Abstract
To identify the controlling factors of the variability of soil respiration (R S) at multiple spatial scales, R S was measured along with environmental factors, tree diameter at breast height (DBH), and soil properties for four typical forest types in northeastern China, including the primary mixed broad-leaved Korean pine (Pinus koraiensis) forest (BKPF), spruce-fir valley forest (SVF), selective cutting of mixed broad-leaved Korean pine forest (SCF), and Korean pine plantation (KPP), throughout the growing season (May–October) in 2013. The variability of R S was quantified and compared at the following three spatial scales: among collars within a plot, among plots within a specific forest stand, and among forest stands within the landscape. The average coefficients of variations of R S within plots (30–52 %) were significantly higher than those of R S among plots (20–25 %) in each forest stand (P < 0.05). The water-filled pore space and mean DBH of trees within 8 m of the measurement collars explained 72 % of the variability of R S within the BKPF. The variabilities of R S within the SVF, SCF, and KPP were explained by the soil organic C content, soil C:N ratio, and mean DBH and total basal area of trees within a few meters of the measurement collars. The variability of R S across the four forest stands was best explained by the soil C:N ratio (R 2 = 0.63, P = 0.001).
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Quantifying the carbon (C) source/sink strength of terrestrial ecosystems is a prerequisite to evaluating the influence of terrestrial ecosystems on atmospheric CO2 concentrations and predicting climate change (Oishi et al. 2013; Luo et al. 2015). The net ecosystem exchange reflects the balance between two large CO2 fluxes, the gross photosynthesis and total ecosystem CO2 efflux (Exbrayat et al. 2013). Soil respiration (R S) accounts for 60–90 % of ecosystem CO2 efflux in forest ecosystems (Liang et al. 2004). Thus, understanding the patterns and controls of R S is of importance.
The temporal variation of R S at different scales is well understood and is affected by the supply of photosynthate, soil temperature (T), and soil water content (Kuzyakov and Gavrichkova 2010; Joo et al. 2012; Hanpattanakit et al. 2015). In contrast, the spatial variability and controlling factors of R S remain poorly understood due to technical limitations and its complex component origin (rhizospheric respiration and heterotrophic respiration) of evolved CO2 (Dore et al. 2014; Prolingheuer et al. 2014). Rhizospheric respiration is mainly controlled by gross primary productivity, the supply of photosynthate to roots from the canopy, root biomass distribution, and root nitrogen (N) concentration (Ruehr and Buchmann 2010; Kuzyakov and Gavrichkova 2010; Hopkins et al. 2013). Heterotrophic respiration is supposed to depend on the activity of soil microbes; substrate availability; litter chemical structure; and the biophysical environment of soil, such as the bulk density, field moisture capacity, and water-filled pore space (Fierer et al. 2009; Karhu et al. 2010; Luan et al. 2012; Tian and Shi 2014; Chen et al. 2015). The role of these controlling factors depends on the spatial scale. Some of these factors may only be appropriate at certain scales, and additional factors may overshadow others as spatial scales change (Martin and Bolstad 2009). For example, at the within-stand level, variation between individual R S measurements is strongly affected by the water-filled pore space, soil C content, soil C:N ratio, fine root biomass, and stand structure, such as tree proximity and leaf area index (Luan et al. 2012; Dore et al. 2014). At the landscape scale, topography, stand development (age), land use change, and plant productivity coupled with substrate availability and the C input to the soil drive R S across the forest types (Saurette et al. 2006; Wang et al. 2006; Martin and Bolstad 2009; Sheng et al. 2010). The quantification and understanding of the controlling factors of the variability of R S at various spatial scales in a study region can reduce errors in scaling up from flux measurements to the stand, landscape, and regional scales (Oishi et al. 2013). However, most studies focus on the variability of R S at a single spatial scale, e.g., at the intra-plot scale (Bréchet et al. 2011), among different plots/treatments in a forest (Ngao et al. 2012; Cheng et al. 2014) or among forest stands at the landscape scale (Sheng et al. 2010; Yoon et al. 2014). Our knowledge of the key factors that regulate the variability of R S at multiple spatial scales is limited.
Over the last few decades, large tracts of primary mixed broad-leaved Korean pine (Pinus koraiensis) forest in northeastern China have been harvested and subsequently transformed into secondary forest and plantations due to human interference or management. The Liangshui National Nature Reserve contains a variety of forest types with different management regimes and thus provides an opportunity to identify the controlling factors of the variability of R S at various scales within a restricted geographic region where air temperature, precipitation, and soil types are similar. In this study, a primary mixed broad-leaved Korean pine forest (BKPF), spruce-fir valley forest (SVF), selective cutting of mixed broad-leaved Korean pine forest (SCF), and Korean pine plantation (KPP) were selected to answer the above questions. Our specific objectives were to (1) quantify and compare the variability of R S at three spatial scales, among collars within a plot, among plots within a specific forest stand, and among forest stands within the landscape, and (2) identify the roles of biotic and abiotic factors that determine the variability of R S within a specific forest stand and among four forest stands.
Materials and methods
Study sites and experimental design
The study was conducted in the Liangshui National Nature Reserve (47° 10′ 50″ N, 128° 53′ 20″ E), Heilongjiang Province, northeastern China. The site has a continental monsoon climate regime, characterized by cold, dry winters and warm, wet summers. The mean annual temperature is −0.3 °C, and the mean annual precipitation is 676 mm, which is concentrated in the summer. The frost-free period is 100–120 days, and the snow period is 130–150 days. The soil is classified as dark-brown forest soil in the Chinese soil classification, which is equivalent to Humaquepts or Cryoboralfs according to the American Soil Taxonomy (Soil Survey Staff 1999).
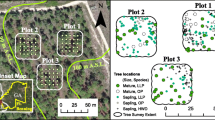
The area has a variety of forest stands at different successional stages. It has one of the most concentrated and well-conserved primary mixed broad-leaved Korean pine forests (zonal climax vegetation) in China. The selective cutting of mixed broad-leaved Korean pine forest was regenerated naturally after selective cutting of Korean pine (intensity 30 %) in 1971. The Korean pine plantation was established in 1954 following the clear-cutting of the BKPF. These three forest stands are similar in topography and microclimate, with elevations ranging from 378 to 510 m and slopes ranging from 10 to 15°. The spruce-fir valley forest was the virgin non-zonal climax forest, mainly distributed along the river or streams valley at relatively low altitudes. This area is its southern boundary of natural distribution. The elevation in this forest ranges from 339 to 350 m, and the slope is almost flat. All sites are within a circular area with a radius of 3 km. The basic characteristics and stand compositions of the four forest stands in this study are summarized in Table 1 and 2.
Three 20 × 30-m survey plots for each forest stand were established in May 2010. Tens to hundreds meters separated plots. A 10 × 10-m subplot was placed within the plot with each R S sample location located at the center of the subplot. A total of six sample locations were selected within each plot.
Soil respiration measurement
The polyvinyl chloride collars (6 cm high and 10.4 cm in diameter) were inserted 4 cm into the soil (including the litter layer) at each sample location at the beginning of May 2013, 1 week before the first measurement campaign. R S was measured approximately every other week during the growing season, from May to October 2013, using an LI-6400 portable CO2 infrared gas analyzer (IRGA; LI-COR Inc., Lincoln, NE, USA) for a total of nine measurement campaigns. The measurements were performed from 10:00 to 16:00 on rainless days to limit the effect of daily fluctuations of soil temperature and rainfall on R S. No measurements were carried out in the winter because the analyzer cannot be used at low temperatures. The soil temperature (T, measured at 5 cm below the ground surface using an Li-6000-09 TC; LI-COR Inc.) and soil water content (measured at 0–10-cm soil depth using a time-domain reflectometry probe; IMKO, Ettlingen, Germany) were measured next to each collar to coincide with R S measurements.
Stand parameters and soil properties
The diameter at breast height (DBH) and position (coordinate) were measured on all trees with a DBH greater than 1 cm within the 20 × 30-m plot and within a 5-m radius outside the plot boundary in May 2010. Based on the DBH and coordinates of all trees and the coordinates of each sampling location (collar), the following stand structural parameters were calculated for each sampling location: mean DBH (mean DBHR), maximum DBH (max DBHR), and cumulative basal area (BAR) of the trees located within 2–10 m (radius) of each R S measurement point (R represent radius).
Samples for fine (<2 mm in diameter) and medium root (2–5 mm in diameter) mass were collected at the end of July and September 2011 and once at the end of each month from May to September 2012 using 10 randomly located soil cores (5-cm diameter × 40-cm depth) in each plot. Next, live fine and medium root in the samples were distinguished, collected, dried at 60 °C to a constant mass, and weighed.
Soil samples were collected using a soil corer (5-cm diameter) within the top 10 cm of soil in September 2013 at three different locations near each sample location. Soil samples were pooled to create one sample to analyze soil chemical factors. The light fraction of soil organic matter was isolated using the method described by Janzen et al. (1992). Subsequently, the bulk soil (soil organic carbon (SOC)) and light fraction organic C contents were analyzed using a multi-N/C 2100 analyzer (Analytik Jena AG, Jena, Germany). The total N (TN) content of the soil was determined by a Hanon K9840 auto Kjeldahl analyzer (Jinan Hanon Instruments Co., Ltd, China). The soil C:N ratio was calculated from the measured SOC and TN. The soil pH was measured in water (1:2.5 w/v). In addition, two soil samples were collected from the top of the soil near each sample location using 100-ml (50.46-mm diameter and 50-mm height) cylinders to analyze the physical characteristics of the soil based on the soil water-retention characteristics (Klute 1986), including bulk density, field moisture capacity, capillary porosity, and non-capillary porosity. The water-filled pore space (WFPS) of the soil was calculated according to the following equation (Linn and Doran 1984):
where W is the volumetric water content (cm3/cm3); BD is the soil bulk density (g/cm3); and PD is the soil particle density, which was assumed to be 2.65 g/cm3 (Klute 1986).
Data analysis
The relationship between seasonal R S and T was examined by using an exponential model
where R S is the soil respiration (μmol CO2 m−2 s−1), T is the soil temperature at a depth of 5 cm, α is the intercept of CO2 efflux when T is zero, and β is the fitted parameter. Q 10 is a quotient of change in R S caused by change in temperature by 10 °C, can be indicative of the sensitivity of R S to temperature, and was calculated as follows:
The coefficient of variation (CV) was calculated as the standard deviation divided by the mean to represent the relative variations in R S, T, and soil water content. These values were calculated at the following three spatial scales: among collars within a plot (n = 6), among three plots within a specific forest stand (n = 3), and among the four forest stands within this landscape (n = 4).
A regression analysis was performed to examine the similarities in the variability of R S among the nine measurement campaigns. The significant correlations among the R S of nine measurement campaigns indicated that spatial patterns of R S remained constant across the growing season (Tables S1, S2, S3, and S4, Electronic supplementary material). Thus, the R S was averaged over the nine measurement campaigns at each sample location to identify controlling factor determining the variability of R S through the growing season in each forest stand.
The relationships between R S and soil microclimate, stand parameters, and soil properties were examined by linear regression within a specific forest stand and across the four forest stands. Subsequently, all variables that were significantly correlated with R S within a specific forest stand were used in a stepwise multiple regression analysis. Due to non-simultaneous R S and differences in T among the four forest stands, an ANCOVA test was carried out by including T as a covariate with LSD comparisons for the mean R S among the four forest stands. One-way analysis of variance (ANOVA) was performed for comparisons of fine and medium root mass, field moisture capacity, capillary porosity, light fraction organic C contents, SOC, TN, C:N ratio, and pH among the four forest types. Statistical analyses were performed with SPSS software (SPSS 18.0 for Windows, Chicago, USA). Graphs were created using SigmaPlot 12.5 software (Systat Software Inc., San Jose, CA, USA).
Results
Temporal variability of R S
The seasonal variability of R S for all forest stands followed a bell-shaped curve and depended on T at the stand levels (Fig. 1). The exponential models of R S against T explained 52–86 % of the seasonal variability in R S across the four forest stands (Fig. 2). The Q 10 values (the sensitivity of R S to temperature) for R S were 2.46, 2.05, 2.4, and 2.69 in BKPF, SVF, SCF, and KPP, respectively.
Seasonal changes in soil respiration (a), soil temperature (b), and soil water content (c) for the four forest stands. BKPF primary mixed broad-leaved Korean pine forest, SVF spruce-fir valley forest, SCF selective cutting of mixed broad-leaved Korean pine forest, KPP Korean pine plantation. The error bars represent standard errors of means (n = 3)
Factors that influences R S within a specific forest stand
The average CVs of R S among plots ranged from 18 % in SCF to 25 % in SVF (Fig. 3). The average CV of R S within plots in KPP (30 %) was significantly lower than that of R S in BKPF (46 %), SVF (44 %), and SCF (52 %; P < 0.01). The average CVs of R S within plots were significantly higher than those of R S among plots in each forest stand (P < 0.05). We found a significant positive correlation among the R S of almost all measurement campaigns for each forest stand (n = 18, P < 0.05; Table S1, S2, S3, and S4, Electronic supplementary material).
The average coefficient of variation of soil respiration (R S) among and within plots for the four forest stands. Different letters indicate significant differences among different forest stands (P < 0.05). BKPF primary mixed broad-leaved Korean pine forest, SVF spruce-fir valley forest, SCF selective cutting of mixed broad-leaved Korean pine forest, KPP Korean pine plantation
There were correlations between R S and forest structural parameters (mean DBH, max DBH, and BA), and the correlations depended on forest stand and the distance between trees and collars (Table 3 and Fig. 4). The maximum significant correlation (R max) was found at a distance of 8 m for the mean DBH, max DBH, and BA (R max = 0.48, 0.75, and 0.69, respectively) in BKPF. The R max was found at a distance of 4 m for the max DBH and BA (R max = 0.62 and 0.61, respectively) in SCF. In contrast, the correlation of R S with mean DBH8 and max DBH8 was strongest and negative (R max = −0.5 and −0.67, respectively) in SVF. The correlation of R S with mean DBH5 was strongest and negative (R max = −0.59) in KPP. Subsequently, the forest structural parameters that had the strongest relationship with R S and the physical and chemical characteristics of the soil that had a significant relationship with R S were used in a stepwise multiple regression analysis (Tables 3 and 4). A general model with both WFPS and mean DBH8 performed best in BKPF. The final model indicated that SOC and mean DBH8 explained 50 % of the variation in R S in SVF. The soil C:N ratio and forest structural parameters, such as BA4 and mean DBH5, explained the variability of R S in SCF and KPP.
Correlations between maximum DBH (max DBH), mean DBH, and basal area (BA) at various distances (from 2 to 10 m) from each measurement point and soil respiration for the four forest stands. BKPF primary mixed broad-leaved Korean pine forest, SVF spruce-fir valley forest, SCF selective cutting of mixed broad-leaved Korean pine forest, KPP Korean pine plantation
Factors influencing R S of the four forest stands
The seasonal changes in the CVs of R S of the four forest stands ranged from 8 to 25 % (Fig. 5). The mean R S in SVF (3.36 μmol CO2 m−2 s−1) was significantly higher than in BKPF (2.16 μmol CO2 m−2 s−1, P < 0.05; Table 2). The mean T differed significantly among forest stands, with the lowest T in SVF (P < 0.05), but the mean soil water content did not differ significantly among the four forest stands (P > 0.05). We found significant differences among forest stands for the physical and chemical properties of soil, such as field moisture capacity, capillary porosity, light fraction organic C contents, SOC, soil C:N ratio, and pH (P < 0.05). In contrast, TN did not differ significantly among the four forest stands (P > 0.05). The correlation analyses between R S and stand characteristics revealed that R S was significantly and linearly correlated with the physical characteristics of the soil, such as field moisture capacity and capillary porosity (P < 0.05; Fig. 6). In addition, the variability of the mean R S across the four forest stands depended on contents of light fraction organic C and SOC, soil C:N ratio, and pH (P < 0.05). The variability of the mean R S across the four forest stands was best explained by the soil C:N ratio (R 2 = 0.63, P = 0.001; Fig. 6c).
Relationships between soil respiration and the soil organic C content (a), light fraction organic C content (b), C:N ratio (c), field moisture capacity (d), capillary porosity (e), and pH (f) at a depth of 0–10 cm across the four forest stands. BKPF primary mixed broad-leaved Korean pine forest, SVF spruce-fir valley forest, SCF selective cutting of mixed broad-leaved Korean pine forest, KPP Korean pine plantation
Discussion
Sources of the variability in R S within a specific forest stand
Our results showed that the minimum CV of R S within plots occurred in KPP. The CVs of forest structure parameters (e.g., max DHB4, mean DBH5, and mean DBH8) for BKPF, SVF, and SCF were higher than that for KPP (Table S5, Electronic supplementary material). This result indicates that the forest structures of the two primary forests (BKPF and SVF) and the selective cutting of mixed forest (SCF) were highly heterogeneous compared to that of plantation (KPP), which could cause higher spatial variability of the rhizospheric component of R S through underground C allocation (Katayama et al. 2009; Luan et al. 2012).
Although there was large temporal differences for R S of a given sample location, the variability of R S remained constant across the growing season within a specific forest stand. This result indicated that some relatively stable factors drive the typically high or low R S of a given sample location. R S originates from rhizospheric respiration and heterotrophic respiration, and the two components are controlled independently by different biotic and abiotic factors (Scott-Denton et al. 2006). These various controlling factors affect the variability of R S (Prolingheuer et al. 2014). Our previous studies and other reports have shown that rhizospheric respiration was correlated with medium or fine root biomass (Pregitzer et al. 2008; Shi et al. 2015). However, root biomass measurements cannot be conducted easily. In contrast, the tree size distribution may account for the variability in R S due to significant relationships between root biomass and forest structure parameters (mean DBH, max DBH, and BA at a certain radius) (Katayama et al. 2009; Bréchet et al. 2011; Luan et al. 2012). In this study, the final model for explaining the variability of R S in BKPF and SCF confirmed this inference. The final model indicated that the forest structure parameters explained the variability of R S in BKPF and SCF. The reason could be that the tree size distribution regulated fine and medium root distribution and biomass, further controlling the variability of R S within the forest stand. Furthermore, we found that R S was negatively correlated with forest structure parameters, such as mean DBH8 and mean DBH5 in SVF and in KPP. This result indicated that larger mean DBH within a few meters (radius) of R S measurement point reduced R S, which appears contradictory to previous inference. But, it is noticeable that the dominant tree species, such as spruce and fir in SVF and Korean pine in KPP, were evergreen coniferous tree species. Thus, the amount of solar radiation reaching the litter and soil layers is small. The sizes of forest gaps or leaf area index may affect R S more strongly when compared to fine and medium root distribution in SVF and KPP. Oishi et al. (2013) reported that within a Pinus taeda plantation, higher leaf area resulted in lower R S, which was attributed to the fact that increased solar radiation associated with lower leaf area index, along with increased T that appears to lead to increases in R S. However, we did not find significant relationships between R S and T (P > 0.05) in SVF and KPP. Several other ecologically driven processes could explain an inverse relationship between R S and forest structure parameters in SVF and KPP, and more research is needed to explain the association. Although the study has several limitations, our results that R S had a significant relationship with forest structural parameters enable us to extrapolate R S spatially using forest structural parameters and have implications for an optimum R S sampling scheme. For example, the selection of R S sampling position should take into account the spatial distribution of tree size.
The heterotrophic component of R S is mainly controlled by several physical and chemical factors of soil and the substrate availability (Luan et al. 2011; Olsson et al. 2012; Jagadamma et al. 2014). Our results showed that WFPS is a good proxy for the physical factors of soil that explain the variability of R S in BKPF. The variability of R S in BKPF depended on the physical properties of soil, whose variability was partly due to the high heterogeneity of microtopography compared to that of the other forest types. The high WFPS that not only inhibits microbial activity as a result of the lack of oxygen (Wiaux et al. 2014) but also limits CO2 emissions due to water filling the soil pores (Ball 2013) could explain the negative relationship between R S with WFPS. Positive relationships were found between the R S and soil C:N ratio in the topsoil in SVF, SCF, and KPP. Our result agrees with that reported by Ngao et al. (2012), who found that the positive relationship between basal soil respiration at 10 °C and the C:N ratio at the topsoil organo-mineral layer probably reflects better C consumption efficiency with increasing C:N ratios rather than the effect of the C supply under non-limiting N conditions. This is due to the fact that litter decomposes more completely under non-limiting N conditions when decomposers are sufficiently supplied with labile C from the humus than when they are C limited (d’Annunzio et al. 2008). All of the above findings suggest that controlling factors for regulating R S depend on forest stands and sites.
The importance of scale on the variability of R S
The seasonal patterns of R S in the four forest stands were strongly related to changes in T, which was consistent with previous studies (Ma et al. 2014). The Q 10 values for R S ranged from 2.05 to 2.69 and showed the varied response of R S to temperature among the four forest stands. The temporal variability of R S is straightforward to capture using an automated soil chamber system at diurnal, seasonal, and annual timescales (Wang et al. 2013; Hanpattanakit et al. 2015). However, the spatial variability of R S and its driving factors are under-researched and complicated at different spatial scales (Buczko et al. 2015). In the present study, the average CVs of R S within plots (30–52 %) were significantly higher than those of the R S among plots (20–25 %) in each forest type (P < 0.05), and this is similar to previous findings (Wang et al. 2006; Tang et al. 2009). Tang et al. (2009) found that the variations in R S within a stand were higher than among stands (four stand replications for the young, intermediate, and mature forests) in a Great Lakes forest chronosequence. These results indicated that finer-scaled heterogeneity in R S is important and should be of concern.
Many of the drivers of R S at small scales may be overpowered by stronger influences at larger scales. Martin and Bolstad (2009) reported that at the scales of 1–10 m, variation between individual R S measurements could be explained by positive relationships between the forest floor mass, root mass, C and N pools, or root N concentration, whereas the topography strongly influenced soil moisture and soil properties and created spatial patterns of R S on the landscape. Oishi et al. (2013) showed that spatially, among three proximate temperate forest ecosystems, R S increased with leaf production, whereas within a P. taeda plantation, R S decreased with increasing leaf production. In this study, forest structure parameters (mean DBH, max DBH, and BA at a certain radius) and physical and chemical factors of soil were strong determinants of the variability in R S within a specific forest stand. However, no significant correlation between the mean R S and the BA, mean DBH, and fine and medium root mass was found across the four forest stands (P > 0.05). It is probable that the forest structure regulated fine and medium root distribution and amount of solar radiation reaching the forest floor (Hilker et al. 2008; Bréchet et al. 2011), further influencing the variability of R S at fine scales within a specific forest stand. However, at larger scales, the fine and medium root biomass did not differ significantly among the four forest stands. The variation of R S across the four forest stands was explained by physical and chemical factors of soil. These results indicate that at fine scales, within a specific forest stand, the biotic and abiotic factors, such as at a distance of a few meters for the mean DBH, max DBH, and BA and soil C:N ratio, determine the variability of R S together, whereas at larger scales, the factors controlling the variability of R S are mostly abiotic in nature (e.g., SOC, soil C:N ratio, and field moisture capacity). In addition, the soil pH was significantly and negatively correlated with R S across the four forest stands (P < 0.05), while there was no significant correlation between soil pH and R S within a specific forest stand, except within SCF as a result of the minor range of soil pH within a specific forest stand. These findings suggest that spatial scales have an important impact on the measuring and modeling of R S.
Conclusions
Our results showed that the variability of R S did not show significant changes with the season within each forest stand. The variability of R S within a specific forest stand depended on the forest structure parameters (i.e., the mean DBH, max DBH, and BA within a few meters (radius) of R S measurement point) and physical and chemical properties of soil, such as WFPS, SOC, and soil C:N ratio. The physical and chemical properties of soil, such as SOC, soil C:N ratio, and field moisture capacity, contributed to the variation of R S across the four forest stands. Furthermore, the variability of R S within plots was higher than that of R S among plots in each forest stand, and thus, fine-scale heterogeneity in R S should be of greater concern. These results imply that the selection of R S sampling position should take into account the spatial distribution of tree size and that sampling scheme with a small number of collars are liable to generate biased average R S within the plot. The sufficient number of replicates that is required to obtain an unbiased average R S within the plot needs to be further studied. Quantifying and understanding the variability of R S at different spatial scales can help reduce errors in the estimates of R S when scaling up from plot-scale measurements to stand, landscape, and regional scales.
References
Ball BC (2013) Soil structure and greenhouse gas emissions: a synthesis of 20 years of experimentation. Eur J Soil Sci 64:357–373
Bréchet L, Ponton S, Alméras T, Bonal D, Epron D (2011) Does spatial distribution of tree size account for spatial variation in soil respiration in a tropical forest? Plant Soil 347:293–303
Buczko U, Bachmann S, Gropp M, Jurasinski G, Glatzel S (2015) Spatial variability at different scales and sampling requirements for in situ soil CO2 efflux measurements on an arable soil. Catena 131:46–55
Chen Y, Sun J, Xie F, Wang X, Cheng G, Lu X (2015) Litter chemical structure is more important than species richness in affecting soil carbon and nitrogen dynamics including gas emissions from an alpine soil. Biol Fertil Soils 51:791–800
Cheng X, Han H, Kang F, Liu K, Song Y, Zhou B, Li Y (2014) Short-term effects of thinning on soil respiration in a pine (Pinus tabulaeformis) plantation. Biol Fertil Soils 50:357–367
d’Annunzio M, Zeller B, Nicolas M, Dhote JF, Saint-Andre L (2008) Decomposition of European beech (Fagus sylvatica) litter: combining quality theory and 15N labelling experiments. Soil Biol Biochem 40:322–333
Dore S, Fry DL, Stephens SL (2014) Spatial heterogeneity of soil CO2 efflux after harvest and prescribed fire in a California mixed conifer forest. For Ecol Manag 319:150–160
Exbrayat JF, Pitman AJ, Abramowitz G, Wang YP (2013) Sensitivity of net ecosystem exchange and heterotrophic respiration to parameterization uncertainty. J Geophys Res-Atmos 118:1640–1651
Fierer N, Grandy AS, Six J, Paul EA (2009) Searching for unifying principles in soil ecology. Soil Biol Biochem 41:2249–2256
Hanpattanakit P, Leclerc MY, Mcmillan AM, Limtong P, Maeght JL, Panuthai S, Chidthaisong A (2015) Multiple timescale variations and controls of soil respiration in a tropical dry dipterocarp forest, western Thailand. Plant Soil 390:167–181
Hilker T, Coops NC, Schwalm CR, Jassal RPS, Black TA, Krishnan P (2008) Effects of mutual shading of tree crowns on prediction of photosynthetic light-use efficiency in a coastal Douglas-fir forest. Tree Physiol 28:825–834
Hopkins F, Gonzalez-Meler MA, Flower CE, Lynch DJ, Czimczik C, Tang JW, Subke JA (2013) Ecosystem-level controls on root-rhizosphere respiration. New Phytol 199:339–351
Jagadamma S, Steinweg JM, Mayes MA, Wang G, Post WM (2014) Decomposition of added and native organic carbon from physically separated fractions of diverse soils. Biol Fertil Soils 50:613–621
Janzen HH, Campbell CA, Brandt SA, Lafond GP, Townley-Smith L (1992) Light-fraction organic matter in soils from long-term crop rotations. Soil Sci Soc Am J 56:1799–1806
Joo SJ, Park SU, Park MS, Lee CS (2012) Estimation of soil respiration using automated chamber systems in an oak (Quercus mongolica) forest at the Nam-San site in Seoul, Korea. Sci Total Environ 416:400–409
Karhu K, Fritze H, Tuomi M, Vanhala P, Spetz P, Kitunen V, Liski J (2010) Temperature sensitivity of organic matter decomposition in two boreal forest soil profiles. Soil Biol Biochem 42:72–82
Katayama A, Kume T, Komatsu H, Ohashi M, Nakagawa M, Yamashita M, Otsuki K, Kumagai T (2009) Effect of forest structure on the spatial variation in soil respiration in a Bornean tropical rainforest. Agric For Meteorol 149:1666–1673
Klute A (1986) Methods of soil analysis: physical and mineralogical methods. American Society of Agronomy, Madison
Kuzyakov Y, Gavrichkova O (2010) Time lag between photosynthesis and carbon dioxide efflux from soil: a review of mechanisms and controls. Glob Chang Biol 16:3386–3406
Liang N, Nakadai T, Hirano T, Qu L, Koike T, Fujinuma Y, Inoue G (2004) In situ comparison of four approaches to estimating soil CO2 efflux in a northern larch (Larix kaempferi Sarg.) forest. Agric For Meteorol 123:97–117
Linn DM, Doran JW (1984) Effect of water-filled pore space on carbon dioxide and nitrous oxide production in tilled and nontilled soils. Soil Sci Soc Am J 48:1267–1272
Luan JW, Liu SR, Wang JX, Zhu XL, Shi ZM (2011) Rhizospheric and heterotrophic respiration of a warm-temperate oak chronosequence in China. Soil Biol Biochem 43:503–512
Luan JW, Liu SR, Zhu XL, Wang JX, Liu K (2012) Roles of biotic and abiotic variables in determining spatial variation of soil respiration in secondary oak and planted pine forests. Soil Biol Biochem 44:143–150
Luo Y, Keenan TF, Smith M (2015) Predictability of the terrestrial carbon cycle. Glob Chang Biol 21:1737–1751
Ma Y, Piao S, Sun Z, Lin X, Wang T, Yue C, Yang Y (2014) Stand ages regulate the response of soil respiration to temperature in a Larix principis-rupprechtii plantation. Agric For Meteorol 184:179–187
Martin JG, Bolstad PV (2009) Variation of soil respiration at three spatial scales: components within measurements, intra-site variation and patterns on the landscape. Soil Biol Biochem 41:530–543
Ngao J, Epron D, Delpierre N, Bréda N, Granier A, Longdoz B (2012) Spatial variability of soil CO2 efflux linked to soil parameters and ecosystem characteristics in a temperate beech forest. Agric For Meteorol 154–155:136–146
Oishi AC, Palmroth S, Butnor JR, Johnsen KH, Oren R (2013) Spatial and temporal variability of soil CO2 efflux in three proximate temperate forest ecosystems. Agric For Meteorol 171:256–269
Olsson BA, Hansson K, Persson T, Beuker E, Helmisaari HS (2012) Heterotrophic respiration and nitrogen mineralisation in soils of Norway spruce, Scots pine and silver birch stands in contrasting climates. For Ecol Manag 269:197–205
Pregitzer KS, Burton AJ, King JS, Zak DR (2008) Soil respiration, root biomass, and root turnover following long-term exposure of northern forests to elevated atmospheric CO2 and tropospheric O3. New Phytol 180:153–161
Prolingheuer N, Scharnagl B, Graf A, Vereecken H, Herbst M (2014) On the spatial variation of soil rhizospheric and heterotrophic respiration in a winter wheat stand. Agric For Meteorol 195:24–31
Ruehr NK, Buchmann N (2010) Soil respiration fluxes in a temperate mixed forest: seasonality and temperature sensitivities differ among microbial and root-rhizosphere respiration. Tree Physiol 30:165–176
Saurette DD, Chang SX, Thomas BR (2006) Some characteristics of soil respiration in hybrid poplar plantations in northern Alberta. Can J Soil Sci 86:257–268
Scott-Denton LE, Rosenstiel TN, Monson RK (2006) Differential controls by climate and substrate over the heterotrophic and rhizospheric components of soil respiration. Glob Chang Biol 12:205–216
Sheng H, Yang Y, Yang Z, Chen G, Xie J, Guo J, Zou S (2010) The dynamic response of soil respiration to land-use changes in subtropical China. Glob Chang Biol 16:1107–1121
Shi BK, Gao WF, Jin GZ (2015) Effects on rhizospheric and heterotrophic respiration of conversion from primary forest to secondary forest and plantations in northeast China. Eur J Soil Biol 66:11–18
Soil Survey Staff (1999) Soil taxonomy: a basic system of soil classification for making and interpreting soil surveys. USDA Natural Resources Conservation Service, Washington, D.C
Tang JW, Bolstad PV, Martin JG (2009) Soil carbon fluxes and stocks in a Great Lakes forest chronosequence. Glob Chang Biol 15:145–155
Tian L, Shi W (2014) Soil peroxidase regulates organic matter decomposition through improving the accessibility of reducing sugars and amino acids. Biol Fertil Soils 50:785–794
Wang CK, Yang JY, Zhang QZ (2006) Soil respiration in six temperate forests in China. Glob Chang Biol 12:2103–2114
Wang CK, Han Y, Chen JQ, Wang XC, Zhang QZ, Bond-Lamberty B (2013) Seasonality of soil CO2 efflux in a temperate forest: biophysical effects of snowpack and spring freeze-thaw cycles. Agric For Meteorol 177:83–92
Wiaux F, Vanclooster M, Cornelis JT, Van Oost K (2014) Factors controlling soil organic carbon persistence along an eroding hillslope on the loess belt. Soil Biol Biochem 77:187–196
Yoon TK, Noh NJ, Han S, Lee J, Son Y (2014) Soil moisture effects on leaf litter decomposition and soil carbon dioxide efflux in wetland and upland forests. Soil Sci Soc Am J 78:1804–1816
Acknowledgments
This work was financially supported by Fundamental Research Funds for the Central Universities (DL13EA05) and the Program for Changjiang Scholars and Innovative Research Team in Universities (IRT_15R09). We would like to thank the anonymous reviewers and the Editor-in-Chief for their constructive and helpful comments that helped to improve the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shi, B., Jin, G. Variability of soil respiration at different spatial scales in temperate forests. Biol Fertil Soils 52, 561–571 (2016). https://doi.org/10.1007/s00374-016-1100-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00374-016-1100-1