Abstract
Object
This study compares endoscopic third ventriculostomy (ETV) and ventriculoperitoneal shunt (VPS) in the treatment of pediatric patients with marked obstructive hydrocephalus due to midline posterior fossa tumors.
Methods
Fifty-three pediatric patients with a midline posterior fossa tumor (32 medulloblastomas and 21 ependymomas) associated with marked hydrocephalus were studied. Patients were divided into two groups: group A (32 patients) operated by ETV with a mean follow-up of 27.4 months and group B (21 patients) operated by VPS with a mean follow-up of 25 months.
Results
Both procedures proved to be effective clinically and radiologically. In group A, intraoperative bleeding occurred in two cases (6.2%) and cerebrospinal fluid leakage in one case (3.1%). In group B, shunt infection occurred in two cases (9.4%), one of these two cases died 4.5 months postoperatively from ventriculitis. Subdural collection occurred in two cases (9.4%), epidural hematoma in one case (4.7%), and upward brain herniation in one case (4.7%). Endoscopic third ventriculostomy proved to be superior due to shorter duration of surgery (15 min versus 35 min), lower incidence of morbidity (9.3% versus 38%), no mortality (0% versus 4.7%), and lower incidence of procedure failure (6.2% versus 38%).
Conclusion
The shorter duration of surgery, the lower incidence of morbidity, the absence of mortality, the lower incidence of procedure failure, and the significant advantage of not becoming shunt dependent make ETV be recommended as the first choice in the treatment of pediatric patients with marked obstructive hydrocephalus due to midline posterior fossa tumors.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Posterior fossa is a common site for various tumors to occur during childhood. It was estimated that they comprise 54–60% of childhood brain tumors. Medulloblastomas represent approximately 20% of all childhood central nervous system tumors [49]. Ependymomas represent approximately 10% of all reported primary intracranial tumors in children [19]. Neurosurgeons are still divided in their opinions concerning the best way to manage obstructive hydrocephalus due to posterior fossa tumors.
Placement of a shunt was the operative “state of the art” over years [1, 2, 10, 26, 27]. However, the technological advances and changes in the availability of neuroimaging systems have allowed for earlier diagnosis and have led most pediatric neurosurgeons to use a combination of corticosteroids, direct approach to the tumor, and external ventricular drainage (EVD) [17, 43, 48, 51, 63]. The obstructive nature of the associated hydrocephalus makes internal cerebrospinal fluid (CSF) diversion to be a promising alternative. There are many studies which discuss the role of ETV in the treatment of obstructive hydrocephalus of different etiologies including some cases due to posterior fossa tumors [20, 28, 68], and only few studies specific for posterior fossa tumors in children [8, 55, 58].
Many articles have been published concerning the morbidity and mortality as well as the outcome of both CSF shunts and ETV, but the comparison between both procedures has rarely been addressed in the literature [13, 24, 31, 67]. The purpose of this study is to evaluate the role of ETV as compared to shunting procedure in the management of obstructive hydrocephalus due to posterior fossa tumors in children. This study compares the outcome and morbidity of both procedures in the management of these cases. It is the first study which is specific for midline posterior fossa tumors in pediatric patients with advanced hydrocephalus.
Material and methods
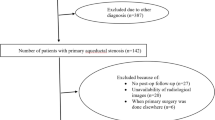
Fifty-three pediatric patients with obstructive hydrocephalus due to posterior fossa tumors were studied during the period between September 2003 and March 2009. The study included only patients with midline posterior fossa tumors (medulloblastomas and ependymomas), whereas cerebellar, brain stem, and cerebellopontine angle tumors were excluded. Cases of medulloblastomas and ependymomas are especially selected due to their vulnerability to cause marked obstructive hydrocephalus and in order to be able to compare homogeneous patients. A full history was taken, and all patients were subjected to a complete neurological examination. Preoperative computed tomography (CT) scan and magnetic resonance (MR) imaging were reviewed with special attention being paid to the degree of ventricular enlargement, the presence of periventricular edema, and location of the tumor. The degree of hydrocephalus (mild, moderate, marked) was defined in all cases based on a preoperative CT scan. The maximum width of the lateral ventricles at the frontal horns was divided by the largest transverse diameter of the skull’s internal table, producing a standardized ratio (Evans Ratio or Ventricular Index) [59]. Mild, moderate, and marked ventriculomegaly was designated for ratios of 0.27 to 0.34, 0.35 to 0.40, and >0.40, respectively. A ratio of <0.27 was considered to be normal. All the patients had severe hydrocephalus, whereas patients with mild–moderate hydrocephalus and nonhydrocephalic patients were excluded from the study.
The patients were grouped into two groups: group A included 32 cases operated by ETV and group B included 21 cases operated by VPS. Surgical failure means that the CSF diversion procedure could not be completed successfully. The patients were assessed postoperatively clinically regarding any improvement or worsening of preoperative symptoms and occurrence of new neurological deficits or postoperative complications. Follow-up CT scan was done for all the patients 3–5 days postoperatively (unless needed earlier), and it was examined regarding any improvement of hydrocephalus or periventricular edema or any change in size of subarachnoid space. Magnetic resonance imaging with CSF flow analysis and cine flow images were obtained prior to tumor excision in 12 cases included in group A. The tumor was directly approached 1–2 weeks after the CSF diversion procedure.
Any recurrence of intracranial hypertension symptoms among the follow-up period was considered to be an indication for repeating the CT scan and/or MR imaging. Any recurrence of hydrocephalus was considered to be a postoperative failure and it was an indication to perform a revision procedure. Revision means repeating the same CSF diversion procedure which was performed initially. The follow-up period ranged from 6.5 months to 6 years with a mean of 27.4 months in group A, and from 4.5 months to 5 years with a mean of 25 months in group B. Any documented mortality was recorded to be either hydrocephalus-related or nonhydrocephalus-related.
Data were collected and summarized in both groups regarding the duration of surgery, improvement of symptomatology, improvement of hydrocephalus, incidence of surgical failure, incidence of surgical complications, postoperative deficits, postoperative failure, incidence of shunt-free patients, and incidence of revision of the CSF diversion procedure. Data of both groups were analyzed and compared using the Student’st test, chi-square test, and Kaplan–Meier survival analysis with logrank test.
Demographic data
Among 32 patients included in group A, 18 patients were males and 14 were females. Their age ranged from 2 to 12 years with a mean of 6.5 years. In group B, 12 patients were males and nine patients were females. Their age ranged from 2.5 to 11.5 years with a mean of 7.2 years. The most common symptoms were increased intracranial pressure (ICP) symptoms in the form of headache, vomiting, and/or blurring of vision which were present in all patients included in both groups. Gait ataxia was present in 10 out of 32 patients (31.2%) included in group A, and in 8 out of 21 patients (38%) included in group B. The incidence of sixth nerve palsy, deteriorated conscious level, bulbar symptoms were 28.1%, 12.5%, 6.2%, respectively, in group A, compared to 28.5%, 9.4%, 14.2%, respectively, in group B. The duration of symptoms ranged from 1 to 24 weeks with a mean of 14.4 weeks in group A, and from 1.5 to 36 weeks with a mean of 18.6 weeks in group B (Table 1). The posterior fossa tumor was midline in all the patients included in both groups and it was associated with severe hydrocephalus.
Results
Group A
This group consisted of 32 pediatric patients with obstructive hydrocephalus due to posterior fossa tumors (20 medulloblastomas, 12 ependymomas) who were operated by ETV. The procedure time ranged from 10 to 30 min with a mean of 15 min. Arterial bleeding occurred in two patients (6.2%), it was minor, did not necessitate termination of the procedure, and it stopped with continuous irrigation. The procedure was completed successfully, and an external ventricular drain was left for few days; however, both patients needed a repeated ETV later on.
The intracranial hypertension symptoms improved postoperatively in 31 out of 32 cases (96.9%), ataxia improved in four out of ten cases (40%), sixth nerve palsy improved in six out of nine cases (66.7%). All the four cases presented with deteriorated conscious level became fully conscious (100%) immediately postoperatively. Postoperative CT scan showed mild improvement of hydrocephalus in 28 out of 32 cases (87.5%). The presence of CSF flow void across the ventriculostomy with an apparent systolic/diastolic flow in postoperative MRI confirmed its patency.
Cerebrospinal fluid leakage occurred in one patient (3.1%), and it stopped spontaneously on the 4th postoperative day. Gross total tumor excision was performed in 26 cases (81.3%), whereas in the remaining six cases (18.7%), the tumor was excised subtotally (Table 2). During surgery for excision of the posterior fossa tumor, the dura was lax enough in all the cases and there was no need for intraoperative CSF tapping. Retraction of the cerebellum was surprisingly easy and harmless, which gave us a good exposure and facilitated tumor dissection in most of the cases. Recurrence of hydrocephalus occurred in two patients at 6.5 and 14 months postoperatively, respectively (both patients had intraoperative bleeding), and a re-ventriculostomy was performed. No permanent procedure related morbidity or mortality was seen in this group of patients. Among the mean duration of follow-up of this group (27.4 months), all the 32 patients (100%) were shunt free.
Group B
This group consisted of 21 pediatric patients with obstructive hydrocephalus due to posterior fossa tumors (12 medulloblastomas, 9 ependymomas) who were operated by VPS. The procedure time ranged from 20 to 60 min with a mean of 35 min. The intracranial hypertension symptoms improved postoperatively in all cases (100%), ataxia improved in two out of eight cases (25%), and sixth nerve palsy improved in three out of six cases (50%). Both patients presented with deteriorated conscious level became fully conscious (100%) immediately postoperatively. Postoperative CT scan showed marked improvement of hydrocephalus in all cases (100%).
Shunt infection occurred in two patients (9.4%, Table 3), 1.5 and 5.5 months postoperatively, respectively. Subdural collection occurred in two cases (9.4%), it was bilateral and was evacuated surgically in one case because it had a significant mass effect and led to clinical symptoms. It was unilateral and resolved conservatively in the other case. Epidural hematoma occurred in one case (4.7%) and it was surgically evacuated. Deterioration of consciousness and Parinaud’s syndrome occurred in one case (4.7%), it was attributed to upward herniation and tumor excision was performed in this case on an urgent basis, and the patient recovered completely. The tumor was totally excised in 14 cases (66.7%), whereas in the remaining seven cases (33.3%), the excision was subtotal. Two patients (9.4%) developed pseudomeningocele after tumor excision, and it resolved spontaneously after shunt revision.
Among the mean follow-up duration of this group (25 months), postoperative failure was encountered in eight cases. Shunt revision was performed 15 times in these eight patients. The cause of first failure was proximal occlusion in three cases, distal occlusion in two cases, infection in two cases, and disconnection in one case. The number of revisions per patient among these eight cases with postoperative failure was 1.8, and the mean duration which lapsed between the VPS procedure and the first revision procedure was 5.6 months (Table 4). All the 21 patients (100%) included in this group became shunt dependent. There is only a single case of mortality (4.7%), and it was hydrocephalus-related. This is one of the two patients with shunt infection, who developed ventriculitis and died 4.5 months postoperatively.
Discussion
Cerebrospinal fluid shunts
The development of hydrocephalus in children with posterior fossa tumors is one of the main factors influencing the outcome. Children are very sensitive to ICP elevation, thus management of hydrocephalus has an utmost priority before any considerations are given to the surgical approach of the posterior fossa tumor itself. It is claimed that a precraniotomy shunt provides improvement in the patient’s physiological condition, a “slack posterior fossa,” and a smooth postoperative course [2, 50]. In one series [2], the operative mortality rate was decreased with preoperative shunting. However, following tumor excision, the obstructive component of the hydrocephalus is relieved in most patients, and the flow of CSF will be restored, thus the shunt will be no more necessary, but the patient now becomes shunt dependent. As long-term survival becomes more frequent in children with posterior fossa tumors, issues pertaining to the quality of life, such as postoperative shunt dependency become increasingly important and must be taken into consideration. Moreover, placement of shunt is a blind procedure and shunt systems are prone to several complications.
External ventricular drainage
Improvements in the availability and type of neuroimaging systems have resulted in establishing a markedly earlier diagnosis. This, coupled with the extensive list of potential complications associated with ventricular shunting in this patient population, resulted in another strategy recommending the use of corticosteroid therapy, early surgery, and EVD [17, 43, 48, 51, 63]. Nevertheless, EVD is associated with a significant risk of infection [59] and hemorrhage [58]. Moreover, such treatment might not be appropriate in young children with advanced hydrocephalus because these patients usually need a permanent diversion procedure. It was estimated that children who have advanced hydrocephalus and children who are younger than 3 years of age had persistent hydrocephalus which required a postoperative shunt in about 80% of cases [16]. Approximately, one third of patients overall will eventually require placement of a shunt [41]. Many factors have been reported to be associated with required shunt placement including a young age (<10 years), midline tumors, more severe ventricular enlargement at diagnosis, incomplete tumor resection, CSF-related infection, prolonged use of EVD, cadaveric dural grafts, and persistent pseudomeningocele [3, 9, 12, 14, 41, 48, 57].
Endoscopic third ventriculostomy
Jones et al. [33] appear to be the first who attempted ETV prior to resection of a case of posterior fossa tumor (medulloblastoma) [34]. The procedure was successful in controlling the intracranial hypertension, but the patient experienced increased ataxia. We did not observe this phenomenon in any of our patients. The rational basis of ETV is provided by the obstructive nature of hydrocephalus due to the presence of blockage of CSF pathway at level of the fourth ventricle outlets or at the aqueduct of Sylvius. Endoscopic third ventriculostomy creates a communication between the ventricular system and the subarachnoid spaces at the level of floor of the third ventricle (Fig. 1a).
a Magnetic resonance imaging T1-weighted sagittal view of a case of posterior fossa tumor (medulloblastoma), demonstrating communication between third ventricle and prepontine cistern after ETV. b Preoperative axial CT scan image showing severe obstructive hydrocephalus due to posterior fossa tumor. The presence of massive periventricular edema signifies acutely occurring hydrocephalus. c Postoperative axial CT scan image obtained 3 days after ETV showing mild reduction in ventricular size, resolution of periventricular edema, and opening of subarachnoid space
Operative findings
An argument was existing regarding the feasibility of performing ETV in obstructive hydrocephalus due to posterior fossa tumors, which was believed to be technically more difficult than in cases of congenital aqueduct stenosis. This is because the tumor bulk may displace the brain stem forward and obliterate the interpeduncular cistern, thus making ETV impossible. Our findings support what was previously reported [58], that posterior fossa tumors tend to elevate the brain stem as they push it forward. Thus, the floor of the third ventricle is displaced largely above the posterior clinoid, making enough room for the fenestration. Endoscopic third ventriculostomy was technically easy and successful in all the cases (100%) included in the present study, and this makes us conclude that the presence of anatomical distortion of the floor of the third ventricle due to posterior fossa tumors does not make the ETV procedure technically more difficult than in other cases. However, a recent study reported abandonment of ETV due to severely distorted anatomy in 1 out of 37 pediatric patients (2.7%) with posterior fossa tumors [8]. The mean duration of surgery in the ETV group was 15 min compared to 35 min in the VPS group (p < 0.01).
Operative outcome
In the current study, both procedures (ETV and VPS) proved to be effective clinically and radiologically. Improvement of intracranial hypertension symptoms occurred in 96.9% of ETV group and in 100% of the VPS group. Postoperative CT scan showed improvement of hydrocephalus in 87.5% of ETV group (Fig. 1b and c) and in 100% of VPS group, however, the decrease in ventricular size was marvelous in the VPS group. Another two studies [28, 68] have reported a significant experience in 17 and 21 cases, respectively, both with a 76% success rate of ETV in controlling hydrocephalus. More recent studies reported better results, in a study including 67 pediatric patients with obstructive hydrocephalus due to posterior fossa tumors operated by ETV, immediate postoperative symptomatic resolution occurred in 98.5% of patients [58]. In another series of 20 pediatric patients with posterior fossa tumors, ETV successfully resolved the intracranial hypertension before tumor removal in 95% of patients [55].
I believe that obstructive hydrocephalus secondary to posterior fossa tumors in children with good absorption of CSF from the subarachnoid space is a good selection for ETV. This is completely different from the long standing hydrocephalus associated with thickened third ventricular floor, possible significant anatomical anomalies [46, 53], and nonoptimal CSF absorption from the subarachnoid space in cases of congenital hydrocephalus, which undoubtedly will adversely affect the outcome and increase the incidence of morbidity. Although ETV achieves complete clinical recovery without reducing ventricular size in cases of aqueduct stenosis [42, 64], it achieves both clinical recovery and a definite improvement of hydrocephalus in pediatric patients with posterior fossa tumors.
Morbidity and mortality
The placement of a shunting device entails significant long-term risk of infection and malfunction. The development of shunt infection in a pediatric patient represents a particularly morbid condition with up to 20% infection related mortality [71]. A 22% infection rate per patient and 6% infection rate per procedure has been reported over a 10-year period study [4]. Migration and dissemination of tumor cells via shunt catheter to the peritoneal cavity is possible in both medulloblastomas and ependymomas, an incidence of up to 19% of extracranial metastasis was reported after shunting [27]. Overdrainage with possible occurrence of subdural, extradural, or intracerebral hematomas [29], upward transtentorial herniation [17], and intratumoral hemorrhage [70] may occur following shunting in advanced hydrocephalus. A 10% rate of upward herniation has been reported in cases of posterior fossa tumors subjected to preliminary shunting [17].
The overall complication rate of ETV in the literature is about 5–10% [65]. It includes uncontrollable bleeding, memory disturbance from injury to the fornix [23, 33], focal neurological deficits, third nerve palsy [33], and hypothalamic dysfunction [40]. Cerebrospinal fluid leakage was the commonest complication in one study [56].
In the current study, intraoperative bleeding was an important predictor for a bad outcome among the ETV group patients. Its incidence among this group was 6.2% compared to 0% in the VPS group. Both patients who were subjected to arterial bleeding needed a re-ventriculostomy operation later due to postoperative closure of the initial ventriculostomy. We agree with what was reported previously that increased experience in endoscopy leads to a reduction of surgery-related complications [32].
Postoperative complications in the VPS group proved to be more fatal than in the ETV group. Postoperative complications in the ETV group included CSF leak in one patient (3.1%), it was transient and was resolved conservatively. On the other hand, postoperative shunt infection occurred in two cases (9.4%) and it ended in mortality due to ventriculitis in one case (4.7%), and the other case was subjected to three shunt revision procedures. Subdural collection occurred in two cases (9.4%) after VPS (one of these two cases was bilateral and needed surgical evacuation), one case (4.7%) developed epidural hematoma which was evacuated surgically, and one case (4.7%) suffered a presumed “upward herniation” but recovered completely after urgently excising the posterior fossa tumor.
The increased incidence of subdural collection, epidural hematoma, and upward herniation in group B may be related to the marked decrease in ventricular size encountered after VPS, which makes us conclude that ETV is the more physiological procedure. After tumor excision, pseudomeningocele occurred in two cases (9.4%) and it was resolved after performing a shunt revision procedure. The overall incidence of complications was 9.3% in group A compared to 38% in group B (p < 0.05).
In a study including 20 pediatric patients with obstructive hydrocephalus due to posterior fossa tumors operated by ETV, there was one intraoperative complication due to severe intraventricular bleeding (5%), which resulted in a permanent morbidity. After tumor excision, peudomeningocele occurred in three cases (15%), and CSF leak occurred in one case (5%) [55].
Other studies did not report any mortality or permanent morbidity related to the ETV procedure in the treatment of obstructive hydrocephalus due to posterior fossa tumors in children [8, 58]. Among a series of 67 patients [58], only one patient suffered “brain herniation” and made a complete recovery after immediate tumor removal. Complications included CSF leak (3%), subdural collection (2%), pseudomeningocele (2%), and epidural hematoma (1%). Another study including 37 patients [8] reported CSF infection and meningitis in two cases (5.4%, both resolved with antibiotics), and bleeding in one case (2.7%, spontaneously resolved with no sequelae).
In a study comparing both procedures in the treatment of pediatric obstructive hydrocephalus where the ETV group included 24 patients, and VPS group included 31 patients, subdural hematoma occurred in three cases operated by ETV and in one case operated by VPS. There were three unrelated deaths, one in the ETV group and two in the VPS group; in all cases they were at the end point of tumor progression [13].
Failure of the procedure
There is large amount of variability in the literature concerning the failure rate of both procedures in the management of pediatric obstructive hydrocephalus. For ETV, the failure rate ranges from 8% to 69% [6, 7, 11, 18, 21, 24, 25, 31, 33, 35, 37, 38, 44, 47, 52, 60, 61, 67, 72, 74] or from 8% to 47% when the series including infants are excluded [6, 7, 18, 21, 25, 31, 35, 38, 60]. When the follow-up is greater than 2 years, the lowest failure rate is 15% [74]. For VPS, the failure rate ranges between 27% and 70% [5, 24, 36, 54, 66, 67, 69, 73]. When analyzing studies with follow-up greater than 2 years, the failure rate ranges between 56% and 70% [24, 54, 66, 67, 69]. The failure rate is on the higher side when infants are considered (63%) [54].
In a series of 20 pediatric patients with obstructive hydrocephalus due to posterior fossa tumors, ETV had been successful in controlling hydrocephalus in 16 patients (80%). Of the four failures (20%), one patient (aged 2 months) had intraoperative bleeding. The other three cases had relief of intracranial hypertension symptoms preoperatively and developed hydrocephalus after posterior fossa surgery (7 days, 16 days, and 2 months, respectively). Failure of ETV in these four patients was treated by VPS insertion. Among a mean follow-up period of 21 months (range 5–39 months), 13 patients (65%) were shunt free [55].
In a study including 67 pediatric patients with posterior fossa tumors operated by ETV, the procedure was technically successful in 65 patients (97%). In two patients (3%), ETV had to be aborted due to poor visibility: one because of a mild intraventricular hemorrhage and the other because of markedly xanthochromic CSF. This later patient underwent an immediate posterior fossa tumor resection and a repeated ETV 10 days later. Four patients (6%) ultimately required the insertion of a VPS for the treatment of progressive hydrocephalus. The incidence of shunt-free patients was 94% among a mean follow-up period of 26 months [58].
Among 37 pediatric patients with posterior fossa tumors, preresectional ETV was successful in controlling hydrocephalus in 32 cases (86.5%). In five cases (13.5%), ETV failed with redevelopment of hydrocephalus postoperatively at some point over the course of follow-up (up to 7.5 years) [8]. It has been concluded that ETV has a curative effect on intracranial hypertension and a prophylactic effect by preventing the development of hydrocephalus after tumor removal. In cases where CSF has caused spread of tumor at presentation and chemotherapy has to be undertaken prior to tumor excision, ETV is considered to be a better choice than VPS because any need for reoperation related to shunt failure during the phase of bone marrow transplantation represents a life-threatening situation [58].
Timing of ventriculostomy
The role of ETV in the management of persistent hydrocephalus following excision of posterior fossa tumors has already been confirmed with a success rate reported to be 90% in one study [62], and 81% in another study [45]. However, some neurosurgeons debated whether routine preoperative ETV is actually necessary claiming that the low rate of persistent hydrocephalus does not justify adopting a routine preoperative ETV [21, 45]. Others recommend its preresectional use in patients with medulloblastomas and ependymomas claiming that tumor resection alone has a low cure rate of hydrocephalus [15]. An incidence between 10% and 62% of persistent or progressive hydrocephalus after primary tumor resection without CSF diversion has been reported in the literature [21, 30, 39, 45]. Re-establishing the patency of CSF flow at the time of tumor resection is not always enough to ensure that postoperative CSF diversion will not be needed [12].
In my opinion, ETV should be performed prior to tumor excision in those patients at particularly high risk of developing postoperative hydrocephalus, such as pediatric patients who have midline posterior fossa tumors associated with marked hydrocephalus. It is also a good choice for patients presenting with acute hydrocephalus to many centers in which emergency surgery for posterior fossa tumors could not be easily performed. In such case, relieving symptoms and signs of acute hydrocephalus allows definitive tumor resection to occur on an elective basis at a time when all experienced personnel are available.
Comparing both procedures
The comparison between ETV and cerebrospinal fluid shunts has rarely been addressed in the literature. In a Swiss series of pediatric patients with obstructive hydrocephalus of different etiologies, where 24 cases (four of them had posterior fossa tumors) were managed by ETV with a mean follow-up of 45 months and 31 cases (five of them had posterior fossa tumors) were managed by VPS with a mean follow-up of 57 months, 33% of patients operated by ETV needed a revision, compared to 52% of the VPS group. The number of revisions per patient was higher for VPS group. The failure rates at 2, 5, and 10 years were 26%, 26%, and 30%, respectively, for the ETV group, compared to 34%, 42%, and 51%, respectively, for the VPS group. The mean time until first revision was 18 months for ETV, and it was 24 months for VPS [13]. Failure of ETV in such cases might be due to some technical difficulties such as small space in front of the basilar artery or arachnoid membranes lower down the clivus. The pathophysiology of the associated hydrocephalus is not completely understood, and some resorption malfunction might be also added to the obstructive etiology [22].
Among 32 patients operated by ETV included in the current study, recurrence of hydrocephalus occurred in two patients at 6.5 and 14 months postoperatively, respectively (both patients had intraoperative bleeding), and a repeated ETV was performed. Among 21 patients operated by VPS, shunt revision was performed 15 times in 8 patients. The incidence of recurrence of hydrocephalus was 6.2% among ETV patients compared to 38% among VPS patients (p < 0.01). All the 32 patients (100%) included in group A were shunt free, among the mean follow-up duration of this group (27.4 months). All the patients (100%) included in group B were shunt dependent among the mean follow-up duration of this group (25 months). The number of revisions per patient (among the eight cases with postoperative failure included in group B) was 1.8, and the mean duration which lapsed between the VPS procedure and the first revision procedure was 5.6 months. Kaplan–Meier survival curve for both groups shows a more progressive decrease of the cumulative survival in the VPS group. The mean survival time in the ETV group is higher than the VPS group (the logrank test is highly significant, p < 0.01; Fig. 2).
Our results emphasize that ETV is superior to shunt systems in terms of safety, simplicity, and resultant outcome. It renders the patient independent from a failure-prone shunt system, offers a good opportunity to become shunt free, and makes safe total resection of the tumor feasible. By performing ETV, we gained the benefits of shunt systems in relieving increased ICP but without its acute or long-term postoperative complications. It is a simple, minimally invasive, and physiological method for bypassing obstruction in CSF circulation due to posterior fossa tumors in children.
Conclusion
The shorter duration of surgery, the lower incidence of morbidity, the absence of mortality, the lower incidence of procedure failure of endoscopic third ventriculostomy as compared to ventriculoperitoneal shunt, and the significant advantage of not becoming shunt dependent make endoscopic third ventriculostomy to be recommended as the first choice in the treatment of pediatric patients with marked obstructive hydrocephalus due to midline posterior fossa tumors. It is a preliminary, simple, safe, effective, physiological, minimally invasive procedure for the relief of elevated intracranial pressure before direct tumor removal.
References
Albright AL (1983) The value of precraniotomy shunts in children with posterior fossa tumors. Clin Neurosurg 30:278–285
Albright AL, Reigel DH (1977) Management of hydrocephalus secondary to posterior fossa tumors. J Neurosurg 46:52–55
Allen JC, Epstein F (1982) Medulloblastoma and other primary malignant neuroectodermal tumors of the CNS. The effects of patients’ age and extent of disease on prognosis. J Neurosurg 57:446–451
Ammirati M, Raimondi AJ (1987) Cerebrospinal fluid infections in children: a study on the relationship between the etiology of hydrocephalus, age at the time of shunt placement, and infection rate. Childs Nerv Syst 3:106–109
Aryan HE, Meltzer HS, Park MS, Bennett RL, Jandial R, Levy ML (2005) Initial experience with antibiotic-impregnated silicone catheters for shunting of cerebrospinal fluid in children. Childs Nerv Syst 21:56–61
Baykan N, Isbir O, Gercek A, Dagcnar A, Ozek MM (2005) Ten years of experience with pediatric neuroendoscopic third ventriculostomy: features and perioperative complications of 210 cases. J Neurosurg Anesthesiol 17:33–37
Beems T, Grotenhuis JA (2002) Is the success rate of endoscopic third ventriculostomy age-dependent? An analysis of the results of endoscopic third ventriculostomy in young children. Childs Nerv Syst 18:605–608
Bhatia R, Tahir M, Chandler CL (2009) The management of hydrocephalus in children with posterior fossa tumors: the role of pre-resectional endoscopic third ventriculostomy. Pediatr Neurosurg 45:186–191
Bognar L, Borgulya G, Benke P, Madarassy G (2003) Analysis of CSF shunting procedure requirement in children with posterior fossa tumors. Childs Nerv Syst 19:332–336
Casotto A, Buoncristiani P (1982) Medulloblastoma in childhood. Multidisciplinary treatment. Childs Brain 9:299–308
Cinalli G, Sainte-Rose C, Chumas P, Zerah M, Brunelle F, Lot G, Pierre-Kahn A, Renier D (1999) Failure of third ventriculostomy in the treatment of aqueductal stenosis in children. Neurosurg Focus 6:E3
Culley DJ, Berger MS, Shaw D, Geyer R (1994) An analysis of factors determining the need for ventriculoperitoneal shunts after posterior fossa tumor surgery in children. Neurosurgery 34:402–408
De Ribaupierre S, Rilliet B, Vernet O, Regli L, Villemure G (2007) Third ventriculostomy vs ventriculoperitoneal shunt in pediatric obstructive hydrocephalus: results from a Swiss series and literature review. Childs Nerv Syst 23:527–533
Dias MS, Albright AL (1989) Management of hydrocephalus complicating childhood posterior fossa tumors. Pediatr Neurosci 15:283–290
Due-Tonnessen BJ, Helseth E (2007) Management of hydrocephalus in children with posterior fossa tumors: role of tumor surgery. Pediatr Neurosurg 43:92–96
Epstein FJ (1993) Medulloblastoma: indications for shunt placement. Pediatr Neurosurg 19:300–302
Epstein FJ, Murali R (1978) Pediatric posterior fossa tumors: hazards of the “preoperative” shunt. Neurosurgery 3:348–350
Etus V, Ceylan S (2005) Success of endoscopic third ventriculostomy in children less than 2 years of age. Neurosurg Rev 28:284–288
Farwell JR, Dohrmann GJ, Flannery JT (1977) Central nervous system tumors in children. Cancer 40:3123–3132
Feng H, Huang G, Liao X, Fu K, Tan H, Pu H, Cheng Y, Liu W, Zhao D (2004) Endoscopic third ventriculostomy in the management of obstructive hydrocephalus: an outcome analysis. J Neurosurg 100:626–633
Fritsch MJ, Doerner L, Kienke S, Mehdorn HM (2005) Hydrocephalus in children with posterior fossa tumors: role of endoscopic third ventriculostomy. J Neurosurg 103(1Suppl):40–42
Fukuhara T, Vorster SJ, Luciano MG (2000) Risk factors for failure of endoscopic third ventriculostomy for obstructive hydrocephalus. Neurosurgery 46:1100–1111
Gaab MR, Shroeder HW (1998) Neuroendoscopic approach to intraventricular lesions. J Neurosurg 88:496–505
Garton HJ, Kestle JR, Cochrane DD, Steinbok P (2002) A cost-effectiveness analysis of endoscopic third ventriculostomy. Neurosurgery 51:69–78
Gorayeb RP, Cavalheiro S, Zymberg ST (2004) Endoscopic third ventriculostomy in children younger than 1 year of age. J Neurosurg 100(5 Suppl):427–429
Gross P, Goat M, Knoblich OE (1978) Disorders of CSF circulation after interventions in the area of the posterior cranial fossa with prior shunt operation. Adv Neurosurg 5:199–202
Hoffman HJ, Hendrick EB, Humphreys RP (1976) Metastasis via ventriculoperitoneal shunt in patients with medulloblastoma. J Neurosurg 44:562–566
Hopf NJ, Grunert P, Fries G, Resch KD, Perneczky A (1999) Endoscopic third ventriculostomy: outcome analysis of 100 consecutive procedures. Neurosurgery 44:795–806
Hoppe-Hirsch E, Sainte-Rose C, Renier D, Hirsch JF (1987) Pericerebral collections after shunting. Childs Nerv Syst 3:97–102
Imielinski BL, Kloc W, Wasilewski W, Liczbik W, Puzyrewski R, Karwacki Z (1998) Posterior fossa tumors in children—indications for ventricular drainage and for V-P shunting. Childs Nerv Syst 14:227–229
Javadpour M, Mallucci C, Brodbelt A, Golash A, May P (2001) The impact of endoscopic third ventriculostomy on the management of newly diagnosed hydrocephalus in infants. Pediatr Neurosurg 35:131–135
Jimenez DF (1998) Third ventriculostomy. In: Jimenez DF (ed) Intracranial endoscopic neurosurgery. American Association of Neurological Surgeons, Park Ridge, pp 101–110
Jones RF, Stening WA, Brydon M (1990) Endoscopic third ventriculostomy. Neurosurgery 26:86–92
Jones RF, Kwok BC, Stening WA, Vonau M (1994) The current status of endoscopic third ventriculostomy in the management of non-communicating hydrocephalus. Minim Invasive Neurosurg 37:28–36
Kadrian D, van Gelder J, Florida D, Jones R, Vonau M, Teo C, Stening W, Kwok B (2008) Long-term reliability of endoscopic third ventriculostomy. Neurosurgery 62(2 Suppl):614–621
Kestle JR, Walker ML (2005) A multicenter prospective cohort study of the Strata valve for the management of hydrocephalus in pediatric patients. J Neurosurg 102(2 Suppl):141–145
Kim SK, Wang KC, Cho BK (2000) Surgical outcome of pediatric hydrocephalus treated by endoscopic III ventriculostomy: prognostic factors and interpretation of postoperative neuroimaging. Childs Nerv Syst 16:161–169
Koch D, Wagner W (2004) Endoscopic third venticulostomy in infants of less than 1 year of age: which factors influence the outcome? Childs Nerv Syst 20:405–411
Kumar V, Phipps K, Harkness W, Hayward RD (1996) Ventriculo-peritoneal shunt requirement in children with posterior fossa tumors: an 11 year audit. Br J Neurosurg 10:467–470
Kunz U, Goldmann A, Bader C, Waldbaur H, Oldenkott P (1994) Endoscopic fenestration of the 3rd ventricular floor in aqueductal stenosis. Minim Invasive Neurosurg 37:42–47
Lee M, Wisoff JH, Abbott R, Freed D, Epstein FJ (1994) Management of hydrocephalus in children with medulloblastoma: prognostic factors for shunting. Pediatr Neurosurg 20:240–247
Lewis AI, Crone KR (1994) Advances in neuroendoscopy. Contemp Neurosurg 16:1–6
McLaurin RL (1983) Disadvantages of the preoperative shunt in posterior fossa tumors. Clin Neurosurg 30:286–292
Mohanty A, Vasudev MK, Sampath S, Radhesh S, Sastry Kolluri VR (2002) Failed endoscopic third ventriculostomy in children: management options. Pediatr Neurosurg 37:304–309
Morelli D, Pirotte B, Lubansu A, Detemmerman D, Aeby A, Fricx C, Berre J, David P, Brotchi J (2005) Persistent hydrocephalus after early surgical management of posterior fossa tumors in children: is routine preoperative endoscopic third ventriculostomy justified? J Neurosurg 103(3 Suppl):247–252
Morota N, Watabe T, Inukai T, Hongo K, Nakagawa H (2000) Anatomical variants in the floor of the third ventricle; implications for endoscopic third ventriculostomy. J Neurol Neurosurg Psychiatry 69:531–534
Navarro R, Gil-Parra R, Reitman AJ, Olavarria G, Grant JA, Tomita T (2006) Endoscopic third ventriculostomy in children: early and late complications and their avoidance. Childs Nerv Syst 22:506–513
Papo I, Caruselli G, Luongo A (1982) External ventricular drainage in the management of posterior fossa tumors in children and adolescents. Neurosurgery 10:13–15
Polednak AP, Flannery JT (1995) Brain, other central nervous system, and eye cancer. Cancer 75:330–337
Raimondi AJ, Tomita T (1981) Hydrocephalus and infratentorial tumors. Incidence, clinical picture, and treatment. J Neurosurg 55:174–182
Rappaport ZH, Shalit MN (1989) Perioperative external ventricular drainage in obstructive hydrocephalus secondary to infratentorial brain tumors. Acta Neurochir (Wien) 96:118–121
Ray P, Jallo GI, Kim RY, Kim BS, Wilson S, Kothbauer K, Abbott R (2005) Endoscopic third ventriculostomy for tumor-related hydrocephalus in a pediatric population. Neurosurg Focus 19:E8
Rhode V, Gilsbach JM (2000) Anomalies and variants of the endoscopic anatomy for third ventriculostomy. Minim Invasive Neurosurg 43:111–117
Robinson S, Kaufman BA, Park TS (2002) Outcome analysis of initial neonatal shunts: does the valve make a difference? Pediatr Neurosurg 37:287–294
Ruggiero C, Cinalli G, Spennato P, Aliberti F, Cianciulli E, Trischitta V, Maggi G (2004) Endoscopic third ventriculostomy in the treatment of hydrocephalus in posterior fossa tumors in children. Childs Nerv Syst 20:828–833
Sacko O, Boetto S, Lauwers-Cances V, Dupuy M, Roux FE (2010) Endoscopic third ventriculostomy: outcome analysis in 368 procedures. J Neurosurg Pediatr 5:68–74
Santos de Oliveira R, Barros Jucá CE, Valera ET, Machado HR (2008) Hydrocephalus in posterior fossa tumors in children. Are there factors that determine a need for permanent cerebrospinal fluid diversion? Childs Nerv Syst 24:1397–1403
Sainte-Rose C, Cinalli G, Roux FE, Maixner W, Chumas PD, Mansour M, Carpentier A, Bourgeois M, Zerah M, Pierre-Kahn A, Renier D (2001) Management of hydrocephalus in pediatric patients with posterior fossa tumors: the role of endoscopic third ventriculostomy. J Neurosurg 95:791–797
Schmid UD, Seiler RW (1986) Management of obstructive hydrocephalus secondary to posterior fossa tumors by steroids and subcutaneous ventricular catheter reservoir. J Neurosurg 65:649–653
Singh D, Gupta V, Goyal A, Singh H, Sinha S, Singh AK, Kumar S (2003) Endoscopic third ventriculostomy in obstructed hydrocephalus. Neurol India 51:39–42
Smyth MD, Tubbs RS, Wellons JC III, Oakes WJ, Blount JP, Grabb PA (2003) Endoscopic third ventriculostomy for hydrocephalus secondary to central nervous system infection or intraventricular hemorrhage in children. Pediatr Neurosurg 39:258–263
Tamburrini G, Pettorini BL, Massimi L, Caldarelli M, Di Rocco C (2008) Endoscopic third ventriculostomy: the best option in the treatment of persistent hydrocephalus after posterior cranial fossa tumor removal? Childs Nerv Syst 24:1405–1412
Taylor WA, Todd NV, Leighton SE (1992) CSF drainage in patients with posterior fossa tumors. Acta Neurochir (Wien) 117:1–6
Teo C (1998) Third ventriculostomy in the treatment of hydrocephalus: experience with more than 120 cases. In: Hellwig D, Bauer BL (eds) Minimally invasive techniques for neurosurgery. Springer, Berlin, pp 73–76
Teo C, Rahman S, Boop FA, Cherny B (1996) Complications of endoscopic neurosurgery. Childs Nerv Syst 12:248–253
Tuli S, Alshail E, Drake J (1999) Third ventriculostomy versus cerebrospinal fluid shunt as a first procedure in pediatric hydrocephalus. Pediatr Neurosurg 30:11–15
Tuli S, Tuli J, Drake J, Spears J (2004) Predictors of death in pediatric patients requiring cerebrospinal fluid shunt. J Neurosurg 100(5 suppl):442–446
Valenzuela S, Trellez A (1999) Pediatric neuroendoscopy in Chile. Analysis of the first 100 cases. Childs Nerv Syst 15:457–460
Vinchon M, Fichten A, Delestret I, Dhellemmes P (2003) Shunt revision for asymptomatic failure: surgical and clinical results. Neurosurgery 52:347–356
Waga S, Shimizu T, Shimosaka S, Tochio H (1981) Intratumoral hemorrhage after a ventriculoperitoneal shunting procedure. Neurosurgery 9:249–252
Walters BC, Hoffman HJ, Hendrick EB, Humphreys RP (1984) Cerebrospinal fluid shunt infection. Influences on initial management and subsequent outcome. J Neurosurg 60:1014–1021
Warf BC (2005) Comparison of endoscopic third ventriculostomy alone and combined with choroid plexus cauterization in infants younger than 1 year of age: a prospective study in 550 African children. J Neurosurg 103(6 suppl):475–481
Warf BC (2005) Comparison of 1-year outcomes for the Chhabra and Codman-Hakim Micro Precision shunt systems in Uganda: a prospective study in 195 children. J Neurosurg 102(4 Suppl):358–362
Wellons JC III, Tubbs RS, Banks JT, Grabb B, Blount JP, Oakes WJ, Grabb PA (2002) Long-term control of hydrocephalus via endoscopic third ventriculostomy in children with tectal plate gliomas. Neurosurgery 51:63–68
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
El-Ghandour, N.M.F. Endoscopic third ventriculostomy versus ventriculoperitoneal shunt in the treatment of obstructive hydrocephalus due to posterior fossa tumors in children. Childs Nerv Syst 27, 117–126 (2011). https://doi.org/10.1007/s00381-010-1263-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-010-1263-2