amplitude of the force fluctuations attributable to the NMES-evoked asynchronous activation of intramuscular motor axons.
Abbreviations
- ANOVA:
-
Analysis of variance
- CV:
-
Coefficient of variation
- MVC:
-
Maximal voluntary contraction
- NMES:
-
Neuromuscular electrical stimulation
- TENS:
-
Transcutaneous electrical nerve stimulation
References
Adams GR, Harris RT, Woodard D, Dudley GA (1993) Mapping of electrical muscle stimulation using MRI. J Appl Physiol 74:532–537
Arpin DJ, Davies BL, Kurz MJ (2016) Multiple sclerosis influences the precision of the ankle plantarflexion muscular force production. Gait Posture 45:170–174
Baldwin ERL, Klakowicz PM, Collins DF (2006) Wide-pulse width, high-frequency neuromuscular electrical stimulation: implications for functional electrical stimulation. J Appl Physiol 101:228–240
Baudry S (2016) Aging changes the contribution of spinal and corticospinal pathways to control balance. Exerc Sport Sci Rev 44:104–109
Baweja HS, Kennedy DM, Vu J, Vaillancourt DE, Christou EA (2010) Greater amount of visual feedback decreases force variability by reducing force oscillations from 0 to 1 and 3–7 Hz. Eur J Appl Physiol 108:935–943
Baweja HS, Kwon M, Christou EA (2012) Magnified visual feedback exacerbates positional variability in older adults due to altered modulation of the primary agonist muscle. Exp Brain Res 222:355–364
Bergquist AJ, Clair JM, Lagerquist O, Mang CS, Okuma Y, Collins DF (2011) Neuromuscular electrical stimulation: implications of the electrically evoked sensory volley. Eur J Appl Physiol 111:2409–2226
Bilodeau M, Keen DA, Sweeney PJ, Shields RW, Enoka RM (2000) Strength training can improve steadiness in persons with essential tremor. Muscle Nerve 23:771–778
Buckmire AJ, Lockwood DR, Doane CJ, Fuglevand AJ (2018) Distributed stimulation increases force elicited with functional electrical stimulation. J Neural Eng 15:026001
Carville SF, Perry MC, Rutherford OM, Smith IC, Newham DJ (2006) Steadiness of quadriceps contractions in young and older adults with and without a history of falling. Eur J Appl Physiol 100:527–533
Chipchase LS, Schabrun SM, Hodges PW (2011) Corticospinal excitability is dependent on the parameters of peripheral electric stimulation: a preliminary study. Arch Phys Med Rehabil 92:1423–1430
Clair-Auger JM, Collins DF, Dewald JPA (2012) The effects of wide pulse neuromuscular electrical stimulation on elbow flexion torque in individuals with chronic hemiparetic stroke. Clin Neurophysiol 123:2247–2255
Collins DF, Burke D, Gandevia SC (2002) Sustained contractions produced by plateau-like behaviour in human motoneurones. J Physiol 538:289–301
De Nunzio AM, Yavuz US, Martinez-Valdes E, Farina D. Falla D (2018) Electro-tactile stimulation of the posterior neck induces body anteropulsion during upright stance. Exp Brain Res 236:1471–1478
Dean JC, Yates LM, Collins DF (2007) Turning on the central contribution to contractions evoked by neuromuscular electrical stimulation. J Appl Physiol 103:170–176
Farina D, Negro F (2015) Common synaptic input to motor neurons, motor unit synchronization, and force control. Exerc Sport Sci Rev 43:23–33
Farina D, Negro F, Muceli S, Enoka RM (2016) Principles of motor unit physiology evolve with advances in technology. Physiology 31:83–94
Feeney DF, Meyer FG, Noone N, Enoka RM (2017) A latent low-dimensional common input drives a pool of motor neurons: a probabilistic latent state-space model. J Neurophysiol 118:2238–2250
Feeney DF, Mani D, Enoka RM (2018) Variability in common synaptic input to motor neurons modulates both force steadiness and pegboard time in young and older adults. J Physiol 596:3793–3806
Galganski ME, Fuglevand AJ, Enoka RM (1993) Reduced control of motor output in a human hand muscle of elderly subjects during submaximal contractions. J Neurophysiol 69:2108–2115
Gomes-Osman J, Tibbett JA, Poe BP, Field-Fote EC (2017) Priming for improved hand strength in persons with chronic tetraplegia: a comparison of priming-augmented functional task practice, priming alone, and conventional exercise training. Front Neurol 242:1–13
Gueugneau N, Grosprêtre S, Stapley P, Lepers R (2017) High-frequency neuromuscular electrical stimulation modulates interhemispheric inhibition in healthy humans. J Neurophysiol 117:467–475
Hultman E, Sjöholm H, Jäderholm-Ek I, Krynicki J (1983) Evaluation of methods for electrical stimulation of human skeletal muscle in situ. Pflügers Archiv 398:139–141
Hyngstrom AS, Kuhnen HR, Kirking KM, Hunter SK (2014) Functional implications of impaired control of submaximal hip flexion following stroke. Muscle Nerve 49:225–232
Jones KE, Hamilton AF, Wolpert DM (2002) Sources of signal-dependent noise during isometric force production. J Neurophysiol 88:1533–1544
Kobayashi H, Koyama Y, Enoka RM, Suzuki S (2014) A unique form of light-load training improves steadiness and performance on some functional tasks in older adults. Scand J Med Sci Sports 24:98–110
Kouzaki M, Kimura T, Yoshitake Y, Hayashi T, Moritani T (2012) Subthreshold electrical stimulation reduces motor unit discharge variability and decreases the force fluctuations of plantar flexion. Neurosci Lett 513:146–150
Lagerquist O, Collins DF (2010) Influence of stimulus pulse width on M-waves, H-reflexes, and torque during tetanic low-intensity neuromuscular stimulation. Muscle Nerve 42:886–893
Lagerquist O, Walsh LD, Blouin JS, Collins DF, Gandevia SC (2009) Effect of a peripheral nerve block on torque produced by repetitive electrical stimulation. J Appl Physiol 107:161–167
Laidlaw DH, Kornatz KW, Keen DA, Suzuki S, Enoka RM (1999) Strength training improves the steadiness of slow lengthening contractions performed by old adults. J Appl Physiol 87:1786–1795
Laidlaw DH, Bilodeau M, Enoka RM (2000) Steadiness is reduced and motor unit discharge is more variable in old adults. Muscle Nerve 23:600–612
Lodha N, Christou EA (2017) Low-frequency oscillations and control of motor output. Front Physiol 8:1–9
Maffiuletti NA (2010) Physiological and methodological considerations for the use of neuromuscular electrical stimulation. Eur J Appl Physiol 110:223–234
Magladery JW, McDougal DB Jr (1950) Electrophysiological studies of nerve and reflex activity in normal man. I. Identification of certain reflexes in the electromyogram and the conduction velocity of peripheral nerve fibers. Bull Johns Hopkins Hosp 86:265–290
Moon H, Kim C, Kwon M, Chen YT, Onushko T, Lodha N, Christou EA (2014) Force control is related to low-frequency oscillations in force and surface EMG. PLoS One 9:e109202
Moran F, Leonard T, Hawthrone S, Hughes CM, McCrum-Gardner E, Johnson MI, Rakel BA, Sluka KA, Walsh DM (2011) Hypoalgesia in response to transcutaneous electrical nerve stimulation (TENS) depends on stimulation intensity. J Pain 12:929–935
Negro F, Holobar A, Farina D (2009) Fluctuations in isometric muscle force can be described by one linear projection of low-frequency components of motor unit discharge rates. J Physiol 587:5925–5938
Negro F, Yavuz US, Farina (2016) The human motor neuron pools receive a dominant slow-varying common synaptic input. J Physiol 594:5491–5505
Okuma Y, Bergquist AJ, Hong M, Chan KM, Collins DF (2013) Electrical stimulation site influences the spatial distribution of motor units recruited in tibialis anterior. Clin Neurophysiol 124:2257–2263
Oldfield RC (1971) The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 9:97–113
Pantaleao MA, Laurino MF, Gallego NL, Cabral CM, Rakel B, Vance C, Sluka KA, Walsh DM, Liebano RE (2011) Adjusting pulse amplitude during transcutaneous electrical nerve stimulation (TENS) application produces greater hypoalgesia. J Pain 12:581–590
Park SH, Casamento-Moran A, Yacoubi B, Christou EA (2017a) Voluntary reduction of force variability via modulation of low-frequency oscillations. Exp Brain Res 235:2717–2727
Park SH, Kwon M, Christou EA (2017b) Motor output oscillations with magnification of visual feedback in older adults. Neurosci Lett 24:647–648
Proske U, Gandevia SC (2012) The proprioceptive senses: their roles in signaling body shape, body position and movement, and muscle force. Physiol Rev 92:1651–1697
Puksa L, Stålberg E, Falck B (2003) Reference values of F wave parameters in healthy subjects. Clin Neurophysiol 114:1079–1090
Radhakrishnan R, Sluka KA (2005) Deep tissue afferents, but not cutaneous afferents, mediate transcutaneous electrical nerve stimulation-induced antihyperalgesia. J Pain 6:673–680
Salonikidis K, Amiridis IG, Oxyzoglou N, de Villareal ES, Zafeiridis A, Kellis E (2009) Force variability during isometric wrist flexion in highly skilled and sedentary individuals. Eur J Appl Physiol 107:715–722
Schieppati M (1987) The Hoffmann reflex: a means of assessing spinal reflex excitability and its descending control in man. Prog Neurobiol 28:345–376
Schmidt MW, Hinder MR, Summers JJ, Garry MI (2011) Long-lasting contralateral motor cortex excitability is increased by unilateral hand movement that triggers electrical stimulation of opposite homologous muscles. Neurorehabilit Neural Repair 25:521–530
Sluka KA, Walsh D (2003) Transcutaneous electrical nerve stimulation: basic science mechanisms and clinical effectiveness. J Pain 4:109–121
Thompson CK, Lewek MD, Jayaraman A, Hornby TG (2011) Central excitability contributes to supramaximal volitional contractions in human incomplete spinal cord injury. J Physiol 589:3739–3752
Thompson CK, Negro F, Johnson MD, Holmes MR, McPherson LM, Powers RK, Farina D, Heckman CJ (2018) Robust and accurate decoding of motoneuron behavior and prediction of the resulting force output. J Physiol 596:2643–2659
Tracy BL, Enoka RM (2002) Older adults are less steady during submaximal isometric contractions with the knee extensor muscles. J Appl Physiol 92:1004–1012
Vanderthommen M, Duchateau J (2007) Electrical stimulation as a modality to improve performance of the neuromuscular system. Exerc Sport Sci Rev 35:180–185
Veldman MP, Gondin J, Place N, Maffiuletti N (2016) Effects of neuromuscular electrical stimulation training on endurance performance. Front Physiol 7:1–5
Walker ER, Hyngstrom AS, Schmit BD (2014) Sensory electrical stimulation improves foot placement during targeted stepping post-stroke. Exp Brain Res 232:1137–1143
Wegrzyk J, Fouré A, Vilmen C, Ghattas B, Maffiuletti NA, Mattei JP, Place N, Bendahan D, Gondin J (2015) Extra forces induced by wide-pulse, high-frequency electrical stimulation: occurrence, magnitude, variability and underlying mechanisms. Clin Neurophysiol 126:1400–1412
Yu D, Yin H, Han T, Jiang H, Cao X (2016) Intramuscular innervations of lower leg skeletal muscles: applications in their clinical use in functional muscular transfer. Surg Radiol Anat 38:675–685
Acknowledgements
We thank Melissa Mazzo and Ryan Price for drawing Fig. 1, and Professors Evangelos Christou and Allison Hyngstrom for providing comments on a draft of the manuscript.
Funding
This work was supported by the Rocky Mountain American College of Sports Medicine Research Grant awarded to Diba Mani and the American Society of Biomechanics Graduate Student Grant-In-Aid awarded to Daniel Feeney.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no financial conflict of interests exists.
Additional information
Communicated by Nicolas Place.
Rights and permissions
About this article
Cite this article
Mani, D., Feeney, D.F. & Enoka, R.M. The modulation of force steadiness by electrical nerve stimulation applied to the wrist extensors differs for young and older adults. Eur J Appl Physiol 119, 301–310 (2019). https://doi.org/10.1007/s00421-018-4025-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-018-4025-6