Abstract
Background
Cataract surgery has been shown to reduce intraocular pressure (IOP) in eyes with primary angle closure glaucoma (PACG). In this study, we examined IOP fluctuations and circadian IOP patterns before and after cataract surgery in PACG patients using the contact lens sensor (CLS) Triggerfish®.
Methods
This was a prospective open-label study. Ten consecutive patients with PACG were included. The patients underwent phacoemulsification and intraocular lens implantation (PEA+IOL) surgery. In each eye, IOP fluctuations over 24 h were measured with the CLS before and at three months after the surgery. Changes in corneal curvatures and anatomic parameters of the anterior chamber were measured by anterior segment optical coherence tomography (AS-OCT).
Results
The mean IOP was significantly reduced from 14.7 ± 1.5 mmHg to 11.2 ± 2.2 mmHg at three months after the surgery in PACG patients (P = 0.002). Although the mean range of 24-h IOP fluctuations was not significantly changed after the surgery (P = 0.49), the mean range of IOP fluctuations during the nocturnal period was significantly decreased from 246 ± 61 mVeq to 179 ± 64 mVeq after the surgery (P = 0.02). After the surgery, seven eyes of the ten (70 %) showed the same circadian IOP patterns. AS-OCT showed significant improvements in the anatomic parameters of the anterior chamber after the surgery.
Conclusions
Cataract surgery decreased IOP fluctuations during the nocturnal period in PACG patients. These effects might partly prevent the progression of PACG.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Background
Glaucoma is a leading cause of irreversible blindness worldwide. It is estimated that primary angle closure glaucoma (PACG) affects 3.9 million people worldwide, has a higher prevalence in Asia and is responsible for half of the blindness from glaucoma [1]. Although PACG accounts for only a quarter of all primary glaucoma cases, it is a severe condition and the number of patients who become blind is almost equal to the number of primary open angle glaucoma (POAG) cases [1]. In addition, the chronic PACG patients have been reported to show more generalized field loss than the POAG patients [2].
It has been speculated that the visual field loss in PACG patients may be related, at least in part, to the elevated fluctuations in circadian IOP in PACG patients [3]. Large fluctuations in circadian IOP are also a significant risk factor for progression of visual field loss in POAG patients [3, 4]. Therefore, it is important to reduce the range of circadian IOP fluctuations for glaucoma treatment. Recently, the SENSEIMED Triggerfish® (SENSIMED AG, Lausanne, Switzerland) contact lens sensor (CLS) was made available. This CLS can continuously monitor IOP fluctuations over 24 h in an ambulatory setting for patients (habitual IOP fluctuations) [5–7].
As potentially effective treatments for PACG, phacoemulsification and intraocular lens implantation (PEA+IOL) have been studied [8, 9]. Previous studies showed that PEA+IOL not only improved the visual acuity but also reduced IOP and the number of anti-glaucoma medications in PACG patients [9–11].
In this study, we measured IOP fluctuations using a CLS in patients with PACG before and after PEA+IOL and evaluated the effects of PEA+IOL on IOP fluctuations and circadian IOP patterns in PACG patients. To the best of the authors’ knowledge, this is the first study to employ 24-h continuous measurement of IOP fluctuations in eyes with PACG before and after PEA+IOL.
Methods
This was a prospective, open-label study. Ten consecutive patients diagnosed with PACG at Toyama University Hospital were enrolled in this study from November 2012 to February 2013. All patients underwent a comprehensive ophthalmic examination including refraction, static and dynamic Goldmann gonioscopy, Goldmann applanation tonometry (GAT), fundus examination, and swept-source anterior segment optical coherence tomography (AS-OCT) (CASIA SS-1000, Tomey Corporation, Nagoya, Japan), and automated perimetry (Humphrey Field Analyzer; Carl Zeiss Meditec, Inc., Dublin, CA). One glaucoma specialist diagnosed PACG based on the criteria of Foster et al. [12].
A diagnosis of PACG was made if all of the following criteria were satisfied: (1) narrow angles (defined as eyes in which at least 270° of the posterior pigmented trabecular meshwork was not visible in gonioscopy); (2) presence of glaucomatous optic disc neuropathy (a cup/disc ratio of ≥0.7, or the presence of notching) accompanied by corresponding visual field defects; (3) a threshold examination of SITA 30–2 showing a glaucoma hemifield test “outside normal limits,” and a cluster of three contiguous points on the pattern deviation plot depressed at the P < 5 % level (versus age-matched normal subjects) not crossing the horizontal meridian. We confirmed at least two reproducible results of glaucomatous defects by the visual field tests.
Patients were required to meet all of the following criteria for inclusion in this study: (1) best-corrected visual acuity ≥0.2; (2) spherical equivalent ≤ −6 diopters; (3) diagnosis of PACG. Patients with any of the following were excluded: (1) pseudo exfoliation syndrome; (2) neovascular glaucoma; (3) steroid glaucoma; (4) primary open angle glaucoma; (5) history of ocular trauma; (6) retinal disease; (7) post vitrectomy; (8) ocular inflammatory disease; and (9) prior laser trabeculoplasty or other prior glaucoma surgery except for laser iridotomy.
The baseline IOP was the average of the last three readings obtained with GAT at the same time of the day prior to PEA+IOL in each patient. The postoperative IOP was similarly obtained with GAT at three months after PEA+IOL.
Twenty four-hour IOP fluctuations were monitored in each eye before and after PEA+IOL with a Triggerfish® CLS (SENSIMED AG, Lausanne, Switzerland) as previously described [5]. Preoperative IOP fluctuations were measured within three days before PEA+IOL. Postoperative IOP fluctuations were measured at three months after PEA+IOL. Briefly, the CLS consisted of a highly oxygen-permeable soft contact lens, whose key elements were two sensing-resistive strain gauges that were capable of recording circumferential changes in the area of the corneoscleral junction. The device uses a novel approach to IOP monitoring in which changes in the corneal curvature and circumference are assumed to correspond to changes in IOP [13]. A change in 1 mm Hg (mmHg) of IOP was shown to produce a change in approximately 3 μm of the central corneal curvature radius [14]. The unit used in monitoring IOP with Triggerfish® is not mmHg but millivolt equivalent (mVeq), which is unique to Triggerfish®. The spiking increases in IOP monitoring with a CLS caused mainly by eye blinks, and the ocular pulsation frequency according to cardiac activity during sleeping. In addition, other factors such as temperature difference and rapid eye movements during sleeping may lead to peak increases.
We evaluated IOP fluctuations separately in the diurnal period and the nocturnal period as previously described [5, 15]. The nocturnal/sleep period was defined through the observation of blink cessation (identified as short and high amplitude spikes that were displayed by the software) on the CLS signals. We also verified the diurnal/wakefulness period and nocturnal/sleep period within the 24-h period by referring to the logs of the individual patients, in which they were instructed to record the time that they returned home, installed eye drops, went to bed, and woke up. Although antibacterial eye drop and anti-inflammatory eye drop were stopped at three months after PEA+IOL, the eye drops for glaucoma were not changed throughout the study periods.
To analyze the IOP fluctuation, the range was defined as the difference between the maximum and minimum values over 24 h, the diurnal period, and the nocturnal period. The patients were classified into three pattern groups, which were the diurnal acrophase, nocturnal acrophase and no significant acrophase groups [15]. The diurnal acrophase group consisted of patients, for whom the average value (mVeq) in the diurnal period was higher than that in the nocturnal period, and the peak value (mVeq) occurred in the diurnal period. The average values (mVeq) were calculated manually. The nocturnal acrophase group consisted of patients, for whom the average value (mVeq) in the nocturnal period was higher than that in the diurnal period, and the peak value (mVeq) occurred in the nocturnal period. The not significant acrophase group consisted of patients who were not assigned to either the diurnal acrophase or nocturnal acrophase group [15]. To evaluate the patterns of IOP fluctuations for 24 h in each patient, the CLS signals were reconstituted by using the hourly mean value (mVeq).
The central corneal thickness and corneal meridians, which may influence IOP measurement, were measured with AS-OCT before and after 24-h IOP measurement in each eye. We also measured the central anterior chamber depth, lens vault and axial length with AS-OCT in each eye. The algorithm then automatically calculated the lens vault, defined as the perpendicular distance between the anterior pole of the crystalline lens and the horizontal line joining the 2 scleral spurs (Fig. 1a). The anterior opening distance at 500 μm from the scleral spur and the trabecular iris angle at 500 μm from the scleral spur were measured in the AS-OCT images (Fig. 1b).
a Image of a PACG eye obtained by anterior segment-optical coherence tomography (AS-OCT).b Magnification of the square at the angle in A. The anterior opening distance is the perpendicular distance between the corneal endothelium and the anterior iris surface at 500 μm anterior to the scleral spur. The trabecular iris angle is defined as the angle between the line of the corneal endothelium and the line of the anterior iris surface. CCT central corneal thickness, CACD central anterior chamber depth, SS scleral spur
A Wilcoxon signed-rank test was used for all statistical analyses. Statistical significance was defined at P < 0.05. The difference of IOP fluctuation range before and after cataract surgery was assumed to be about 50mVeq. A standard deviation was supposed to be 50mVeq. With a power of 80 %, a total of 10 patients was necessary to detect the decrease in the range of IOP fluctuation when conducting a paired t-test with two-sided 5 % type I error.
The research protocol was approved by the Institutional Review Board of the University of Toyama and the procedures used conformed to the tenets of the Declaration of Helsinki. After the nature and possible consequences of the study were explained to the patients, written informed consent was obtained from each patient.
Results
Ophthalmic data and cataract surgery
Table 1 shows the ophthalmic data of the patients. The study was performed on ten eyes of ten patients (one male and nine females: mean age 72 ± 6 years). No patients took oral Acetazolamide. The average values of the central anterior chamber depth, lens vault, central corneal thickness (CCT), and axial length were 2.01 ± 0.28 (mm), 1,057 ± 221 (μm), 537 ± 36 (μm), and 22.30 ± 0.57 (mm), respectively. All surgeries were performed successfully and no complications occurred.
The mean visual acuity improved from 0.036 ± 0.13 (logMAR±standard deviation) before PEA+IOL to −0.047 ± 0.07 after PEA+IOL (P = 0.003). The mean baseline IOP was 14.6 ± 1.5 mmHg. The mean IOP at 3 months after PEA+IOL was significantly reduced to 11.1 ± 2.2 mmHg, compared to the mean baseline IOP (P = 0.002). By analysis of AS-OCT images, the mean anterior opening distance at 500 μm from the scleral spur and the mean trabecular iris angle at 500 μm from the scleral spur before PEA+IOL were 0.20 ± 0.1 mm and 16.4 ± 8.2°, respectively. These significantly improved to 0.47 ± 0.14 mm (P < 0.001) and 40.0 ± 11.1° (P < 0.001) after PEA+IOL.
Circadian IOP patterns
IOP fluctuations over 24 h in each patient were successfully recorded using a CLS. Patients showed no serious complications related to the Triggerfish® CLS. We converted the original IOP fluctuation graphs recorded every 5 min to IOP fluctuation graphs with mean fluctuations of every hour (Fig. 2a, b) and evaluated the circadian patterns of IOP changes. Before PEA+IOL, six of ten eyes showed peak values during the nocturnal period (nocturnal acrophase group) and two eyes showed peak values during the diurnal period (diurnal acrophase group) (Table 2). Two eyes were included in the no significant acrophase group because these two eyes did not show any specific patterns before and after PEA+IOL. At three months after PEA+IOL, eight of the ten eyes (80 %) showed the same circadian IOP pattern. One eye changed from the nocturnal acrophase to the diurnal acrophase, and another one eye changed from the diurnal acrophase to the nocturnal acrophase.
a Diagram of 24-h continuous IOP fluctuations recorded with a Triggerfish® CLS before PEA+IOL. The diagram was redrawn with the mean IOP fluctuations every hour. This was the result of case No.10. b Diagram of continuous IOP fluctuations in the same eye over 24 h recorded with a Triggerfish® CLS after PEA+IOL. The dotted line represents the nocturnal period and the solid line represents the diurnal period. The peak IOP and IOP fluctuations were decreased after PEA+IOL
Measurement of IOP fluctuations
The ranges of IOP fluctuations over 24 h before and after PEA+IOL are shown in Table 3. The range of IOP fluctuations over 24 h decreased after PEA+IOL in five of the ten eyes. The ranges of IOP fluctuations in the nocturnal period were smaller than those in the diurnal period in nine of the ten eyes before PEA+IOL and all eyes after PEA+IOL (Table 3).
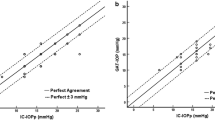
As shown in Fig. 3, the mean ranges of preoperative IOP fluctuations were 425 ± 54 mVeq (24 h), 407 ± 72 mVeq (diurnal period), and 241 ± 61 mVeq (nocturnal period). The mean ranges of postoperative IOP fluctuations were 410 ± 73 mVeq (24 h), 409 ± 74 mVeq (diurnal period), and 179 ± 64 mVeq (nocturnal period). The mean range of IOP fluctuations over 24 h was 425 ± 54 mVeq before PEA+IOL and 410 ± 73 mVeq after PEA+IOL (P = 0.49) (Fig. 3a). During the diurnal period, the mean range of IOP fluctuations was 407 ± 72 mVeq before PEA+IOL and 409 ± 73 mVeq after PEA+IOL (P = 0.92) (Fig. 3b). On the other hand, during the nocturnal period, PEA+IOL significantly decreased the range of IOP fluctuations from 241 ± 61 mVeq to 179 ± 64 mVeq (P = 0.02) (Fig. 3c). Nine of the ten eyes decreased the amplitude of IOP fluctuations during the nocturnal period after PEA+IOL (Table 3).
Corneal effects of the CLS
We chose a size of the CLS from three types of the CLS according to the base curve of the cornea in each eye. In this study, all patients wore the medium size of the CLS. To examine the effects of the CLS on the cornea, we analyzed corneal changes before and after measurement with the CLS. The average steeper meridian was largely unchanged, being 45.2 ± 1.9 diopter (D) before measurement and 45.8 ± 2.2 D after measurement (P = 0.07). The average flatter meridian was also basically unchanged: 44.4 ± 1.6 D before measurement and 44.6 ± 2.0 D after measurement (P = 0.59). The average CCT was also unchanged: 540 ± 33 (μm) before measurement and 547 ± 38 (μm) after measurement (P = 0.06).
Discussion
Cataract surgery in PACG patients has been shown to improve the anterior chamber depth and aqueous humor outflow, resulting in an improvement of IOP control and visual acuity [8–11, 16]. Similarly to the previous studies [9–11], we also demonstrated that PEA+IOL significantly improved the mean IOP and anatomical parameters of the anterior chamber, such as the lens vault and the mean trabecular iris angle, in the PACG patients.
The reproducibility of the measurement of the circadian IOP pattern with the CLS was proved acceptable [7, 15, 17, 18], and our results supported it because the circadian IOP patterns were the same in eight of the ten eyes before and after PEA+IOL in this study. The CLS is the only method to measure habitual IOP fluctuations for 24 h at present. Mansouri et al., reported that the nocturnal acrophase was seen in 62.9 % of glaucoma patients by analysis with the CLS [15]. Pajic B et al., reported that IOP fluctuations measured by the CLS were flattened by medication in the patients with normal tension glaucoma [18]. Even though the CLS does not measure IOP of mmHg, measurement of IOP fluctuations with the CLS is reproducible and useful to evaluate circadian IOP rhythm and effects on glaucoma treatments.
Cataract surgeries were shown to reduce diurnal IOP fluctuations in patients of pseudo exfoliation with both open and occludable angles as well as in patients with primary open angle glaucoma [19, 20]. Our results showed a significant decrease of IOP fluctuations in the nocturnal period only at three months after PEA+IOL. Further studies are needed to examine long-term effects of cataract surgery on IOP fluctuations in glaucoma patients.
The measurement of IOP with the CLS has some limitations. First, it is not possible to convert the recorded units of mVeq to units of mmHg. Since the values of mVeq recorded with a Triggerfish® CLS did not directly show the values of mmHg, we evaluated only relative changes of mVeq. Secondly, artifacts of measurements by the CLS method could have an impact on the results. Changes in the corneal curvatures due to corneal hypoxia may have affected the values of mVeq during the 24-h measurement of IOP fluctuations. In this study, the mean steeper meridian was not significantly increased (P = 0.07), but it was reported that central corneal thickness and corneal curvature irregularities slightly increased with the Triggerfish® CLS [21].
In conclusion, continuous 24-h IOP monitoring with the CLS appears to be useful in the management of patients with glaucoma. Measurement of IOP fluctuations with the CLS revealed that cataract surgery resulted in a significant reduction of the range of IOP fluctuations during the nocturnal period in PACG patients. These results support the idea that PEA+IOL would contribute to the prevention of visual field loss in PACG patients.
Abbreviations
- AS-OCT:
-
Anterior segment optical coherence tomography
- CLS:
-
Contact lens sensor
- IOP:
-
Intraocular pressure
- PACG:
-
Primary angle closure glaucoma
- PEA+IOL:
-
Phacoemulsification and intraocular lens
- POAG:
-
Primary open angle glaucoma
References
Quigley HA, Broman AT (2006) The number of people with glaucoma worldwide in 2010 and 2020. Br J Ophthalmol 90:262–267
Rhee K, Kim YY, Nam DH, Jung HR (2001) Comparison of visual field defects between primary open-angle glaucoma and chronic primary angle-closure glaucoma in the early or moderate stage of the disease. Korean J Ophthalmol 15:27–31
Baskaran M, Kumar RS, Govindasamy CV, Htoon HM, Wong CY, Perera SA, Wong TT, Aung T (2009) Diurnal intraocular pressure fluctuation and associated risk factors in eyes with angle closure. Ophthalmology 116:2300–2304
Asrani S, Zeimer R, Wilensky J, Gieser D, Vitale S, Lindenmuth K (2000) Large diurnal fluctuations in intraocular pressure are an independent risk factor in patients with glaucoma. J Glaucoma 9:134–142
Tojo N, Oka M, Miyakoshi A, Ozaki H, Hayashi A (2013) Comparison of Fluctuations of Intraocular Pressure Before and After Selective Laser Trabeculoplasty in Normal-tension Glaucoma Patients. J Glaucoma Nov 16. [Epub ahead of print]
De Smedt S, Mermoud A, Schnyder C (2012) 24-hour intraocular pressure fluctuation monitoring using an ocular telemetry Sensor: tolerability and functionality in healthy subjects. J Glaucoma 21:539–544
Mansouri K, Medeiros FA, Tafreshi A, Weinreb RN (2012) Continuous 24-hour monitoring of intraocular pressure patterns with a contact lens sensor: safety, tolerability, and reproducibility in patients with glaucoma. Arch Ophthalmol 130:1534–1539
Amerasinghe N, Aung T (2008) Angle-closure: risk factors, diagnosis and treatment. Prog Brain Res 173:31–45
Lam DS, Leung DY, Tham CC, Li FC, Kwong YY, Chiu TY, Fan DS (2008) Randomized trial of early phacoemulsification versus peripheral iridotomy to prevent intraocular pressure rise after acute primary angle closure. Ophthalmology 115:1134–1140
Yudhasompop N, Wangsupadilok B (2012) Effects of phacoemulsification and intraocular lens implantation on intraocular pressure in primary angle closure glaucoma (PACG) patients. J Med Assoc Thai 95:557–560
Liu XQ, Zhu HY, Su J, Hao XJ (2013) Effects of phacoemulsification on intraocular pressure and anterior chamber depth. Exp Ther Med 5:507–510
Foster PJ, Buhrmann R, Quigley HA, Johnson GJ (2002) The definition and classification of glaucoma in prevalence surveys. Br J Ophthalmol 86:238–242
Leonardi M, Leuenberger P, Bertrand D, Bertsch A, Renaud P (2004) First steps toward noninvasive intraocular pressure monitoring with a sensing contact lens. Invest Ophthalmol Vis Sci 45:3113–3117
Leonardi M, Pitchon EM, Bertsch A, Renaud P, Mermoud A (2009) Wireless contact lens sensor for intraocular pressure monitoring: assessment on enucleated pig eyes. Acta Ophthalmol 87:433–437
Mansouri K, Liu JH, Weinreb RN, Tafreshi A, Medeiros FA (2012) Analysis of continuous 24-hour intraocular pressure patterns in glaucoma. Invest Ophthalmol Vis Sci 53:8050–8056
Lai JS, Tham CC, Chan JC (2006) The clinical outcomes of cataract extraction by phacoemulsification in eyes with Primary Angle-Closure Glaucoma (PACG) and co-existing cataract: a prospective case series. J Glaucoma 15:47–52
Mottet B, Aptel F, Romanet JP, Hubanova R, Pépin JL, Chiquet C (2013) 24-hour intraocular pressure rhythm in young healthy subjects evaluated with continuous monitoring using a contact lens sensor. JAMA Ophthalmol 131:1507–1516
Pajic B, Pajic-Eggspuchler B, Haefliger I (2011) Continuous IOP fluctuation recording in normal tension glaucoma patients. Curr Eye Res 36:1129–1138
Rao A (2012) Diurnal curve after phacoemulsification in patients with pseudoexfoliation syndrome and cataract. Semin Ophthalmol 27:1–5
Sacca S, Marletta A, Pascotto A, Barabino S, Rolando M, Giannetti R, Calabria G (2001) Daily tonometric curves after cataract surgery. Br J Ophthalmol 85:24–29
Hubanova R, Aptel F, Chiquet C, Mottet B, Romanet JP (2013) Effect of overnight wear of the Triggerfish® sensor on corneal thickness measured by Visante® anterior segment optical coherence tomography. Acta Ophthalmol 92:e119–e123
Conflicts of interest
The authors declare that they have no conflicts of interest that are relevant to the content of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tojo, N., Otsuka, M., Miyakoshi, A. et al. Improvement of fluctuations of intraocular pressure after cataract surgery in primary angle closure glaucoma patients. Graefes Arch Clin Exp Ophthalmol 252, 1463–1468 (2014). https://doi.org/10.1007/s00417-014-2666-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-014-2666-7