Abstract
Objectives
To systematically review the published literature and evaluate the efficacy and safety of core needle biopsy (CNB) for initially detected thyroid nodules.
Methods
The Ovid-MEDLINE and Embase databases were searched for studies evaluating CNB for the diagnosis of initially detected thyroid nodules. A meta-analysis was performed to evaluate non-diagnostic results, inconclusive results and diagnostic accuracy for a diagnosis of malignancy with CNB. To overcome heterogeneity, multiple subgroup analyses were performed. The complication rate was also evaluated.
Results
Thirteen eligible studies, which included a total sample size of 9,166 patients with 13,585 nodules, were included. The pooled proportions were 3.5% (95% CI 2.4–5.1) for non-diagnostic results and 13.8% (95% CI 9.1–20.3) for inconclusive results. Considerable heterogeneity was observed among the studies in terms of the pooled proportions for CNB (I2=92.9%, 97%). With regard to the diagnostic performance for malignancy, the sensitivity was 80% (95% CI 75–85) and the specificity was 100% (95% CI 93–100). Only two major complications of CNB were observed.
Conclusions
CNB demonstrates a low non-diagnostic result rate and high diagnostic accuracy for initially detected thyroid nodules and a low major complication rate. These findings indicate that CNB may be a feasible diagnostic tool for patients with initially detected thyroid nodules.
Key Points
-
CNB demonstrates high diagnostic accuracy for initially detected thyroid nodules.
-
CNB demonstrates a low major complication rate.
-
CNB may be a feasible diagnostic tool for patients with initially detected thyroid nodules.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Ultrasound-guided fine-needle aspiration (FNA) is widely accepted as the primary diagnostic tool for the evaluation of thyroid nodules. FNA is a safe, simple, and reliable method for evaluating the thyroid nodule and has reduced the number of unnecessary thyroid surgeries. Although FNA has high diagnostic accuracy for thyroid nodules, it has several limitations: the non-diagnostic result rate ranges from 10% to 36% [1,2,3,4] and the indeterminate result rate ranges from 3% to 18% [2, 3, 5]. Furthermore, the false-negative rate of thyroid nodules with a benign FNA result reaches 13.6–56.6% when the thyroid nodules have suspicious features on ultrasound [6]. Poor specimen quality due to insufficient cellularity and preservation is considered to be the cause of misdiagnoses using FNA [7].
Core needle biopsy (CNB) has been suggested as an additional diagnostic tool to FNA. Many studies have demonstrated the successful use of CNB for thyroid nodules after initial non-diagnostic or indeterminate results on FNA [8,9,10,11,12,13,14,15]. Recent systematic reviews and meta-analyses have examined the value of CNB as a complementary diagnostic tool in thyroid nodules with initially non-diagnostic or indeterminate results on FNA [16, 17]. In addition to the complementary role of CNB, several studies have published data on the use of CNB as a primary diagnostic tool for initially detected thyroid nodules [18,19,20,21]. However, some physicians still doubt the value of CNB for the evaluation of thyroid nodules due to insufficient evidence. No systematic review with meta-analysis has assessed the role of CNB in the evaluation of initially detected thyroid nodules. Accordingly, the aim of this study was to systematically review the published literature and evaluate the prevalence of non-diagnostic results and inconclusive results and the diagnostic performance of CNB for initially detected thyroid nodules.
Materials and methods
Literature search strategy
A computerised search of the MEDLINE and Embase databases was performed to identify relevant original articles on the use of CNB as a first-line diagnostic tool for initially detected thyroid nodules until 17 August 2017. We used the following search terms: ((thyroid)) AND ((core-needle biopsy) OR (core needle biopsy) OR (CNB)). Only studies published in English were included. The bibliographies of the selected articles were screened to identify other relevant articles. Endnote version X8 (Thomson Reuters, New York, NY, USA) was used to manage the literature.
Inclusion criteria
Studies investigating the use of CNB as a first-line diagnostic tool for initially-detected thyroid nodules were eligible for inclusion. Studies or subsets of studies satisfying all of the following criteria were included:
-
(a)
Population: Patients with thyroid nodules who underwent CNB as a first-line diagnostic tool without a previous FNA procedure.
-
(b)
Reference standard: Because the diagnostic criteria for CNB of thyroid nodules have not been standardised, the histological results of CNB were categorised into the six categories of the Bethesda System [8, 11, 22]. The six categories of the Bethesda System include non-diagnostic, benign, atypia of undetermined significance (AUS) or follicular lesion of undetermined significance (FLUS), follicular neoplasm or suspicious for follicular neoplasm, suspicious for malignancy and malignant (21). Non-diagnostic results included the absence of any identifiable follicular thyroid tissue [23], follicular cells are present [24], but those are regarded as normal thyroid tissue only [25], and tissues containing only a few follicular cells insufficient for diagnosis [3]. Inconclusive results were defined as Bethesda category 1 and category 3. The diagnostic criteria for malignancy were defined as Bethesda category 6 (malignancy). Malignant nodules were diagnosed after surgery or after biopsy. Benign nodules were diagnosed after surgery, after at least two sets of benign findings on FNA and/or CNB, or after benign cytology findings on FNA or CNB with a stable nodule size after 1 year.
-
(c)
Study designs: All observational studies (retrospective or prospective).
-
(d)
Outcomes: Results reported in sufficient detail to evaluate the prevalence of non-diagnostic results, inconclusive result and diagnostic performance of CNB.
Exclusion criteria
The exclusion criteria were as follows: (a) case reports and case series with a sample size smaller than ten patients; (b) review articles, editorials, letters, comments and conference proceedings; (c) studies that did not focus on the use of CNB as the first-line diagnostic tool for thyroid nodules; (d) studies with insufficient data to be included in a meta-analysis of rates of non-diagnostic results and diagnostic performance; and (e) studies with overlapping patients and data. Two reviewers (S.R.C. and C.H.S.) independently selected literature reports using a standardised form.
Data extraction
One reviewer (S.R.C.) extracted data from the studies, and the second reviewer (C.H.S.) double-checked the accuracy of the extracted data and resolved any uncertainty through discussion. The following data were extracted from each of the selected studies onto standardised data forms: (a) Study characteristics: authors, year of publication, hospital or medical school, years of patient recruitment, sample size and study design; (b) demographic and clinical characteristics of patients: mean age, nodule size and patient reference standards; (c) rates of non-diagnostic and inconclusive results of CNB; (d) diagnostic performance of CNB for the diagnosis of malignancy; and (e) complications.
Quality assessment
The methodological quality of the included studies was assessed independently by two reviewers (S.R.C. and C.H.S.) using tailored questionnaires devised according to Quality Assessment of Diagnostic Accuracy Studies-2 (QUADAS-2) criteria [23]. Disagreements were very minor and were resolved by consensus.
Data synthesis and analyses
The pooled proportions for non-diagnostic and inconclusive results of CNB for initially-detected thyroid nodules were adopted as the main indices for this meta-analysis. The meta-analytic pooling was conducted by the inverse variance method for calculating weights. The overall proportion was obtained using fixed and random effects meta-analysis of single proportions and logit transformation of proportions [26, 27]. We also obtained the confidence interval (CI) using the Clopper-Pearson interval for individual studies and used a continuity correction of 0.5 in studies with zero cell frequencies. Heterogeneity among the studies was determined by using the following methods: the Cochran Q-test for pooled estimates (p < .05 indicating significant heterogeneity) and the Higgins inconsistency index (I2) test (0–40%, may not be important; 30–60%, may represent moderate heterogeneity; 50–90%, may represent substantial heterogeneity; 75–100%, may represent considerable heterogeneity) [28, 29]. Publication bias was visually assessed by funnel plots, and statistical significance was evaluated by Egger’s test [30]. Thereafter, publication bias-adjusted pooled estimates – that is, adjusted pooled proportions – were obtained by using the trim-and-fill method [31]. If the original unadjusted pooled proportions and the trim-and-fill–adjusted pooled proportions were in agreement, the results were regarded as robust for publication bias. We also carried out multiple subgroup analyses according to the design of the study (retrospective or prospective), origin of the study (Asia vs. outside Asia) and who performed the CNB (radiologist or not).
The secondary index of this study was the diagnostic performance of CNB in diagnosing malignancy. A threshold effect was visually assessed using coupled forest plots of sensitivity and specificity. The Spearman correlation coefficient between the sensitivity and a false-positive rate was obtained, and > 0.6 was considered a considerable threshold effect [32]. Hierarchical logistic regression modeling (bivariate modeling and hierarchical summary receiver operating characteristic [HSROC] modeling) was used to calculate pooled summary estimates of sensitivity and specificity [33,34,35]. An HSROC curve with a 95% confidence region and prediction region was plotted. Publication bias was evaluated using a Deeks’ funnel plot and Deeks’ asymmetry test [36].
All statistical analyses were performed by one reviewer (C.H.S., with 4 years of experience in performing systematic reviews and meta-analyses) using the ‘meta’ package in R v. 3.4.1 (R Foundation for Statistical Computing, Vienna, Austria) and the ‘metandi’ and ‘midas’ modules in Stata 10.0 (StataCorp, College Station, TX, USA).
Results
Literature search
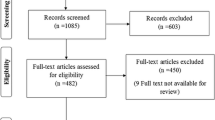
Our study selection process is illustrated in Fig. 1. The literature search of the Ovid-MEDLINE and Embase databases generated 639 initial articles; after duplicates were removed, 398 articles were screened for eligibility. Of the remaining articles, we excluded 379 after reviewing the titles and abstracts: 167 articles that were not in the field of interest; 126 conference abstracts; 47 review articles; 30 case reports; and nine editorial letters. The full texts of the remaining 19 articles were reviewed; an additional search of the bibliographies of these articles identified no further eligible studies. Of these 19 articles, we further excluded six after full text review: four articles that were not in the field of interest [37,38,39,40], one article with partially overlapping patient cohorts [41] and one article with insufficient data [42]. Finally, 13 eligible studies, which included a total sample size of 9,166 patients with 13,585 nodules, were included in this meta-analysis [18,19,20,21, 43,44,45,46,47,48,49,50,51].
Characteristics of the included studies
The characteristics of the 13 included studies are detailed in Table 1. The 13 original articles included 11 retrospective studies [18,19,20,21, 43, 45, 46, 48,49,50,51] and two prospective studies [44, 47]. All included studies had clear descriptions of the CNB technique and equipment. There were six studies from Asian countries (South Korea, 5; Japan, 1) and seven from outside Asian countries (USA, 4; UK, 1; Spain, 1; Denmark, 1). The mean patient age was 51.3 years (range, 13–92 years) and the mean nodule size was 2 cm (range, 0.2–13cm). CNB was performed by a radiologist in 11 studies and by a surgeon in two studies. Nodule composition was described in six studies; most nodules were solid [20, 21, 47, 49,50,51].
Quality assessment
The QUADIA-2 quality of the included studies was moderate overall, and all of the studies satisfied at least four of the seven items (Supplementary Fig. 1). Eight studies had a high risk of bias in the reference standard due to a poor description or inappropriate definition in the pathological report of the CNB [18, 19, 43,44,45,46,47,48].
Pooled proportions of non-diagnostic results and inconclusive results of CNB
The prevalence of non-diagnostic results and inconclusive results of CNB was described in 12 [18,19,20,21, 44,45,46,47,48,49,50,51] and five [20, 21, 49,50,51] studies, respectively. The pooled proportions of non-diagnostic results and inconclusive results on CNB are summarised in Table 2, and the corresponding forest plots are shown in Fig. 2. The pooled proportion of non-diagnostic results was 3.5% (95% CI 2.4–5.1), and the pooled proportion of inconclusive results was 13.8% (95% CI 9.1–20.3). Considerable heterogeneity was observed among the studies in terms of the pooled proportions of CNB (I2=92.9%, 97%). The funnel plots showed no publication bias for the pooled proportion of non-diagnostic results on CNB (p = .1945).
Multiple subgroup analyses
A summary of the multiple subgroup analyses of the non-diagnostic results of CNB is presented in Table 3. The pooled non-diagnostic results of CNB were significantly higher in retrospective studies than in prospective studies (p = .01). There was no significant difference in the pooled non-diagnostic results of CNB between studies originating within Asia and outside Asia (p = .347). The pooled non-diagnostic results of CNBs performed by a radiologist were 3.5%.
Diagnostic performance of CNB for malignancy
The diagnostic performance of CNB for malignancy was described in nine studies involving 5,010 nodules [19, 20, 43, 45, 46, 48,49,50,51]. The diagnostic criteria for malignancy involved a classification of Bethesda category 6 (malignancy). The coupled forest plots of the sensitivity and specificity of CNB for malignancy diagnosis are shown in Fig. 3. CNB demonstrated a summary sensitivity of 80% (95% CI 75–85) and specificity of 100% (95% CI 93–100). The area under the HSROC curve is shown in Fig. 4 and was 0.93 (95% CI 0.91–0.95). Regarding the linear regression test of funnel plot asymmetry, the statistically insignificant value (p = .07) for the slope coefficient suggested symmetry in the data and a low likelihood of publication bias (Supplementary Fig. 2).
Complications
Twelve of the 13 studies reported complications of CNB [18,19,20, 43,44,45,46,47,48,49,50,51]. In these 12 studies, only two major complications occurred in 6,979 patients [19, 48]. One patient had some bleeding after CNB and was admitted to the hospital for overnight observation, without any intervention [48]. Another patient had a major complication of recurrent laryngeal nerve damage after CNB [19]. The damage to the recurrent laryngeal nerve occurred due to direct puncture of the nerve and caused permanent dysphonia. This complication was caused by a lateral approach of the needle. No procedure-related deaths or need for intervention were reported. All other studies reported the occurrence of minor complications including haematoma (n = 73), soft tissue infection (n = 1) and parenchymal oedema (n = 9) [18,19,20, 43,44,45,46,47,48,49,50,51].
Discussion
Our present meta-analysis revealed that CNB is an acceptable diagnostic tool for initially-detected thyroid nodules. In this present study, we found pooled proportions of 3.5% (95% CI 2.4–5.1) for non-diagnostic results and 13.8% (95% CI 9.1–20.3) for inconclusive results. With regard to the diagnostic performance for malignancy, CNB showed a summary sensitivity of 80% (95% CI 75–85) and specificity of 100% (95% CI 93–100). There were only two major complications associated with CNB among 6,979 patients. These results suggest that CNB can be used as a primary diagnostic tool for initially-detected thyroid nodules as well as a subsequent diagnostic tool to FNA.
Over the last few decades, many original articles have described the role of CNB for thyroid nodules. Many studies have investigated the role of CNB in thyroid nodules previously diagnosed as non-diagnostic or inconclusive in FNA [8,9,10,11,12,13,14,15, 52,53,54]. Two systematic reviews and meta-analyses have examined the value of CNB as a complementary diagnostic tool in thyroid nodules with initially non-diagnostic or indeterminate results on FNA [16, 17]. The results showed low non-diagnostic results of 1.8% (95% CI 0.4–3.2) and high specificity (100%) for the diagnosis of malignancy in nodules with an initially indeterminate result on FNA [16]. For nodules with an initially non-diagnostic result on FNA, CNB also showed lower non-diagnostic results (6.4% vs. 36.5%) and higher diagnostic accuracy (sensitivity, 89.8% vs. 60.6%) than repeat FNA [17]. In addition, current guidelines recommend that CNB be considered when the cytological results of FNA are repeatedly inadequate or inconclusive [55,56,57]. However, because the role of indeterminate lesions is still unsettled, routine use of CNB is not currently recommended by guidelines [56].
Several recent studies have reported the potential of CNB as a primary diagnostic tool for patients with thyroid nodules [18,19,20, 51]. However, CNB is not routinely used in thyroid biopsy. This is partly due to limited evidence of the efficacy of CNB and concerns about safety [56, 58, 59]. Our meta-analysis revealed that the non-diagnostic result rate was 3.5% (95% CI 2.4–5.1), which is somewhat lower than the 2–16% of FNA [58, 60, 61].The sensitivity for diagnosis of malignancy was 80%, which is slightly higher than the reported sensitivity of FNA of 74% [20]. The advantages of CNB may be explained by its ability to sample large amounts of tissue, assess histological architecture (rather than cytology), and function with a low rate of operator dependence, if targeting of thyroid nodules is successful [8]. In addition, our data confirm the safety of the CNB procedure, with only two reported major complications in 6,979 patients who underwent CNB. One patient had some bleeding after CNB and was admitted to the hospital for overnight observation, without the need for an intervention [48]. Another patient had a major complication of recurrent laryngeal nerve damage after CNB [19]. The recurrent laryngeal nerve damage was due to direct puncture of the nerve caused by a lateral approach of the needle. To prevent this complication, the transisthmic approach is recommended [62]. By using a transisthmic approach, the operator can prevent direct injury to the recurrent laryngeal nerve and can carefully measure the safe distance from the needle tip before the stylet is fired.
This meta-analysis also showed variable heterogeneities regarding the pooled proportions for CNB, which affects the general applicability of the summary estimates. To overcome the heterogeneity, we performed subgroup analyses according to the operator, study design and origin of the study. In subgroup analysis, the non-diagnostic result rate of retrospective studies was significantly higher than that of prospective studies, which seems to be due to the fact that the two prospective studies were published in 1994 and 2001, before the development of modern techniques and devices of CNB. There was no significant difference in the pooled non-diagnostic results of CNB between studies originating within Asia and outside Asia. Thus, with modern techniques and devices, CNB may be a more effective diagnostic tool for thyroid nodules, regardless of the region.
This study had some limitations. First, the diagnostic categories of CNB specimens have not been standardised. Eight studies had a high risk of bias in the reference standard due to a poor description or inappropriate definition in the pathological report of the CNB. However, the pathological criteria for non-diagnostic results and malignancy of CNB specimen were identical in almost all studies. Second, ten of the included studies were retrospective studies. However, we used validated systematic review methods and reported our data according to standard reporting guidelines, including the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) [63] and the guidelines of the Handbook for Diagnostic Test Accuracy Reviews published by the Cochrane Collaboration [64]. Third, our meta-analysis showed considerable heterogeneity in the pooled proportions. Other variables that might affect the CNB result, such as operator experience, nodule characteristics and number of needle passes were not evaluated by subgroup analysis because it was difficult to extract accurate data for our meta-analysis. Finally, we only included articles in English, which could have resulted in an overestimation or underestimation of the results. In addition, we excluded grey literature, such as letters, case reports, conference abstracts and unpublished data, which may have caused a publication bias. However, it was difficult to extract accurate data for the meta-analysis from these types of publications.
In conclusion, our present systematic review and meta-analysis indicated that CNB has a low non-diagnostic result and high diagnostic accuracy for initially detected thyroid nodules and a low major complication rate. CNB may therefore be a feasible diagnostic tool for patients with initially detected thyroid nodules.
Abbreviations
- AUS:
-
Atypia of undetermined significance
- CNB:
-
Core needle biopsy
- FNA:
-
Fine needle aspiration
- US:
-
Ultrasound
References
Gharib H, Papini E, Paschke R et al (2010) American Association of Clinical Endocrinologists, Associazione Medici Endocrinologi, and EuropeanThyroid Association Medical Guidelines for clinical practice for the diagnosis and management of thyroid nodules. Endocr Pract 16 Suppl 1:1–43
Nayar R, Ivanovic M (2009) The indeterminate thyroid fine-needle aspiration: experience from an academic center using terminology similar to that proposed in the 2007 National Cancer Institute Thyroid Fine Needle Aspiration State of the Science Conference. Cancer 117:195–202
Yassa L, Cibas ES, Benson CB et al (2007) Long-term assessment of a multidisciplinary approach to thyroid nodule diagnostic evaluation. Cancer 111:508–516
Degirmenci B, Haktanir A, Albayrak R et al (2007) Sonographically guided fine-needle biopsy of thyroid nodules: the effects of nodule characteristics, sampling technique, and needle size on the adequacy of cytological material. Clin Radiol 62:798–803
Yang J, Schnadig V, Logrono R, Wasserman PG (2007) Fine-needle aspiration of thyroid nodules: a study of 4703 patients with histologic and clinical correlations. Cancer 111:306–315
Chung SR, Baek JH, Park HS et al (2017) Ultrasound-pathology discordant nodules on core-needle biopsy: malignancy risk and management strategy. Thyroid 27:707–713
Raab SS, Vrbin CM, Grzybicki DM et al (2006) Errors in thyroid gland fine-needle aspiration. Am J Clin Pathol 125:873–882
Na DG, Kim JH, Sung JY et al (2012) Core-needle biopsy is more useful than repeat fine-needle aspiration in thyroid nodules read as nondiagnostic or atypia of undetermined significance by the Bethesda system for reporting thyroid cytopathology. Thyroid 22:468–475
Na DG, Min HS, Lee H, Won JK, Seo HB, Kim JH (2015) Role of core needle biopsy in the management of atypia/follicular lesion of undetermined significance thyroid nodules: comparison with repeat fine-needle aspiration in subcategory nodules. Eur Thyroid J 4:189–196
Park KT, Ahn SH, Mo JH et al (2011) Role of core needle biopsy and ultrasonographic finding in management of indeterminate thyroid nodules. Head Neck 33:160–165
Choi YJ, Baek JH, Ha EJ et al (2014) Differences in risk of malignancy and management recommendations in subcategories of thyroid nodules with atypia of undetermined significance or follicular lesion of undetermined significance: the role of ultrasound-guided core-needle biopsy. Thyroid 24:494–501
Jang EK, Kim WG, Kim EY et al (2016) Usefulness of NRAS codon 61 mutation analysis and core needle biopsy for the diagnosis of thyroid nodules previously diagnosed as atypia of undetermined significance. Endocrine 52:305–312
Kim TH, Jeong DJ, Hahn SY et al (2016) Triage of patients with AUS/FLUS on thyroid cytopathology: effectiveness of the multimodal diagnostic techniques. Cancer Med 5:769–777
Choi SH, Baek JH, Lee JH et al (2014) Thyroid nodules with initially non-diagnostic, fine-needle aspiration results: comparison of core-needle biopsy and repeated fine-needle aspiration. Eur Radiol 24:2819–2826
Lee SH, Kim MH, Bae JS, Lim DJ, Jung SL, Jung CK (2014) Clinical outcomes in patients with non-diagnostic thyroid fine needle aspiration cytology: usefulness of the thyroid core needle biopsy. Ann Surg Oncol 21:1870–1877
Suh CH, Baek JH, Park C, Choi YJ, Lee JH (2017) The role of core needle biopsy for thyroid nodules with initially indeterminate results on previous fine-needle aspiration: a systematic review and meta-analysis. AJNR Am J Neuroradiol 38:1421–1426
Suh CH, Baek JH, Kim KW et al (2016) The role of core-needle biopsy for thyroid nodules with initially nondiagnostic fine-needle aspiration results: a systematic review and meta-analysis. Endocr Pract 22:679–688
Chen BT, Jain AB, Dagis A et al (2015) Comparison of the efficacy and safety of ultrasound-guided core needle biopsy versus fine-needle aspiration for evaluating thyroid nodules. Endocr Pract 21:128–135
Paja M, del Cura JL, Zabala R et al (2016) Ultrasound-guided core-needle biopsy in thyroid nodules. A study of 676 consecutive cases with surgical correlation. Eur Radiol 26:1–8
Suh CH, Baek JH, Lee JH et al (2016) The role of core-needle biopsy as a first-line diagnostic tool for initially detected thyroid nodules. Thyroid 26:395–403
Ahn SH, Park SY, Choi SI (2017) Comparison of consecutive results from fine needle aspiration and core needle biopsy in thyroid nodules. Endocr Pathol 28:332–338
Yeon JS, Baek JH, Lim HK et al (2013) Thyroid nodules with initially nondiagnostic cytologic results: the role of core-needle biopsy. Radiology 268:274–280
Lee YH, Baek JH, Jung SL, Kwak JY, Kim JH, Shin JH (2015) Ultrasound-guided fine needle aspiration of thyroid nodules: a consensus statement by the korean society of thyroid radiology. Korean J Radiol 16:391–401
Renshaw AA (2010) Should “atypical follicular cells” in thyroid fine-needle aspirates be subclassified? Cancer Cytopathol 118:186–189
Jung CK, Min HS, Park HJ et al (2015) Pathology reporting of thyroid core needle biopsy: a proposal of the Korean Endocrine Pathology Thyroid Core Needle Biopsy Study Group. J Pathol Transl Med 49:288–299
Borenstein M, Hedges LV, Higgins JP, Rothstein HR (2010) A basic introduction to fixed-effect and random-effects models for meta-analysis. Res Synth Methods 1:97–111
DerSimonian R, Kacker R (2007) Random-effects model for meta-analysis of clinical trials: an update. Contemp Clin Trials 28:105–114
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327:557–560
Higgins J, Green S (2011) Cochrane handbook for systematic reviews of interventions version 5.1.0. The Cochrane Collaboration, London. Available via http://handbook.cochrane.org. Accessed 10 Oct 2017
Egger M, Davey Smith G, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315:629–634
Duval S, Tweedie R (2000) Trim and fill: a simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics 56:455–463
Deville WL, Buntinx F, Bouter LM et al (2002) Conducting systematic reviews of diagnostic studies: didactic guidelines. BMC Med Res Methodol 2:9
Suh CH, Park SH (2016) Successful publication of systematic review and meta-analysis of studies evaluating diagnostic test accuracy. Korean J Radiol 17:5–6
Kim KW, Lee J, Choi SH, Huh J, Park SH (2015) Systematic review and meta-analysis of studies evaluating diagnostic test accuracy: a practical review for clinical researchers-Part I. General guidance and tips. Korean J Radiol 16:1175–1187
Lee J, Kim KW, Choi SH, Huh J, Park SH (2015) Systematic review and meta-analysis of studies evaluating diagnostic test accuracy: a practical review for clinical researchers-Part II. Statistical methods of meta-analysis. Korean J Radiol 16:1188–1196
Deeks JJ, Macaskill P, Irwig L (2005) The performance of tests of publication bias and other sample size effects in systematic reviews of diagnostic test accuracy was assessed. J Clin Epidemiol 58:882–893
Guler B, Kiran T, Arici DS, Aysan E, Sonmez FC (2016) Should core needle biopsy be used in the evaluation of thyroid nodules? Endocr Pathol 27:352–358
Kim SY, Lee HS, Moon J et al (2017) Fine-needle aspiration versus core needle biopsy for diagnosis of thyroid malignancy and neoplasm: a matched cohort study. Eur Radiol 27:801–811
Silverman JF, West RL, Finley JL et al (1986) Fine-needle aspiration versus large-needle biopsy or cutting biopsy in evaluation of thyroid nodules. Diagn Cytopathol 2:25–30
Trimboli P, Nasrollah N, Guidobaldi L et al (2014) The use of core needle biopsy as first-line in diagnosis of thyroid nodules reduces false negative and inconclusive data reported by fine-needle aspiration. World J Surg Oncol 12:61
Ha EJ, Baek JH, Lee JH et al (2014) Core needle biopsy can minimise the non-diagnostic results and need for diagnostic surgery in patients with calcified thyroid nodules. Eur Radiol 24:1403–1409
Munn JS, Castelli M, Prinz RA, Walloch JL (1988) Needle biopsy of nodular thyroid disease. Am Surg 54:438–443
Lo Gerfo P, Colacchio T, Caushaj F, Weber C, Feind C (1982) Comparison of fine-needle and coarse-needle biopsies in evaluating thyroid nodules. Surgery 92:835–838
Quinn SF, Nelson HA, Demlow TA (1994) Thyroid biopsies: fine-needle aspiration biopsy versus spring-activated core biopsy needle in 102 patients. J Vasc Interv Radiol 5:619–623
Liu Q, Castelli M, Gattuso P, Prinz RA (1995) Simultaneous fine-needle aspiration and core-needle biopsy of thyroid nodules. Am Surg 61:628–632 discussion 632-623
Taki S, Kakuda K, Kakuma K et al (1997) Thyroid nodules: evaluation with US-guided core biopsy with an automated biopsy gun. Radiology 202:874–877
Karstrup S, Balslev E, Juul N, Eskildsen PC, Baumbach L (2001) US-guided fine needle aspiration versus coarse needle biopsy of thyroid nodules. Eur J Ultrasound 13:1–5
Harvey JN, Parker D, De P, Shrimali RK, Otter M (2005) Sonographically guided core biopsy in the assessment of thyroid nodules. J Clin Ultrasound 33:57–62
Sung JY, Na DG, Kim KS et al (2012) Diagnostic accuracy of fine-needle aspiration versus core-needle biopsy for the diagnosis of thyroid malignancy in a clinical cohort. Eur Radiol 22:1564–1572
Kim HC, Kim YJ, Han HY et al (2017) First-line use of core needle biopsy for high-yield preliminary diagnosis of thyroid nodules. AJNR Am J Neuroradiol 38:357–363
Suh CH, Baek JH, Choi YJ et al (2017) Efficacy and safety of core-needle biopsy in initially detected thyroid nodules via propensity score analysis. Sci Rep 7:8242
Samir AE, Vij A, Seale MK et al (2012) Ultrasound-guided percutaneous thyroid nodule core biopsy: clinical utility in patients with prior nondiagnostic fine-needle aspirate. Thyroid 22:461–467
Trimboli P, Nasrollah N, Amendola S et al (2015) A cost analysis of thyroid core needle biopsy vs. diagnostic surgery. Gland Surg 4:307–311
Yoo WS, Choi HS, Cho SW et al (2014) The role of ultrasound findings in the management of thyroid nodules with atypia or follicular lesions of undetermined significance. Clin Endocrinol 80:735–742
Shin JH, Baek JH, Chung J et al (2016) Ultrasonography diagnosis and imaging-based management of thyroid nodules: revised Korean Society of Thyroid Radiology Consensus Statement and Recommendations. Korean J Radiol 17:370–395
Gharib H, Papini E, Garber JR et al (2016) American Association of Clinical Endocrinologists, American College of Endocrinology, and Associazione Medici Endocrinologi Medical Guidelines for clinical practice for the diagnosis and management of thyroid nodules--2016 update. Endocr Pract 22:622–639
Baek JH (2017) Current status of core needle biopsy of the thyroid. Ultrasonography 36:83–85
Haugen BR, Alexander EK, Bible KC et al (2016) 2015 American Thyroid Association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: The American Thyroid Association Guidelines Task Force on thyroid nodules and differentiated thyroid cancer. Thyroid 26:1–133
Lo Gerfo P (1994) The value of coarse needle biopsy in evaluating thyroid nodules. Thyroidology 6:1–4
Theoharis CG, Schofield KM, Hammers L, Udelsman R, Chhieng DC (2009) The Bethesda thyroid fine-needle aspiration classification system: year 1 at an academic institution. Thyroid 19:1215–1223
Bongiovanni M, Spitale A, Faquin WC, Mazzucchelli L, Baloch ZW (2012) The Bethesda System for reporting thyroid cytopathology: a meta-analysis. Acta Cytol 56:333–339
Na DG, Baek JH, Jung SL et al (2017) Core needle biopsy of the thyroid: 2016 consensus statement and recommendations from Korean Society of Thyroid Radiology. Korean J Radiol 18:217–237
Liberati A, Altman DG, Tetzlaff J et al (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol 62:e1-34
Deeks JJ, Bossuyt PM, Gatsonis C (2013) Cochrane handbook for systematic reviews of diagnostic test accuracy version 1.0.0. The Cochrane Collaboration, London. Available via http://srdta.cochrane.org/handbook-dtareviews. Accessed 10 Oct 2017
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Jung Hwan Baek.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
One of the authors has significant statistical expertise.
Informed consent
Written informed consent was not required for this study because it is systematic review and meta-analysis.
Ethical approval
Institutional Review Board approval was not required because it is systematic review and meta-analysis.
Methodologys
• Meta-analysis
Electronic supplementary material
ESM 1
(DOCX 2250 kb)
Rights and permissions
About this article
Cite this article
Chung, S.R., Suh, C.H., Baek, J.H. et al. The role of core needle biopsy in the diagnosis of initially detected thyroid nodules: a systematic review and meta-analysis. Eur Radiol 28, 4909–4918 (2018). https://doi.org/10.1007/s00330-018-5494-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-018-5494-z