Abstract
Late gadolinium enhancement (LGE) with cardiac magnetic resonance (CMR) can predict ventricular arrhythmia and poor prognosis in hypertrophic cardiomyopathy (HCM) patients. Although myocardial T2-high signal has been reported to appear within LGE in those patients, its clinical significance remains unclear. We investigated the relationship between the T2-high signal and nonsustained ventricular tachycardia (NSVT) in HCM patients. Eighty-one HCM patients who underwent Holter ECG and CMR including T2-weighted and LGE imaging were retrospectively recruited. They were divided into NSVT-positive and NSVT-negative groups. We compared the clinical and CMR characteristics between both of the groups, and assessed predictors of NSVT with multivariate analysis. Myocardial T2-high signal was observed in 15/81 (18.5 %) patients. Each T2-high signal was localized within LGE. Significantly in the NSVT-positive group, the prevalence of atrial fibrillation [5/17 (29.4 %) vs. 2/64 (3.1 %), p = 0.0006] and T2-high signal [9/17 (52.9 %) vs. 6/64 (9.4 %), p < 0.0001] and the left ventricular (LV) end-systolic volume index (32.2 ± 15.9 ml/m2 vs. 23.3 ± 14.9 ml/m2, p = 0.034) and the number of segments with LGE (5.8 ± 3.3 vs. 2.7 ± 2.7, p < 0.0001) was increased, and the LV ejection fraction (54.8 ± 10.9 % vs. 65.1 ± 10.6 %, p = 0.0007) was decreased, compared to the NSVT-negative group. On multivariate analysis, the presence of atrial fibrillation (OR 29.49, p = 0.0025) and DM (OR 7.36, p = 0.0455) and T2-high signal (OR 14.96, p = 0.0014) and reduced LV ejection fraction (OR 0.93, p = 0.0222) were significantly associated with NSVT. The presence of myocardial T2-high signal is a significant independent predictor of NSVT in HCM patients.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In patients with ischemic heart disease, multidetector computed tomography is useful for the identification of coronary plaque morphology and cardiac magnetic resonance (CMR) is valuable in that of myocardial tissue characteristics [1, 2]. Furthermore, CMR imaging is a noninvasive useful modality for structural and functional evaluations in patients with hypertrophic cardiomyopathy (HCM) [3]. With contrast-enhanced CMR, the presence of myocardial late gadolinium enhancement (LGE) can indicate the risk of ventricular arrhythmia and predict poor prognosis [4–7]. Since Higgins et al. [8] first reported a relationship between myocardial water content and T2 relaxation time, myocardial high signal on T2-weighted imaging (T2-high signal) has been considered to represent myocardial edema. A few reports in recent years have described the presence of myocardial T2-high signal in some patients with HCM [9, 10]. Melacini et al. [10] reported that the T2-high signal areas were localized within the LGE areas in those patients. To our knowledge, the relationship between the T2-high signal and ventricular arrhythmia remains unknown in HCM patients. The objective of the present study was to investigate the relationship between the T2-high signal and nonsustained ventricular tachycardia (NSVT) and to clarify the predictors of NSVT by multiple logistic regression analysis in those patients.
Materials and methods
Subjects and procedures
From 7 January 2008 to 31 March 2009, 81 HCM patients who obtained T2-weighted and LGE images on CMR and underwent Holter ECG examination within 3 months before or after the CMR examination were retrospectively recruited into this study. Firstly, we explained the use of patient’s clinical data for retrospective study to all the patients. Afterward, we obtained the patients’ approvals in writing.
HCM was diagnosed when echocardiography and/or CMR showed left ventricular (LV) wall thickness of 15 mm or greater in the absence of other cardiac or systemic diseases that could account for the hypertrophy [11].
The study patients were divided into two groups: those showing NSVT on Holter ECG (NSVT-positive group) and those without such findings (NSVT-negative group). NSVT was defined as sustained ventricular beats of more than three consecutive beats faster than 120 beats per minute. The clinical background, including medication, results of blood biochemistry tests, and CMR findings, were compared between these two groups.
CMR protocols and image analysis
CMR studies were performed with a 1.5-T MR scanner (Magnetom Sonata; Siemens Medical Solutions, Erlangen, Germany) using a six-channel phased-array body and spine coil. All images were acquired by ECG-gated breath-hold technique. First, steady-state free-precession cine images were acquired in three long-axis (2-, 3-, and 4-chamber) and five short-axis views covering the LV from base to apex (TR = 56.8 ms, TE = 1.2 ms). Subsequently, black-blood T2-weighted multi-echo spin-echo images with fat suppression (short tau inversion recovery) were acquired (TR = 2 heartbeats, TE = 104 ms, TI = 170 ms, field of view = 340 mm, matrix = 192 × 256, slice thickness = 8 mm, echo-train length = 21, band width = 399 Hz/pixel) in the same views used in cine imaging. Finally, LGE images were acquired 10 min after intravenous administration of 0.1 mmol/kg of gadodiamide hydrate (Omniscan; Daiichi Sankyo, Tokyo, Japan), using the inversion recovery technique in the identical views (TR = 600 ms, TE = 1.26 ms, TI was individually optimized to null normal myocardial signal using a TI-scout sequence).
Experienced radiology technicians performed LV function analysis with dedicated software (Argus Function VA30, Siemens Medical Solutions, Erlangen, Germany). Epicardial and endocardial borders of the LV myocardium were manually traced during the whole cardiac phase on each cine short-axis image to obtain LV end-diastolic volume (EDV), end-systolic volume (ESV), ejection fraction (EF), and myocardial mass. The EDV index was calculated as the EDV divided by the body surface area of each patient, and ESV index, the ESV divided by the body surface area. The myocardial mass was calculated by multiplying volume of the myocardium measured at end-diastole with the specific gravity of the myocardium (1.05 g/ml). The LV mass index was calculated as the myocardial mass divided by the body surface area of each patient.
Two cardiologists and one radiologist with 5-or-more-years experience in interpreting CMR evaluated T2-weighted images for the presence of myocardial T2-high signal areas and LGE images for the presence of LGE areas within the LV myocardium for each patient in consensus. We measured the signal intensity at the site of suspected T2-high signal area (minimum ROI size 27 mm2). Furthermore, if the signal intensity of the suspected site was more than 3 SD above that of the reference myocardium, we defined it as the positive of T2-high signal. LGE segments were defined as those with image intensities of 2 SD above the mean of image intensities in a remote myocardial region in the same image.
The presence of any area of the T2-high signal was defined as T2-positive, and the presence of any LGE area as LGE-positive. The spatial distribution of the T2-high signal and LGE was also assessed referring to the 17-segment model [12]. The maximal LV wall thickness was determined by measuring the minimal thickness of the thickest LV myocardium in the cine image at the end-diastole, in consensus, also by the three readers.
Statistical analysis
Data are expressed as mean ± SD for continuous variables and as frequency with percentage for categorical data. Differences between means were tested by unpaired Student’s t test. Frequencies were compared by Chi-square test or Fisher’s exact test, as appropriate. The spatial distribution of the T2-high signal and LGE was also assessed referring to the 17-segment model. The relation between the extent of LGE and T2-high signal was assessed with Cochran–Armitage test. Predictors of NSVT were assessed with logistic regression analysis. We performed the univariate analysis on the variables considered to be correlated with NSVT in clinical practice. For multivariate analysis, those variables with a p value <0.1 on univariate analysis were entered into the model. Furthermore, we performed logistic regression analysis with stepwise variable selection. A p value <0.05 was considered to be statistically significant. Analysis was performed using the SAS ver.9.1 (SAS Institute, Cary, NC, USA).
Results
Patient background and clinical characteristics
NSVT was present in 17 (21.0 %) of our 81 patients. The mean episodes of NSVT run in 24 h were 28 times. The numbers of the longest burst in NSVT was 3–19 beats (average 6.8 ± 4.3). The fastest ventricular rate was 120–211 beats/min (average 156 ± 28).
Table 1 shows the clinical background of our patients divided into the NSVT-positive and NSVT-negative groups. The two groups did not differ significantly in age, family history of HCM and sudden cardiac death, history of syncope, New York Heart Association function class, medications, and blood biochemistry data. Male gender ratio was significantly higher in the NSVT-positive group (15/17 patients, 88.2 %) than in the NSVT-negative group (37/64 patients, 57.8 %) (p = 0.02). The prevalence of atrial fibrillation (AF) was significantly higher in the NSVT-positive group (5/17 patients, 29.4 %) than in the NSVT-negative group (2/64 patients, 3.1 %) (p = 0.0006).
CMR findings
Table 2 shows the CMR findings in our patients divided into the NSVT-positive and NSVT-negative groups. The two groups did not differ significantly in the LVEDV index, LV mass index, and maximal LV wall thickness. However, the LVESV index was significantly greater in the NSVT-positive group (32.2 ± 15.9 ml/m2) than in the NSVT-negative group (23.3 ± 14.9 ml/m2) (p = 0.034). The LVEF was significantly lower in the NSVT-positive group (54.8 ± 10.9 %) than in the NSVT-negative group (65.1 ± 10.6 %) (p = 0.0007).
Myocardial T2-high signal was less frequently observed in 15/81 (18.5 %) patients, whereas LGE was identified in 63/81 (77.8 %) patients (p < 0.0001). The prevalence of LGE was similar in patients with and without NSVT [16/17 (94.1 %) vs. 47/64 (73.4 %), p = 0.07]. On the other hand, the number of segments with LGE was significantly higher in the NSVT-positive group (5.8 ± 3.3) than in the NSVT-negative group (2.7 ± 2.7) (p < 0.0001). Furthermore, the prevalence of T2-high signal was significantly higher in the NSVT-positive group (9/17 patients, 52.9 %) than in the NSVT-negative group (6/64 patients, 9.4 %) (p < 0.0001). The number of segments with T2-high signal was significantly higher in the NSVT-positive group (0.9 ± 1.1) than in the NSVT-negative group (0.2 ± 0.8) (p = 0.0042).
Distribution and correlation of T2-high signal and LGE
Figure 1a, b shows the distribution of T2-high signal and LGE areas for all our patients using the LV 17-segment model. Myocardial T2-high signal areas were the most frequently identified in the ventricular septum at the basal to mid portion, such as the basal anteroseptal, mid inferoseptal, and mid anteroseptal segments. LGE areas were frequently recognized not only in the ventricular septum at the basal to mid portion but also in the apical region.
Distributions of T2-high signal (a) and late gadolinium enhancement (LGE) (b) in the myocardium of the left ventricle for all our patients with hypertrophic cardiomyopathy. a The distribution of a total of 30 segments of the T2-high signal is shown based on the 17-segment model with the total positive number in each segment. b The distribution of a total of 270 segments of the LGE is shown based on the 17-segment model with the total positive number in each segment. The segments of myocardial T2-high signal are the most frequently identified in the ventricular septum at the basal to mid portion, such as the basal anteroseptal, mid inferoseptal, and mid anteroseptal segments. Those of LGE are frequently recognized not only in the ventricular septum at the basal to mid portion but also in the apical region
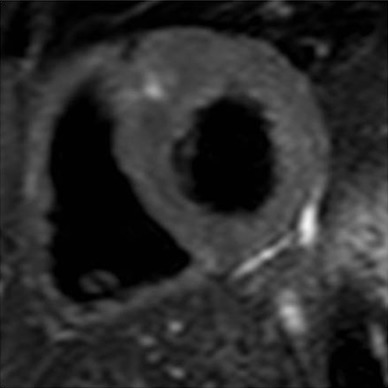
In the 15 patients with myocardial T2-high signal, a total of 30 segments of the high signal were observed (mean 2.0 ± 1.3 sites per patient) referring to the 17-segment model. All the segments of T2-high signal corresponded to those of LGE in each patient, and each T2-high signal area was localized within the corresponding LGE area, as shown in Fig. 2a, b.
Cardiac magnetic resonance images of late gadolinium enhancement (LGE) and T2-weighted imaging in two different patients (a, b) with hypertrophic cardiomyopathy showing both LGE and T2-high signal in the myocardium of the left ventricle. The LGE images (upper panel) and T2-weighted images (lower panel) are shown at the identical slice location in the same view for each patient. The areas of T2-high signal corresponded well to and are localized within those of LGE in all these patients
Relation between LGE and T2-high signal
Table 3 shows the relation between the extent of LGE and T2-high signal. We performed semi-quantitative analysis of the extent of LGE by using the number of segments with LGE referring to the 17-segment model. We divided the extent of LGE for tertile (number of segments; low: ≤1, mid: 2–4, high: 5≤) because of the small number of study patients, and examined the relation between the extent of LGE and T2-high signal using Cochran–Armitage test. A statistically significant correlation was observed between the extent of LGE and T2-high signal (p < 0.0001). This result suggested that the extent of LGE and T2-high signal were same direction to predict the NSVT.
Predictors for NSVT
Tables 4 and 5 show the results of logistic regression analysis. We performed the univariate analysis on the variables considered to be correlated with NSVT in clinical practice. For multivariate analysis, those variables with a p value <0.1 on univariate analysis were entered into the model. Furthermore, we performed logistic regression analysis with stepwise variable selection. On the logistic regression with stepwise variable selection, the odds ratio for LGE (tertile) and T2-high signal were obtained separately because of the significant correlation between the extent of LGE and presence of T2-high signal. Table 5 shows two types of analysis (model 1 and model 2). We excluded LGE (tertile) as variable in model 1, and excluded T2 high signal in model 2.
On the logistic regression with stepwise variable selection (model 1), the four variables that were found to be independently associated with NSVT consisted of the presence of DM (OR 7.36, 95 % CI 1.04–51.99, p = 0.0455), the presence of AF (OR 29.49, 95 % CI 3.27–265.70, p = 0.0025), the presence of T2-high signal(OR 14.96, 95 % CI 2.85–78.39, p = 0.0014), and the reduced LVEF (OR 0.93, 95 % CI 0.87–0.99, p = 0.0222). The odds ratio of T2 high signal was higher than that of LGE (tertile) (OR 14.96 vs. 3.54).
Discussion
In HCM patients, Adabag et al. [13] observed that NSVT had high sensitivity but low specificity as a risk of sudden death. They simultaneously pointed out that this result was probably influenced by the low incidence of sudden death in their study patients. According to recent large-scale studies regarding the prognosis of HCM patients, no significant relationship was recognized between the number of conventional risk factors including NSVT and the rate of appropriate intracardiac defibrillator (ICD) therapy for secondary prevention of sudden death [14]. In contrast, NSVT was identified as an independent predictor of sustained ventricular tachycardia or fibrillation and appropriate ICD therapy by multivariate analysis [15]. Thus, there is as yet no consensus on the clinical significance of NSVT. However, ventricular arrhythmia including NSVT is still considered to be an important risk factor related to sudden death in HCM patients.
In the present study, the presence of AF, the presence of DM, reduced LVEF, and the presence of myocardial T2-high signal were identified as significant independent predictors of NSVT by multivariate analysis. Together with ventricular tachycardia, AF is an important arrhythmia for HCM patients [16]. AF has been reported to be a frequent concurrent condition in HCM patients with advanced myocardial impairments, such as enlarged ventricular cavity and reduced contraction [17]. Thus, it is not surprising that AF and reduced LVEF are independent predictors of NSVT. Diabetes mellitus has been shown to be a significant risk factor of ischemic heart disease unquestionably. Chen-Scarabelli et al. [18] reported that the high level hemoglobin A1c had a significant association with spontaneous VT. They speculated the mechanisms of hyperglycemia-induced ventricular instability were increasing sympathetic activity and free radical production. Thus, these mechanisms may be applicable to our study patients.
Also for HCM patients, recent advances in CMR imaging technique allow the accurate measurement of the LV volume, systolic and diastolic function, and myocardial mass and the detailed morphological evaluation of cardiac hypertrophy. Importantly, LGE on CMR is widely considered to pathologically represent myocardial fibrosis [19, 20] and has been reported to be related to ventricular arrhythmia and poor prognosis in HCM patients [4–7]. Choudhry et al. [21] reported LGE on CMR was observed only in hypertrophied regions, as shown in our patients, and correlated inversely with regional contraction. Extensive LGE has been observed in patients with the dilated-phase HCM [22]. Interestingly, myocardial T2-high signal was an independent predictor with a high odds ratio of 14.96 in our study.
Myocardial T2-high signal is related to the water content of myocardium. In patients with acute myocardial infarction, since the edematous portion of the myocardium shows high signal [23], T2-weighted imaging has been reported to be useful for identifying the myocardial area at risk [24]. T2-high signal is also observed and diagnostically useful in patients with acute myocarditis and Takotsubo cardiomyopathy [25, 26]. Recently, myocardial T2-high signal has been observed in some patients with HCM [9, 10]. Melacini et al. [10] reported that the T2-high signal areas were localized within the LGE areas in those patients. They speculated that since, on CMR first pass perfusion image, perfusion defects were detected in the T2-high signal areas, the T2-high signal may indicate acute or subacute ischemic event or microvascular damage. In the present study, myocardial T2-high signal was always observed in the LGE areas or the patients with the LGE, as reported in previous studies [9, 10]. The study of HCM patients by Knaapen et al. [27] using positron emission tomography as a reference showed that LGE may not simply represent fibrosis as in patients with old myocardial infarction, but involve other pathologic changes. Histopathological studies reported a mixture of cellular components, necrosis, and small blood vessels within the LGE areas in HCM [28, 29]. We speculate that myocardial T2-high signal may reflect a certain pathological alternation among the tissue mixture within LGE areas, and this alternation may trigger NSVT. Thus, the T2-high signal may be more directly related to NSVT than LGE. In all those reports, however, the clinical assumptions could not be verified by histopathological findings, and the definite cause of the T2-high signal remains unknown.
Rubinshtein et al. [30] reported that the comparison study of the incidence of cardiovascular event rate in HCM patients with and without LGE did not achieve a statistically significant difference, although this result was considered to be influenced by the low event rate and short follow-up period. Bruder et al. [31] reported cardiac mortality in HCM patients with LGE exceeded that without LGE but that there was no significant difference in the incidence of sudden cardiac death between them. Appelbaum et al. [32] reported the magnitude of LGE may be more important for the identification of patients at risk for the generation of ventricular tachyarrhythmias than only the presence of LGE. For screening high-risk HCM patients, T2-high signal is expected to be clinically useful as a new predictor for ventricular tachyarrhythmias from our results. In addition, T2-weighted imaging is superior to LGE study because of no need of the use of gadolinium contrast medium, especially when examining patients with impaired renal function who have a high risk of nephrogenic systemic fibrosis and who are not indication for LGE study.
Limitations
The present study had several limitations. First, the definition of myocardial T2-high signal has not been universally determined, and our definition may not be acceptable. Myocardial signal intensity on T2-weighted imaging may be variable depending on several factors, including CMR hardware and imaging technique. Thus, based on previous CMR experience at our institution, the three readers, in consensus, originally determined the definition of myocardial T2-high signal in this study. By our definition, the T2-high signal was clearly identified as a bright area in the LV myocardium to all those readers. In contrast, some mild but significant T2-high signal areas may be overlooked. Second, we could not perform a quantitative analysis of LGE extension in detail. We assessed only a 17-segment-based semi-quantitative evaluation of LGE. Third, we did not investigate pathological findings of myocardial T2-high signal. The definite mechanism of T2-high signal in the relation to NSVT remains unclear. Fourth, this study was retrospective and observational using the relatively small number of patients. Finally, we only assessed predictors of NSVT recorded by Holter ECG at once. In the future, the relationship between the presence of T2-high signal and the long-term prognosis or various cardiac complications should be prospectively studied using a large number of HCM patients.
Conclusions
Myocardial T2-high signal is a significant independent predictor of NSVT in patients with HCM. Although the exact pathological mechanism of this high signal remains unknown, this finding appears to be clinically interesting and T2-weighted imaging on CMR may be useful for the risk stratification of those patients.
References
Utsunomiya D, Fukunaga T, Oda S, Awai K, Nakaura T, Urata J, Yamashita Y (2011) Multidetector computed tomography evaluation of coronary plaque morphology in patients with stable angina. Heart Vessels 26(4):392–398
Olimulder MA, Kraaier K, Galjee MA, Scholten MF, van Es J, Wagenaar LJ, van der Palen J, von Birgelen C (2012) Infarct tissue characteristics of patients with versus without early revascularization for acute myocardial infarction: a contrast-enhancement cardiovascular magnetic resonance imaging study. Heart Vessels 27(3):250–257
Hendel RC, Patel MR, Kramer CM, Poon M, Carr JC, Gerstad NA, Gillam LD, Hodgson JM, Kim RJ, Lesser JR, Martin ET, Messer JV, Redberg RF, Rubin GD, Rumsfeld JS, Taylor AJ, Weigold WG, Woodard PK, Brindis RG, Douglas PS, Peterson ED, Wolk MJ, Allen JM (2006) ACCF/ACR/SCCT/SCMR/ASNC/NASCI/SCAI/SIR 2006 appropriateness criteria for cardiac computed tomography and cardiac magnetic resonance imaging: a report of the American College of Cardiology Foundation Quality Strategic Directions Committee Appropriateness Criteria Working Group, American College of Radiology, Society of Cardiovascular Computed Tomography, Society for Cardiovascular Magnetic Resonance, American Society of Nuclear Cardiology, North American Society for Cardiac Imaging, Society for Cardiovascular Angiography and Interventions, and Society of Interventional Radiology. J Am Coll Cardiol 48:1475–1497
Moon JCC, McKenna WJ, McCrohon JA, Elliott PM, Smith GC, Pennell DJ (2003) Toward clinical risk assessment in hypertrophic cardiomyopathy with gadolinium cardiovascular magnetic resonance. J Am Coll Cardiol 41:1561–1567
Teraoka K, Hirano M, Ookubo H, Sasaki K, Katsuyama H, Amino M, Abe Y, Yamashina A (2004) Delayed contrast enhancement of MRI in hypertrophic cardiomyopathy. Magn Reson Imaging 22:155–161
Adabag AS, Maron BJ, Appelbaum E, Harrigan CJ, Buros JL, Gibson CM, Lesser JR, Hanna CA, Udelson JE, Manning WJ (2008) Occurrence and frequency of arrhythmias in hypertrophic cardiomyopathy in relation to delayed enhancement on cardiovascular magnetic resonance. J Am Coll Cardiol 51:1369–1374
Leonardi S, Raineri C, De Ferrari GM, Ghio S, Scelsi L, Pasotti M, Tagliani M, Valentini A, Dore R, Raisaro A, Arbustini E (2009) Usefulness of cardiac magnetic resonance in assessing the risk of ventricular arrhythmias and sudden death in patients with hypertrophic cardiomyopathy. Eur Heart J 30:2003–2010
Higgins CB, Herfkens R, Lipton MJ, Sievers R, Sheldon P, Kaufman L, Crooks LE (1983) Nuclear magnetic resonance imaging of acute myocardial infarction in dogs: alterations in magnetic relaxation times. Am J Cardiol 52:184–188
Abdel-Aty H, Cocker M, Strohm O, Filipchuk N, Friedrich MG (2008) Abnormalities in T2-weighted cardiovascular magnetic resonance images of hypertrophic cardiomyopathy: regional distribution and relation to late gadolinium enhancement and severity of hypertrophy. J Magn Reson Imaging 28:242–245
Melacini P, Corbetti F, Calore C, Pescatore V, Smaniotto G, Pavei A, Bobbo F, Cacciavillani L, Iliceto S (2008) Cardiovascular magnetic resonance signs of ischemia in hypertrophic cardiomyopathy. Int J Cardiol 128:364–373
Maron BJ, McKenna WJ, Danielson GK, Kappenberger LJ, Kuhn HJ, Seidman CE, Shah PM, Spencer WH 3rd, Spirito P, Ten Cate FJ, Wigle ED (2003) American College of Cardiology/European Society of Cardiology clinical expert consensus document on hypertrophic cardiomyopathy. A report of the American College of Cardiology Foundation Task Force on Clinical Expert Consensus Documents and the European Society of Cardiology Committee for Practice Guidelines. Eur Heart J 24:1965–1991
Cerqueira MD, Weissman NJ, Dilsizian V, Jacobs AK, Kaul S, Laskey WK, Pennell DJ, Rumberger JA, Ryan T, Verani MS (2002) Standardized myocardial segmentation and nomenclature for tomographic imaging of the heart: a statement for healthcare professionals from the Cardiac Imaging Committee of the Council on Clinical Cardiology of the American Heart Association. Circulation 105:539–542
Adabag AS, Casey SA, Kuskowski MA, Zenovich AG, Maron BJ (2005) Spectrum and prognostic significance of arrhythmias on ambulatory Holter electrocardiogram in hypertrophic cardiomyopathy. J Am Coll Cardiol 45:697–704
Maron BJ, Spirito P, Shen WK, Haas TS, Formisano F, Link MS, Epstein AE, Almquist AK, Daubert JP, Lawrenz T, Boriani G, Estes NA 3rd, Favale S, Piccininno M, Winters SL, Santini M, Betocchi S, Arribas F, Sherrid MV, Buja G, Semsarian C, Bruzzi P (2007) Implantable cardioverter-defibrillators and prevention of sudden cardiac death in hypertrophic cardiomyopathy. JAMA 298:405–412
O’Hanlon R, Grasso A, Roughton M, Moon JC, Clark S, Wage R, Webb J, Kulkarni M, Dawson D, Sulaibeekh L (2010) Prognostic significance of myocardial fibrosis in hypertrophic cardiomyopathy. J Am Coll Cardiol 56:867–874
Kubo T, Kitaoka H, Okawa M, Hirota T, Hayato K, Yamasaki N, Matsumura Y, Yabe T, Takata J, Doi YL (2009) Clinical impact of atrial fibrillation in patients with hypertrophic cardiomyopathy. Results from Kochi RYOMA Study. Circ J 73:1599–1605
Melacini P, Basso C, Angelini A, Calore C, Bobbo F, Tokajuk B, Bellini N, Smaniotto G, Zucchetto M, Iliceto S, Thiene G, Maron BJ (2010) Clinicopathological profiles of progressive heart failure in hypertrophic cardiomyopathy. Eur Heart J 31:2111–2123
Chen-Scarabelli C, Scarabelli TM (2006) Suboptimal glycemic control, independently of QT interval duration, is associated with increased risk of ventricular arrhythmias in a high-risk population. 29(1):9–14
Moon JCC, Reed E, Sheppard MN, Elkington AG, Ho S, Burke M, Petrou M, Pennell DJ (2004) The histologic basis of late gadolinium enhancement cardiovascular magnetic resonance in hypertrophic cardiomyopathy. J Am Coll Cardiol 43:2260–2264
Papavassiliu T, Schnabel P, Schroder M, Borggrefe M (2005) CMR scarring in a patient with hypertrophic cardiomyopathy correlates well with histological findings of fibrosis. Eur Heart J 26:2395
Choudhury L, Mahrholdt H, Wagner A, Choi KM, Elliott MD, Klocke FJ, Bonow RO, Judd RM, Kim RJ (2002) Myocardial scarring in asymptomatic or mildly symptomatic patients with hypertrophic cardiomyopathy. J Am Coll Cardiol 40:2156–2164
Harris KM, Spirito P, Maron MS, Zenovich AG, Formisano F, Lesser JR, Mackey-Bojack S, Manning WJ, Udelson JE, Maron BJ (2006) Prevalence, clinical profile, and significance of left ventricular remodeling in the end-stage phase of hypertrophic cardiomyopathy. Circulation 114:216–225
Garcia-Dorado D, Oliveras J, Gili J, Sanz E, Perez-Villa F, Barrabes J, Carreras MJ, Solares J, Soler-Soler J (1993) Analysis of myocardial oedema by magnetic resonance imaging early after coronary artery occlusion with or without reperfusion. Cardiovasc Res 27:1462–1469
Wright J, Adriaenssens T, Dymarkowski S, Desmet W, Bogaert J (2009) Quantification of myocardial area at risk with T2-weighted CMR comparison with contrast-enhanced CMR and coronary angiography. JACC Cardiovasc Imaging 2:825–831
Abdel-Aty H, Boye P, Zagrosek A, Wassmuth R, Kumar A, Messroghli D, Bock P, Dietz R, Friedrich M, Schulzmenger J (2005) Diagnostic performance of cardiovascular magnetic resonance in patients with suspected acute myocarditis comparison of different approaches. J Am Coll Cardiol 45:1815–1822
Joshi SB, Chao T, Herzka DA, Zeman PR, Cooper HA, Lindsay J, Fuisz AR (2010) Cardiovascular magnetic resonance T2 signal abnormalities in left ventricular ballooning syndrome. Int J Cardiovasc Imaging 26:227–232
Knaapen P, van Dockum WG, Bondarenko O, Kok WE, Gotte MJ, Boellaard R, Beek AM, Visser CA, van Rossum AC, Lammertsma AA, Visser FC (2005) Delayed contrast enhancement and perfusable tissue index in hypertrophic cardiomyopathy: comparison between cardiac MRI and PET. J Nucl Med 46:923–929
Aso H, Takeda K, Ito T, Shiraishi T, Matsumura K, Nakagawa T (1998) Assessment of myocardial fibrosis in cardiomyopathic hamsters with gadolinium-DTPA enhanced magnetic resonance imaging. Invest Radiol 33:22–32
Basso C, Thiene G, Corrado D, Buja G, Melacini P, Nava A (2000) Hypertrophic cardiomyopathy and sudden death in the young: pathologic evidence of myocardial ischemia. Hum Pathol 31:988–998
Rubinshtein R, Glockner JF, Ommen SR, Araoz PA, Ackerman MJ, Sorajja P, Bos JM, Tajik AJ, Valeti US, Nishimura RA, Gersh BJ (2010) Characteristics and clinical significance of late gadolinium enhancement by contrast-enhanced magnetic resonance imaging in patients with hypertrophic cardiomyopathy. Circ Heart Fail 3:51–58
Bruder O, Wagner A, Jensen CJ, Schneider S, Ong P, Kispert E-M, Nassenstein K, Schlosser T, Sabin GV, Sechtem U (2010) Myocardial scar visualized by cardiovascular magnetic resonance imaging predicts major adverse events in patients with hypertrophic cardiomyopathy. J Am Coll Cardiol 56:875–887
Appelbaum E, Maron BJ, Adabag S, Hauser TH, Lesser JR, Haas TS, Riley AB, Harrigan CJ, Delling FN, Udelson JE, Gibson CM, Manning WJ, Maron MS (2012) Intermediate-signal-intensity late gadolinium enhancement predicts ventricular tachyarrhythmias in patients with hypertrophic cardiomyopathy. Circ Cardiovasc Imaging 5:78–85
Acknowledgments
We gratefully thank Mr. Kenji Horie, Mr. Tatsunori Niwa, Mr. Naokazu Mizuno, and Miss Airi Shibata for their technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hen, Y., Iguchi, N., Machida, H. et al. High signal intensity on T2-weighted cardiac magnetic resonance imaging correlates with the ventricular tachyarrhythmia in hypertrophic cardiomyopathy. Heart Vessels 28, 742–749 (2013). https://doi.org/10.1007/s00380-012-0300-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00380-012-0300-3