Abstract
Purpose
According to the World Health organization (WHO), more than 10% in people older than 60 years suffer from osteoarthritis (OA). Over the last years, there has been an increased interest around regenerative medicine, especially regarding stem cell treatments and related applications. We hypothesize that stem cell therapies can represent a feasible option for idiopathic knee OA, delaying or even avoiding the joint replacement. To emphasize the potential of percutaneous injections of mesenchymal stem cells for knee OA, a comprehensive systematic review of the literature was conducted.
Material and methods
Two independent authors (FM, GC) performed the literature search. This study was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines (PRISMA). The main databases were accessed: Pubmed, Embase, Google Scholar, Cochrane Systematic Reviews, Scopus, AMED. For this systematic review, all articles treating percutaneous injections of mesenchymal stem cells for knee OA were considered. Because of the rapid advancements promoted by the scientific progress on stem cell expansion and processing, only articles published within the last five years were included. Solely articles reporting the outcomes of interest across 6- and 12-month follow-up were recruited for eligibility. We included only studies reporting quantitative data under the outcomes of interest. We referred for the quality assessment to the Coleman Methodology Score (CMS). The statistical analysis was performed with Review Manager Software 5.3 (The Nordic Cochrane Centre, Copenhagen).
Results
A total of 18 studies were enrolled in the present study, comprising 1069 treated knees. The mean age of the samples was 57.39 ± 7.37 years. 72% of the included studies harvested the stem cells from the iliac crest (bone marrow-derived MSCs), the remaining 28% from the adipose tissue (adipose-derived MSCs). The mean visual analogic scale improved from 18.37 to 30.98 and 36.91 at 6- and 12-month follow-up, respectively. The mean WOMAC score improved from 25.66 to 25.23 and 15.60 at 6- and 12-month follow-up, respectively. The mean walking distance improved from 71.90 to 152.22 and 316.72 at 6- and 12-month follow-up, respectively. The mean Lequesne scale improved from 33.76 to 12.90 at 12-month follow-up. The KOOS score improved from 41.07 to 8.47% and 18.94 at 6- and 12-month follow-up. All the KOOS subscales improved significantly from the baseline. A total of 136 (12.7%) local complications were detected.
Conclusion
According to the current evidences and the main findings of this systematic review, we reported that MSC infiltrations for knee OA can represent a feasible option, leading to an overall remarkable improvement of all clinical and functional considered outcomes, regardless of the cell source. Patients treated at earlier-degeneration stages reported statistically significant greater outcomes. The pain and function scores were improved considerably, thus, leading to a significant improvement of patient participation in recreational activities and quality of life.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
According to the World Health organization (WHO), more than 10% of people older than 60 years suffer from osteoarthritis (OA) [1]. The first approach for symptomatic knee OA is represented by analgesics and nonsteroidal anti-inflammatory drugs (NSAIDs). These drugs lead to gastrointestinal and cardiovascular adverse events in the long run [2]. Furthermore, they are not able to stop the OA cascade, or even to guarantee a long-term pain relief [3]. Platelet-rich plasma, hyaluronic, corticosteroids, and local anaesthetic injections represent additional options for early stage joint degeneration; however, the long-term results are poor and lack professional consensus [4,5,6]. Knee arthroplasties (partial or total) remain the gold standard treatments for end stage OA, reporting high clinical and functional outcomes and being cost-effective [7, 8]. On the downside, they expose the patients to the risk of several complications and further revision surgeries [9].
Regenerative medicine, especially stem cell therapies, attracts more attention from the scientific communities than ever by achieving promising results [10, 11]. From a theoretical point of view, stem cells can be committed in every cell lineage in order to replace and repair damaged human tissues [12, 13]. Stem cell therapies are closely connected/linked to the progress of other disciplines, such as molecular biology, which is essential to understand signalling pathways, proliferations, differentiations and expansion patterns [14, 15].
We hypothesize that stem cell therapies may represent feasible options for idiopathic knee OA, delaying joint replacement. Several studies have attempted to delineate these therapies, but there is no recent study to have reviewed the latest evidence, indications, and outcomes. This review aims to update the current state of research concerning the potential of percutaneous injections of mesenchymal stem cells for knee OA. We tried to clarify the current indications and to summarize biological pathways supporting these infiltrations, along with the outcomes and criteria of patient selection.
Materials and methods
Search strategy
Two independent authors (FM, GC) performed the literature search. This study was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines (PRISMA) [16]. A preliminary protocol was carried out to guide the initial research:
-
(a)
Population: knee OA;
-
(b)
Intervention: percutaneous mesenchymal stem cells injection;
-
(c)
Outcomes: clinical and functional scores, further complications.
Data extraction
The main databases were accessed in December 2018: Pubmed, Embase, Google Scholar, Cochrane Systematic Reviews, Scopus, AMED. The following keywords were used in combination: knee osteoarthritis and/ or degeneration combined with mesenchymal stem cells (MSC) and/ or bone marrow-derived or adipose-derived or peripheral blood-derived combined with injection and/ or percutaneous. If the title matched, the abstract was carefully examined and, if suitable, the full-text was accessed. Furthermore, the bibliographies of all relevant studies were evaluated for inclusion as well.
Eligibility criteria
Two independent authors (FM, GC) screened the articles resulting from the search for suitability/eligibility. For this systematic review, all articles treating percutaneous injections of mesenchymal stem cells for knee OA were considered. In accordance with the authors’ capabilities, articles in English, German, Italian, Spanish, and French were considered. Correspondent to the Oxford Centre of Evidence-Based Medicine [17], levels of evidence I, II, and III were included. Due to rapid advancements promoted by the scientific progress on stem cells expansion and processing, only articles published in the last 5 years were included. Articles discussing infiltrations with proteins, collagens, fibrins or other components of the extracellular matrix were excluded. Studies considering infiltrations with chondrocytes, osteocytes, synoviocytes, erythrocytes of platelets or another committed lineage were rejected. Studies performing infiltration of bone marrow aspirate (BMA) or platelet-rich plasma (RPR) were excluded. Studies addressing allogeneic or heterogenic transplants, along with studies discussing embryonal or umbilical cord stem cells were excluded. Moreover, studies involving totipotent as pluripotent and other less committed stem lineages were excluded. Additionally, papers treating patients with previous or planned knee surgery, along with studies infiltrating cells under arthroscopic guidance and studies treating patients suffering from acute traumas, chondropathies, focal or multiple chondral defects were rejected. Solely articles reporting the outcomes of interest across 6- and 12-month follow-up were regarded as suitable. We enrolled studies treating patients suffering from OA with percutaneous injections of mesenchymal stem cells only. Similarly, they were required to report quantitative data under the outcomes of interest.
Outcomes of interest
Two independent authors (FM, GC) performed the data collection. For each article, the following data were extracted: author, year, type of study, mean age and follow-up, number of patients, control group, cell source, dose injected, inclusion and exclusion criteria, and further complications. The following scores were considered: visual analogic scale (VAS), WOMAC [18], Knee injury and Osteoarthritis Outcome Score (KOOS) and related subscales [19], Walking distance (meters), and Lequensne index [20]. We divided all studies into two groups, depending on the length of the follow-ups of either 6 or 12 months. The following subgroup analyses was performed: according to the donor-tissue source (adipose- versus bone marrow-derived MSCs) and according to the Kellgren and Lawrence scale (grade II to III versus II to IV).
Methodological quality assessment
For the quality assessment, we referred to the Coleman Methodology Score (CMS). The values related to each article were assessed independently by two authors (FM, GC). This score evaluates the included studies under different points of view: number of enrolled patients, mean follow-up, type of approach and study, description of diagnosis, surgical technique and post-operative rehabilitation. Furthermore, the criteria, the procedures, and the selection process are evaluated. For final evaluation, the CMS results in a value ranging from 0 (poor) to 100 (excellent).
Statistical analysis
The statistical analysis was performed with Review Manager Software 5.3 (The Nordic Cochrane Centre, Copenhagen). To evaluate continuous data and related overall effect estimate (EE), the arithmetic mean and standard deviation (SD) were calculated. The inverse variance (IV) statistical method with the mean difference was used to evaluate the level of improvement across the follow-ups. The confidence interval (CI) was set to 95% for the entire comparison. Both \(\chi\)2 and I2 (Higgins) tests were performed to assess the heterogeneity. A fixed effect method was initially used. If \(\chi\)2 resulted in P > 0.5 and the I2 > 50%, the comparison was analysed under a random effect analysis method. Values of P > 0.5 were considered statistically significant.
Results
Search result
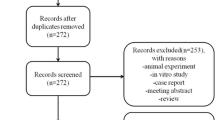
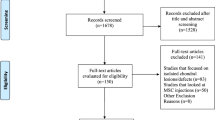
The literature research resulted in 3512 articles. After removing duplicates, 3288 articles in total were screened for inclusion. A total of 2474 was excluded because they did not match the eligibility criteria. Another 537 were excluded due to lack of data in regard to the outcomes of interest. Further, 239 articles were excluded because of insufficient quantitative data. An additional 13 studies were rejected due to poor quality or ambiguous results. 7 articles were excluded because no data was reported regarding the mentioned follow-up periods (6 and/or 12 months). This left 18 articles for this systematic review. The literature flow-chart is shown in Fig. 1.
Methodological quality assessment
The CMS scored 59.8 ± 7.96 (good quality). Discussing and contextualizing this result are immensely important, since the CMS was negatively influenced by the lack of randomization, which was merely applied to 11% of the included studies. However, only one included study was retrospective, while the other 83% were prospective, representing the improved methodological quality of this work. In consequence of the inept blinding and randomization of the abovementioned treatments, it becomes apparent that the score underestimates their overall quality. In conclusion, we validated the superior quality of the methodological assessment. The results of the CMS of each study are shown in Table 1.
Patients demographic
A total of 1069 knees were enrolled in the present study. The mean age of the samples was 57.39 ± 7.37 years. Three studies took advantage of a control group. 72% of all studies harvested the stem cells from the iliac crest (bone marrow-derived MSCs), whereas 28% harvested from the adipose tissue (adipose-derived MSCs). The mean volume injected into the joint was 39.01 ml. The demographic baseline of the studied groups is shown in Table 1.
Outcomes of interest
The mean visual analogic scale improved from a baseline of 55.20 ± 18.37 to 30.98 and 36.91 at 6- and 12-month follow-up, respectively. The mean WOMAC score improved from a baseline of 25.66 ± 15.10 to 25.23 and 15.60 at 6- and 12-month follow-up, respectively. Likewise, the mean walking distance improved from a baseline of 71.90 ± 28.41 m to 152.22 and 316.72 m at 6- and 12-month follow-up, respectively. The mean Lequesne scale improved from a baseline of 33.76 ± 19.72 to 12.90 at 12-month follow-up. The KOOS score improved from a baseline of 41.07 ± 12.17 to 8.47 and 18.94 at 6- and 12-month follow-up. In addition, all the KOOS subscales improved significantly from the baseline to both 6- and 12-month follow-up. The overall results of the comparison are shown in Table 2.
Subgroup analysis
Due to lack of quantitative data, in the subgroup analysis only the VAS, WOMAC and walking distance were evaluated. Concerning donor source (adipose vs bone marrow), no statistical differences were found concerning VAS (EE 3.97; 95% CI 0.01–5.15, P = 0.68), WOMAC (EE 5.12; 95% CI 3.56–6.99; P = 0.21) and walking distance (EE 2.17; 95% CI 1.36–3.16; P = 0.48). Concerning the degree of degeneration, the Kellgren and Lawrence II to III evidenced statistically significant greater VAS (EE 15.79; 95% CI 11.91–16.77, P = 0.03), WOMAC (EE 9.94; 95% CI 5.40–11.99; P = 0.05) and walking distance (EE 27.51; 95% CI 18.49–33.15; P = 0.004).
Complications
A total of 136 (12.7%) local complications were detected. In 130 cases, pain and swelling were reported: of these, 35 were rated as mild, 2 as moderate, and 2 as intense knee pain. Other complications included one case of skin reaction, two cases of allergic reactions, and two hematomas. Complications requiring surgery during the follow-up time were: one total knee replacement and one acute meniscus lesion.
Discussion
This systematic found that MSC infiltrations for knee OA can represent a feasible option, leading to an overall remarkable improvement of all clinical and functional considered outcomes with a very low complication rate during the follow-up duration. Any statistically significant difference among adipose- and bone marrow-derived MSCs were found. Patients treated at earlier-degeneration stages reported statistically significant greater VAS, WOMAC and walking distance. The pain and function scores were improved considerably, thus, leading to a significant improvement of patient participation in recreational activities and quality of life.
Several options for knee infiltration have been suggested as conservative treatment for OA. Corticosteroids (CCS) infiltrations have been used as palliative treatment for advanced OA for many years [40]. The CCS inhibit the inflammatory cascade, causing a temporary relief from OA symptoms [41]. However, destructive effects on the articular cartilage have been extensively documented [42,43,44,45]. On the contrary, the MSCs encourage the differentiation and proliferation, negatively modulating the inflammatory cascade promoting the articular healing [46,47,48,49]. Moreover, CCS are not recommended in concomitance with MSCs infiltrations. The CCS can dose-dependently reverse the therapeutic effect of MSCs [50]. This is supported by in vitro and in vivo observations [51, 52]. However, the correlation between CCS and MSCs is still not completely clarified and requires further studies [53]. In addition, the authors excluded patients who previously underwent local anaesthetic injections since they have a cytotoxic effect on MSCs [54, 55]. Platelet rich plasma (PRP) have been extensively used in the orthopaedic and trauma surgery. Platelets are committed leucocytes derived from the fragmentation of the precursor megakaryocytes [56]. They represent a source of growth factors, promoting tissue healing and regenerative processes [57,58,59]. However, results concerning PRP are contrasting and no consensus has been reached [60,61,62]. Platelet are extracted, concentrated and re-implanted in the same day, requiring minimal cell manipulation: same characteristics for the bone marrow aspirate (BMA) [23]. The BMA is a niche of cells with multiple degree of differentiation and lineage commitment. However, the quality and quantity of cells present within the aspiration are not adequate. The estimated amount of MSCs in BMA is between the 0.01% and 0.001% [63, 64]. On the opposite, MSCs injection shows several methods of processing, culture preparation/expansion and delivery, and the various adjuvants and diluents involved. MSCs being not committed, have high proliferation and differentiation potential, can modulate the immune answer and tissue trophism [65,66,67].
The authors of said studies referred to different stages of the Kellgren and Lawrence Scale in their criteria [68]. Some patients were treated at early stages of osteoarthritic knee degeneration. The MSCs could potentially reverse these stages of degeneration, differentiating into every cell derived from the mesoderm germ layer including chondroblasts, adipocytes, and osteocytes [69]. The process of allocating stem cells is called “homing” and is followed by differentiation and proliferation, regenerating the damaged tissue, and healing the intraarticular cascade [46]. These processes are characterized by a wide production of growth factors, cytokines, and chemokines, giving life to a signalling pattern between the environment and the MSCs [47, 70]. In the early stages of the Kellgren and Lawrence Scale, a minimally viable substrate can still be recognized: the required condition to generate the signalling pattern. In animal models, MSCs have been successfully transplanted, reporting considerable clinical improvement and better outcomes compared to controls [71,72,73,74]. In accordance with the Kellgren and Lawrence Scale [68], other authors injected their patients at advanced or end-stage degeneration. If the environment is irreparably damaged, however, how can stem cells interact with them? What is the role of stem cell infiltration? In addition to their homing ability, stem cells showed intrinsic immunomodulation ability [75]. Stem cells interact with the NK cells, macrophages and lymphocytes, inhibiting the proliferation, chemiotaxis, and promoting cytotoxic action of immune cells [46, 48, 49, 76, 77]. With the OA also being characterized by the activation of inflammatory and catabolic cascades [78, 79], it becomes apparent that patients suffering from knee OA can still experience relief and improvement of the aforementioned scores.
This study has several limitations; therefore, data must be interpreted with caution. The most important limitation of the present study are the heterogeneous methods of processing, culture preparation/expansion and delivery, and the various adjuvants and diluents involved. This underlines how our competences are not yet sufficient to understand which is the most effective methods of dealing with MSCs. Furthermore, the different legislations of certain countries that limit or prohibit the use of MSCs in humans, having a negative impact on the overall development and knowledges of MSCs. Other considerable limitations were the heterogeneous inclusion and exclusion criteria and lack of appropriate controls, representing remarkable sources of selection bias, purposely done to increase the pooling data. Further significant limitations exist due to the low level of evidence of the included studies and the limited follow-up duration. Based on a lack of existing data, it was not possible to analyse other follow-up terms. Points of strength of this study are the comprehensive nature of the research, along with the strict eligibility criteria. We excluded several works to ensure the best evidence possible concerning these increasingly expanding therapies. In the literature there are contrasting evidences and a lack of consensus regarding the best cell source (bone marrow, adipose, peripheral blood) and further studies should be addressed to clarify this point. Due to lack of evidences and data in the literature, we only focused in adipose- and bone marrow-derived stem cells. This represent a limitation of this study. Further study should also provide randomization and blinding methods, along with a longer follow-up and group control.
Conclusion
According to the current evidences and the main findings of this systematic review, we reported that MSC infiltrations for knee OA can represent a feasible option, leading to an overall remarkable improvement of all clinical and functional considered outcomes with a very low complication rate during the follow-up duration. No difference among adipose- and bone marrow-derived MSCs were found. Patients treated at earlier-degeneration stages reported statistically significant greater outcomes. The pain and function scores were improved considerably, thus, leading to a significant improvement of patient participation in recreational activities and quality of life.
References
Runhaar J, van Middelkoop M, Reijman M, Willemsen S, Oei EH, Vroegindeweij D, van Osch G, Koes B, Bierma-Zeinstra SM (2015) Prevention of knee osteoarthritis in overweight females: the first preventive randomized controlled trial in osteoarthritis. Am J Med 128(8):888–895. https://doi.org/10.1016/j.amjmed.2015.03.006
Harirforoosh S, Asghar W, Jamali F (2013) Adverse effects of nonsteroidal antiinflammatory drugs: an update of gastrointestinal, cardiovascular and renal complications. J Pharm Pharm Sci 16(5):821–847
Felson DT, Lawrence RC, Hochberg MC, McAlindon T, Dieppe PA, Minor MA, Blair SN, Berman BM, Fries JF, Weinberger M, Lorig KR, Jacobs JJ, Goldberg V (2000) Osteoarthritis: new insights Part 2: treatment approaches. Ann Intern Med 133(9):726–737
Fibel KH, Hillstrom HJ, Halpern BC (2015) State-of-the-Art management of knee osteoarthritis. World J Clin Cases 3(2):89–101. https://doi.org/10.12998/wjcc.v3.i2.89
Pourcho AM, Smith J, Wisniewski SJ, Sellon JL (2014) Intraarticular platelet-rich plasma injection in the treatment of knee osteoarthritis: review and recommendations. Am J Phys Med Rehabil 93(11 Suppl 3):S108–121. https://doi.org/10.1097/PHM.0000000000000115
Jevsevar D, Donnelly P, Brown GA, Cummins DS (2015) Viscosupplementation for osteoarthritis of the knee: a systematic review of the evidence. J Bone Joint Surg Am 97(24):2047–2060. https://doi.org/10.2106/JBJS.N.00743
Peersman G, Jak W, Vandenlangenbergh T, Jans C, Cartier P, Fennema P (2014) Cost-effectiveness of unicondylar versus total knee arthroplasty: a Markov model analysis. Knee 21(Suppl 1):S37–42. https://doi.org/10.1016/S0968-0160(14)50008-7
Song EK, Agrawal PR, Kim SK, Seo HY, Seon JK (2016) A randomized controlled clinical and radiological trial about outcomes of navigation-assisted TKA compared to conventional TKA: long-term follow-up. Knee Surg Sports Traumatol Arthrosc 24(11):3381–3386. https://doi.org/10.1007/s00167-016-3996-2
Healy WL, Della Valle CJ, Iorio R, Berend KR, Cushner FD, Dalury DF, Lonner JH (2013) Complications of total knee arthroplasty: standardized list and definitions of the Knee Society. Clin Orthop Relat Res 471(1):215–220. https://doi.org/10.1007/s11999-012-2489-y
Aicher WK, Buhring HJ, Hart M, Rolauffs B, Badke A, Klein G (2011) Regeneration of cartilage and bone by defined subsets of mesenchymal stromal cells–potential and pitfalls. Adv Drug Deliv Rev 63(4–5):342–351. https://doi.org/10.1016/j.addr.2010.12.004
Caplan AI, Dennis JE (2006) Mesenchymal stem cells as trophic mediators. J Cell Biochem 98(5):1076–1084. https://doi.org/10.1002/jcb.20886
Akgun I, Unlu MC, Erdal OA, Ogut T, Erturk M, Ovali E, Kantarci F, Caliskan G, Akgun Y (2015) Matrix-induced autologous mesenchymal stem cell implantation versus matrix-induced autologous chondrocyte implantation in the treatment of chondral defects of the knee: a 2-year randomized study. Arch Orthop Trauma Surg 135(2):251–263. https://doi.org/10.1007/s00402-014-2136-z
Reichenbach S, Blank S, Rutjes AW, Shang A, King EA, Dieppe PA, Juni P, Trelle S (2007) Hylan versus hyaluronic acid for osteoarthritis of the knee: a systematic review and meta-analysis. Arthritis Rheum 57(8):1410–1418. https://doi.org/10.1002/art.23103
Pecina M, Vukicevic S (2007) Biological aspects of bone, cartilage and tendon regeneration. Int Orthop 31(6):719–720. https://doi.org/10.1007/s00264-007-0425-7
Lind M, Bunger C (2005) Orthopaedic applications of gene therapy. Int Orthop 29(4):205–209. https://doi.org/10.1007/s00264-005-0650-x
Moher D, Liberati A, Tetzlaff J, Altman DG, Group P (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ 339:b2535. https://doi.org/10.1136/bmj.b2535
Howick JCI, Glasziou P, Greenhalgh T, Heneghan C, Liberati A, Moschetti I, Phillips B, Thornton H, Goddard O, Hodgkinson M (2011) The 2011 Oxford Levels of Evidence. Oxford Centre for Evidence-Based Medicine Available at https://www.cebm.net/index.aspx?o=5653
Walker LC, Clement ND, Bardgett M, Weir D, Holland J, Gerrand C, Deehan DJ (2018) The WOMAC score can be reliably used to classify patient satisfaction after total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc. https://doi.org/10.1007/s00167-018-4879-5
Roos EM, Lohmander LS (2003) The Knee injury and Osteoarthritis Outcome Score (KOOS): from joint injury to osteoarthritis. Health Qual Life Outcomes 1:64. https://doi.org/10.1186/1477-7525-1-64
Lecorney J, Verhoeven F, Chouk M, Guillot X, Prati C, Wendling D (2018) Correlation between catastrophizing and Lequesne index in case of osteoarthritis of the knee: a prospective study. Joint Bone Spine 85(5):605–607. https://doi.org/10.1016/j.jbspin.2017.10.002
Al-Najar M, Khalil H, Al-Ajlouni J, Al-Antary E, Hamdan M, Rahmeh R, Alhattab D, Samara O, Yasin M, Abdullah AA, Al-Jabbari E, Hmaid D, Jafar H, Awidi A (2017) Intra-articular injection of expanded autologous bone marrow mesenchymal cells in moderate and severe knee osteoarthritis is safe: a phase I/II study. J Orthop Surg Res 12(1):190. https://doi.org/10.1186/s13018-017-0689-6
Bastos R, Mathias M, Andrade R, Bastos R, Balduino A, Schott V, Rodeo S, Espregueira-Mendes J (2018) Intra-articular injections of expanded mesenchymal stem cells with and without addition of platelet-rich plasma are safe and effective for knee osteoarthritis. Knee Surg Sports Traumatol Arthrosc. https://doi.org/10.1007/s00167-018-4883-9
Centeno C, Pitts J, Al-Sayegh H, Freeman M (2014) Efficacy of autologous bone marrow concentrate for knee osteoarthritis with and without adipose graft. Biomed Res Int 2014:370621. https://doi.org/10.1155/2014/370621
Davatchi F, Abdollahi BS, Mohyeddin M, Nikbin B (2016) Mesenchymal stem cell therapy for knee osteoarthritis: 5 years follow-up of three patients. Int J Rheum Dis 19(3):219–225. https://doi.org/10.1111/1756-185X.12670
Emadedin M, Aghdami N, Taghiyar L, Fazeli R, Moghadasali R, Jahangir S, Farjad R, Baghaban Eslaminejad M (2012) Intra-articular injection of autologous mesenchymal stem cells in six patients with knee osteoarthritis. Arch Iran Med 15(7):422–428. https://doi.org/012157/AIM.0010
Emadedin M, Ghorbani Liastani M, Fazeli R, Mohseni F, Moghadasali R, Mardpour S, Hosseini SE, Niknejadi M, Moeininia F, Aghahossein Fanni A, Baghban Eslaminejhad R, Vosough Dizaji A, Labibzadeh N, Mirazimi Bafghi A, Baharvand H, Aghdami N (2015) Long-term follow-up of intra-articular injection of autologous mesenchymal stem cells in patients with knee, ankle, or hip osteoarthritis. Arch Iran Med 18(6):336–344. https://doi.org/015186/AIM.003
Fodor PB, Paulseth SG (2016) Adipose derived stromal cell (ADSC) injections for pain management of osteoarthritis in the human knee joint. Aesthet Surg J 36(2):229–236. https://doi.org/10.1093/asj/sjv135
Garay-Mendoza D, Villarreal-Martinez L, Garza-Bedolla A, Perez-Garza DM, Acosta-Olivo C, Vilchez-Cavazos F, Diaz-Hutchinson C, Gomez-Almaguer D, Jaime-Perez JC, Mancias-Guerra C (2018) The effect of intra-articular injection of autologous bone marrow stem cells on pain and knee function in patients with osteoarthritis. Int J Rheum Dis 21(1):140–147. https://doi.org/10.1111/1756-185X.13139
Goncars V, Kalnberzs K, Jakobsons E, Engele I, Briede I, Blums K, Erglis K, Erglis M, Patetko L, Muiznieks I, Erglis A (2018) Treatment of knee osteoarthritis with bone marrow-derived mononuclear cell injection: 12-month follow-up. Cartilage:1947603517746721. https://doi.org/10.1177/1947603517746721
Lamo-Espinosa JM, Mora G, Blanco JF, Granero-Molto F, Nunez-Cordoba JM, Sanchez-Echenique C, Bondia JM, Aquerreta JD, Andreu EJ, Ornilla E, Villaron EM, Valenti-Azcarate A, Sanchez-Guijo F, Del Canizo MC, Valenti-Nin JR, Prosper F (2016) Intra-articular injection of two different doses of autologous bone marrow mesenchymal stem cells versus hyaluronic acid in the treatment of knee osteoarthritis: multicenter randomized controlled clinical trial (phase I/II). J Transl Med 14(1):246. https://doi.org/10.1186/s12967-016-0998-2
Lamo-Espinosa JM, Mora G, Blanco JF, Granero-Molto F, Nunez-Cordoba JM, Lopez-Elio S, Andreu E, Sanchez-Guijo F, Aquerreta JD, Bondia JM, Valenti-Azcarate A, Del Consuelo Del Canizo M, Villaron EM, Valenti-Nin JR, Prosper F (2018) Intra-articular injection of two different doses of autologous bone marrow mesenchymal stem cells versus hyaluronic acid in the treatment of knee osteoarthritis: long-term follow up of a multicenter randomized controlled clinical trial (phase I/II). J Transl Med 16(1):213. https://doi.org/10.1186/s12967-018-1591-7
Orozco L, Munar A, Soler R, Alberca M, Soler F, Huguet M, Sentis J, Sanchez A, Garcia-Sancho J (2013) Treatment of knee osteoarthritis with autologous mesenchymal stem cells: a pilot study. Transplantation 95(12):1535–1541. https://doi.org/10.1097/TP.0b013e318291a2da
Pers YM, Rackwitz L, Ferreira R, Pullig O, Delfour C, Barry F, Sensebe L, Casteilla L, Fleury S, Bourin P, Noel D, Canovas F, Cyteval C, Lisignoli G, Schrauth J, Haddad D, Domergue S, Noeth U, Jorgensen C, Consortium A (2016) Adipose mesenchymal stromal cell-based therapy for severe osteoarthritis of the knee: a phase I dose-escalation trial. Stem Cells Transl Med 5(7):847–856. https://doi.org/10.5966/sctm.2015-0245
Pintat J, Silvestre A, Magalon G, Gadeau AP, Pesquer L, Perozziello A, Peuchant A, Mounayer C, Dallaudiere B (2017) Intra-articular injection of mesenchymal stem cells and platelet-rich plasma to treat patellofemoral osteoarthritis: preliminary results of a long-term pilot study. J Vasc Interv Radiol 28(12):1708–1713. https://doi.org/10.1016/j.jvir.2017.08.004
Rajput B, Kulkarni R, Bopardikar A, Somalapur P, Kumar R (2018) Retrospective analysis of role of autologous bone marrow derived mononuclear stem cells in the management of degenerative arthritis of knee. J Stem Cell Res Ther 4(2):21–27
Soler R, Orozco L, Munar A, Huguet M, Lopez R, Vives J, Coll R, Codinach M, Garcia-Lopez J (2016) Final results of a phase I–II trial using ex vivo expanded autologous Mesenchymal Stromal Cells for the treatment of osteoarthritis of the knee confirming safety and suggesting cartilage regeneration. Knee 23(4):647–654. https://doi.org/10.1016/j.knee.2015.08.013
Soler Rich R, Munar A, Soler Romagosa F, Peirau X, Huguet M, Alberca M, García SA, Sancho JLO (2015) Treatment of knee osteoarthritis with autologous expanded bone marrow mesenchymal stem cells: 50 cases clinical and mri results at one year follow-up. J Stem Cell Res Ther 5(6):1–7
Spasovski D, Spasovski V, Bascarevic Z, Stojiljkovic M, Vreca M, Andelkovic M, Pavlovic S (2018) Intra-articular injection of autologous adipose-derived mesenchymal stem cells in the treatment of knee osteoarthritis. J Gene Med. https://doi.org/10.1002/jgm.3002
Vega A, Martin-Ferrero MA, Del Canto F, Alberca M, Garcia V, Munar A, Orozco L, Soler R, Fuertes JJ, Huguet M, Sanchez A, Garcia-Sancho J (2015) Treatment of knee osteoarthritis with allogeneic bone marrow mesenchymal stem cells: a randomized controlled trial. Transplantation 99(8):1681–1690. https://doi.org/10.1097/TP.0000000000000678
Bellamy N, Campbell J, Robinson V, Gee T, Bourne R, Wells G (2006) Intraarticular corticosteroid for treatment of osteoarthritis of the knee. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD005328.pub2
Caldwell JR (1996) Intra-articular corticosteroids. Guide to selection and indications for use. Drugs 52(4):507–514
Wernecke C, Braun HJ, Dragoo JL (2015) The effect of intra-articular corticosteroids on articular cartilage: a systematic review. Orthop J Sports Med 3(5):2325967115581163. https://doi.org/10.1177/2325967115581163
Barker WD, Martinek J (1976) An ultrastructural evaluation of the effect of hydrocortisone on rabbit cartilage. Clin Orthop Relat Res 115:286–290
Dragoo JL, Danial CM, Braun HJ, Pouliot MA, Kim HJ (2012) The chondrotoxicity of single-dose corticosteroids. Knee Surg Sports Traumatol Arthrosc 20(9):1809–1814. https://doi.org/10.1007/s00167-011-1820-6
Frisbie DD, Kawcak CE, Trotter GW, Powers BE, Walton RM, McIlwraith CW (1997) Effects of triamcinolone acetonide on an in vivo equine osteochondral fragment exercise model. Equine Vet J 29(5):349–359
Yagi H, Soto-Gutierrez A, Parekkadan B, Kitagawa Y, Tompkins RG, Kobayashi N, Yarmush ML (2010) Mesenchymal stem cells: mechanisms of immunomodulation and homing. Cell Transpl 19(6):667–679. https://doi.org/10.3727/096368910X508762
Aggarwal S, Pittenger MF (2005) Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood 105(4):1815–1822. https://doi.org/10.1182/blood-2004-04-1559
Augello A, Tasso R, Negrini SM, Amateis A, Indiveri F, Cancedda R, Pennesi G (2005) Bone marrow mesenchymal progenitor cells inhibit lymphocyte proliferation by activation of the programmed death 1 pathway. Eur J Immunol 35(5):1482–1490. https://doi.org/10.1002/eji.200425405
Plumas J, Chaperot L, Richard MJ, Molens JP, Bensa JC, Favrot MC (2005) Mesenchymal stem cells induce apoptosis of activated T cells. Leukemia 19(9):1597–1604. https://doi.org/10.1038/sj.leu.2403871
Wyles CC, Houdek MT, Wyles SP, Wagner ER, Behfar A, Sierra RJ (2015) Differential cytotoxicity of corticosteroids on human mesenchymal stem cells. Clin Orthop Relat Res 473(3):1155–1164. https://doi.org/10.1007/s11999-014-3925-y
Chen X, Gan Y, Li W, Su J, Zhang Y, Huang Y, Roberts AI, Han Y, Li J, Wang Y, Shi Y (2014) The interaction between mesenchymal stem cells and steroids during inflammation. Cell Death Dis 5:e1009. https://doi.org/10.1038/cddis.2013.537
Nuzzi R, Gunetti M, Rustichelli D, Roagna B, Fronticelli Bardelli F, Fagioli F, Ferrero I (2012) Effect of in vitro exposure of corticosteroid drugs, conventionally used in AMD treatment, on mesenchymal stem cells. Stem Cells Int 2012:946090. https://doi.org/10.1155/2012/946090
Feldman BJ (2009) Glucocorticoids influence on mesenchymal stem cells and implications for metabolic disease. Pediatr Res 65(2):249–251. https://doi.org/10.1203/PDR.0b013e3181909c08
Breu A, Eckl S, Zink W, Kujat R, Angele P (2013) Cytotoxicity of local anesthetics on human mesenchymal stem cells in vitro. Arthroscopy 29(10):1676–1684. https://doi.org/10.1016/j.arthro.2013.06.018
Rahnama R, Wang M, Dang AC, Kim HT, Kuo AC (2013) Cytotoxicity of local anesthetics on human mesenchymal stem cells. J Bone Joint Surg Am 95(2):132–137. https://doi.org/10.2106/JBJS.K.01291
Ahmad Z, Howard D, Brooks RA, Wardale J, Henson FM, Getgood A, Rushton N (2012) The role of platelet rich plasma in musculoskeletal science. JRSM Short Rep 3(6):40. https://doi.org/10.1258/shorts.2011.011148
Sheth U, Simunovic N, Klein G, Fu F, Einhorn TA, Schemitsch E, Ayeni OR, Bhandari M (2012) Efficacy of autologous platelet-rich plasma use for orthopaedic indications: a meta-analysis. J Bone Joint Surg Am 94(4):298–307. https://doi.org/10.2106/JBJS.K.00154
Le ADK, Enweze L, DeBaun MR, Dragoo JL (2018) Current clinical recommendations for use of platelet-rich plasma. Curr Rev Musculoskelet Med 11(4):624–634. https://doi.org/10.1007/s12178-018-9527-7
Johal H, Khan M, Yung SP, Dhillon MS, Fu FH, Bedi A, Bhandari M (2019) Impact of platelet-rich plasma use on pain in orthopaedic surgery: a systematic review and meta-analysis. Sports Health 11(4):355–366. https://doi.org/10.1177/1941738119834972
Franchini M, Cruciani M, Mengoli C, Marano G, Pupella S, Veropalumbo E, Masiello F, Pati I, Vaglio S, Liumbruno GM (2018) Efficacy of platelet-rich plasma as conservative treatment in orthopaedics: a systematic review and meta-analysis. Blood Transfus 16(6):502–513. https://doi.org/10.2450/2018.0111-18
Chen X, Jones IA, Park C, Vangsness CT Jr (2018) The efficacy of platelet-rich plasma on tendon and ligament healing: a systematic review and meta-analysis with bias assessment. Am J Sports Med 46(8):2020–2032. https://doi.org/10.1177/0363546517743746
Fitzpatrick J, Bulsara M, Zheng MH (2017) The effectiveness of platelet-rich plasma in the treatment of tendinopathy: a meta-analysis of randomized controlled clinical trials. Am J Sports Med 45(1):226–233. https://doi.org/10.1177/0363546516643716
Martin DR, Cox NR, Hathcock TL, Niemeyer GP, Baker HJ (2002) Isolation and characterization of multipotential mesenchymal stem cells from feline bone marrow. Exp Hematol 30(8):879–886
Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR (1999) Multilineage potential of adult human mesenchymal stem cells. Science 284(5411):143–147. https://doi.org/10.1126/science.284.5411.143
Baker N, Boyette LB, Tuan RS (2015) Characterization of bone marrow-derived mesenchymal stem cells in aging. Bone 70:37–47. https://doi.org/10.1016/j.bone.2014.10.014
Caplan AI, Correa D (2011) The MSC: an injury drugstore. Cell Stem Cell 9(1):11–15. https://doi.org/10.1016/j.stem.2011.06.008
Murray IR, Corselli M, Petrigliano FA, Soo C, Peault B (2014) Recent insights into the identity of mesenchymal stem cells: Implications for orthopaedic applications. Bone Joint J 96B(3):291–298. https://doi.org/10.1302/0301-620X.96B3.32789
Petersson IF, Boegard T, Saxne T, Silman AJ, Svensson B (1997) Radiographic osteoarthritis of the knee classified by the Ahlback and Kellgren & Lawrence systems for the tibiofemoral joint in people aged 35–54 years with chronic knee pain. Ann Rheum Dis 56(8):493–496
Harichandan A, Buhring HJ (2011) Prospective isolation of human MSC. Best Pract Res Clin Haematol 24(1):25–36. https://doi.org/10.1016/j.beha.2011.01.001
Bartholomew A, Sturgeon C, Siatskas M, Ferrer K, McIntosh K, Patil S, Hardy W, Devine S, Ucker D, Deans R, Moseley A, Hoffman R (2002) Mesenchymal stem cells suppress lymphocyte proliferation in vitro and prolong skin graft survival in vivo. Exp Hematol 30(1):42–48
Desando G, Cavallo C, Sartoni F, Martini L, Parrilli A, Veronesi F, Fini M, Giardino R, Facchini A, Grigolo B (2013) Intra-articular delivery of adipose derived stromal cells attenuates osteoarthritis progression in an experimental rabbit model. Arthritis Res Ther 15(1):R22. https://doi.org/10.1186/ar4156
Guercio A, Di Marco P, Casella S, Cannella V, Russotto L, Purpari G, Di Bella S, Piccione G (2012) Production of canine mesenchymal stem cells from adipose tissue and their application in dogs with chronic osteoarthritis of the humeroradial joints. Cell Biol Int 36(2):189–194. https://doi.org/10.1042/CBI20110304
Jurgens WJ, Kroeze RJ, Zandieh-Doulabi B, van Dijk A, Renders GA, Smit TH, van Milligen FJ, Ritt MJ, Helder MN (2013) One-step surgical procedure for the treatment of osteochondral defects with adipose-derived stem cells in a caprine knee defect: a pilot study. Biores Open Access 2(4):315–325. https://doi.org/10.1089/biores.2013.0024
Jung M, Kaszap B, Redohl A, Steck E, Breusch S, Richter W, Gotterbarm T (2009) Enhanced early tissue regeneration after matrix-assisted autologous mesenchymal stem cell transplantation in full thickness chondral defects in a minipig model. Cell Transpl 18(8):923–932. https://doi.org/10.3727/096368909X471297
Di Ianni M, Del Papa B, De Ioanni M, Moretti L, Bonifacio E, Cecchini D, Sportoletti P, Falzetti F, Tabilio A (2008) Mesenchymal cells recruit and regulate T regulatory cells. Exp Hematol 36(3):309–318. https://doi.org/10.1016/j.exphem.2007.11.007
Benvenuto F, Ferrari S, Gerdoni E, Gualandi F, Frassoni F, Pistoia V, Mancardi G, Uccelli A (2007) Human mesenchymal stem cells promote survival of T cells in a quiescent state. Stem Cells 25(7):1753–1760. https://doi.org/10.1634/stemcells.2007-0068
Zhang W, Ge W, Li C, You S, Liao L, Han Q, Deng W, Zhao RC (2004) Effects of mesenchymal stem cells on differentiation, maturation, and function of human monocyte-derived dendritic cells. Stem Cells Dev 13(3):263–271. https://doi.org/10.1089/154732804323099190
Kafienah W, Al-Fayez F, Hollander AP, Barker MD (2003) Inhibition of cartilage degradation: a combined tissue engineering and gene therapy approach. Arthritis Rheum 48(3):709–718. https://doi.org/10.1002/art.10842
Bondeson J, Lauder S, Wainwright S, Amos N, Evans A, Hughes C, Feldmann M, Caterson B (2007) Adenoviral gene transfer of the endogenous inhibitor IkappaBalpha into human osteoarthritis synovial fibroblasts demonstrates that several matrix metalloproteinases and aggrecanases are nuclear factor-kappaB-dependent. J Rheumatol 34(3):523–533
Acknowledgements
We would like to thank Pia Offermanns B. A. (freelance editor, translator and proof-reader) for her professional editing of this study.
Funding
No external source of funding was used.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
For this type of study informed consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Migliorini, F., Rath, B., Colarossi, G. et al. Improved outcomes after mesenchymal stem cells injections for knee osteoarthritis: results at 12-months follow-up: a systematic review of the literature. Arch Orthop Trauma Surg 140, 853–868 (2020). https://doi.org/10.1007/s00402-019-03267-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00402-019-03267-8