Abstract
Objective
The purpose of this study is to compare the effectiveness and safety of artificial total disc replacement (TDR) with fusion for the treatment of lumbar degenerative disc disease (DDD).
Summary of background data
Spinal fusion is the conventional surgical treatment for lumbar DDD. Recently, TDR has been developed to avoid the negative effects of the fusion by preserving function of the motion segment. Controversy still surrounds regarding whether TDR is better.
Methods
We systematically searched six electronic databases (Medline, Embase, Clinical, Ovid, BIOSIS and Cochrane registry of controlled clinical trials) to identify randomized controlled trials (RCTs) published up to March 2013 in which TDR was compared with the fusion for the treatment of lumbar DDD. Effective data were extracted after the assessment of methodological quality of the trials. Then, we performed the meta-analysis.
Results
Seven relevant RCTs with a total of 1,584 patients were included. TDR was more effective in ODI (MD −5.09; 95 % CI [−7.33, −2.84]; P < 0.00001), VAS score (MD −5.31; 95 % CI [−8.35, −2.28]; P = 0.0006), shorter duration of hospitalization (MD −0.82; 95 % CI [−1.38, −0.26]; P = 0.004) and a greater proportion of willing to choose the same operation again (OR 2.32; 95 % CI [1.69, 3.20]; P < 0.00001). There were no significant differences between the two treatment methods regarding operating time (MD −44.16; 95 % CI [−94.84, 6.52]; P = 0.09), blood loss (MD −29.14; 95 % CI [−173.22, 114.94]; P = 0.69), complications (OR 0.72; 95 % CI [0.45, 1.14]; P = 0.16), reoperation rate (OR 0.83; 95 % CI [0.39, 1.77]; P = 0.63) and the proportion of patients who returned to full-time/part-time work (OR 1.10; 95 % CI [0.86, 1.41]; P = 0.47).
Conclusion
TDR showed significant safety and efficacy comparable to lumbar fusion at 2 year follow-up. TDR demonstrated superiorities in improved physical function, reduced pain and shortened duration of hospitalization. The benefits of operating time, blood loss, motion preservation and the long-term complications are still unable to be proved.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Spinal fusion remains the established gold standard for the treatment of painful degenerative disc disease (DDD) [1–3], but it has the drawbacks of stiffness and adjacent segmental degeneration [4–6]. Recently, artificial total disc replacement (TDR), a motion-preserving option, has been used to treat the patients with symptomatic DDD [7, 8]. But there is still a controversy whether TDR is more effective and safer than lumbar fusion. Previous meta-analysis concluded that TDR showed significant superiority for the treatment of DDD when compared with fusion [9]. However, the meta-analysis was based on a small sample size and insufficient analyses. The need remains for strong evidence based on the latest high-quality RCTs to test the above conclusion. The aim of our meta-analysis is to systematically compare the effectiveness and safety of TDR to fusion for the treatment of lumbar DDD again.
Materials and methods
Search methods
Up to March 2013, all published RCTs comparing TDR with lumbar fusion intervention for DDD were searched for by two authors (MJR and SSC) independently. We performed the research of Medline, Embase, Clinical, Ovid, BIOSIS and Cochrane central registry of controlled trials. A manual search of Spine, European Spine Journal, and the American and British versions of Journal of Bone and Joint Surgery was also performed to identify additional studies. Publication language was limited to English. Key words used for search were as follows: DDD, lumbar fusion, low back pain, TDR and randomized controlled trial.
Criteria for selected trials
Two reviewers (MJR and SSC) checked titles and abstracts identified from the database. For items which could not be decided on the basis of titles and abstracts, the full text was retrieved for second-round selection. All randomized controlled clinical trials (RCTs) comparing the TDR to fusion for the treatment of lumbar DDD were taken into consideration. The indication for surgical treatment was low back pain with or without radicular pain that failed to respond to conservative treatment. Patients older than 18 years of age with lumbar systematic DDD were included in this study. The interventions included various types of TDR and fusion in the lumbar spine. Studies with patients who had acute spinal fracture, infection, tumor, osteoporosis, or rheumatoid arthritis were excluded. The reviewers applied the inclusion criteria to select the potentially appropriate trials. Disagreements between two investigators were resolved by discussion, and a consensus was attempted.
Data extraction
Two reviewers participated in the extraction of relevant data from the included reports. One reviewer (MJR) extracted all relevant data onto a table; a second reviewer (SSC) checked the data. Disagreement was resolved by further discussion. The data extracted to describe characteristics of the investigations were characteristics of participants, intervention details, number of participants in each intervention group, sex radio, follow-up rate and period.
Methodological assessment
The modified Jadad scale was used as the methodological assessment for the study [10]. There are eight items designed to assess randomization, blinding, withdrawals and dropouts, inclusion and exclusion criteria, adverse effects and statistical analysis (Table 1). The score could range from 0 to 8. Scores of 0–3 indicate poor to low quality and 4–8 good to excellent quality.
Outcomes for meta-analysis
Primary outcomes consisted of visual analog scale (VAS), Oswestry disability index (ODI) and the patient satisfaction. Other outcome measures, such as the reoperation rate, employment rate, the operation time and blood loss and the complications etc. were considered as secondary outcome measures.
Assessment of clinical relevance
The clinical relevance of the seven included studies was assessed according to the five questions recommended by the Cochrane Back Review Group [11]. Positive (+) would be recorded if the clinical relevance item is appeared, negative (−) for the irrelevance and unclear (?) suggests that the data are inadequate for answering the question. 20 % of improvement in the pain score [12] and 25 % of improvement in the functioning score are considered to be clinically important [13].
Statistical analysis
The Q- and I 2-statistics were used to test for statistical heterogeneity [14, 15]. The Q-statistic tested the null hypothesis that all studies shared a common effect size with minimal dispersion of the effect size across studies. I 2 can be readily calculated from basic results obtained from a typical meta-analysis as, I 2 = 100 % × (Q–df)/Q, where Q is Cochrane’s heterogeneity statistic and df is the degrees of freedom. An I 2 value <25 % was considered homogeneous, an I 2-statistic between 25 and 50 % as low heterogeneity, an I 2-statistic between 50 and 75 % as moderate heterogeneity, and an I 2-statistic above 75 % as high heterogeneity [15]. Although the random-effects model cannot explain or remove the heterogeneity, for which we still used because it was considered to be more suitable for the statistical combination of LBP trials than the fixed-effect model [16]. Dichotomous variables are presented as relative risk (RR) and continuous variables as mean difference (MD), both with 95 % confidence intervals (CI) and probability value. These data were calculated when one outcome was assessed in different ways in different trials. The meta-analysis was performed by RevMan 5.1 software (Cochrane Collaboration, Oxford, UK) for outcome measures. A level of P ≤ 0.05 was considered statistically significant.
Results
Search results
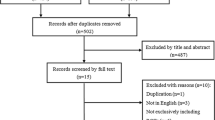
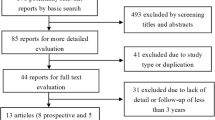
The process of searching relevant literature and the results is shown in Fig. 1. Seven published RCTs [17–23] with a total of 1,584 patients were included according to the inclusion criteria. The characteristics of the studies and participants are listed in Table 2.
Results of methodological quality
As shown in Table 2, it is indicated that most studies achieved high quality by modified Jadad scale. However, the main shortcoming reflected in nearly all studies was the lack of blinding method, which might lead to a certain degree of detection bias. All of the participants in the included studies had performed the follow-up for 2 years. The two studies [18, 21] that scored <4 had inappropriate randomization. The data of the Sasso’s study have not been pooled into the meta-analysis because of the extremely low follow-up rate, with only 18 out of the 67 patients completed the follow-up [18].
Clinical relevance
The results of clinical relevance are shown in Table 3. The patient details and intervention procedures were clearly recoded in all included studies in effort to allow researchers to replicate them in clinical practice. The complication, one of relevant outcomes, was not reported in the study [21]. An improvement more than 20 % in pain scores and improvement more than 25 % in functioning scores were accomplished in all included studies. In other word, the effect of the treatment was clinically important. The consistent outcomes suggested that the treatment benefits were likely worth the potential harms.
Heterogeneity
There were similar demographic characteristics, pain and functioning status baseline for the participants from seven included studies. Four different artificial discs (ProDisc-L, Maverick, CHARITE’ and FlexiCore) were used in these studies. Circumferential fusion was performed in four studies [18, 19, 21, 22], ALIF in Blumethal’s study [17] and instrumented PLF or PLIF in Berg’s study [23]. The surgical data were not pooled together because of the above differences. Most outcomes were measured by the same method in the studies. In random-effects meta-analysis, heterogeneity was observed in duration of hospitalization (I 2 = 91 %, P < 0.00001), proportion of patients choosing the same treatment again (I 2 = 31 %, P = 0.22), operation time (I 2 = 99 %, P < 0.00001), blood loss (I 2 = 95 %, P < 0.00001), reoperation rate (I 2 = 55 %, P = 0.06). The outcomes regarding patient functioning, painfulness and proportion of patients returning to full-time/part-time work (I 2 = 0 %) and the complication (I 2 = 5 %) were consistent.
Meta-analyses results
As revealed in Fig. 2, the patient’s functioning ability measured by ODI in the TDR group was better than that of the fusion group (MD −5.09; 95 % CI [−7.33, −2.84]; P < 0.00001), with statistical significance between the two groups. Unfortunately, the MD of five Owestry points was not clinically relevant. The VAS score of painfulness for TDR group was less than that of the fusion group (MD −5.31; 95 % CI [−8.35, −2.28]; P = 0.0006, Fig. 3), but the MD of five points was also not clinically significant. There was a shorter duration of hospitalization (MD −0.82; 95 % CI [−1.38, −0.26]; P = 0.004, Fig. 4) in TDR group than in fusion group. Besides, A greater proportion of patients in TDR group were willing to choose the same operation again (OR 2.32; 95 % CI [1.69, 3.20]; P < 0.00001, Fig. 5), with 79.2 vs. 63.0 %. However, there was no significant difference in operation time (MD −44.16; 95 % CI [−94.84, 6.52]; P = 0.09, Fig. 6), blood loss (MD −29.14; 95 % CI [−173.22, 114.94]; P = 0.69, Fig. 7), complications (OR 0.72; 95 % CI [0.45, 1.14]; P = 0.16, Fig. 8), reoperation rate (OR 0.83; 95 % CI [0.39, 1.77]; P = 0.63, Fig. 9) and the proportion of patients who returned to full-time/part-time work (OR 1.10; 95 % CI [0.86, 1.41]; P = 0.47, Fig. 10) between TDR group and the fusion group.
Functional assessment
No meta-analysis on functional recovery was carried out because different assessment systems had been used in the studies and few effective data could be extracted and pooled. Outcomes and conclusions concerning functional recovery varied. At the 2 years of follow-up, significant differences were observed in the overall clinical success in TDR and fusion group from the studies reported by Blumenthal et al. [17] (63.6 vs. 56.8 %, P = 0.0004), Zigler et al. [19] (63.5 vs. 45.1 %, P = 0.0053), and Gornet et al. [20] (73.5 vs. 55.3 %, P < 0.001). However, Berg et al. [23] reported that there were no differences in ODI success between TDR group and fusion group. The author still believed that the efficacy of the TDR could be improved with the strictly choosing surgical indications. Overall, there is strong evidence that TDR patients showed satisfactory outcomes than fusion patients in ODI and VAS scores. Therefore, it could be said that the functional recovery in the TDR group was better than in the fusion group.
Discussion
Lumbar fusion is a well-established procedure for the treatment of degenerative lumbar diseases [3, 24–27]. However, the original biomechanics of the spine was altered because of the loss of motion at the fused segments [28]. In addition, spinal fusion is associated with a common complication of adjacent disc degeneration [2, 6, 29–31]. Adjacent segment degeneration can cause significantly stenotic lesion or instability, for which additional operations are often required [32].
TDR has increased in popularity as an alternative for lumbar fusion [33]. The technique is to restore and maintain spinal segment motion, which is attempted to prevent adjacent level degeneration at the operated segments [34, 35]. TDR provides the opportunity to restore normal segmental motion of the spine and normal loading to the adjacent segment, preventing the degeneration progression of the adjacent disc. But excessive forces are concentrated on the facet joints at the level of TDR insertion. Therefore, most problems with the TDR occur at the insertion level and not at the adjacent level. However, there is still debate on the preferred surgical method for the degenerative lumbar spine. The purpose of this study is to compare the effectiveness and the safety of TDR to fusion for the treatment of lumbar DDD.
Results of our meta-analyses confirmed that TDR shows significant safety and efficacy comparable to lumbar fusion at 2 year follow-up. Besides, TDR has significant superiority in improved physical function and reduced pain. We consider that this superiority is associated with maintaining normal spinal segmental motion. These are in accordance with the conclusion proposed by Yajun et al. [9]. However, our meta-analyses offered new findings. TDR also shows significant superiority in shortened duration of hospitalization as compared to the fusion. It suggested that the functional recovery in the TDR group was better than in the fusion group.
Some basic epidemiological information of the participates can be derived from Table 2. It is noticeable that the number of TDR was about two times as the fusion except in Berg et al. [23]. Second, most lumbar DDD patients were in the age group of 35 s–45 s. This indicated that the middle-aged population should be given more attention on heavy manual work because their intervertebral discs are no longer as good as they were in adolescence. Third, sex ratio was 1:1 indicating that there was no correlation with lumbar DDD.
The statistical results of TDR versus fusion were not stated in the previous systematic reviews because of the lack of relevant RCTs [36, 37]. The previous meta-analysis confirmed that the TDR does not show significant superiority for the treatment of lumbar DDD when compared with fusion [9]. The meta-analysis was based on 837 patients with lumbar DDD. The latest two high-quality RCTs [20, 22] which compare TDR to the spinal fusion were added in our study, with a total of 1,584 patients. When effective data from the six high-quality studies were pooled, we find that the patients with TDR had a better function and back or leg pain status and shorter duration of hospitalization stay. There was no significant difference in operation time, blood loss, complications, reoperation rate and the proportion of patients who returned to full-time/part-time work between TDR group and the fusion group.
In our meta-analysis, seven published RCTs on lumbar TDR versus fusion were analyzed. Five studies had good methodological qualities (Jadad scores ≥4), two studies only gained three scores which implied a higher risk of bias. The most prevalent methodological shortcomings appeared to be insufficiency regarding the outcome assessor blinding to intervention. The low number of included studies limited our assessment of potential publication bias by the funnel plot and unpublished researches with negative results cannot be identified. Therefore, publication bias may exist, which could result in the overestimation of the effectiveness of interventions.
Different procedures of the fusion and different types of artificial discs may affect the comparing outcomes between the interventions, although no artificial disc is shown to be superior or inferior to the others [23]. Fusion method also could result in different operative data, even if there is no significant difference in clinical and function results [38, 39]. In addition, the results are affected by heterogeneity caused by random sampling. For example, the results of operating time, blood loss and duration of hospitalization presented significant heterogeneity. Therefore, the results of this meta-analysis should be cautiously accepted. Besides, the benefits of motion preservation and protecting adjacent levels, long-term complications and surgical revisions still remain unproved from the existing data. More independent high-quality RCTs with long-term outcomes are needed to strengthen the quality of evidence and contribute information to complement the findings.
Conclusion
TDR showed significant safety and efficacy comparable to lumbar fusion at 2 year follow-up. TDR demonstrated superiorities in improved physical function, reduced pain and shorten duration of hospitalization. The benefits of operating time, blood loss, motion preservation and the long-term complications are still unable to be proved.
References
Berg S, Tropp HT, Leivseth G (2011) Disc height and motion patterns in the lumbar spine in patients operated with total disc replacement or fusion for discogenic back pain. Results from a randomized controlled trial. Spine J 11:991–998
Kumar A, Beastall J, Hughes J et al (2008) Disc changes in the bridged and adjacent segments after Dynesys dynamic stabilization system after two years. Spine (Phila Pa 1976) 33:2909–2914
Bono CM, Kadaba M, Vaccaro AR (2009) Posterior pedicle fixation-based dynamic stabilization devices for the treatment of degenerative diseases of the lumbar spine. J Spinal Disord Tech 22:376–383
Kalanithi PS, Patil CG, Boakye M (2009) National complication rates and disposition after posterior lumbar fusion for acquired spondylolisthesis. Spine (Phila Pa 1976) 34:1963–1969
Lee SE, Park SB, Jahng TA, Chung CK, Kim HJ (2008) Clinical experience of the dynamic stabilization system for the degenerative spine disease. J Korean Neurosurg Soc 43:221–226
Reyes-Sanchez A, Zarate-Kalfopulos B, Ramirez-Mora I, Rosales-Olivarez LM, Alpizar-Aguirre A, Sanchez-Bringas G (2010) Posterior dynamic stabilization of the lumbar spine with the Accuflex rod system as a stand-alone device: experience in 20 patients with 2-year follow-up. Eur Spine J 19:2164–2170
Auerbach JD, Jones KJ, Milby AH, Anakwenze OA, Balderston RA (2009) Segmental contribution toward total lumbar range of motion in disc replacement and fusions: a comparison of operative and adjacent levels. Spine (Phila Pa 1976) 34:2510–2517
Gamradt SC, Wang JC (2005) Lumbar disc arthroplasty. Spine J 5:95–103
Yajun W, Yue Z, Xiuxin H, Cui C (2010) A meta-analysis of artificial total disc replacement versus fusion for lumbar degenerative disc disease. Eur Spine J 19:1250–1261
Oremus M, Wolfson C, Perrault A, Demers L, Momoli F, Moride Y (2001) Interrater reliability of the modified Jadad quality scale for systematic reviews of Alzheimer’s disease drug trials. Dement Geriatr Cogn Disord 12:232–236
van Tulder M, Furlan A, Bombardier C, Bouter L (2003) Updated method guidelines for systematic reviews in the Cochrane collaboration back review group. Spine (Phila Pa 1976) 28:1290–1299
Ostelo RW, de Vet HC (2005) Clinically important outcomes in low back pain. Best Pract Res Clin Rheumatol 19:593–607
Lauridsen HH, Hartvigsen J, Manniche C, Korsholm L, Grunnet-Nilsson N (2006) Responsiveness and minimal clinically important difference for pain and disability instruments in low back pain patients. BMC Musculoskelet Disord 7:82
Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21:1539–1558
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327:557–560
Furlan AD, Pennick V, Bombardier C, van Tulder M (2009) updated method guidelines for systematic reviews in the Cochrane Back Review Group. Spine (Phila Pa 1976) 34:1929–1941
Blumenthal S, McAfee PC, Guyer RD et al (2005) A prospective, randomized, multicenter food and drug administration investigational device exemptions study of lumbar total disc replacement with the CHARITE artificial disc versus lumbar fusion: part I: evaluation of clinical outcomes. Spine (Phila Pa 1976) 30(1565–1575):E387–E391
Sasso RC, Foulk DM, Hahn M (2008) Prospective, randomized trial of metal-on-metal artificial lumbar disc replacement: initial results for treatment of discogenic pain. Spine (Phila Pa 1976) 33:123–131
Zigler J, Delamarter R, Spivak JM et al (2007) Results of the prospective, randomized, multicenter food and drug administration investigational device exemption study of the ProDisc-L total disc replacement versus circumferential fusion for the treatment of 1-level degenerative disc disease. Spine (Phila Pa 1976) 32:1155–1163
Gornet MF, Burkus JK, Dryer RF, Peloza JH (2011) Lumbar disc arthroplasty with MAVERICK disc versus stand-alone interbody fusion: a prospective, randomized, controlled, multicenter investigational device exemption trial. Spine (Phila Pa 1976) 36:E1600–E1611
Delamarter RB, Bae HW, Pradhan BB (2005) Clinical results of ProDisc-II lumbar total disc replacement: report from the United States clinical trial. Orthop Clin North Am 36:301–313
Delamarter R, Zigler JE, Balderston RA, Cammisa FP, Goldstein JA, Spivak JM (2011) Prospective, randomized, multicenter food and drug administration investigational device exemption study of the ProDisc-l total disc replacement compared with circumferential arthrodesis for the treatment of two-level lumbar degenerative disc disease: results at 24 months. J Bone Joint Surg Am 93:705–715
Berg S, Tullberg T, Branth B, Olerud C, Tropp H (2009) Total disc replacement compared to lumbar fusion: a randomised controlled trial with 2-year follow-up. Eur Spine J 18:1512–1519
Ozer AF, Crawford NR, Sasani M et al (2010) Dynamic lumbar pedicle screw-rod stabilization: two-year follow-up and comparison with fusion. Open Orthop J 4:137–141
Morishita Y, Ohta H, Naito M et al (2011) Kinematic evaluation of the adjacent segments after lumbar instrumented surgery: a comparison between rigid fusion and dynamic non-fusion stabilization. Eur Spine J 20:1480–1485
Cheng BC, Gordon J, Cheng J, Welch WC (2007) Immediate biomechanical effects of lumbar posterior dynamic stabilization above a circumferential fusion. Spine (Phila Pa 1976) 32:2551–2557
Beastall J, Karadimas E, Siddiqui M et al (2007) The Dynesys lumbar spinal stabilization system: a preliminary report on positional magnetic resonance imaging findings. Spine (Phila Pa 1976) 32:685–690
Yu SW, Yen CY, Wu CH, Kao FC, Kao YH, Tu YK (2012) Radiographic and clinical results of posterior dynamic stabilization for the treatment of multisegment degenerative disc disease with a minimum follow-up of 3 years. Arch Orthop Trauma Surg 132:583–589
Zencica P, Chaloupka R, Hladikova J, Krbec M (2010) Adjacent segment degeneration after lumbosacral fusion in spondylolisthesis: a retrospective radiological and clinical analysis. Acta Chir Orthop Traumatol Cech 77:124–130
Harrop JS, Youssef JA, Maltenfort M et al (2008) Lumbar adjacent segment degeneration and disease after arthrodesis and total disc arthroplasty. Spine (Phila Pa 1976) 33:1701–1707
Levin DA, Hale JJ, Bendo JA (2007) Adjacent segment degeneration following spinal fusion for degenerative disc disease. Bull NYU Hosp Jt Dis 65:29–36
Park P, Garton HJ, Gala VC, Hoff JT, McGillicuddy JE (2004) Adjacent segment disease after lumbar or lumbosacral fusion: review of the literature. Spine (Phila Pa 1976) 29:1938–1944
van den Eerenbeemt KD, Ostelo RW, van Royen BJ, Peul WC, van Tulder MW (2010) Total disc replacement surgery for symptomatic degenerative lumbar disc disease: a systematic review of the literature. Eur Spine J 19:1262–1280
Frelinghuysen P, Huang RC, Girardi FP, Cammisa FJ (2005) Lumbar total disc replacement part I: rationale, biomechanics, and implant types. Orthop Clin North Am 36:293–299
McAfee PC, Cunningham B, Holsapple G et al (2005) A prospective, randomized, multicenter food and drug administration investigational device exemption study of lumbar total disc replacement with the CHARITE artificial disc versus lumbar fusion: part II: evaluation of radiographic outcomes and correlation of surgical technique accuracy with clinical outcomes. Spine (Phila Pa 1976) 30:1576–1583 E388–E390
Freeman BJ, Davenport J (2006) Total disc replacement in the lumbar spine: a systematic review of the literature. Eur Spine J 15(Suppl 3):S439–S447
Gibson JN, Waddell G (2005) Surgery for degenerative lumbar spondylosis. Cochrane Database Syst Rev D1352
Han X, Zhu Y, Cui C, Wu Y (2009) A meta-analysis of circumferential fusion versus instrumented posterolateral fusion in the lumbar spine. Spine (Phila Pa 1976) 34:E618–E625
Madan SS, Boeree NR (2003) Comparison of instrumented anterior interbody fusion with instrumented circumferential lumbar fusion. Eur Spine J 12:567–575
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rao, MJ., Cao, SS. Artificial total disc replacement versus fusion for lumbar degenerative disc disease: a meta-analysis of randomized controlled trials. Arch Orthop Trauma Surg 134, 149–158 (2014). https://doi.org/10.1007/s00402-013-1905-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00402-013-1905-4