Abstract
Purpose
Recent reports regarding endoscopic third ventriculostomy (ETV) for pediatric hydrocephalus revealed that ETV could avoid cerebrospinal fluid (CSF) shunting in certain types of hydrocephalus. However, the effectiveness of ETV for “pure” communicating hydrocephalus that has no obstruction through CSF pathway is still unknown. In this study, we report clinical outcome of ETV and CSF shunting for communicating hydrocephalus and discuss the efficacy of ETV for pure communicating hydrocephalus.
Methods
Children less than 15 years old who underwent ETV or CSF shunting for communicating hydrocephalus were retrospectively reviewed. The absence of obstruction through CSF circulation was confirmed by CT cisternography or cine-contrast image in MRI.
Results
Sixty-three patients (45 CSF shunting and 18 ETV) were included. The mean follow-up period was 6.1 years. The success rate was 60% in CSF shunting and 67% in ETV at the last visit (p = 0.867). Normal development was observed in 24 patients (53%) in CSF shunting and 12 patients (67%) in ETV (p = 0.334). There was a significant difference in the mean time to failure (CSF shunting: 51.1 months, ETV 3.6 months, p = 0.004). The factor that affected success rate in ETV was the age at surgery (success 21.6 months, failure 4.4 months, p = 0.024) and ETV success score (success 66.7, failure 50.0, p = 0.047).
Conclusion
Clinical outcomes of ETV were not inferior to those of CSF shunting in patients with communicating hydrocephalus. Further studies is required to elucidate to establish the consensus of ETV as a treatment option for communicating hydrocephalus.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Endoscopic third ventriculostomy (ETV) is a surgical treatment for hydrocephalus alternative to cerebrospinal fluid (CSF) shunting. ETV is efficacious for non-communicating hydrocephalus caused by aqueductal stenosis or infratentorial brain tumors; in other words, it elicits an effect in case of hydrocephalus maintaining absorption capacity of CSF [1, 2]. Meanwhile, ETV for communicating hydrocephalus is less likely to avoid CSF shunting than that for non-communicating hydrocephalus. Therefore, CSF shunting still remains the first-line treatment for communicating hydrocephalus.
Warf reported some favorable results of ETV concurrently with choroid plexus cauterization (CPC) in various types of hydrocephalus including communicating hydrocephalus [3,4,5,6]. However, they were not pure communicating hydrocephalus because some pathology involves the obstruction of fourth ventricle outlet by membranate closure or tonsillar herniation [7]. Success rate after ETV focusing on pure communicating hydrocephalus is still unknown.
It is known that the neurocognitive outcome in patients with hydrocephalus is lower than that in healthy children [8]. Although patients with ETV failure can have a stagnation of development until CSF shunting, the difference in the long-term intellectual outcome between success and failure is still unclear [9].
This study was conducted to compare clinical outcome of ETV and CSF shunting for patients with communicating hydrocephalus, and to analyze the outcome of intellectual development after surgery. In addition, the potential of ETV as a treatment option for communicating hydrocephalus is discussed.
Methods
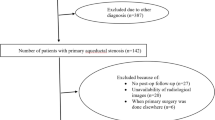
The present study was approved by institutional ethical committee in National Center for Child Health and Development. Written informed consent was obtained from all patients for postoperative data analysis. Subjects were children under 15 years old who underwent ETV or shunting for communicating hydrocephalus between April 2002 and April 2019 in National Center for Child Health and Development. Data were collected from medical records and retrospectively reviewed.
Evaluation of hydrocephalus
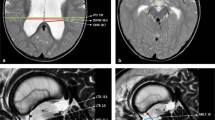
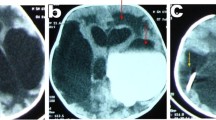
Communicating hydrocephalus was determined by computed tomographic cisternography (CTC) and/or cine-contrast image in MRI. In CTC, a small amount of contrast agent was injected by lumber puncture or from CSF reservoir if placed previously. CT scan was performed after 3, 6, and 24 h after the injection of contrast agent. When the contrast agent diffused both into all ventricular system and into cerebral surface, it was defined as communicating hydrocephalus (Fig. 1). In cine-contrast image in MRI, when CSF flow was observed both in the mesencephalic aqueduct and in the outlet of the fourth ventricle, it was defined as communicating hydrocephalus (Fig. 2). Patients without CTC or cine-contrast image in MRI were excluded from this study.
ETV success score (ETVSS) was calculated by the age, the etiology, and the presence of previous shunt [10]. Ventricular size was measured in CT scan, and the frontal and occipital horn ratio (FOHR) was calculated [11]. Neurocognitive development was evaluated by Kyoto Scale of Psychological Development [12], Tanaka-Binét test [13], or Wechsler Intelligence Scale for Children, third edition (WISC-III) depending on the age. Although normal value in each battery is not strictly defined, in this study, we defined normal development as more than 79 of the developmental or intellectual quotient (DQ/IQ).
Failure of ETV was defined as necessitating CSF shunting determined by progression of head circumference, bulging fontanelle, and/or enlarging ventricle or skull suture in CT scan. Failure of shunting was defined as necessitating shunt revision due to shunt malfunction.
Surgical indication and procedure
We inform parents about treatment options including both CSF shunting and ETV with or without CPC in any type of hydrocephalus. In principle, we usually propose CSF shunting for communicating hydrocephalus. ETV with or without CPC was performed when parents do not accept CSF shunting and strongly desire ETV.
Detailed surgical procedures of ETV were previously described [14]. In brief, a frontal approach was adopted and a small amount of CSF was obtained for a specimen. A flexible endoscope was inserted into the third ventricle through the foramen of Monro. The floor of the third ventricle was perforated using a balloon catheter. When ETV was followed by CPC, choroid plexus was extensively cauterized using an endoscopic monopolar electrode. Septostomy was performed to cauterize choroid plexus in the contralateral ventricle.
Statistical analyses
Each statistical comparison was performed with JMP 15.1 (SAS Institute, Cary, NC, USA). The nonparametric Wilcoxon signed-rank test or the chi-square test was used to compare data between groups. The paired Student’s t-test was used for comparison between preoperative and postoperative values. In multivariate analysis, logistic regression analysis was used to the factors that influenced outcome. The survival time of each treatment was analyzed with the Kaplan–Meier curve and log-rank test. A significance level of p < 0.05 was considered for all analysis.
Results
Patients’ characteristics are summarized in Table 1. Sixty-three patients (36 boys and 27 girls) were included in the study. Gestational age at birth was 37.3 ± 3.5 weeks. The age at surgery was 16.8 ± 18.4 months ranging from 21 days to 6.4 years. The value of protein in CSF obtained during surgery was 31.9 ± 41.1 mg/dl. The mean ETVSS was 58.9 ± 15.1. Preoperative FOHR was 0.49 ± 0.10. Etiology of hydrocephalus was post-intraventricular hemorrhage (IVH) in 5, Blake’s pouch cyst (BPC) in 3, Dandy–Walker syndrome (DWS) in 3, myelomeningocele (MMC) in 3, post-infection (meningitis) in 3, and unknown in 46. Normal development was observed in 36 patients (58.7%). The mean follow-up period was 6.1 years.
CSF shunting vs ETV (Table 2)
The success rate was 60% in CSF shunting and 67% in ETV (p = 0.867). Although pre- and post-FOHR were statistically different, it was more pronounced postoperatively (preoperative: CSF shunting 0.48 ± 0.10, ETV 0.52 ± 0.08, p = 0.035, postoperatively: 0.38 ± 0.08, 0.45 ± 0.08, p = 0.001). The Kaplan–Meier survival curve is shown in Fig. 3. There was no significant difference between the 2 groups according to log-rank test (p = 0.775: log-rank test). ETV failure did not occur after 217 days of surgery. Meanwhile, shunt failure occurred throughout the entire period. The mean time to failure was 3.6 ± 2.8 and 51.4 ± 42.4 months, respectively (p = 0.004). There was no difference in normal development between the 2 groups (CSF shunting 24 (53%), ETV 12 (67%), p = 0.334).
Comparison between success and failure in each group
CSF shunting group (Table 3)
Twenty-seven patients (60%) had not required additional surgery, and 18 (40%) had required revision surgery at the last visit. There were no significant differences between failure and success groups in gestational weeks (37.9 ± 2.7 vs 37.3 ± 2.9, p = 0.504), the age at surgery (13.4 ± 14.3 vs 19.6 ± 17.7 months, p = 0.175), ETVSS (56.7 ± 13.7 vs 58.9 ± 12.3, p = 0.573), preoperative FOHR (0.48 ± 0.11 vs 0.47 ± 0.09, p = 0.871), and CSF protein (13.9 ± 10.1 vs 16.8 ± 16.8 mg/dl, p = 0.904). Regarding etiology, unknown and IVH were observed in both groups whereas post-infection, MMCs were only in success cases. Normal development was in 12 patients (67%) of failure group and in 12 (44%) of success group without statistical significance (p = 0.277).
ETV group
Twelve patients (66.7%) had not required CSF shunting and six (33.3%) had required CSF shunting at the last visit. Among six patients with CSF shunting, interval between ETV and CSF shunting was 110.2 ± 85.8 days. In univariate analysis, the age at surgery was significantly earlier in failure cases than that in success cases (4.4 ± 3.5 vs 21.6 ± 24.8 months, p = 0.024). ETVSS was also associated with success rate of ETV (50.0 ± 15.3 vs 66.7 ± 18.9, p = 0.047). There were no significant differences between failure and success groups in gestational weeks (38.8 ± 2.0 vs 35.5 ± 5.3 weeks, p = 0.19), preoperative FOHR (0.56 ± 0.09 vs 0.50 ± 0.06, p = 0.30), and CSF protein (48.3 ± 57.4 vs 23.8 ± 26.1 mg/dl, p = 0.39). In multivariate analysis, only the age at surgery affected success of ETV (p = 0.037). CPC was added after ETV in 7 (58%) of success group and in 6 (100%) of failure group. Regarding etiology, IVH, BPC, and unknown were observed in both groups whereas DWS, MMC, and post-infection were only in success cases. Normal development was observed in five patients (83%) of failure group and in seven (58%) of success group without statistical significance (p = 0.290).
Discussion
In recent years, some studies on comparison between CSF shunting and ETV have been reported [15, 16]. Those reports advocated that ETV was not inferior to CSF shunting regarding success rate; nevertheless, ETV has not been considered as the first-line treatment for hydrocephalus at present, particularly in hydrocephalus including communicating etiology. According to Warf’s series, success rate of ETV was 63% in post-infectious hydrocephalus [6], 76% in MMC [5], 82% in DWS [4], and 72% in idiopathic hydrocephalus [3]. However, it was not confirmed that obstruction of the fourth ventricle outlet by membranate structure or tonsillar herniation was observed in those series. Therefore, the success rate of “pure” communicating hydrocephalus in those series was unknown. There was a few series of ETV for adult hydrocephalus that patency through CSF pathway was confirmed by CTC or cine-contrast image in MRI [17, 18]. Nonetheless, the pathophysiology of hydrocephalus in adults was different from that in children. The present study revealed that ETV alone or ETV with CPC could avoid CSF shunt in 67% of patients with communicating hydrocephalus, which was roughly equal to previous studies. Given that the outcomes of previous reports and outs were equal, hydrocephalus classified as communicating etiology, such as post-infectious, MMC, or idiopathic, may rarely include obstructive one.
In previous series and the present one, the age at surgery affected the success rate of ETV. Younger patients tended to need additional surgery after failed ETV. Most failures occurred in less than 6 months of age, and reoperation was performed within 1 year after ETV [8, 19]. One of the possible reasons why ETV fails in early infants is a disproportion between CSF production and absorption. In healthy infants, physiological subdural effusion can be observed from birth up to seven month of age because the increase of CSF absorption cannot catch up with CSF production [20]. Particularly in hydrocephalus in which the absorption capacity is essentially low, the CSF production may be more likely to exceed the absorption in early infants. Accordingly, CPC is reasonable to resolve the disproportion of CSF. Warf et al. reported ETV with CPC can succeed in 66% in comparison to ETV alone in 47% [21].
Neurocognitive development
The neurocognitive outcome in children with hydrocephalus is worse than that in healthy children [22, 23]. Neurocognitive deterioration due to progressive hydrocephalus can be prevented by CSF diversion over a long period. In the 40-year cohort of 128 treated hydrocephalus patients, two thirds of patients graduated from a normal school; in addition, 56% were socially independent and 42% were employed [24]. Patients in whom ETV failed may suffer from delay of improvement of hydrocephalus compared to successful patients. It is unclear whether this delay affects developmental outcomes. In the present study, CSF shunting was performed 110 days after ETV on average in failure cases, there was no significant difference in neurocognitive development between success and failure cases. In the literature, ETV can prevent neurocognitive decline, achieving equivalent to CSF shunt 1 year after surgery [25]. In long-term, we previously reported that intelligence was not significantly different between CPC and CSF shunting [26]. There has been no evidence that CSF shunting was better than ETV [27]. Further, long-term outcome of comparison between ETV and CSF shunting is expected.
How does ETV improve communicating hydrocephalus?
It has been believed that CSF is produced from choroid plexus in ventricles, passes through the foramen Magendie or Luschka, and is absorbed into the arachnoid villi, which is so-called bulk flow theory (BFT). BFT can explain how ETV resolves obstructive hydrocephalus by making a new CSF pathway from ventricle to cisternal space. By contrast, improvement of communicating hydrocephalus after ETV alone or ETV with CPC cannot be explained by BFT. Recently, CSF is thought to be absorbed via capillary vessels [28]. Particularly in children less than 2 years old, most CSF absorption depends on capillary vessels of ventricular ependyma because of the immaturity of arachnoid villi [29]. An increase of intraventricular pulsative pressure decreases CSF absorption via capillary vessels [30]. As the absorption via capillary vessels decreases, CSF gradually accumulates, resulting in a decrease of intracranial venous volume that buffers pulsative pressure. Furthermore, it decreases the CSF absorption via capillary vessels and accumulates CSF, which falls in a vicious cycle [3]. ETV terminates this cycle by making a hole to the cisternal space through which a pulsative pressure can be buffered and CPC attenuates pulsative pressure originating from choroid plexus. This theory can be applied to case of noncommunicating hydrocephalus in which pressure gradient exists between intra- and extraventricular space. It is unknown whether pulsative pressure between ventricle and cisternal space is constant. There may be pressure gradient even in case of communicating hydrocephalus. In such case, ETV with/without CPC can be effective. In the failure case, ETV and CPC may have not been able to achieve a sufficient reduction of pulsative pressure, or the CSF absorptive function in the intraventricular glymphatic system may have lost by infection or hemorrhage.
Study limitations
For comparison of intellectual development, the effects of etiology must be taken into account. In the present study, the number of patients in each etiology was too small to compare. There was a considerable selection bias because we substantially offered CSF shunting for communicating hydrocephalus. In order to the consensus for surgical indication should be established, ideally, randomized comparison between ETV and CSF shunting for communicating hydrocephalus is required. In addition, we are still far from elucidation of underlying mechanisms for improvement of communicating hydrocephalus by ETV. Further basic and clinical researches for CSF circulation are necessary.
Conclusion
The clinical outcomes of ETV were comparable to those of CSF shunting for communicating hydrocephalus. Regarding intellectual development, ETV was not inferior to CSF shunting. ETV has a potential to avoid CSF shunting in patients with hydrocephalus even if hydrocephalus does not have any obstruction through CSF pathway. In order that ETV for communicating hydrocephalus to become common, it is necessary to accumulate evidence of noninferiority in case of ETV for communicating hydrocephalus compared to CSF shunting as the first-line treatment in terms of treatment success and future development.
References
Di Rocco F, Juca CE, Zerah M, Sainte-Rose C (2013) Endoscopic third ventriculostomy and posterior fossa tumors. World Neurosurg 79(S18):e15-19
Ogiwara H, Dipatri AJ Jr, Alden TD, Bowman RM, Tomita T (2010) Endoscopic third ventriculostomy for obstructive hydrocephalus in children younger than 6 months of age. Childs Nerv Syst : ChNS : Official Journal of the International Society for Pediatric Neurosurgery 26:343–347
Warf BC (2013) Congenital idiopathic hydrocephalus of infancy: the results of treatment by endoscopic third ventriculostomy with or without choroid plexus cauterization and suggestions for how it works. Childs Nerv Syst : ChNS : Official Journal of the International Society for Pediatric Neurosurgery 29:935–940
Warf BC, Dewan M, Mugamba J (2011) Management of Dandy-Walker complex-associated infant hydrocephalus by combined endoscopic third ventriculostomy and choroid plexus cauterization. J Neurosurg Pediatr 8:377–383
Warf BC, Campbell JW (2008) Combined endoscopic third ventriculostomy and choroid plexus cauterization as primary treatment of hydrocephalus for infants with myelomeningocele: long-term results of a prospective intent-to-treat study in 115 East African infants. J Neurosurg Pediatr 2:310–316
Warf BC, Dagi AR, Kaaya BN, Schiff SJ (2011) Five-year survival and outcome of treatment for postinfectious hydrocephalus in Ugandan infants. J Neurosurg Pediatr 8:502–508
McLone DG, Dias MS (2003) The Chiari II malformation: cause and impact. Childs Nerv Syst : ChNS : Official Journal of the International Society for Pediatric Neurosurgery 19:540–550
Kahle KT, Kulkarni AV, Limbrick DD Jr, Warf BC (2016) Hydrocephalus in children. Lancet 387:788–799
Baird LC (2016) First treatment in infants with hydrocephalus: the case for endoscopic third ventriculostomy/choroid plexus cauterization. Neurosurgery 63(Suppl 1):78–82
Kulkarni AV, Drake JM, Mallucci CL, Sgouros S, Roth J, Constantini S (2009) Endoscopic third ventriculostomy in the treatment of childhood hydrocephalus. J Pediatr 155: 254–259 e251
Kulkarni AV, Drake JM, Armstrong DC, Dirks PB (1999) Measurement of ventricular size: reliability of the frontal and occipital horn ratio compared to subjective assessment. Pediatr Neurosurg 31:65–70
Koyama T, Osada H, Tsujii H, Kurita H (2009) Utility of the Kyoto Scale of Psychological Development in cognitive assessment of children with pervasive developmental disorders. Psychiatry Clin Neurosci 63:241–243
Uno Y, Mizukami H, Ando M, Yukihiro R, Iwasaki Y, Ozaki N (2014) Reliability and validity of the new Tanaka B Intelligence Scale scores: a group intelligence test. PLoS One 9: e100262
Ogiwara H, Uematsu K, Morota N (2014) Obliteration of the choroid plexus after endoscopic coagulation. J Neurosurg Pediatr 14:230–233
Texakalidis P, Tora MS, Wetzel JS, Chern JJ (2019) Childs Nerv Syst : ChNS : Official Journal of the International Society for Pediatric Neurosurgery. Child’s nervous system : ChNS : official journal of the International Society for Pediatric Neurosurgery 35:1283–1293
Riva-Cambrin J, Kestle JRW, Rozzelle CJ, Naftel RP, Alvey JS, Reeder RW, Holubkov R, Browd SR, Cochrane DD, Limbrick DD, Shannon CN, Simon TD, Tamber MS, Wellons JC, Whitehead WE, Kulkarni AV (2019) Predictors of success for combined endoscopic third ventriculostomy and choroid plexus cauterization in a North American setting: a Hydrocephalus Clinical Research Network study. J Neurosurg Pediatr 1–11
Rangel-Castilla L, Barber S, Zhang YJ (2012) The role of endoscopic third ventriculostomy in the treatment of communicating hydrocephalus. World Neurosurg 77:555–560
Hailong F, Guangfu H, Haibin T, Hong P, Yong C, Weidong L, Dongdong Z (2008) Endoscopic third ventriculostomy in the management of communicating hydrocephalus: a preliminary study. J Neurosurg 109:923–930
Stone SS, Warf BC (2014) Combined endoscopic third ventriculostomy and choroid plexus cauterization as primary treatment for infant hydrocephalus: a prospective North American series. J Neurosurg Pediatr 14:439–446
Lam WW, Ai VH, Wong V, Leong LL (2001) Ultrasonographic measurement of subarachnoid space in normal infants and children. Pediatr Neurol 25:380–384
Warf BC (2005) Comparison of endoscopic third ventriculostomy alone and combined with choroid plexus cauterization in infants younger than 1 year of age: a prospective study in 550 African children. J Neurosurg 103:475–481
Lacy M, Pyykkonen BA, Hunter SJ, Do T, Oliveira M, Austria E, Mottlow D, Larson E, Frim D (2008) Intellectual functioning in children with early shunted posthemorrhagic hydrocephalus. Pediatr Neurosurg 44:376–381
Dalen K, Bruaroy S, Wentzel-Larsen T, Laegreid LM (2008) Intelligence in children with hydrocephalus, aged 4–15 years: a population-based, controlled study. Neuropediatrics 39:146–150
Paulsen AH, Lundar T, Lindegaard KF (2015) Pediatric hydrocephalus: 40-year outcomes in 128 hydrocephalic patients treated with shunts during childhood. Assessment of surgical outcome, work participation, and health-related quality of life. J Neurosurg Pediatr 16:633–641
Kulkarni AV, Schiff SJ, Mbabazi-Kabachelor E, Mugamba J, Ssenyonga P, Donnelly R, Levenbach J, Monga V, Peterson M, MacDonald M, Cherukuri V, Warf BC (2017) Endoscopic treatment versus shunting for infant hydrocephalus in Uganda. N Engl J Med 377:2456–2464
Okano A, Ogiwara H (2018) Long-term follow-up for patients with infantile hydrocephalus treated by choroid plexus coagulation. J Neurosurg Pediatr 22:638–645
Azab WA, Mijalcic RM, Nakhi SB, Mohammad MH (2016) Ventricular volume and neurocognitive outcome after endoscopic third ventriculostomy: is shunting a better option? A review. Childs Nerv Syst : ChNS : Official Journal of the International Society for Pediatric Neurosurgery 32:775–780
Greitz D (2007) Paradigm shift in hydrocephalus research in legacy of Dandy’s pioneering work: rationale for third ventriculostomy in communicating hydrocephalus. Childs Nerv Syst : ChNS : Official Journal of the International Society for Pediatric Neurosurgery 23:487–489
Oi S, Di Rocco C (2006) Proposal of “evolution theory in cerebrospinal fluid dynamics” and minor pathway hydrocephalus in developing immature brain. Childs Nerv Syst : ChNS : Official Journal of the International Society for Pediatric Neurosurgery 22:662–669
Di Rocco C, Pettorossi VE, Caldarelli M, Mancinelli R, Velardi F (1978) Communicating hydrocephalus induced by mechanically increased amplitude of the intraventricular cerebrospinal fluid pressure: experimental studies. Exp Neurol 59:40–52
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Usami, K., Ishisaka, E. & Ogiwara, H. Endoscopic third ventriculostomy and cerebrospinal fluid shunting for pure communicating hydrocephalus in children. Childs Nerv Syst 37, 2813–2819 (2021). https://doi.org/10.1007/s00381-021-05242-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-021-05242-4