Abstract
Introduction
Different dissection studies as well as comparative studies about the anterolateral ligament of the knee (ALL) already exist and the structure’s topology and properties have been shown. However, most of the studies investigating the ligament were performed in embalmed knees, which is thought to change the structural integrity of ligaments and thus the topologic and dynamic measurements. Since the biomechanical function of the ALL is not fully understood until today and a correlation with the pivot shift phenomenon is yet speculative, further studies will have to clarify its definitive importance. Its function as a limiter of internal rotation and lateral meniscal extrusion leads to the assumption of a secondary knee stabilizer.
Methods
Twenty paired fresh-frozen cadaveric knees of ten donors have been dissected in a layerwise fashion. After identification of the ALL, topologic measurements were undertaken using a digital caliper.
Results
The ALL could be identified as a tender, pearly structure in front of the anterolateral joint capsule in only 60% of the dissected knee joints. Only 20% of donors had a bilateral ALL while 80% had an ALL only in one side. Mean length, thickness and width as well as topographic measurements were comparable to other available studies investigating fresh-frozen cadavers.
Conclusion
Anatomy and topography of the ALL seem to be highly variable, but consistent within certain borders. Prevalence has to be argued though as it strongly differs between studies. The impact of an ALL absence, even if only unilateral, needs to be investigated in clinical and imaging studies to finally clarify its importance.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A lot of knowledge has been gained on the anatomy and biomechanical function of the anterolateral ligament (ALL) of the knee since the descriptive anatomical study by Claes et al. [1], which led to a complete rethinking of its importance in reconstructive orthopedics. Although known for over a century [2], anatomists and surgeons were not able to attribute a distinct function to this nondescript structure. Therefore, it has been given a lot of different names in the past, but none of them prevailed [3].
Due to results of recent anatomical and biomechanical investigations it is thought to control internal tibial rotation and therefore has an influence on the pivot shift phenomenon [4]. As it supports the thin anterolateral joint capsule [5] it can be compared to the glenohumeral ligaments of the shoulder. In a recent Cochrane review Mohtadi et al. reported of a 19% incidence of pivot shifts II° and higher after anterior cruciate ligament (ACL) reconstruction with either hamstring (single- and double-bundle) or patellar tendon graft [6]. As the cause for such a high incidence of postoperative rotational instabilities remained unclear, a possible link to anterolateral rotational instability (ALRI) can be postulated [7]. This has recently been fortified by results of Parsons et al. showing that an increased tightening of the ALL in internal rotation corresponds with an increased slackening of the ACL which classifies the ALL as an antagonist to the ACL regarding rotational stability especially in flexion higher than 35° [8].
However, the lateral aspect of the knee has been studied intensively during the last decades and a three-layer composition has been recognized, the ALL was not discovered as a singular structure, but merely a thickening of the lateral joint capsule [9]. In recent times different anatomical dissection studies have been published describing the topography of the ALL and several other aspects as length changes during flexion or anatomical variations like additional meniscal fibers [1, 5, 10–15]. Table 1 gives an overview on all of these studies. However, a direct comparison of these studies is very difficult due to a high variability of basic parameters. Three studies investigated the structure in embalmed cadaveric knees [1, 5, 15], five studies used fresh or fresh-frozen specimens [10–14]. Embalming demonstrably changes biomechanical properties of tendons by changing collagen integrity. This may lead to shrinking and a loss of elasticity.
All available studies investigated unpaired cadaveric knees only. An exception is a subseries of the results published by Caterine et al. comprising three paired knees which is, however, not worked up in detail regarding certain differences [13]. Prevalences of the ALL vary between 45.5 and 100% which can be interpreted as either associated with preparation protocol or a positive selection of investigated specimens. Moreover, Shea et al. performed dissection of skeletally immature knee joints (ages 3 months to 10 years) and found an ALL or alike structure in only 7 out of 8 specimens. All other structures outlining anterolateral topography (LCL, PT, ITB) could be identified though [16]. In addition, dissection of different primate cadaveric knee joints did not show any evidence of an ALL regardless of race. This suggests that the structure is also reserved for the human species [17]. This, together with recently reported low prevalences, underlines the hypothesis, that the ALL may not be a preformed anatomical structure, but is either a remnant like the palmaris longus tendon in the lower arm or even evolved throughout a lifetime due to distinct external stimuli.
Aim of the present anatomical dissection study was to investigate ALL prevalence in paired fresh-frozen cadaveric knee joints and to check for possible side differences in close comparison to the literature available.
Methods
Anatomical dissection
Investigations were performed between September and November 2014 at the Institute of Molecular and Cellular Anatomy, RWTH Aachen University, by a single experienced knee surgeon. Written informed consent regarding postmortem scientific use existed for all donors and was available from the institute’s records. Twenty paired fresh-frozen cadaveric knee joints from ten donors were dissected. Average donor age was 79.4 ± 9.4 years (range 54–90) and gender distribution was equal.
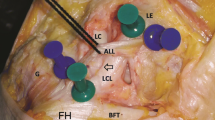
At the beginning knee joints were thawed in a bath of normal saline (NaCl 0.9%) until room temperature (23.5 °C) was reached. Overlying skin and subcutaneous fatty tissue were preparated and reflected carefully, regarding any possible connection to underlying fasciae, until visualization of the biceps femoris dorsally and the patella ventrally. After identification of the quadriceps tendon (QT), lateral patellar edge and patellar tendon (PaT), a conventional lateral parapatellar approach was used to open up the joint and reflect the patella. Surplus parts of the lateral retinaculum were carefully removed preserving the iliotibial band (ITB) in its course. After identification of the tibial insertion of the ITB it was removed sharply and reflected, leaving its fibrous connection to the lateral femoral condyle intact. Remaining parts of the lateral retinaculum were removed sharply from their femoral and tibial insertions as well as from the ITB. If present, the ALL could be visualized as an oblique subtle semi-translucent structure next to the lateral collateral ligament (LCL) lying directly underneath the aforementioned structures. After identification of the ALL the Hoffa fat pad and adjacent joint capsule were removed meticulously to unveil the lateral meniscus and tibial plateau. Furthermore, Gerdy’s tubercle and the fibular head were preparated to unveil the whole insertion area of the ALL and identify clear landmarks for topographic measurements. All distance measurements have been performed using a digital caliper (IP67, Hogetex, Nieder-Olm, Germany).
Length of the ALL was measured in full extension and 90° flexion as well as width and thickness near femoral origin, midportion and tibial insertion. Distances of the closest point of tibial insertion to Gerdy’s tubercle (GT), femoral head (FH) and joint line (JL) were recorded. Areas of origin and insertion were approximated after complete dissection with a micro-scalpel. If present, thickness of linking fibers to the lateral aspect of the meniscus was also measured.
Statistical analysis
All data were processed with Excel 2016 (Microsoft Inc., Redmond, WA, USA) or Graphpad Prism 7 (Graphpad Inc., La Jolla, CA, USA) and are given as mean ± standard deviation (SD) and range if not indicated otherwise. Graphics were produced using the same software packages.
Meta-analysis
To obtain a pooled prevalence of the ALL from all available studies, an anatomical meta-analysis was performed based on suggestions by Henry et al. and Neyeloff et al. [18, 19]. Due to a high heterogeneity of available studies (I 2 = 50.49), a random effects model was chosen. All quantitative and qualitative data extracted from available publications are presented separately in Tables 1 and 2. Individual weights for each study as well as confidence intervals (CI) were calculated using the algorithm named above.
Results
Anatomical dissection
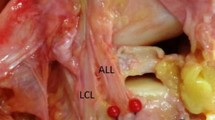
Topographical landmarks of the anterolateral corner (ITB, LCL, PT and GT) could easily be identified in all specimens. An ALL could be identified in only 60% (n = 12) of the dissected knee joints (Fig. 1). Eight out of ten donors (80%) had an ALL only in one side (n = 5 right, n = 3 left). Two donors (20%) exhibited a bilateral ALL. Mean length in extension was 39.63 ± 0.78 mm (range 38.0–40.7) and in 90° flexion 42.91 ± 1.01 mm (range 41.0–44.1). Mean femoral width was 7.63 ± 0.35 mm (range 6.9–8.1), mean midportion width 5.28 ± 0.33 mm (range 4.80–5.70) and mean tibial width 10.17 ± 0.36 mm (range 9.7–10.7). Mean midportion thickness was 1.52 ± 0.31 mm (range 1.1–2.0).
Photograph of a right (a) and left (b) dissected knee pair. The patella has been reflected and the ITB removed giving a free view on the anterolateral structures of the knee joint: an ALL could only be identified on the left side. FCL fibular collateral ligament, ALL anterolateral ligament, PT popliteus tendon, Men lateral meniscus, Fib fibular head [3]. PROX proximal, DIST distal, ANT anterior, POST posterior
Meniscal fibers leaving the ALL in different shapes and thicknesses could be identified in 11 out of 12 cases (91.6%) where an ALL was present. Micro-dissection showed that the femoral origin of the ALL merged with that of the LCL over the lateral femoral epicondyle in all 12 cases. Tibial insertion presented more or less feathered with a firm attachment to the underlying periosteum. Distance of the ALL insertion to joint line were 9.54 ± 0.96 mm (range 7.8–10.6), to lateral center of fibular head 22.06 ± 1.08 mm (range 20.4–23.1) and to Gerdy’s tubercle 22.3 ± 1.08 mm (range 21.3–23.1). Mean ALL/LCL central axis angle in full extension was 29.33° ± 2.50° (range 25.0–33.0).
Meta-analysis
Pooled prevalence of the ALL in all available dissection studies regardless of the preparation technique was 76.25%. A detailed overview on all results is given in Table 3 together with a forest plot (Fig. 2).
Discussion
Results of the presents study indicate that the ALL seems to be a structure that is not ubiquitous among individuals. Furthermore, it was found unilateral in most cases, suggesting that it is not a preformed anatomical structure, but may be part of embryological development. It was found directly above the very thin anterolateral joint capsule in close connection to the lateral meniscus with connecting fibers in a bifidal fashion. Unless described in other studies [5, 10, 20], the ALL does not appear as a thickening of the joint capsule but, due to its orientation, more as a singular fine ligament. So far most of the present results are consistent with current literature.
Descriptive anatomical studies depict a high variability of origin and insertion of the ALL. While Vincent et al. [10] postulated an origin distal-anterior, Dodds et al. [12] described an origin proximal-posterior to that of the LCL over the lateral femoral epicondyle. However, Claes et al. [1] described a merge of ALL and FCL origins which is in line with findings of the present study. Other available studies identified variances of the above named origin topography [13, 14]. Topographic measurements of the tibial insertion, meaning the distances to tibial landmarks (GT, JL and FH) also were in line with other studies; however, variances of means seemed to be larger here. A bifurcation of the ALL with additional meniscal fibers first described by Helito et al. [11, 21] was also largely noticed in the present study as well as in others [15].
Mean measurements of the ALL are highly variable between studies, supposedly also depending on the preparation and fixation methods used. As histomorphology of tendon tissue changes during chemical preservation with agents like formalin, its native properties are also altered with respect to volume and elasticity as well as a fluid loss [22]. If conducted correctly, freeze-thawing does not change tendon histological and biomechanical properties and thus guarantees for more realistic measurement parameters [23, 24]. Therefore, a comparison of available studies in a meta-analysis like fashion does not seem appropriate in case of anatomic dimensions due to different study designs and inconsistent data especially regarding extension/flexion angles during measurements. Tables 1 and 2 provide all data extracted from available dissection studies and also highlight heterogeneity in study designs and measurements methods. Furthermore, it has been disputed that Vincent et al. rather described the so-called capsular ligament instead of the ALL [10]. However, mean ALL lengths recorded by Claes et al. and Caterine et al. [1, 13] are still much shorter than those measured by Dodds et al. [13]. An accurate dimensioning of the ALL can thus only be stated from large cohort studies of fresh cadaveric knees without any means of preservation, which are admittedly difficult to perform. At this stage, however, a subgroup analysis of available data seems obsolete due to a still too little number of cases.
Prevalence of the ALL was 60% in the present study while pooled prevalence (Fig. 2; Table 3) was calculated with 76.25%. Assuming normal distribution among studied collectives this means that about a quarter of individual knee joints do not exhibit an ALL. Reasons for that are only speculative though. However, due to its leading role in controlling internal tibial rotation and thus influencing the pivot shift phenomenon [25, 26], its seems unlikely that about a quarter of investigated knee joints did not show a comparable structure. Anterior cruciate ligament (ACL) insufficiency results in a deviation of the rotatory knee axis and therefore generates instability with altered knee kinematics [27, 28]. Dynamical re-stabilization is seen as a counter reaction in some cases what brought up the following terms in literature: while most individuals experience instability mostly during pivoting action (non-copers), some are able to compensate these effects (copers) while others just adapt their activity level (adapters or avoiders) [29]. Possibility to cope with an isolated ACL deficiency may be due to the presence of an ALL or alike structure limiting the pivot shift, rotatory instability and therefore also subjective symptoms like giving way. In these cases, ALRI will not be due to an isolated ALL rupture or an anterolateral corner injury, but merely due to a missing ALL and a lack of pivoting control. Although lots of confounding variables affecting return to sport after individual ACL injuries [30] as well as gender-specific anatomical topologies [31] have already been identified, large-scale clinical, imaging and in vivo biomechanical studies will be needed to compare copers and non-copers of ACL deficiency and identify possible differences in ALL prevalence among those two groups to verify this hypothesis.
A largely unilateral appearance of the ALL was noted in the present study, which is the first one to investigate paired knees only. Wang et al. postulated that there is a significant difference in knee joint kinematics between dominant and non-dominant knees in individuals after ACL reconstruction especially regarding control of tibial rotation [32]. Findings were in line with earlier reports [33, 34], leading to the hypothesis that ALL prevalence and shaping may also correlate with lower limb dominance although there are possible articular confounders like cartilage degeneration and subsequent joint line height and topography changes, which may also add to an altered kinematic behavior [35]. As the structure is not consistently seen in either developing human individuals [16] or different primate species [17] a possible delayed formation due to external stimuli during neuromotor development seems possible. Moreover, it remains to investigate whether the ALL is more often found in the dominant knee of healthy individuals, which then could be attributed to a general need for increased rotational stability while carrying higher loads and torques [36]. In case the ALL was more often found in the non-dominant knee, it could be discussed as an additional ligamentous stabilizer compensating for less muscular volume and activity [37]. Reconstruction of the ALL is still controversial in the literature, although it might help avoid ALRI and thus concomitant lesions associated with ACL-deficient pivoting knees like meniscal tears and cartilage damages [38]. Of course the present study also has some limitations. First there is a relatively high mean donor age, which is however quite common among anatomical dissection studies. This issue has also been commented on by other authors [15]. Secondly, freezing and thawing may be confounding variables, as they have a considerable influence on tissue properties as reported above. Lastly, the number of specimens is quite low and only one investigator performed the dissection by the fixed protocol. For this reason pooling of the data to achieve a better hint on real prevalence of the structure was performed via meta-analysis for example. The aspect of a possible laterality of the ALL has never been reported before though. However, comparative clinical and imaging studies are needed to prove these hypotheses and concepts with a special attention to kinematic forces in dominant and non-dominant knee joints.
Conclusion
Anatomy and topography of the ALL seems to be highly variable, but consistent within certain borders regarding all available dissection studies for now. Prevalence of the structure has to be argued though, as it strongly differs between studies depending on design, preservation and dissection protocol.
References
Claes S, Vereecke E, Maes M et al (2013) Anatomy of the anterolateral ligament of the knee. J Anat 223:321–328. doi:10.1111/joa.12087
Segond P (1879) Recherches cliniques et expérimentales sur les épanchements sanguins du genou par entorse. Progres Med 7:297–341
Roessler PP, Schüttler KF, Heyse TJ et al (2016) The anterolateral ligament (ALL) and its role in rotational extra-articular stability of the knee joint: a review of anatomy and surgical concepts. Arch Orthop Trauma Surg 136:305–313. doi:10.1007/s00402-015-2395-3
Guenther D, Griffith C, Lesniak B et al (2015) Anterolateral rotatory instability of the knee. Knee Surg Sports Traumatol Arthrosc 23:2909–2917. doi:10.1007/s00167-015-3616-6
Stijak L, Bumbaširević M, Radonjić V et al (2016) Anatomic description of the anterolateral ligament of the knee. Knee Surg Sports Traumatol Arthrosc 24:2083–2088. doi:10.1007/s00167-014-3422-6
Mohtadi NG, Chan DS, Dainty KN, Whelan DB (2011) Patellar tendon versus hamstring tendon autograft for anterior cruciate ligament rupture in adults. Cochrane Database Syst Rev CD005960. doi:10.1002/14651858.CD005960.pub2
Hughston JC, Andrews JR, Cross MJ, Moschi A (1976) Classification of knee ligament instabilities. Part II. The lateral compartment. J Bone Joint Surg Am 58:173–179
Parsons EM, Gee AO, Spiekerman C, Cavanagh PR (2015) The biomechanical function of the anterolateral ligament of the knee. Am J Sports Med 43:669–674. doi:10.1177/0363546514562751
Seebacher JR, Inglis AE, Marshall JL, Warren RF (1982) The structure of the posterolateral aspect of the knee. J Bone Joint Surg Am 64:536–541
Vincent JP, Magnussen RA, Gezmez F et al (2012) The anterolateral ligament of the human knee: an anatomic and histologic study. Knee Surg Sports Traumatol Arthrosc 20:147–152. doi:10.1007/s00167-011-1580-3
Helito CP, Demange MK, Bonadio MB et al (2013) Anatomy and histology of the knee anterolateral ligament. Orthop J Sports Med 1:2325967113513546. doi:10.1177/2325967113513546
Dodds AL, Halewood C, Gupte CM et al (2014) The anterolateral ligament: Anatomy, length changes and association with the Segond fracture. Bone Joint J 96-B:325–331. doi:10.1302/0301-620X.96B3.33033
Caterine S, Litchfield R, Johnson M et al (2015) A cadaveric study of the anterolateral ligament: re-introducing the lateral capsular ligament. Knee Surg Sports Traumatol Arthrosc 23:3186–3195. doi:10.1007/s00167-014-3117-z
Kosy JD, Soni A, Venkatesh R, Mandalia VI (2016) The anterolateral ligament of the knee: unwrapping the enigma. Anatomical study and comparison to previous reports. J Orthop Traumatol. doi:10.1007/s10195-016-0392-0
Runer A, Birkmaier S, Pamminger M et al (2016) The anterolateral ligament of the knee: a dissection study. Knee 23:8–12. doi:10.1016/j.knee.2015.09.014
Shea KG, Polousky JD, Jacobs JC et al (2015) The anterolateral ligament of the knee. J Pediatr Orthop. doi:10.1097/bpo.0000000000000616
Ingham SJM, de Carvalho RT, Martins CAQ et al (2015) Anterolateral ligament anatomy: a comparative anatomical study. Knee Surg Sports Traumatol Arthrosc. doi:10.1007/s00167-015-3956-2
Neyeloff JL, Fuchs SC, Moreira LB (2012) Meta-analyses and Forest plots using a microsoft excel spreadsheet: step-by-step guide focusing on descriptive data analysis. BMC Res Notes 5:52. doi:10.1186/1756-0500-5-52
Henry BM, Tomaszewski KA, Walocha JA (2016) Methods of evidence-based anatomy: a guide to conducting systematic reviews and meta-analysis of anatomical studies. Ann Anat 205:16–21. doi:10.1016/j.aanat.2015.12.002
Kosy JD, Mandalia VI, Anaspure R (2015) Characterization of the anatomy of the anterolateral ligament of the knee using magnetic resonance imaging. Skeletal Radiol 44:1647–1653. doi:10.1007/s00256-015-2218-1
Helito CP, Bonadio MB, Soares TQ et al (2016) The meniscal insertion of the knee anterolateral ligament. Surg Radiol Anat 38:223–228. doi:10.1007/s00276-015-1533-5
Ling Y, Li C, Feng K et al (2016) Effects of fixation and preservation on tissue elastic properties measured by quantitative optical coherence elastography (OCE). J Biomech 49:1009–1015. doi:10.1016/j.jbiomech.2016.02.013
Bitar AC, Santos LAU, Croci AT et al (2010) Histological study of fresh versus frozen semitendinous muscle tendon allografts. Clinics (Sao Paulo) 65:297–303. doi:10.1590/S1807-59322010000300010
Woo SL, Orlando CA, Camp JF, Akeson WH (1986) Effects of postmortem storage by freezing on ligament tensile behavior. J Biomech 19:399–404
Zens M, Niemeyer P, Ruhhammer J et al (2015) Length changes of the anterolateral ligament during passive knee motion: a human cadaveric study. Am J Sports Med 43:2545–2552. doi:10.1177/0363546515594373
Kittl C, Halewood C, Stephen JM et al (2015) Length change patterns in the lateral extra-articular structures of the knee and related reconstructions. Am J Sports Med 43:354–362. doi:10.1177/0363546514560993
Kostogiannis I, Ageberg E, Neuman P et al (2007) Activity level and subjective knee function 15 years after anterior cruciate ligament injury: a prospective, longitudinal study of nonreconstructed patients. Am J Sports Med 35:1135–1143. doi:10.1177/0363546507299238
Defrate LE, Papannagari R, Gill TJ et al (2006) The 6 degrees of freedom kinematics of the knee after anterior cruciate ligament deficiency: an in vivo imaging analysis. Am J Sports Med 34:1240–1246. doi:10.1177/0363546506287299
Walla DJ, Albright JP, McAuley E et al (1985) Hamstring control and the unstable anterior cruciate ligament-deficient knee. Am J Sports Med 13:34–39
Rodríguez-Roiz JM, Caballero M, Ares O et al (2015) Return to recreational sports activity after anterior cruciate ligament reconstruction: a one- to six-year follow-up study. Arch Orthop Trauma Surg 135:1117–1122. doi:10.1007/s00402-015-2240-8
Estes K, Cheruvu B, Lawless M et al (2015) Risk assessment for anterior cruciate ligament injury. Arch Orthop Trauma Surg 135:1437–1443. doi:10.1007/s00402-015-2292-9
Wang H, Fleischli JE, Nigel Zheng N (2012) Effect of lower limb dominance on knee joint kinematics after anterior cruciate ligament reconstruction. Clin Biomech (Bristol, Avon) 27:170–175. doi:10.1016/j.clinbiomech.2011.08.006
Scanlan SF, Chaudhari AMW, Dyrby CO, Andriacchi TP (2010) Differences in tibial rotation during walking in ACL reconstructed and healthy contralateral knees. J Biomech 43:1817–1822. doi:10.1016/j.jbiomech.2010.02.010
Tashman S, Kolowich P, Collon D et al (2007) Dynamic function of the ACL-reconstructed knee during running. Clin Orthop Relat Res 454:66–73. doi:10.1097/BLO.0b013e31802bab3e
Thambyah A, Nather A, Goh J (2006) Mechanical properties of articular cartilage covered by the meniscus. Osteoarthr Cartil 14:580–588. doi:10.1016/j.joca.2006.01.015
de Ruiter CJ, de Korte A, Schreven S, de Haan A (2010) Leg dominancy in relation to fast isometric torque production and squat jump height. Eur J Appl Physiol 108:247–255. doi:10.1007/s00421-009-1209-0
Ergün M, İşlegen Ç, Taşkiran E (2004) A cross-sectional analysis of sagittal knee laxity and isokinetic muscle strength in soccer players. Int J Sports Med 25:594–598. doi:10.1055/s-2004-821116
Hagino T, Ochiai S, Senga S et al (2015) Meniscal tears associated with anterior cruciate ligament injury. Arch Orthop Trauma Surg 135:1701–1706. doi:10.1007/s00402-015-2309-4
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Rights and permissions
About this article
Cite this article
Roessler, P.P., Schüttler, K.F., Stein, T. et al. Anatomic dissection of the anterolateral ligament (ALL) in paired fresh-frozen cadaveric knee joints. Arch Orthop Trauma Surg 137, 249–255 (2017). https://doi.org/10.1007/s00402-016-2617-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00402-016-2617-3