Abstract
The objective of the study was to investigate the association between IL-8 and other biomarkers of endothelial dysfunction (MCP-1, V-CAM, I-CAM) and the disease activity scores in a sample of 54 patients with ankylosing spondylitis (AS) without use of biological agents. Fifty-four AS patients without treatment with anti-TNFs agents between 18 and 80 years old, who met modified New York criteria and at the same time the axial ASAS criteria, were evaluated using an epidemiological questionnaire that included among others clinical data, BASDAI, BASFI, ASQoL, ASDAS and plasma levels of CRP, ESR, MCP-1, IL-8, ICAM-1 and VCAM-1. IL-8 varied in proportion to disease activity rates (BASDAI and ASDAS) p < 0.05, being strongly correlated with the disease activity. The levels of adhesion molecules I-CAM and VCAM, as described in other studies, were positively correlated with predisposing factors for cardiovascular disease. IL-8 has shown to be strongly correlated with clinical markers of disease activity and inflammatory activity and may be an additional variable to the overall assessment of the activity of the AS.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ankylosing spondylitis (AS) is a chronic autoimmune inflammatory disease, typically with axial joint involvement. Its prevalence is higher among young men, Caucasians and HLA-B27 positives [1, 2]. Inflammatory back pain (IBP) associated with morning stiffness is the main symptom of the disease [1, 3]. Although it does not occur in all patients with AS, extra-articular manifestations are responsible for increased morbidity and mortality. High incidence of cardiovascular mortality in patients with AS has stimulated researchers to the study of possible etiologic or risk factors involved [4]. Han et al. [5] found an increased risk of atherosclerosis by 1.5 times in a cohort of patients with AS compared to the controls.
The cardiovascular risk in autoimmune diseases is multifactorial. Traditional cardiovascular risk factors have been reported to be more prevalent in patients with rheumatic diseases as smoking, hypertension, low high-density lipoprotein (HDL)-cholesterol levels, diabetes and obesity. Furthermore, some inflammation factors are being correlated with accelerated atherogenesis. The C-reactive protein is the well-established factor related with the low-grade inflammation of the vessels [6] but reduced plasma levels of lipoprotein-associated phospholipase A2 (Lp-PLA2) and apoprotein A-I also predispose to coronary heart disease [7]. Specifically in AS, the hypertension [8], low levels of HDL-cholesterol and apoA-I [9] are correlated with increased cardiovascular risk.
Currently, the endothelium is considered the main regulator of vascular homeostasis and pathogenesis of atherosclerosis is closely related to systemic inflammation, autoimmunity, chronic diseases and dyslipidemia [10]. Lipoproteins, activated neutrophil, platelets and other pro-inflammatory factors play an important role in the inflammation in autoimmune diseases [11, 12]. Atherosclerosis has been associated with markers of systemic inflammation such as CRP, fibrinogen, and recently, biomarkers such as cytokines, chemokines, adhesion molecules and proteases [13, 14]. Due to biological variability and availability, many of these biomarkers, such as MCP-1, IL-8, V-CAM, I-CAM, have shown contradictory results, which somehow still limits their measurements to clinical research. IL-8, a member of the superfamily of chemokines CXC [15], was found increased in studies approaching the role of cytokines in the atherosclerotic process [16]. Its production is stimulated by the action of TNF alpha, and most of its actions depend on the interaction with neutrophils [17] and monocytes [18]. IL-8 also stimulates proliferation and migration of smooth muscle endothelial cells [19]. MCP-1 (monocyte chemoattractant protein-1) is a molecule belonging to the family of C–C chemokines β, which plays an important role in inflammation because of its power attractor on monocytes. MCP-1 interacts with the CCR2 receptor of these cells stimulating tropism to the inflammatory site [20], and it is secreted by endothelial cells, smooth muscle cells, fibroblasts and immune cells such as T lymphocytes and macrophages located in the stimulus place [21]. The adhesion molecules VCAM-1 and ICAM-1 are stimulated by inflammatory cytokines, expressed in the luminal surface of endothelial cells or soluble in plasma, and mediate the adhesion of various types of leukocytes to the endothelial surface. The objective of this study was to investigate the association between the levels of IL-8 and other biomarkers of endothelial dysfunction such as MCP-1, V-CAM, I-CAM with disease activity scores (BASDAI, ASDAS); the index of quality of life ASQoL, factors related to cardiovascular disease and the functional activity score (BASFI) in a sample of patients with AS.
Materials and methods
Study population
Fifty-four patients with AS followed between December 2009 and February 2010 at the Outpatient Spondylarthritis Clinic of Federal University of Paraná (UFPR) were initially recruited for this study and submitted to an informed consent (IC). Inclusion criteria were age between 18 and 80 years, the presence of AS according to the modified New York criteria [22] and also axial ASAS at the same time [23]. The exclusion criteria were the use of anti-TNF agents or other biologics, signs of acute inflammation or infectious disease, the presence of other concomitant autoimmune diseases, the presence of malignancy, liver dysfunction or diabetes. At the recruitment, patients were categorized according to age, sex, weight, height, smoking, diabetes mellitus (DM), hypertension (HBP), cardiovascular disease, dyslipidemia, cerebrovascular disease, peripheral vascular disease and the use of concomitant medication. No control group was included because the main purpose of the study was to correlate the level of these endothelial markers, mainly IL-8 with specific disease activity scores in AS. All patients were using NSAIDs for more than 1 year, and no one was using methotrexate for axial disease. Two patients were being treated with sulfasalazine, and none of them was taking steroids.
Measures
After the initial consultation, patients who agreed and signed the IC were submitted to the proposed methodology. The assessment of disease activity and functional status were performed by using protocols BASDAI and BASFI. A questionnaire of quality of life in AS (ASQoL) was also applied. After responding to the questionnaires, all of them answered a clinical and epidemiological survey. Then, patients were examined with a specific focus for clinical signs of AS. After the physical examination, two blood samples were collected from each patient. The tubes with blood samples were stored in a refrigerator at −80 °C and were later transported to the laboratory of the Health Sciences Research Center at the Catholic University of Paraná.
Samples of patients serum were separated by immediately centrifugation (1,500g for 10 min), divided into tubes and stored at −80 °C until the analysis. Plasma levels of MCP-1, IL-8, ICAM-1 and VCAM-1 were measured by ELISA method (enzyme-linked immunosorbent assay) using commercial kits with antibodies (R&D Systems, Minneapolis, Minn., USA). The concentrations of chemokines (pg/ml) and adhesion molecules (ng/ml) were calculated with reference to standard curves performed with the corresponding recombinant molecule. The measurement range of the ELISA system was 31.25–2,000 pg/ml for MCP-1 and IL-8 and 0.03–2 ng/ml for ICAM-1 and VCAM-1. When the values were outside the expected range, the samples were previously diluted. The intra-assay coefficients of variation (CV) for MCP-1, IL-8, ICAM and VCAM-1 were 6.0. 8.0. 7.9 and 8.2 %, respectively, and inter-assay CV 6.2, 7.7, 7.7 and 8.1 %, respectively. There were also assayed in the same blood tests CRP, ESR, total cholesterol, HDL-cholesterol, triglycerides and glucose. ASDAS CRP was calculated only after the release of ultra-sensitive CRP result.
Statistical analysis
The results obtained in the study were expressed as averages, median, minimum values, maximum values and standard deviation (quantitative variables) or as frequencies and percentages (qualitative variables). All variables were normally distributed according to Shapiro–Wilk test. To compare the groups regarding quantitative variables, it was considered the nonparametric Mann–Whitney test or the Kruskal–Wallis nonparametric test. To assess the association between two quantitative variables, it was estimated the Spearman correlation coefficient. p values < 0.05 were considered statistically significant. The data were organized into an Excel spreadsheet and analyzed with the computer program Statistica v.8.0.
Results
Fifty-four patients were evaluated (09 women and 45 men). The average age was 43 ± 11.6 years. Forty-six patients were regularly taking NSAIDs, and none was using anti-TNF. The sample general characteristics are presented in Table 1. The measurements of the 4 biomarkers were correlated to the indexes of disease activity such as CRP, VAS, ASDAS and BASDAI. There were also correlated with factors related to cardiovascular disease such as HBP, systolic blood pressure (SBP), diastolic blood pressure (DBP), body mass index (BMI), waist circumference (WC), total cholesterol (TC), DM, dyslipidemia, history of coronary artery disease (CAD Hx), smoking, alcohol consumption, as well as the functional activity BASFI score.
We observed that the rate of MCP-1 showed an inverse correlation to the measurement of CRP (Spearman correlation coefficients of −0.33 with p = 0.018) and factors related to cardiovascular disease, such as HBP (p = 0.01) and CAD Hx (p = 0.058).
V-CAM was increased in hypertensive patients, but not correlated with CRP or BASDAI (Table 2).
I-CAM was positively correlated with the presence of factors such as HBP, DBP, DBP, dyslipidemia, smoking and CAD Hx, but not with disease activity.
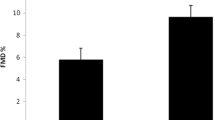
IL-8 varied proportionally with two indices of disease activity analyzed, BASDAI and ASDAS (Table 3).
Discussion
No control group was included in this study. The main purpose was to correlate the level of these endothelial markers, mainly IL-8 with disease activity in a sample of patients with AS without use of anti-TNFs agents.
Our selected sample did not include patients using anti-TNFs in order to facilitate the analysis of the criteria for inflammatory activity, although most of them were in regular use of NSAIDs. There is considerable evidence that the biomarkers evaluated in our study are very well correlated with endothelial dysfunction in case controlled studies in AS and other rheumatic diseases as well. The link between high disease activity and the onset of cardiovascular disease in patients with ankylosing spondylitis is under investigation.
While employed as a marker of changes and activity in the endothelium, IL-8 has been presented with conflicting and inconclusive results in the medical literature. While some studies show correlation between patients with angina and increased plasma levels of IL-8 [19], or even high levels of IL-8 in patients who later came to coronary artery disease [20], other studies suggest a protective effect of IL-8, delivering little attraction and little migration of neutrophils to the chemotactic site of atherosclerotic plaques via modulation by VCAM-1 [24].
IL-8 has also been studied in patients with AS, and its levels seem to be elevated when compared with controls, but it has not been compared with specific AS disease activity scores [25, 26].
In this study, IL-8 did not correlate with factors related to cardiovascular disease, SBP, DBP, BMI and TC (descriptive levels: 0.531, 0.585, 0.860, 0.463 and 0.934). IL-8 also did not correlate with BASFI functional activity score (p = 0.403). However, levels of IL-8 were positively correlated and statistically significant to ASDAS and BASDAI (p value = 0.015) and curiously not statistically significant related to CRP (0.728) alone, VAS back pain (0.539) or ASQol (0,221) (Table 2). Based on our results, we consider that IL-8 may be an additional useful marker of disease activity although it has not had a correlation with acute inflammatory activity when measured only by CRP.
Hwang et al. [27] demonstrated that ICAM-1 and E-selectin are potent biomarkers of atherosclerosis and cardiovascular damage, yet not so strong correlation was found with VCAM-1. A meta-analysis conducted by Malik et al. [28] demonstrated a significant association between levels of ICAM-1 and VCAM-1 and traditional risk factors for coronary disease. Corroborating with the same idea, the prospective study of Haim et al. [29] showed significant association between the concentration of soluble ICAM-1 and increased risk for future coronary events, but did not take into account several other factors. Thus, although it was not our primary objective, our results also reinforce what has been described in the literature, that is, the unquestionable quality of these molecules, particularly ICAM-1, as early markers of cardiovascular lesions.
In this study, we have shown that VCAM-1 is a good marker for HBP, while ICAM-1 is a good marker for DBP, smoking, HBP, dyslipidemia and CAD Hx but do not have association with specific disease activity scores of AS. Finally and the most important, IL-8 proved to be a good marker for disease activity, since it is associated only with BASDAI and ASDAS.
Conclusion
Our results should be interpreted cautiously, all the observations are cross-sectional and do not provide evidence of causality or directionality. Some limitations include the small sample size and the lack of analyses between medications used in the treatment and IL-8. On the other hand, we believe that the absence of a control group does not jeopardize our results. At the same time, a single temporary measure of four biomarkers and of three criteria for disease activity was performed to determine correlations between variables. The results of this study demonstrated that IL-8 can be used as a marker of inflammatory activity due to its statistically significant correlation with BASDAI and ASDAS.
References
Dougados M (1995) Diagnostic features of ankylosing spondylitis. Br J Rheumatol 34:301–305
Van Der Linden S, Van Der Heijde D (1998) Ankylosing spondylitis: clinical features. Rheum Dis Clin North Am 24(4):663–676
Sampaio-Barros P, Azevedo VF, Bonfiglioli R, Campos WR, Carneiro SCS, Carvalho MAP et al (2007) Consenso brasileiro de espondiloartropatias: espondilite anquilosante e artrite psoriásica diagnóstico e tratamento—primeira revisão. Rev Bras Reumatol 47(4):233–242
Peters MJ, van der Horst-Bruinsma IE et al (2004) Cardiovascular risk profile of patients with spondylarthropathies, particularly ankylosing spondylitis and psoriatic arthritis. Semin Arthritis Rheum 34:585–592
Han C, Robinson DW Jr, Hackett MV, Paramore LC, Fraeman KH, Bala MV (2006) Cardiovascular disease and risk factors in patients with rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis. J Rheumatol 33(11):2167–2172
Gasparyan AY, Stavropoulos-Kalinoglou A, Mikhailidis DP, Toms TE, Douglas KM, Kitas GD (2010) The rationale for comparative studies of accelerated atherosclerosis in rheumatic diseases. Curr Vasc Pharmacol 8(4):437–449
Onat A, Direskeneli H (2012) Excess cardiovascular risk in inflammatory rheumatic diseases: pathophysiology and targeted therapy. Curr Pharm Des 18(11):1465–1477
Kang JH, Chen YH, Lin HC (2010) Comorbidity profiles among patients with ankylosing spondylitis: a nationwide population-based study. Ann Rheum Dis 69(6):1165–1168 (Epub 2010 Apr 7)
Joven J, Rubiés-Prat J, Ras MR, de la Figuera M, Lience E, Masdeu S (1984) High density lipoprotein cholesterol subfractions and apoprotein A-I in patients with rheumatoid arthritis and ankylosing spondylitis. Arthritis Rheum 27(10):1199–1200
Azevedo VF, Pecoits-Filho R (2010) Atherosclerosis and endothelial dysfunction in patients with ankylosing spondylitis. Rheumatol Int 30(11):1411–1416
Wright HL, Moots RJ, Bucknall RC, Edwards SW (2010) Neutrophil function in inflammation and inflammatory diseases. Rheumatology (Oxford) 49(9):1618–1631 (Epub 2010 Mar 24)
Gasparyan AY, Stavropoulos-Kalinoglou A, Mikhailidis DP, Douglas KM, Kitas GD (2011) Platelet function in rheumatoid arthritis: arthritic and cardiovascular implications. Rheumatol Int 31(2):153–164
Maksimowicz-McKinnon K, Bhatt DL, Calabrese LH (2004) Recent advances in vascular inflammation: C-reactive protein and other inflammatory biomarkers. Curr Opin Rheumatol 16(1):18–24
Hahn BH, Grossman J, Chen W, McMahon M (2007) The pathogenesis of atherosclerosis in autoimmune rheumatic diseases: roles of inflammation and dyslipidemia. J Autoimmun 28(2–3):69–75
Yue TL, Wang X, Sung CP, Olson B, McKenna PJ, Gu JL, Feuerstein GZ (1994) Interleukin-8. A mitogen and chemoattractant for vascular smooth muscle cells. Circ Res 75(1):1–7
Apostolakis S, Papadakis EG, Krambovitis E, Spandidos DA (2006) Chemokines in vascular pathology. Int J Mol Med 17:691–701
Baggiolini M, Clark-Lewis I (1992) Interleukin-8, a chemotactic and inflammatory cytokine. FEBS Lett 307(1):97–101
Gerszten RE, Garcia-Zepeda EA, Lim YC, Yoshida M, Ding HA, Gimbrone MA et al (1999) MCP-1 and IL-8 trigger firm adhesion of monocytes to vascular endothelium under flow conditions. Nature 398:718–723
Yue TL, Wang X, Sung CP, Olson B, McKenna PJ, Gu JL et al (1994) Interleukin-8. A mitogen and chemoattractant for vascular smooth muscle cells. Circ Res 75:1–7
O’Hayre M, Salanga CL, Handel TM, Allen SJ (2008) Chemokines and cancer: migration, intracellular signalling and intercellular communication in the microenvironment. Biochem J 409:635–649
Conti P, DiGioacchino M (2001) MCP-1 and RANTES are mediators of acute and chronic inflammation. Allergy Asthma Proc 22:133–137
van der Linden S, Valkenburg HA, Cats A (1984) Evaluation of diagnostic criteria for ankylosing spondylitis. A proposal for modification of the New York criteria. Arthritis Rheum 27(4):361–368
Rudwaleit M, van der Heijde D, Landewé R, Listing J, Akkoc N, Brandt J, Braun J, Chou CT, Collantes-Estevez E, Dougados M, Huang F, Gu J, Khan MA, Kirazli Y, Maksymowych WP, Mielants H, Sørensen IJ, Ozgocmen S, Roussou E, Valle-Oñate R, Weber U, Wei J, Sieper J (2009) The development of Assessment of Spondylarthritis international Society classification criteria for axial spondylarthritis (part II): validation and final selection. Ann Rheum Dis 68(6):777–783 (Epub 2009 Mar 17)
Taylor PC, PetersAM Paleolog E, ChapmanPT Elliott MJ, McCloskey R et al (2000) Reduction of chemokine levels and leukocyte traffic to joints by tumor necrosis factor alpha blockade in patients with rheumatoid arthritis. Arthrit Rheum 43:38–47
Limón-Camacho L, Vargas-Rojas MI, Vázquez-Mellado J, Casasola-Vargas J, Moctezuma JF, Burgos-Vargas R, Llorente L (2012) In vivo peripheral blood proinflammatory T cells in patients with ankylosing spondylitis. J Rheumatol 39(4):830–835
Azuz-Lieberman N et al (2005) The involvement of NK cells in ankylosing spondylitis. Int Immunol 17(7):837–845
Hwang SJ, Ballantyne CM, Sharrett AR, Smith LC, Davis CE, Gotto AM Jr, Boerwinkle E (1997) Circulating adhesion molecules VCAM-1, ICAM-1, and E-selectin in carotid atherosclerosis and incident coronary heart disease cases: the atherosclerosis risk in communities (ARIC) study. Circulation 96:4219–4225
Malik I, Danesh J, Whincup P, Bhatia V, Papacosta O, Walker M, Lennon L et al (2001) Soluble adhesion molecules and prediction of coronary heart disease: a prospective study and meta-analysis. Lancet 358:971–976
Haim M, Tanne D, Boyko V, Reshef T, Goldbourt U, Leor J, Mekori YA et al (2002) Soluble intercellular adhesion molecule-1 and long-term risk of acute coronary events in patients with chronic coronary heart disease. Data from the bezafibrate infarction prevention (BIP) Study. J Am Coll Cardiol 39:1133–1138
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Azevedo, V.F., Faria-Neto, J.R., Stinghen, A. et al. IL-8 but not other biomarkers of endothelial damage is associated with disease activity in patients with ankylosing spondylitis without treatment with anti-TNF agents. Rheumatol Int 33, 1779–1783 (2013). https://doi.org/10.1007/s00296-012-2631-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-012-2631-x