Abstract
Objectives
To analyse the development of MRI-guided vacuum-assisted biopsy (VAB) in Switzerland and to compare the procedure with stereotactically guided and ultrasound-guided VAB.
Methods
We performed a retrospective analysis of VABs between 2009 and 2011. A total of 9,113 VABs were performed. Of these, 557 were MRI guided.
Results
MRI-guided VAB showed the highest growth rate (97 %) of all three procedures. The technical success rates for MRI-guided, stereotactically guided and ultrasound-guided VAB were 98.4 % (548/557), 99.1 % (5,904/5,960) and 99.6 % (2,585/2,596), respectively. There were no significant differences (P = 0.12) between the MRI-guided and the stereotactically guided procedures. The technical success rate for ultrasound-guided VAB was significantly higher than that for MRI-guided VAB (P < 0.001). There were no complications using MRI-guided VAB requiring open surgery. The malignancy diagnosis rate for MRI-guided VAB was similar to that for stereotactically guided VAB (P = 0.35).
Conclusion
MRI-guided VAB is a safe and accurate procedure that provides insight into clinical breast findings.
Key points
• Three vacuum-assisted breast biopsy (VAB) procedures were compared.
• Technical success rates were high for all three VAB procedures.
• Medical complications were relatively low using all three VAB procedures.
• The use of MRI-guided vacuum-assisted breast biopsy is growing.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Vacuum-assisted biopsy (VAB) devices came onto the market in the mid-1990s with the goal of eliminating the sampling difficulties associated with core needle biopsy. The larger tissue volume resulting from VAB facilitates removal of microcalcifications and is associated with less frequent underdiagnosis of malignancies [1, 2]. VAB is currently recommended for stereotactic and MRI-guided interventions [3, 4]. Ultrasound-guided VAB is not generally indicated, because the less expensive core biopsy procedure frequently achieves the same objective [5]. Ultrasound-guided VAB might be indicated as a therapeutic procedure for histology proven benign lesions in selected situations.
Stereotactically guided VABs have reduced the repeat-biopsy rate (9 % versus 14.9 %) and the percentage of unusable samples (0 % versus 9.5 %) [6]. VAB also leads to a higher diagnostic yield and greater accuracy in MRI-guided procedures [4] and permits the retention of the needle in the breast during cylinder collection, resulting in a lower risk of needle dislocation in comparison to core needle biopsy. Another benefit of VAB is the possibility of direct aspiration of blood in the event of bleeding complications.
Several indications for MRI of the breast, including screening the high-risk population, have been recommended by the American College of Radiology (ACR) [7]. Because of the method’s high sensitivity, MRI can detect suspected breast malignancies that usually escape clinical, ultrasound and mammographic detection.
A result of the wide use of MRI in breast diagnostics is an increase in incidental findings. Because of the high sensitivity and lower specificity of breast MRI, it is imperative that these findings be histologically assessed before any surgical intervention [8–10]. MRI-guided VAB is indicated for MRI findings from patients in BI-RADS categories 4 and 5, as well as BI-RADS 3 in individual cases (e.g. for high-risk patients or after breast-conserving treatment) without correlating findings on mammography or ultrasound [11]. Depending on patient selection, the correlation rate of MRI findings with second-look ultrasound ranges from 23 to 83 % [12–15]. In positive ultrasound correlation of the findings, ultrasound intervention is preferable to MRI-guided intervention because it is simpler, faster and more cost-effective than MRI. However, MRI-guided VAB is a safe, highly accurate method which rarely leads to complications [16].
In Switzerland, as a legal requirement stipulated by the Swiss Federal Office of Public Health and a prerequisite for reimbursement, all data of all minimally invasive breast biopsies (MIBB) have to be entered in a central database
The purpose of our study was to analyse the development of MRI-guided VAB and to compare the procedure with stereotactically guided and ultrasound-guided VAB in Switzerland. The study also examined the technical success rate, the complication rates and the completeness and usability of histological results of MRI-guided VAB compared with the other two procedures.
Materials and methods
Origin of the data
Data from all 53 MIBB centres in Switzerland were collected by the image-guided MIBB Working Group of the Swiss Society of Senology (SGS) [5], on behalf of the Swiss Federal Office of Public Health (Bundesamt für Gesundheit [BAG]). According to the quality guidelines of the MIBB group, the operator for VAB procedures must be a specialist in radiology, gynaecology, obstetrics or surgery. Before being allowed to operate independently, the operator must complete a workshop with successful interventions on a phantom and five supervised interventions when the method is established at their institute or 20 interventions under supervision when the method is newly introduced at their institution. For maintenance of qualifications, each operator must perform at least 12 interventions per year, with a minimum of 20 interventions per institution per year. Using an 11-G needle or bigger depending of the size of the lesion at least 12 samples should be taken [5].
Adjumed Services AG (Zurich, Switzerland) provides an online questionnaire (www.mibb.ch) that has to be completed by the institutions. Accreditation as an MIBB operator and complete data collection are prerequisites for reimbursement by the health insurance companies. Patients were informed before a vacuum biopsy that the collected data would be made available in anonymised form for further data analysis and gave written informed consent [17]. The identities of patients, surgeons and institutions could not be ascertained. The responsible ethics committee approved this data-sharing plan.
Raw data processing
This study was based on 9,113 records of VABs of the breast with biopsies performed in Switzerland between 2009 and 2011. The full data set contains 9,153 records and will be used in a second study focusing on histopathological B3 lesions. In this study we excluded 40 records that lacked information regarding the biopsy method. All mammographically guided biopsies performed using either a stereotactic table with the patient in a prone position (n = 5,340) or in an “upright” device with the patient in a seated position (n = 564) were included in the stereotactically guided VAB method group. A distinction between these two procedural variations was maintained only for the analysis of complications and the chronological development of the biopsy methods. During the analysis of the indications, complications and histological results, multiple answers were converted as often as possible into single answers.
With regard to multiple answers for histological diagnoses, the degree of malignancy was arranged in descending order and the most degenerate diagnosis was retained.
A positive family history was recorded when a patient’s mother, sister or daughter (1st degree of kinship) had a positive history of breast carcinoma.
If there was no information or unclear information provided for the individually analysed data types, this was interpreted as a missing answer and the data were excluded from the respective analysis.
Implementation of image-guided VAB
There are many reports in the literature relating to the technical implementation of image-guided VAB [2, 18]. Figure 1 shows images of MRI-guided vacuum biopsies from a patient who had negative second-look ultrasound.
A 77-year-old female patient with diffuse hardening of the right breast showing infiltration of an invasive lobular, predominantly pleomorphic breast carcinoma. a–d Mammography images showed bilateral benign microcalcifications and slight volume asymmetry on the left side, as well as densification of the interstitial structures on the right side; BI-RADS 3 right side and BI-RADS 2 left side. e, f T1-weighted MRI and post-contrast subtraction (left side): an incidental T1-hypointense (e) stellate lesion (arrows) and on the first subtraction early contrast-enhanced image (f) (BI-RADS 4). g T1 biopsy image. h Histological section showing invasive lobular, predominantly pleomorphic breast carcinoma (haematoxylin and eosin; magnification × 100) (doi: 10.1594/ecr2013/C-0554)
Histological evaluation
Tissue sample preparation was carried out according to the quality standards of the Swiss Society of Pathology (Schweizer Gesellschaft für Pathologie (SGPath)) [19].
Statistical methods
Categorical variables of the groups were compared using the chi-squared test. IBM SPSS Statistics version 20 (SPSS Inc., Chicago, IL, USA) was used for statistical analyses. Values of P < 0.05 were considered statistically significant. The Bonferroni correction was used when single VAB methods were compared. Because MRI-guided VAB was compared with the other two VAB methods (stereotactically guided and ultrasound-guided VAB), a P value of less than 0.025 was considered statistically significant. In the analysis of the complication rates, the three VAB methods (using a stereotactic table, an upright device and ultrasound) were compared to the MRI-guided VAB method (P < 0.017 was considered statistically significant). Sensitivity, specificity, negative (NPV) and positive (PPV) predictive values were presented as percentages with 95 % Wilson confidence intervals (CI).
Results
VAB developments in Switzerland
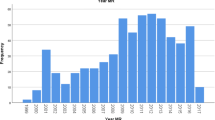
Of the 9,113 VABs, 557 (6 %) were performed using MRI guidance, 5,391 (59 %) using stereotactic guidance (by means of a dedicated stereotactic table), 569 (6 %) using a stereotactic upright device and 2,596 (28 %) using ultrasound guidance. Developments in the use of VAB techniques are shown in Fig. 2.
Developments in image-guided vacuum-assisted biopsy (VAB) in Switzerland between 2009 and 2011. a Total number of VABs performed. b Number of MRI-guided VABs performed. c The number of VABs undertaken using a stereotaxis table. d Number of VABs undertaken using an upright device. e Number of ultrasound-guided VABs. The percentage change in image-guided VAB usage (red lines) is also shown (doi: 10.1594/ecr2013/C-0554)
VAB technical success rates
Seventy six VABs were unsuccessful because of no histology in 69 (MRI 0.9 % [5/557], stereotactically 0.9 % [53/5,960], ultrasound 0.4 % [11/2,596]) and no collection of biopsy material was possible in 7 cases (MRI 0.7 % [4/557], stereotactic 0.05 % [3/5,960], ultrasound 0 % [0/2,596]) leading to an overall success rate of 99.2 % (9,037/9,113). The technical success rate for MRI-guided VAB was 98.4 % (548/557), 99.1 % for stereotactically guided VAB (5,904/5,960) and 99.6 % (2,585/2,596) for ultrasound-guided VAB. There was no significant difference (P = 0.12) in the technical success rates between the MRI-guided procedures and the stereotactic procedures. As expected this rate for ultrasound guidance, with its benefits of real-time imaging, was significantly higher than that for MRI (P < 0.001). The 9,037 successfully implemented VABs formed the basis for further analyses.
Family and carcinoma history
A family history of breast cancer was positive in 2,014 (22 %) cases (Table 1). There was no significant difference in positive family history between the MRI-guided and stereotactically guided VAB procedures (P = 0.23). The occurrence of a positive family history was more frequent in patients who underwent MRI-guided VAB than in those who underwent ultrasound-guided VAB (P < 0.001).
A positive personal history of breast carcinoma was present in a total of 1,227/9,037 (14 %) of the biopsy cases (Table 1).The MRI-guided procedure was used in a significantly higher proportion of these patients than the stereotactically guided (P < 0.001) or ultrasound-guided (P < 0.001) procedures.
Indications
No specific indication was listed in 107/9,037 (1 %) of the cases; this was true in 18/548 of the MRI-guided VAB cases, in 8/5,904 of the stereotactically guided VAB cases and in 81/2,585 of the ultrasound-guided VAB cases.
Seventy nine per cent (421/530) of the findings clarified using MRI-guided VAB were detected by MRI only. The rationale for the intervention was based purely on mammographic findings in 94 % (5,527/5,896) of the stereotactically guided VAB cases and purely on ultrasound findings in 69 % (1,718/2,504) of the ultrasound-guided VAB cases
Clip insertion
In MRI-guided VAB a clip marker was used in 84 % of cases (460/548), in stereotactically guided VAB in 86 % of cases (5,091/5,904) and in ultrasound-guided VAB in 23 % (603/2,585) of cases. There was no significant difference in clip marker usage between the MRI-guided and stereotactically guided VAB procedures, but a significantly higher number of clip markers were used in MRI-guided procedures than in ultrasound-guided procedures (P < 0.001).
Complications
The complication rate of MRI-guided VAB was compared to VAB using stereotaxis table, upright device and ultrasound.
Complications were divided into medical (haemorrhages with or without open revision and infection) and non-medical (technical complications, missed lesions and lesion inaccessibility). No specific complications were reported in 52/9,037 of cases. Terminated intervention was reported in 19/9,037 cases without stating the medical or technical grounds on which the decision was based.
In total, 8,966 records were available for evaluation of the complication rate, and complications were recorded in 6 % (511/8,966). The distribution of the complications is shown in Table 2.
The overall complication rates for the VAB procedure using a stereotaxis table (P = 0.08) or using the upright device (P = 0.02) were not significantly different from that for the MRI-guided procedure. The overall complication rate for ultrasound-guided VAB was significantly lower (P < 0.001) relative to that for MRI-guided VAB.
There was no case of bleeding requiring surgical intervention by MRI-guided VAB.
Histological evaluation
Information on the histological assessment of the vacuum biopsy cylinders was unavailable in 54/9,037 cases. For the entire study population, the material obtained was specified by the pathologist as representative in 97.6 %: samples from MRI-guided, stereotactically guided and ultrasound-guided VABs were representative in 96.6 % (512/530), 97.3 % (5,724/5,884) and 98.5 % (2,530/2,569) of cases, respectively.
Because the histological findings for the VABs were specified as “other” in 175/9,037 (2 %) of cases, a distinction between malignant, “high-risk” or benign histology was impossible and these data were excluded from the malignancy rate analysis.
For the entire study population, VAB revealed malignant lesions in 1,853/8,862 cases (21 %; 37 % invasive carcinoma and 63 % DCIS), benign lesions in 5,524/8,862 cases (62 %) and high-risk lesions in 1,485/8,862 cases (17 %) (Table 3). The rate of malignancy in stereotactically guided VABs was not significantly different from that for MRI-guided VAB (P = 0.35), but the malignancy rate for the MRI-guided procedure was significantly higher than that for the ultrasound-guided procedure (P = 0.001).
Histology of subsequent surgical excisions
Of the 8,862 VAB samples with known histological results, a total of 2,434 (27 %) lesions were further clarified by means of subsequent surgery and histological analysis of the tissue samples obtained. No histological data from the surgery were available in 131 cases. Ninety per cent (1,660/1,853) of the malignant lesions, 5 % (255/5,524) of the benign lesions and 35 % (519/1,485) of the high-risk or B3 lesions were surgically excised.
The rates of upgrade to malignant disease after histological analysis of open surgical excision samples for the various image-guided VAB procedures are listed in Table 4. The number of false negatives was 3 %, 1 % and 0.4 % for the MRI-guided, stereotactically guided and ultrasound-guided VAB procedures, respectively. Benign and high-risk lesions were upgraded at a significantly higher rate after open surgical excision for the MRI-guided procedure than was the case for the stereotactically guided (P < 0.001) and the ultrasound-guided (P < 0.001) procedures.
Surgical specimens of the 1,853 malignant lesions no longer revealed malignancy in 115 cases (6 %) post-VAB; full removal of the lesion during VAB was thus assumed. Absence of malignancy in the surgical specimens occurred in 3 % (4/137) of cases after MRI-guided VAB, in 7 % (99/1,408) after stereotactically guided VAB and in 4 % (12/308) after ultrasound-guided VAB.
Sensitivity of VAB
In the calculation of the sensitivity and specificity, as well as the positive and negative predictive values (Table 5), the assumption of complete removal of the lesion during VAB in the event of missing evidence of malignancy in the surgical specimen was not considered.
Discussion
To our knowledge, the present study is the largest comparative study involving VABs with MRI, stereotactic and ultrasound guidance to date. We have shown that in Switzerland throughout the study period the number of MRI-guided VABs rose significantly by 97 %, whereas the number of the other two image-guided VAB techniques performed remained mostly constant. The increase in the number of MRI-guided vacuum biopsies is not due to any decrease in ultrasound-guided biopsies. An “MRI only” finding was the indication for MRI-guided biopsy in 79 % of the cases and an “ultrasound only” finding was the indication for 69 % of ultrasound-guided biopsies. The increase in MRI-guided VABs can be explained by the increasing use of MRI in breast diagnostics, and the resultant increasing number of findings requiring clarification.
The technical success rate for MRI-guided VAB was slightly lower than that for the stereotactically guided and ultrasound-guided procedures, most likely because more complex technical requirements and longer intervention periods are involved in the use of MRI-guided VAB. The technical success rates for image-guided VABs were comparable with or superior to those previously reported [13, 20–29].
There is a lack of significant difference in family histories between the MRI-guided and stereotactically guided procedures. We would have expected a higher percentage of patients with positive family history for the MRI-guided procedure. However, the lack of difference is probably explained by the fact that MRI, although recommended by the ACR guidelines, has not yet been established in Switzerland as the screening method for high-risk population [7].
Because the indication for diagnostic MRI includes screening for contralateral extension in newly diagnosed breast cancer, determination of the spread of the disease or follow-up examinations [7], patients undergoing MRI-guided biopsy have a higher have a higher rate of previous breast cancer.
The MRI-guided VAB shows, despite higher technical requirements, a comparable complication rate compared to VAB under stereotactic guidance. The majority of complications of MRI-guided VAB were haemorrhages, none requiring surgical intervention. Therefore it is a safe procedure.
The total complication rate of MRI-guided VAB lies within the published range of complication rates (2–14 %) [16, 23, 26–28, 30].
No significant difference in the rates of diagnosis of malignancy between MRI-guided and stereotactically guided procedures was found. The rate of diagnosis of malignancy using ultrasound-guided VAB was significantly lower because of the lack of distinction between diagnostic and therapeutic ultrasound-guided VABs and the generally lower pretest likelihood of malignancy of the BI-RADS 3 lesions usually selected for ultrasound-guided VAB. The rate of malignancy detection achieved using MRI-guided VAB corresponds with those (22–33 %) previously reported [4, 22, 24, 28, 30–32].
The upgrade rate of histologically benign and high-risk lesions to malignancies for the MRI-guided VAB [31] was significantly higher than that for the stereotactically guided group. This leads to a lower sensitivity and NPV of the MRI-guided VAB in comparison to the stereotactically guided procedure, whereby the MRI-guided VAB shows a higher specificity and PPV. Both the specificity and PPV of MRI-guided VAB have been reported to be 100 % [20, 27]. These parameters had lower values in the current study because although complete removal of the lesion by VAB prior surgical excision might be an explanation in the event of missing proof of malignancy in the final specimen, we did not include this option in the calculation. The sensitivity of MRI-guided VAB has been reported to be 92.3–93 % and the NPV 93–96.7 % [20, 27]. It was not possible to verify whether the biopsied lesion correlated with the excised lesion in all cases. Although in most cases (84 %) of MRI-guided VAB the biopsy site was marked by a clip and we expect that the clip was part of the resected tissue, it is conceivable that the histology of the surgical specimen, e.g. multifocal lesions, did not relate to the same lesion as the histology of the VAB specimen; thus, in this respect the analysis is limited. Therefore, especially in MRI-guided VAB, it is necessary to mark the biopsy site for surgical correlation or follow-up with a clip and, in cases of benign results and mismatch with the imaging findings, to discuss the possibility of a repeat biopsy.
Another limiting factor of our study was that the data were recorded by the individual MIBB centres in Switzerland, making errors in data input conceivable. On the other hand, because of the large number of participating centres, a very large amount of data was available for analysis, providing an advantage over other studies with considerably smaller case numbers.
In summary, MRI-guided VAB is a safe, specific and widely accepted, growing procedure. As a result of a slightly lower sensitivity, in cases with benign histology a re-evaluation and potentially a repeat biopsy should be discussed if findings persist or if there is a mismatch between histology and imaging.
References
O'Flynn EA, Wilson AR, Michell MJ (2010) Image-guided breast biopsy: state-of-the-art. Clin Radiol 65:259–270
Vandromme MJ, Umphrey H, Krontiras H (2011) Image-guided methods for biopsy of suspicious breast lesions. J Surg Oncol 103:299–305
Albert US, Altland H, Duda V et al (2008) Summary of the updated stage 3 guideline for early detection of breast cancer in Germany 2008. Rofo 180:455–465
Meeuwis C, Veltman J, van Hall HN et al (2012) MR-guided breast biopsy at 3T: diagnostic yield of large core needle biopsy compared with vacuum-assisted biopsy. Eur Radiol 22:341–349
Köchli OR, Rageth JC, Brun del Re R, Bürge M, Caduff R, Kinkel K, Oehlschlegel C (2009) Bildgesteuerte minimalinvasive Mammaeingriffe: Konsensusstatements für die Schweizerische Gesellschaft für Senologie (SGS) und die Arbeitsgruppe “Bildgesteuerte minimalinvasive Mammaeingriffe”. Senologie 6:181–184
Philpotts LE, Shaheen NA, Carter D, Lange RC, Lee CH (1999) Comparison of rebiopsy rates after stereotactic core needle biopsy of the breast with 11-gauge vacuum suction probe versus 14-gauge needle and automatic gun. AJR Am J Roentgenol 172:683–687
Elizabeth A, Morris M (2008) ACR practice guideline for the performance of contrast-enhanced magnetic resonance imaging (MRI) of the breast. http://www.acr.org/~/media/2A0EB28EB59041E2825179AFB72EF624.pdf. Accessed 23 Aug 2012
Kuhl CK, Schmutzler RK, Leutner CC et al (2000) Breast MR imaging screening in 192 women proved or suspected to be carriers of a breast cancer susceptibility gene: preliminary results. Radiology 215:267–279
Peters NH, Borel Rinkes IH, Zuithoff NP, Mali WP, Moons KG, Peeters PH (2008) Meta-analysis of MR imaging in the diagnosis of breast lesions. Radiology 246:116–124
Sardanelli F, Podo F, D'Agnolo G et al (2007) Multicenter comparative multimodality surveillance of women at genetic-familial high risk for breast cancer (HIBCRIT study): interim results. Radiology 242:698–715
Heywang-Kobrunner SH, Sinnatamby R, Lebeau A et al (2009) Interdisciplinary consensus on the uses and technique of MR-guided vacuum-assisted breast biopsy (VAB): results of a European consensus meeting. Eur J Radiol 72:289–294
Abe H, Schmidt RA, Shah RN et al (2010) MR-directed ("Second-Look") ultrasound examination for breast lesions detected initially on MRI: MR and sonographic findings. AJR Am J Roentgenol 194:370–377
Carbognin G, Girardi V, Brandalise A et al (2011) MR-guided vacuum-assisted breast biopsy in the management of incidental enhancing lesions detected by breast MR imaging. Radiol Med 116:876–885
Demartini WB, Eby PR, Peacock S, Lehman CD (2009) Utility of targeted sonography for breast lesions that were suspicious on MRI. AJR Am J Roentgenol 192:1128–1134
LaTrenta LR, Menell JH, Morris EA, Abramson AF, Dershaw DD, Liberman L (2003) Breast lesions detected with MR imaging: utility and histopathologic importance of identification with US. Radiology 227:856–861
Perlet C, Heywang-Kobrunner SH, Heinig A et al (2006) Magnetic resonance-guided, vacuum-assisted breast biopsy: results from a European multicenter study of 538 lesions. Cancer 106:982–990
Senology Mibb-wgotSSo AUFKLÄRUNGSPROTOKOLL ZUR MRI-GESTEUERTEN MINIMALINVASIVEN BRUSTBIOPSIE (Vakuumbiopsie). http://www.mibb.ch/Documents/consensus_mri_deu- %281 %29.aspx. Accessed 25 Jul 2012
Eby PR, Lehman CD (2008) Magnetic resonance imaging-guided breast interventions. Top Magn Reson Imaging 19:151–162
Dirnhofer ST, Bubendorf L, Lehr H-A, Landau B, Zenklusen H-R (2011) Qualitätsrichtlinien SGPath. SGPath/SSPath. http://www.sgpath.ch/docs/QR_SGPath_DE_2011.pdf. Accessed 9 Feb 2013
Crystal P, Sadaf A, Bukhanov K, McCready D, O'Malley F, Helbich TH (2011) High-risk lesions diagnosed at MRI-guided vacuum-assisted breast biopsy: can underestimation be predicted? Eur Radiol 21:582–589
Fischer U, Schwethelm L, Baum FT, Luftner-Nagel S, Teubner J (2009) Effort, accuracy and histology of MR-guided vacuum biopsy of suspicious breast lesions–retrospective evaluation after 389 interventions. Rofo 181:774–781
Han BK, Schnall MD, Orel SG, Rosen M (2008) Outcome of MRI-guided breast biopsy. AJR Am J Roentgenol 191:1798–1804
Hauth EA, Jaeger HJ, Lubnau J et al (2008) MR-guided vacuum-assisted breast biopsy with a handheld biopsy system: clinical experience and results in postinterventional MR mammography after 24 h. Eur Radiol 18:168–176
Lehman CD, Deperi ER, Peacock S, McDonough MD, Demartini WB, Shook J (2005) Clinical experience with MRI-guided vacuum-assisted breast biopsy. AJR Am J Roentgenol 184:1782–1787
Liberman L, Bracero N, Morris E, Thornton C, Dershaw DD (2005) MRI-guided 9-gauge vacuum-assisted breast biopsy: initial clinical experience. AJR Am J Roentgenol 185:183–193
Mahoney MC (2008) Initial clinical experience with a new MRI vacuum-assisted breast biopsy device. J Magn Reson Imaging 28:900–905
Malhaire C, El Khoury C, Thibault F et al (2010) Vacuum-assisted biopsies under MR guidance: results of 72 procedures. Eur Radiol 20:1554–1562
Orel SG, Rosen M, Mies C, Schnall MD (2006) MR imaging-guided 9-gauge vacuum-assisted core-needle breast biopsy: initial experience. Radiology 238:54–61
Sakamoto N, Tozaki M, Higa K, Abe S, Ozaki S, Fukuma E (2010) False-negative ultrasound-guided vacuum-assisted biopsy of the breast: difference with US-detected and MRI-detected lesions. Breast Cancer 17:110–117
Perretta T, Pistolese CA, Bolacchi F, Cossu E, Fiaschetti V, Simonetti G (2008) MR imaging-guided 10-gauge vacuum-assisted breast biopsy: histological characterisation. Radiol Med 113:830–840
Rauch GM, Dogan BE, Smith TB, Liu P, Yang WT (2012) Outcome analysis of 9-gauge MRI-guided vacuum-assisted core needle breast biopsies. AJR Am J Roentgenol 198:292–299
Tozaki M, Yamashiro N, Sakamoto M, Sakamoto N, Mizuuchi N, Fukuma E (2010) Magnetic resonance-guided vacuum-assisted breast biopsy: results in 100 Japanese women. Jpn J Radiol 28:527–533
Acknowledgments
Parts of the study were included in an EPOS (Electronic Presentation Online System) poster at European Congress of Radiology (ECR) 2013 (doi: 10.1594/ecr2013/C-0554).
This study was performed on behalf of the Minimally Invasive Breast Biopsies Working Group Switzerland.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Imschweiler, T., Haueisen, H., Kampmann, G. et al. MRI-guided vacuum-assisted breast biopsy: comparison with stereotactically guided and ultrasound-guided techniques. Eur Radiol 24, 128–135 (2014). https://doi.org/10.1007/s00330-013-2989-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-013-2989-5