Abstract
Background
Central neurocytoma (CN) represents a rare, relatively recently described primary central nervous system tumor. It ranks among intraventricular tumors due to its predominant location within the lateral brain ventricles. CN occurs mostly in young adults around the 3rd decade of life; almost a fifth of the cases are children under 18 years of age.
Objectives
The authors present three cases of patients with histopathologically confirmed CN, emphasizing diagnostic imaging issues. A review of the literature concerning differential diagnosis and clinical and therapeutic aspects is also presented.
Conclusion
Literature reports of CN comprise most likely case reports, small cohorts of patients, and meta-analytic studies due to the generally low incidence of this tumor. In the current paper, the authors summarize up-to-date knowledge of this rare disease on the background of their own observations. CN should be included in the list for differential diagnostics of intraventricular brain tumors, especially those located in lateral ventricles.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Central neurocytoma (CN) represents an uncommon central nervous system (CNS) tumor usually found in lateral ventricles of the brain in young adults. It is mostly connected with good prognosis for the patients. Hassoun et al. [11] in 1982 were the first authors to describe two cases of central neurocytoma, which was recognized as a separate pathologic entity for the first time. Since then, the number of literature reports of this tumor has increased considerably, which reflects its increasing recognition and importance. To date, more than 500 cases of CN have been published [17, 24].
According to the available data, CN comprises less than 1% of all primary CNS tumors (0.25–0.5%) [26]. CN is typically found in young adults around the third decade of life, almost 75% of described cases occur in patients between 20 and 40 years of age [6]. However, CN can also occur in patients younger than 18 years or older than 50 years [9, 25]. Children under 18 years of age are affected in approximately 17% of the cases [24]. The incidence in men and women is approximately the same, although some studies mention slight male predominance [24].
Regarding the fact that literature data published on this topic usually represent case studies or small cohorts of patients, the diagnostics and treatment of this ailment cannot yet be considered fully established [26].
In the current paper, the authors report three cases of patients with histopathologically confirmed CN. The aspects of radiological diagnosis of CN are emphasized together with differential diagnostic issues of intraventricular tumor lesions. A brief review of therapeutic management of this rare primary CNS tumor is also presented.
Case reports
Case 1
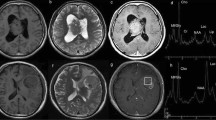
The 22-year-old patient had suffered an injury with a lacerated wound to the head. Subsequent X-ray proved no injury of the scull but showed intracranial calcifications as secondary findings. Computed tomography (CT) examination confirmed a calcified intraventricular mass with obstructive hydrocephalus. Magnetic resonance (MR) examination revealed an intraventricular tumor with heterogeneous signal intensities located in the third ventricle and part of the lateral ventricles (Fig. 1a–c). By the time of admission to the neurosurgical clinic, no neurological focal symptoms were found.
Central neurocytoma in the supratentorial brain ventricles. a MR T2-weighted axial scan shows a nonhomogeneous tumor mass with hypointense areas corresponding to calcifications. The ventricles are dilated due to obstructive hydrocephalus; the frontal horn of the right lateral ventricle is filled with a cyst of hyperintense content (arrow). b, c Sagittal and coronal MR T1-weighted image after gadolinium contrast agent administration with evident nodular enhancement. d CT examination after ventricular drainage shows striking calcifications of the tumor. The drain converges to the trigone of the left lateral ventricle (arrow). Small pneumocephalus are indicated (arrowheads)
A ventricular endoscopy was performed and Torkildsen drainage established as an acute treatment for hydrocephalus and at the same time a biopsy of the tumor tissue was performed (Fig. 1d). The histology revealed a tumor consisting of uniform cells with round nuclei, lumpy chromatin, and indistinct nucleoli; mitoses were very rare. The case was diagnosed as a CN. Within the following 4 months, the patient underwent two operations using a transcortical approach, during which the tumor mass was subtotally removed. The postsurgery course was complicated by the formation of a lateral ventricle cyst, which was treated by endoscopic fenestration and internal drainage. The follow-up MR examinations elicited suspicion of a tiny tumor residuum, without any signs of progression for 15 months at the time of preparation of this manuscript. Regarding this fact, radiotherapy of the patient was not indicated by committee decision.
Case 2
The 42-year-old man experienced recurrent headaches for several months, the sensation of some pressure in the head, overall animosity, and transient paralysis in the lower extremities. After neurological investigation, a CT examination was performed with findings of an intraventricular mass that caused obstructive hydrocephalus by bilateral blockade of the Monroe's foramen. The finding was subsequently confirmed by MR examination (Fig. 2a–c). The patient was treated surgically; the tumor was subtotally resected through a left frontal transcortical approach. Because of the high risk of injury to the deep veins and fornices, the residuum of the tumor was left in the third ventricle and a part of the lateral ventricles (Fig. 2d). Histological examination led to classification of the tumor as a CN consisting of uniform cells with round centrally located nuclei and tinted cytoplasm. Mitoses were sporadic and proliferative activity according to Ki-67 was less than 1%.
MR examination exhibits CN in a 42-year-old man. a, b T2- and T1-weighted axial images reveal a tumor mass located predominantly in the left lateral ventricle. Signal intensities are considerably nonhomogeneous due to the presence of calcifications and small cysts (arrow). c Sagittal T1-weighted image after Gd contrast administration demonstrates a marked enhancement of the tumor. d Postoperative T2-weighted MR examination in the axial plane. Small residuum is seen in the original tumor site (arrow)
After the surgery, a mild expressive fatic disorder and right-side hemiparesis were noted; however, these sequelae gradually diminished.
The indicated adjuvant therapy consisted of stereotactic radiotherapy (54 Gy in 18 fractions). Follow-up MR examinations 5 and 10 months after irradiation indicate stationary size and appearance of the tumor residuum and the patient has remained event free for 18 months since the initial surgery.
Case 3
A girl, 13 years of age, had complained of back and neck pain, gradually altered vision, and also sporadic vomitus, which were assumed to be a gastrointestinal disorder. The patient underwent ophthalmological examination with the finding of papilledema; an acute CT examination revealed an intraventricular tumor (Fig. 3a). The finding was subsequently confirmed by MR, which showed a tumor mass located mainly in the right lateral ventricle with nonhomogeneous signal intensities and mild enhancement after intravenous gadolinium contrast administration (Fig. 3b–d). The girl underwent surgical treatment, during which the tumor was removed through a right frontal transcortical approach. The resection was stated as complete by perioperative finding and follow-up MR examinations (Fig. 4).
CT and MR examinations showing a central neurocytoma in a 13-year-old girl. a An acute CT examination revealed an intraventricular tumor mass with nonhomogeneous, rather high density. b Native T1-weighted MR image. The tumor mass (arrowheads) is remarkable by a characteristic “sponge-like” structure due to the presence of multiple small cysts. c Coronal FLAIR image. The tumor is hyperintense, trenches to both lateral ventricles (arrowheads). d Sagittal T1-weighted MR examination after Gd contrast administration. Enhancement is rather weak, and it occurs predominantly in the posterior part of the tumor (arrow)
MR examination after the complete resection of the CN. a Axial T1-weighted postcontrast scan. The intraventricular mass was removed through a right frontal transcortical approach. Postoperative meningeal enhancement is visible in the right frontal area (arrow) together with small subdural fluid accumulation (arrowheads). b Coronal FLAIR MR image. Hyperintensity around the right lateral ventricle (arrowheads) represents postoperative edema
The histopathological examination revealed a tumor consisting of uniform cells with round nuclei and smooth chromatin, synaptophysine positive. Increased mitotic activity was noted in some parts of the tumor (under 5% according to Ki-67). The final diagnosis is stated as grade II “atypical” CN with increased mitotic activity.
No adjuvant radiotherapy was indicated. The patient has been observed for 5 years after surgery with no clinical or MR signs of tumor recurrence.
Imaging features
CN is in most of the cases found in an intraventricular location, situated in the septum pellucidum or the walls of the lateral ventricles [27]; however, it can also sporadically occur in an extraventricular location. Lenzi et al. [19] described an unusual case of intraventricular CN that massively infiltrated the brain stem and basal ganglia. There are also a few reports of tumors histomorphologically identical to CN that were located within the cervical or thoracic spinal cord [10, 28, 33]. A few authors have also reported cases of CN with extraventricular secondary dissemination despite the fact that the primary tumor exhibited no histopathological signs of malignant potential. [8, 32].
CT and MR imaging depict CN as a heterogeneous mass, typically located in the anterior half of the lateral ventricles in close proximity to the Monroe's foramen, where it often causes obstructive hydrocephalus. Propagation of the tumor mass into the 3rd ventricle is less common; only 3% of CN occur in the third ventricle as an isolated location [16]. Also, the isolated presence of CN in the 4th ventricle is only rarely observed and has been described in only a few case reports [13, 34]. The tumor is usually attached with a broad base to the roof or lateral wall of the lateral ventricle [26].
In CT images, the tumor tissue usually appears iso or hyperdense with variable, largely heterogeneous enhancement after administration of an iodinated contrast agent. Hypodense cystic areas and calcifications are common and can be found in about half of the cases [12]. The MR depiction of the tumor is analogous, showing considerably nonhomogeneous signal intensities, especially when using T2 weighting, depending on the content of cysts, calcifications, and greater vessels [3]. Signal intensities of the tumor are usually low in unenhanced T1-weighted images; after gadolinium intravenous contrast injection, variable low to medium enhancement occurs with respect to the level of vascularity [16]. This fact agrees with diverse angiographic findings that can provide images of highly vascular lesions as well as entirely negative findings.
MR spectroscopy can document elevated levels of choline to creatine and choline to NAA ratios. Several studies also mention a signal peak at 3.55 ppm that may correspond to elevated glycine content [15, 36]. These findings can be considered comparatively specific for CN; thus, in this field, MR spectroscopy may contribute to the improvement of preoperational diagnostics.
Differential diagnosis
The topic of differential diagnosis of intraventricular brain tumors is rather extensive and accordingly fairly complicated. CT and MR image of several tumor entities is quite similar, making differentiation disputable or even impossible; thus, biopsy and histopathological examination may be the only way to obtain an accurate diagnosis. Nevertheless, important factors for the correct differential diagnosis based on imaging methods are the consideration of the patient's age and the exact localization of the tumor mass within the brain ventricular system. Central neurocytoma is characterized by occurrence within lateral ventricles in young patients. In terms of CN differential diagnosis, it is necessary to consider several benign and few malignant tumors.
Subependymal giant cell astrocytoma, similar to CN among young patients, is commonly found within lateral ventricles of the brain [14, 21]. It occurs usually in patients suffering from tuberous sclerosis; other signs of this disease should allow the differentiation from CN and formulation of the correct diagnosis. Subependymoma (Fig. 5) can be distinguished by weak or no enhancement in postcontrast CT or MR images; however, a differentiation from CN may be impossible. A clue for a correct diagnosis may be its typical incidence in elderly patients [4]. Ependymoma is in most of the cases located within the 4th ventricle. Supratentorial location is reported in one third of the patients, in this case with common extension into the periventricular white matter [22]. Choroid plexus papilloma (Fig. 6) is a more common finding in childhood than in adults; its clinical manifestation starts early; the diagnosis is made before 5 years of age in 85% of the patients. The tumor is localized in the lateral ventricles of the brain in about half of the cases; occurrence in the 4th ventricle is more common in adults [29]. Meningioma is rarely localized within the ventricular system (less than 5%), but because of this possibility it is necessary to mention this benign tumor within the differential diagnosis of CN. It is typically found in elderly patients in the lateral ventricles or the 4th ventricle. The finding of meningioma is generally uncommon in childhood; if this occurs, an intraventricular location of the tumor is relatively more frequent [20]. The lesion, as in all meningiomas, is characterized by strong homogeneous enhancement after contrast medium application in CT and MR examination [18]. Metastases (Fig. 7) in ventricular locations can be found more likely in elderly patients; a clue for a correct diagnosis might be knowledge of the primary malignancy. Colloid cyst arises usually from the 3rd ventricle roof; as a pseudotumor lesion it lacks any postcontrast enhancement. The nomenclature itself suggests a predominantly cystic character of the lesions. The content of the cysts commonly appears hyperdense in CT images [1]. Several other lesions like oligodendroglioma or pilocytic astrocytoma may sporadically be found within the brain ventricular system. The finding of intraventricular schwannoma is extremely rare; it has been described in only a few case reports [2, 7].
Subependymoma found on MR examination of a 62-year-old woman. a Axial T2-weighted image shows hyperintense tumor mass located in the frontal horn of the right lateral ventricle (arrow). The ventricle is dilated due to the Monroe's foramen obstruction. b Mild nodular enhancement is apparent in the coronal T1-weighted image after Gd contrast administration (arrowheads). Such appearance is very similar to CN
MR examination reveals choroid plexus papilloma in a 45-year-old woman. a Axial T2-weighted image. The tumor is located within the 4th ventricle (arrow) with only discrete hyperintensities. b FLAIR image in the coronal plane. The tumor is generally isointense compared with the gray matter with few tiny hypointense cysts (arrowheads). Surprisingly, no ventricles dilatation is present. c, d T1-weighted axial and sagittal image after Gd contrast administration with apparent strong enhancement of the lesion
Neuroendocrine tumor metastasis of unknown origin in a 60-year-old man. a, b T2- and T1-weighted axial MR image showing tumor mass located within the 3rd and left lateral ventricle (arrowheads). Signal intensities are close to the gray matter. c Coronal T1-weighted MR scan after Gd contrast administration demonstrates intermediate inhomogeneous enhancement. d The lesion is hyperdense in native CT image, which is due to the high cellularity of the tumor
Clinical and therapeutic aspects
According to the WHO 2007 classification, CN belongs among the primary neuronal tumors (ICD-O 9506/1, WHO grade II). It is a well-differentiated tumor consisting of a uniform cell population showing immunochemical and ultrastructural signs of neuronal differentiation. However, in approximately one fifth of the cases, we find so-called atypical neurocytoma, which exhibits some anaplastic signs such as focal necrosis, vascular endothelial proliferation, increased mitotic activity, or hallmarks of cell atypia. Increased tumor proliferation activity (stated by Ki-67 labeling index >3%) is considered as a prognostic sign linked with worse local control of the disease [23].
The therapy for CN is based on surgical treatment, which should be as radical as possible, but not at the price of injury to eloquent structures. As it could be anticipated, the extent of the resection strongly influences the overall prognosis for the patients. Complete resection (CTR) is associated with better consecutive local control of the disease as well as patient survival compared to incomplete resection (ITR). Regarding the predominantly intraventricular location of CNs, the diagnosis is often made after the tumor reaches large proportions. The tumor tissue usually has a good vascular supply and frequently infiltrates ventricle walls. Consequently, CTR is often technically demanding and sometimes may require a second operation. The choice of the appropriate surgical approach depends on the tumor localization within the ventricular system: in general for lateral ventricle expansion a transcortical frontal or occipital approach is used, and entry to the 3rd ventricle provides an interhemispheric transcallous approach [30, 31, 35]. Disturbance of the cerebrospinal fluid circulation may appear as another surgical complication, which subsequently requires additional surgical intervention.
Adjuvant radiotherapy following surgical tumor resection is another treatment option. As a consequence of the low incidence of CN, there are no randomized clinical studies that allow an evaluation of the role of adjuvant radiotherapy following neurosurgery. The current knowledge of the optimal treatment management is mostly based on retrospective evaluation of rather small groups of patients, thus resulting mainly from meta-analyses. Therefore, the optimal role of adjuvant radiotherapy in CN treatment is still being assessed.
The therapeutic recommendations have been summarized in the largest worldwide study to date by Rades and Schild [24]. They analyzed more than 400 previously described cases of CN according to age of the patients, radicality of the resection (CTR vs. ITR), and biological nature of the tumor (typical vs. atypical CN). In patients after CTR, no statistically significant benefit of radiotherapy was documented in terms of overall outcome and local control. In incompletely resected CNs in adults, adjuvant radiotherapy significantly improved local control of the tumor as well as overall survival for both typical and atypical CNs. For typical CN, a 50–54-Gy dose is sufficient, while for atypical lesions a higher dose of 56–60 Gy is required. An alternative for radiotherapy may involve stereotactic radiosurgery in cases of small residua [5]. In children, radiotherapy after ITR improved local control of the disease but did not significantly affect the overall survival; only a trend to better outcome was observed [24]. However, the conclusions made for pediatric CN were based on a limited number of cases and warrant further evaluation. Thus, the application of adjuvant radiotherapy should be considered with regard to the tumor biology, patient's age, radicality of preceding surgical resection as well as to the potential neurotoxicity of the radiotherapy and the overall patient's performance.
Chemotherapy is not widely used; there are no long-term results of this treatment available, which should accordingly be considered entirely experimental.
Conclusion
CN represents an uncommon, relatively recently described tumor, which is usually found in young adults within the lateral ventricles of the brain. Because of the generally small rate of CNs among CNS tumors, the lesion has been described only in the form of case reports and small sets of patients. In the present paper, we report three case studies, review the literature, describe neuroimaging issues, and discuss differential diagnostics and treatment management of CN.
References
Armao D, Castillo M, Chen H, Kwock L (2000) Colloid cyst of the third ventricle: imaging pathologic correlation. AJNR Am J Neuroradiol 21:1470–1477
Benedict WJ Jr, Brown HG, Sivarajan G, Prabhu VC (2008) Intraventricular schwannoma in a 15-year-old adolescent: a case report. Childs Nerv Syst 24:529–532
Chang KH, Han MH, Kim DG, Chi JG, Suh DC, Kim SJ, Cha SH, Han MC (1993) MR appearance of central neurocytoma. Acta Radiol 34:520–526
Chiechi MV, Smirniotopoulos JG, Jones RV (1995) Intracranial subependymomas: CT and MR imaging features in 24 cases. AJR Am J Roentgenol 165:1245–1250
Cobery ST, Noren G, Friehs GM, Chougule P, Zheng Z, Epstein MH, Taylor W (2001) Gamma knife surgery for treatment of central neurocytomas. Report of four cases. J Neurosurg 94:327–330
De Tommasi A, D, Urso PI, De Tommasi C, Sanguedolce F, Cimmino A, Ciappetta P (2006) Central neurocytoma: two case reports and review of the literature. Neurosurg Rev 29:339–347
Dow GR, Hussein A, Robertson IJ (2004) Supratentorial intraventricular schwannoma. Br J Neurosurg 18:561–562
Eng DY, DeMonte F, Ginsberg L, Fuller GN, Jaeckle K (1997) Craniospinal dissemination of central neurocytoma. Report of two cases. J Neurosurg 86:547–552
Figarella-Branger D, Pellissier JF, Daumas-Duport C, Delisle MB, Pasquier B, Parent M, Gambarelli D, Rougon G, Hassoun J (1992) Central neurocytomas. Critical evaluation of a small-cell neuronal tumor. Am J Surg Pathol 16:97–109
Giangaspero F, Cenacchi G, Losi L, Cerasoli S, Bisceglia M, Burger PC (1997) Extraventricular neoplasms with neurocytoma features. A clinicopathological study of 11 cases. Am J Surg Pathol 21:206–212
Hassoun J, Gambarelli D, Grisoli F, Pellet W, Salamon G, Pellissier JF, Toga M (1982) Central neurocytoma. An electron-microscopic study of two cases. Acta Neuropathol 56:151–156
Hassoun J, Söylemezoglu F, Gambarelli D, Figarella-Branger D, von Ammon K, Kleihues P (1993) Central neurocytoma: a synopsis of clinical and histological features. Brain Pathol 3:297–306
Hsu PW, Hsieh TC, Chang CN, Lin TK (2002) Fourth ventricle central neurocytoma: case report. Neurosurgery 50:1365–1367
Jelinek J, Srnirniotopoulos JG, Parisi JE, Kanzer M (1990) Lateral ventricular neoplasms of the brain: differential diagnosis based on clinical, CT and MR findings. AJR Am J Roentgenol 155:365–372
Kim DG, Choe WJ, Chang KH, Song IC, Han MH, Jung HW, Cho BK (2000) In vivo proton magnetic resonance spectroscopy of central neurocytomas. Neurosurgery 46:329–333
Koeller KK, Sandberg GD (2002) From the archives of the AFIP. Cerebral intraventricular neuroplasms: radiologic-pathologic correlation. Radiographics 22:1473–1505
Korshunov A, Sycheva R, Golanov A (2007) Recurrent cytogenetic aberrations in central neurocytomas and their biological relevance. Acta Neuropathol 113:303–312
Latschaw RE, Rai AT, Branstetter BF IV, Hogg JP (2005) Extra-axial tumors of the head: diagnostic imaging, physiologic testing and embolization. In: Latchaw RE, Kucharczyk J, Moseley ME (eds) Imaging of the nervous system: diagnostic and therapeutic applications. Elsevier, Philadelphia, p 784
Lenzi J, Salvati M, Frati A, Raco A, Pichierri A, Giangaspero F, Delfini R (2006) Intraventricular neurocytoma with massive brain stem involvement in a 5-year-old child. Childs Nerv Syst 22:95–98
Nakamura M, Roser F, Bundschuh O, Vorkapic P, Samii M (2003) Intraventricular meningiomas: a review of 16 cases with reference to the literature. Surg Neurol 59:491–503
Nishio S, Morioka T, Suzuki S, Fukui M (2002) Tumours around the foramen of Monro: clinical and neuroimaging features and their differential diagnosis. J Clin Neurosci 9:137–141
Jones BV (2004) Ependymoma. In: Osborn AG, Blaser S, Salzman KL (eds) Diagnostic imaging: brain. Elsevier, Philadelphia, pp I6–52
Rades D, Fehlauer F, Schild SE (2004) Treatment of atypical neurocytomas. Cancer 100:814–817
Rades D, Schild SE (2006) Treatment recommendations for the various subgroups of neurocytomas. J Neurooncol 77:305–309
Robbins P, Segal A, Narula S, Stokes B, Lee M, Thomas W, Caterina P, Sinclair I, Spagnolo D (1995) Central neurocytoma. A clinicopathological, immunohistochemical and ultrastructural study of 7 cases. Pathol Res Pract 191:100–111
Schmidt MH, Gottfried ON, von Koch CS, Chang SM, McDermott MW (2004) Central neurocytoma: a review. J Neurooncol 66:377−384
Sgouros S, Carey M, Aluwihare N, Barber P, Jackowski A (1998) Central neurocytoma: a correlative clinicopathologic and radiologic analysis. Surg Neurol 49:197–204
Singh A, Chand K, Singh H, Sarkar C, Sharma MC (2007) Atypical neurocytoma of the spinal cord in a young child. Childs Nerv Syst 23:207–211
Smirniotopoulos JG, Meltzer CC (2005) Primary intra-axial neoplasms. In: Latchaw RE, Kucharczyk J, Moseley ME (eds) Imaging of the nervous system: diagnostic and therapeutic applications. Elsevier, Philadelphia, pp 763–764
Smrcka V, Smrcka M, Maca K (2000) Guzy pierwotne ukladu komorowego mozgu. Valetudinaria Post Med Klin Wojsk 5:84–86 (in Polish)
Smrcka V, Smrcka M, Schroder R (2005) Transkalozni nebo transventrikularni pristup do III. mozkove komory? Cesk Slov Neurol Neurochir 68/101:175–178 (in Czech)
Takao H, Nakagawa K, Ohtomo K (2003) Central neurocytoma with craniospinal dissemination. J Neurooncol 61:255–259
Tatter SB, Borges LF, Louis DN (1994) Central neurocytomas of the cervical spinal cord. Report of two cases. J Neurosurg 81:288–293
Warmuth-Metz M, Klein R, Sörensen N, Solymosi L (1999) Central neurocytoma of the fourth ventricle. Case report. J Neurosurg 91:506–509
Yasargil MG, von Ammon K, von Deimling A, Valavanis A, Wichmann W, Wiestler OD (1992) Central neurocytoma: histopathological variants and therapeutic approaches. J Neurosurg 76:32–37
Yeh IB, Xu M, Ng WH, Ye J, Yang D, Lim CC (2008) Central neurocytoma: typical magnetic resonance spectroscopy findings and atypical ventricular dissemination. Magn Reson Imaging 26:59–64
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Keřkovský, M., Zitterbart, K., Svoboda, K. et al. Central neurocytoma: the neuroradiological perspective. Childs Nerv Syst 24, 1361–1369 (2008). https://doi.org/10.1007/s00381-008-0655-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-008-0655-z