Abstract
Rheumatoid arthritis (RA) is a chronic autoimmune disease characterized by the influxation of synovia and synovial compartments with immune cells including dendritic cells (DCs). DCs that induce autoimmune tolerance are called tolerogenic DCs (tolDCs). As a promising immunotherapeutic strategy for RA, tolDCs have received increasing attention. In this review, we first introduce the significant role of tolDCs in autoimmune regulation and then describe the manipulation strategies to generate tolDCs; next, we summarize recent progress in the experimental application of tolDCs for RA therapy, and finally we discuss the perspectives of tolerogenic vaccination for the treatment for RA in clinic.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rheumatoid arthritis (RA) is a chronic autoimmune disease characterized by the inflation of synovia and synovial compartments with dendritic cells (DCs), monocytes, T cells, B cells, neutrophils and natural killer (NK) cells [1]. RA affects multiple joints such as hands, wrists and feet and is one important cause of disability.
RA is currently treated with immunosuppressive drugs and biological agents. However, these therapeutic agents may induce a generalized immune suppression that increases the risk of infectious diseases [2]. Therefore, new therapeutic approaches should aim at the suppression of inflammation and establishment of tolerance toward arthritogenic antigens without compromising the patients’ immune system [3]. Recent research has shown that a particular subset of DCs could modulate immune responses [4]. These DCs play a key role in maintaining both central and peripheral autoimmune tolerance, and the constitutive ablation of DCs destroys self-tolerance, resulting in spontaneous fatal autoimmunity [5]. The DCs that induce autoimmune tolerance are called tolerogenic DCs (tolDCs). As a promising immunotherapeutic strategy for RA, tolDCs have received considerable attention [6]. In this review, we first introduce the significant role of tolDCs in autoimmune regulation and then describe the strategies to generate tolDCs; next, we summarize recent progress in the experimental application of tolDCs for RA therapy, and finally we discuss the perspectives of tolerogenic vaccination for the treatment for RA in clinic.
Features of DCs associated with tolDCs: subsets and maturation stage
TolDCs are derived from DCs which exhibit tolerogenic phenotype. It has been found that the subsets and maturation stage of DCs are closely related to the generation of tolDCs. In this part, we will introduce several features of DCs that are associated with tolDCs.
Subset of DCs and tolDCs
DCs constitute a heterogeneous population of cells characterized by the differences in tissue distribution, phenotype and function [7]. Human peripheral blood DCs can be divided into two major subsets according to the source they derive from: myeloid DCs (mDCs) and plasmacytoid DCs (pDCs) [8]. In addition to these two subsets, there is another type of DCs that is derived from monocyte and plays an important role in innate and adaptive immunity and is named monocyte-derived DCs (mo-DCs) [9]. Detailed information on these subsets of DCs and their applications in RA has been described in several reviews [10].
Mature mDCs can induce the differentiation of naïve T cells into T helper (Th) cells with increased expression of adhesion molecules and cytokine receptors as well as the production of cytokines, which can activate autoimmune responses [11]. On the contrary, immature mDCs have been commonly loaded with antigen and manipulated to generate tolDCs and suppress autoimmune responses both in vivo and in vitro [12]. TolDCs derived from mDCs have been widely used in the therapy of RA and experimental arthritis [13].
pDCs represent a naturally occurring regulatory DC subset with tolerogenic phenotype. Under certain circumstances, pDCs appear to induce the differentiation of regulatory T cells (Tregs) both in mice models and in human [14]. Thus, the use of pDCs to induce immune tolerance may offer new opportunities in autoimmunity and transplantation [15]. Recent studies have demonstrated that pDCs exhibited tolerogenic phenotype and modulated anti-inflammatory function in RA patients through IDO pathway [16].
mo-DCs are by far the most common type of cells used in clinical immunotherapeutics [17]. Data based on the use of mo-DCs have shown that mo-DCs can be manipulated with certain biological agents to generate tolDCs [18]. Moreover, tolDCs derived from mo-DCs are proposed as a promising cellular therapeutic tool for tailoring immunomodulation in the treatment for RA [19].
Maturation stage and tolDCs
Immature DCs do not express maturation markers nor produce proinflammatory cytokines. The natural function of immature DCs is to create conditions for self-tolerance either via the generation of Treg or via the induction of apoptosis or anergy of autoreactive effector cells [20]. During the past years, immature DCs have been widely used to generate tolDCs and utilized in the treatment for autoimmune diseases including RA [21]. Strikingly, while mature DCs are considered immunogenic as professional antigen-presenting cells, recent evidence suggested that tolDCs can be generated from mature DCs by genetic engineering [22], which provides additional means of generating tolDCs for RA treatment.
Interestingly, an independent subgroup of DCs has been recognized as semi-mature DCs, which express maturation markers but do not produce inflammatory cytokines, and they appear to be tolerogenic in autoimmune diseases including RA [23]. However, other reports suggest that semi-mature DCs become immunogenic when inoculated at a high dose in CIA mice [24]. Therefore, further characterization of the role of semi-mature DCs in RA pathogenesis is required.
Generation of tolDCs: strategies
Manipulations of DCs to generate tolerogenic phenotype have been extensively studied and reported. In this review, we classify the commonly used strategies into three groups: immunoregulatory drugs and biological agents, coculture with apoptotic cells, and genetic engineering. Most of the strategies have been practiced in studies aiming at RA therapy, although several of them are practiced in other autoimmune diseases.
Regulation of molecular targets on DCs
During the past years, various biological agents and pharmacological agents have been used to confer tolerogenic properties on DCs and regarded as a clinically applicable option [25]. On the basis of recent data, we will introduce the molecules that serve as targets for tolerogenic phenotype of DCs with pharmacological drugs and other agents.
RelB component of NF-κB has been shown to be critical for DCs maturation in vivo [26]. Tumor necrosis factor-α (TNF-α) is another molecular target on DCs because anti-TNF-α therapies diminish DCs maturation and their ability to produce proinflammatory cytokines and chemokines and are effective in treating patients with RA [27].
On the contrary, induction or upregulation of other molecules could be employed to generate tolDCs. PD-L1 signaling has been shown to negatively regulate T-cell response and contribute to tolerogenic phenotype of these DCs [28]. The immunoglobulin-like transcript (ILT) family consists of a group of activating and inhibitory receptors, and some of them play a role in tolerance induction [29]. The upregulation of ILT3 and ILT4 receptors on human DCs renders them tolerogenic, along with reduced expression of costimulatory molecules and induction of antigen-specific unresponsiveness in CD4+ T cells. IDO is an immunosuppressive protein expressed on DCs, and upregulation of IDO expression on DCs also makes them tolerogenic [30]. In addition, upregulation of ICOSL, a molecule that induces anergy of T cells, could induce tolerogenic phenotype on immature DCs [31].
Uptake of apoptotic cells
Apoptotic cells induce tolerogenic properties of innate immune cells including DCs, which then recognize and phagocytose the apoptotic cells [32]. DCs produce IL-10 after ingestion of apoptotic cells and induce T-cell tolerance via immunosuppressive cytokines [32]. Immature DCs was rendered tolerogenic in vitro by pre-exposure to autologous apoptotic cells [33]. Recently, it has been found that the culture of apoptotic DCs with immature DCs in vitro results in their uptake by immature DCs, which subsequently turn into tolDCs [34]. In vivo study in mice models also demonstrated that apoptotic DCs can be taken up by viable DCs, which suppress the ability of viable DCs to undergo maturation and subsequent migration to the lymph nodes [35]. Therefore, apoptotic cells are a promising agent to induce tolDCs, which could possibly be used for therapy for RA in the future.
Genetic engineering strategy
Recently, genetic engineering was practiced as a novel strategy to induce tolDCs regardless of their maturation stages [36]. Moreover, applications aimed at treatment for RA have been practiced in experimental murine models. Both knockdown of costimulatory factors such as CD40, CD80 and CD86 and expression of immunosuppressive molecules in DCs have been exploited to generate tolDCs, which effectively suppressed the onset of collagen-induced arthritis [37].
Possible mechanisms of tolDCs function in RA therapy
Past studies have shown that tolDCs induce immunotolerance through a variety of mechanisms that have been extensively investigated, and the readers are referred to recent reviews and articles [28, 31, 38]. On the basis of literature published recently, here we highlight the potential mechanisms of tolDCs function in RA therapy.
Reduction in Th17
Th17 is a new and unique subset of T cells that plays a critical role in host defense against certain extracellular pathogens and also contributes to the pathogenesis of various autoimmune diseases including RA [39–43]. Notably, Th17 produces inflammatory cytokines such as IL-17 and is a subset of osteoclastogenic Th cells, which was demonstrated to induce tissue destruction in RA [44–46].
IL-23 is essential for the expansion of Th17 cells, and IL-23 receptor is expressed on DCs [47]. Moreover, DCs that are tolerogenic demonstrated reduction in IL-23, which gives rise to lower number of Th17 [48]. Recently, studies have shown that tolDCs led to the reduction in Th17 responses in experimental RA models, which is a potential mechanism by which tolDCs help treat RA [49].
Anergy and apoptosis in effector T cells
As an autoimmune disease, RA is partially characterized by the excessive activation and infiltration of T cells toward synovium and synovial compartments [1]. DCs induce naive T cells to differentiate into T helper cells in process of RA, which results in inflammation and bone destruction [2]. Therefore, suppression of mature DC-induced T-cell differentiation and activation is a crucial pathway for the therapy for RA with tolDCs. It has been reported that tolDCs could lead to incomplete signaling to T cells through inhibition of IL-12 production and generation of TGF-β, which induce alloantigen-specific T-cell hyporesponsiveness, anergy or apoptosis in vitro and suppress immune reactivity [50]. During the past years, studies have shown that tolDCs suppress effector T-cell response via upregulation of molecules including IDO, FasL, PD-L1 and CTLA-4 Ig that are associated with immunoregulation and apoptosis and downregulation of self-peptide-MHC complex in couple with limited costimulatory molecules (especially CD86) [51]. These mechanisms have been recapitulated in experimental model of RA and arthritis and provide support for therapy of RA with tolDCs [13].
TolDCs suppress the activation of memory T cells
RA is characterized by the accumulation of CD4(+) T cells in the inflamed synovium, and most of them are CD4+ memory T cells. Recent evidence suggests that steady-state immature DCs, which constitutively present an endogenously expressed antigen, can inactivate fully differentiated memory CD8+ T cells in vivo through deletion and inactivation [52]. Moreover, studies have shown that tolDCs that are generated with different immunosuppressive agents and cytokines can induce antigen-specific anergy and regulatory properties in CD4+ memory T cells [48, 53]. Recently, it is reported that tolDCs can regulate CD4+ memory T-cell differentiation. Xu X et al. found that coculture of tolDCs with CD4+ T cells results in the secretion of Th2 cytokines (IL-4, IL-5, IL-10 and IL-13) and negative immune regulation by memory T cells [54]. Taken together, these studies suggest that inhibition of the activation of memory T cells is another possible mechanism for tolDCs function in RA treatment.
Switch of Th1/Th2 balance to Th2 cells selectively
RA is characterized by a marked shift toward the Th1 phenotype, which is described as proinflammatory, with overproduction of IFN and inadequate production of Th2 cytokines such as IL-4 and IL-13 [55]. The immune deviation (skewing of T cells toward the Th2 type) and the role of Th2 cytokines in immune tolerance have been discussed [56]). Th2 cells induce processes involved in cartilage repair, including collagen synthesis [57]. Studies have shown that therapies aimed at switch of Th1/Th2 balance attenuated RA and CIA. Accumulating evidence has shown that prophylactic treatment with tolDCs is associated with a reduced collagen II-specific IgG2a/IgG1 ratio, indicating a switch from a Th1- toward a Th2-driven collagen II-specific immune response, which inhibits CIA in experimental arthritis models [3, 58].
Effect of tolDCs on regulatory T cells (Treg)
It has been well established that Foxp3+ Treg cells could lead to the suppression of autoimmune response. Interestingly, recent data showed that induction of Treg cells facilitates the restoration of immune tolerance in RA [59]. The potential role of Treg cells in RA was reviewed very recently [60].
Tregs can be induced by repeated stimulation with allogeneic immature human or mouse mDCs. In addition, pDCs express tolerogenic phenotype and induce Treg cell through the expression of IDO [61–64]. In vivo study further showed that selective ablation of DCs led to the loss of FoxP3-expressing Treg and the development of proinflammatory autoreactive T effectors, resulting in excessive autoimmunity [65]. Recent studies have shown that tolDCs can attenuate RA and experimental arthritis through induction of Tregs [66], which represents another promising mechanism for treatment for RA with tolDCs.
Treatment for RA with tolDCs: current situation and perspectives
Current situation of tolDCs for RA
Up to now, the therapeutic application of tolDCs is practiced mostly in animal models of RA. The development of animal models in which DCs are selectively depleted will help characterize the specific role of these cells in the pathogenesis of RA. Studies have shown that repetitive injection of immature DCs or DCs, which were modulated with TNF, IL-10, dexamethasone or LF 15-0195, could induce tolerance to autoimmunity and result in the amelioration of inflammation and destruction in experimental RA models. Furthermore, DCs transduced with Fas ligand or IL-4 could prevent CIA and inhibit arthritic symptoms in mice with established disease [3, 67, 68]. In addition, tolDCs modified by other drugs or cytokines have been used successfully to prevent the onset of CIA and alleviate established arthritis in the antigen-induced arthritis model [69].
TolDCs induced via drugs or other agents have also been evaluated for therapeutic effects in RA patients. Anti-TNF therapy has been proven to be effective in treating patients with RA clinically, which ameliorates clinical symptoms partially through induction of tolDCs in vivo. Moreover, analysis of the immunophenotypes of circulating DCs in RA patients before and after treatment with infliximab demonstrated that tolerogenic phenotype of DCs is closely correlated with clinical outcome of RA [70].
Perspectives
The utilization of tolDCs as a promising cellular vaccine for tumor, infection and some autoimmune diseases has been well described in the literature [71–75]. Fortunately, the application of tolDCs as potential vaccine for the clinical treatment for RA is getting more attention [22, 76, 77].
TolDCs for immunotherapy must be safe, standardized and controlled [78, 79]. Recently, it was reported that tolDCs generated in one study exhibited high-level expression of a certain receptor TLR-2, which is an appropriate quantity control marker [19] for safety purposes. This is encouraging for the therapeutic use of tolDC vaccines in RA clinically. In conclusion, we can see that although there are still challenges for its clinical use, tolDC-based vaccination has a promising future to treat RA safely, conveniently and effectively (Fig. 1).
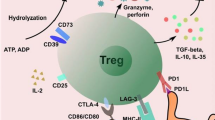
Manipulation strategies to generate tolDCs and potential mechanisms underlying tolDC-induced autoimmune tolerance. Various cytokines (such as ILs, TNF and TGF-β), and immunoregulatory molecules (such as IDO, PD-L1, ICOSL and ILTs) participate in all this process during which tolDCs induce special differentiation of naïve T cells, which cause tolerance of autoimmunity and protect and improve RA. IL interleukin, MHC major histocompatibility complex, TGF-β transforming growth factor-β, Th helper T cells, PD-L1 programmed death 1 ligand, DC dendritic cell, lIDO indoleamine 2,3-dioxygenase, RA rheumatoid arthritis; NF-kβ nuclear factor-kappa B; Treg regulatory T cell; ILT immunoglobulin-like transcript, ICOSL inducible costimulator ligand.  Prevention/inhibition;
Prevention/inhibition;  upregulation;
upregulation;  downregulation. BOX-1 (1) Genetic engineering as a novel tool for the generation of tolDCs by suppressing expression of costimulatory molecules (CD40, CD80 and CD86) and promoting expression of immunosuppressive proteins (e.g. IDO). (2) Uptake of ACs leads to ligation of individual receptors on human DCs that recognize apoptotic cells (such as CR3 and CR4), during which process DCs get tolerogenic phenotype to inhibit IL-12 production, generate transforming growth factor β (TGF-β) and produce IL-10. (3) Drugs and biological agents that target inhibition of costimulatory molecules such as MHC, CD40, CD80 and CD86, and upregulation of immunosuppressive molecules including IDO, PD-L1, ILTs and ICOSL can induce tolerogenic phenotype of DCs. BOX-2 (1) Naive T cells can differentiate into Th17, which produces inflammatory cytokines and results in tissue destruction in RA. TolDCs lead to the reduction in IL-23, which is essential for the expansion of Th17 cells. (2) Effector T cells comprise main part of autoimmune reactions in RA. TolDCs could inhibit IL-12 production, generate TGF-β and induce hyporesponsiveness, anergy or apoptosis of effector T cells. (3) CD4+ memory T cells accumulate in RA synovium and take part in T-cell inflammatory response. TolDCs can produce cytokines (e.g. IL-10), which induce antigen-specific anergy in CD4+ memory T cells. (4) RA is characterized by a marked shift toward the Th1 phenotype, which is proinflammatory, with overproduction of IFN. TolDCs lead to a switch from Th1 to Th2 response with more Th2 cytokines (e.g. IL-4, IL-13) and result in autoimmune tolerance. (5) Tregs have been proved to attenuate RA via secretion of cytokines such as IL-25. TolDCs can express IDO and produce TGF-β and IL-10, which facilitate Foxp3+ Tregs
downregulation. BOX-1 (1) Genetic engineering as a novel tool for the generation of tolDCs by suppressing expression of costimulatory molecules (CD40, CD80 and CD86) and promoting expression of immunosuppressive proteins (e.g. IDO). (2) Uptake of ACs leads to ligation of individual receptors on human DCs that recognize apoptotic cells (such as CR3 and CR4), during which process DCs get tolerogenic phenotype to inhibit IL-12 production, generate transforming growth factor β (TGF-β) and produce IL-10. (3) Drugs and biological agents that target inhibition of costimulatory molecules such as MHC, CD40, CD80 and CD86, and upregulation of immunosuppressive molecules including IDO, PD-L1, ILTs and ICOSL can induce tolerogenic phenotype of DCs. BOX-2 (1) Naive T cells can differentiate into Th17, which produces inflammatory cytokines and results in tissue destruction in RA. TolDCs lead to the reduction in IL-23, which is essential for the expansion of Th17 cells. (2) Effector T cells comprise main part of autoimmune reactions in RA. TolDCs could inhibit IL-12 production, generate TGF-β and induce hyporesponsiveness, anergy or apoptosis of effector T cells. (3) CD4+ memory T cells accumulate in RA synovium and take part in T-cell inflammatory response. TolDCs can produce cytokines (e.g. IL-10), which induce antigen-specific anergy in CD4+ memory T cells. (4) RA is characterized by a marked shift toward the Th1 phenotype, which is proinflammatory, with overproduction of IFN. TolDCs lead to a switch from Th1 to Th2 response with more Th2 cytokines (e.g. IL-4, IL-13) and result in autoimmune tolerance. (5) Tregs have been proved to attenuate RA via secretion of cytokines such as IL-25. TolDCs can express IDO and produce TGF-β and IL-10, which facilitate Foxp3+ Tregs
References
Chen M, Wang YH, Wang Y, Huang L, Sandoval H, Liu YJ, Wang J (2006) Dendritic cell apoptosis in the maintenance of immune tolerance. Science 311(5764):1160–1164. doi:10.1126/science.1122545
Imperato AK, Bingham CO III, Abramson SB (2004) Overview of benefit/risk of biological agents. Clin Exp Rheumatol 22(5 Suppl 35):S108–S114
Flores-Borja F, Mauri C, Ehrenstein MR (2008) Restoring the balance: harnessing regulatory T cells for therapy in rheumatoid arthritis. Eur J Immunol 38(4):934–937. doi:10.1002/eji.200738107
Rutella S, Danese S, Leone G (2006) Tolerogenic dendritic cells: cytokine modulation comes of age. Blood 108(5):1435–1440. doi:10.1182/blood-2006-03-006403
Rescigno M (2010) Dendritic cells in tolerance induction for the treatment of autoimmune diseases. Eur J Immunol 40(8):2119–2123. doi:10.1002/eji.201040474
Khan S, Greenberg JD, Bhardwaj N (2009) Dendritic cells as targets for therapy in rheumatoid arthritis. Nat Rev Rheumatol 5(10):566–571. doi:10.1038/nrrheum.2009.185
Piccioli D, Tavarini S, Borgogni E, Steri V, Nuti S, Sammicheli C, Bardelli M, Montagna D, Locatelli F, Wack A (2007) Functional specialization of human circulating CD16 and CD1c myeloid dendritic-cell subsets. Blood 109(12):5371–5379. doi:10.1182/blood-2006-08-038422
Jongbloed SL, Lebre MC, Fraser AR, Gracie JA, Sturrock RD, Tak PP, McInnes IB (2006) Enumeration and phenotypical analysis of distinct dendritic cell subsets in psoriatic arthritis and rheumatoid arthritis. Arthritis Res Ther 8(1):R15. doi:10.1186/ar1864
Geissmann F, Manz MG, Jung S, Sieweke MH, Merad M, Ley K (2010) Development of monocytes, macrophages, and dendritic cells. Science 327(5966):656–661. doi:10.1126/science.1178331
Lebre MC, Tak PP (2009) Dendritic cells in rheumatoid arthritis: which subset should be used as a tool to induce tolerance? Hum Immunol 70(5):321–324. doi:10.1016/j.humimm.2009.02.006
Banchereau J, Steinman RM (1998) Dendritic cells and the control of immunity. Nature 392(6673):245–252. doi:10.1038/32588
Falcon C, Carranza F, Martinez FF, Knubel CP, Masih DT, Motran CC, Cervi L (2010) Excretory-secretory products (ESP) from Fasciola hepatica induce tolerogenic properties in myeloid dendritic cells. Vet Immunol Immunopathol 137(1–2):36–46. doi:10.1016/j.vetimm.2010.04.007
Arce F, Breckpot K, Stephenson H, Karwacz K, Ehrenstein MR, Collins M, Escors D (2010) Selective ERK activation suppresses inflammatory arthritis via tolerogenic dendritic cells and antigen-specific regulatory T cells. Arthritis Rheum. doi:10.1002/art.30099
Wei S, Kryczek I, Zou L, Daniel B, Cheng P, Mottram P, Curiel T, Lange A, Zou W (2005) Plasmacytoid dendritic cells induce CD8+ regulatory T cells in human ovarian carcinoma. Cancer Res 65(12):5020–5026. doi:10.1158/0008-5472.CAN-04-4043
Tang Q, Henriksen KJ, Bi M, Finger EB, Szot G, Ye J, Masteller EL, McDevitt H, Bonyhadi M, Bluestone JA (2004) In vitro-expanded antigen-specific regulatory T cells suppress autoimmune diabetes. J Exp Med 199(11):1455–1465. doi:10.1084/jem.20040139
Kavousanaki M, Makrigiannakis A, Boumpas D, Verginis P (2010) Novel role of plasmacytoid dendritic cells in humans: induction of interleukin-10-producing Treg cells by plasmacytoid dendritic cells in patients with rheumatoid arthritis responding to therapy. Arthritis Rheum 62(1):53–63. doi:10.1002/art.25037
Steinman RM, Banchereau J (2007) Taking dendritic cells into medicine. Nature 449(7161):419–426. doi:10.1038/nature06175
Rossetti M, Gregori S, Roncarolo MG (2010) Granulocyte-colony stimulating factor drives the in vitro differentiation of human dendritic cells that induce anergy in naive T cells. Eur J Immunol 40(11):3097–3106. doi:10.1002/eji.201040659
Harry RA, Anderson AE, Isaacs JD, Hilkens CMU (2010) Generation and characterisation of therapeutic tolerogenic dendritic cells for rheumatoid arthritis. Ann Rheum Dis 69(11):2042–2050. doi:10.1136/ard.2009.126383
Vosters O, Neve J, De Wit D, Willems F, Goldman M, Verhasselt V (2003) Dendritic cells exposed to nacystelyn are refractory to maturation and promote the emergence of alloreactive regulatory t cells. Transplantation 75(3):383–389. doi:10.1097/01.TP.0000043924.09647.61
Healy LJ, Collins HL, Thompson SJ (2008) Systemic administration of tolerogenic dendritic cells ameliorates murine inflammatory arthritis. Open Rheumatol J 2:71–80. doi:10.2174/1874312900802010071
Zheng X, Suzuki M, Ichim TE, Zhang X, Sun H, Zhu F, Shunnar A, Garcia B, Inman RD, Min W (2010) Treatment of autoimmune arthritis using RNA interference-modulated dendritic cells. J Immunol 184(11):6457–6464. doi:10.4049/jimmunol.0901717
Fu BM, He XS, Yu S, Hu AB, Ma Y, Wu LW, Tam NL, Huang JF (2009) Tolerogenic semimature dendritic cells induce effector T-cell hyporesponsiveness by the activation of antigen-specific CD4+ CD25+ T-regulatory cells. Exp Clin Transplant 7(3):149–156
Lim DS, Kang MS, Jeong JA, Bae YS (2009) Semi-mature DC are immunogenic and not tolerogenic when inoculated at a high dose in collagen-induced arthritis mice. Eur J Immunol 39(5):1334–1343. doi:10.1002/eji.200838987
Thomson AW, Robbins PD (2008) Tolerogenic dendritic cells for autoimmune disease and transplantation. Ann Rheum Dis 67(Suppl 3):iii90–iii96. doi:10.1136/ard.2008.099176
Zanetti M, Castiglioni P, Schoenberger S, Gerloni M (2003) The role of relB in regulating the adaptive immune response. Ann N Y Acad Sci 987:249–257
Tracey D, Klareskog L, Sasso EH, Salfeld JG, Tak PP (2008) Tumor necrosis factor antagonist mechanisms of action: a comprehensive review. Pharmacol Ther 117(2):244–279. doi:10.1016/j.pharmthera.2007.10.001
Wang L, Pino-Lagos K, de Vries VC, Guleria I, Sayegh MH, Noelle RJ (2008) Programmed death 1 ligand signaling regulates the generation of adaptive Foxp3+ CD4+ regulatory T cells. Proc Natl Acad Sci USA 105(27):9331–9336. doi:10.1073/pnas.0710441105
Kabalak G, Dobberstein SB, Matthias T, Reuter S, The YH, Dörner T, Schmidt RE, Witte T (2009) Association of immunoglobulin-like transcript 6 deficiency with Sjögren’s syndrome. Arthritis Rheum 60(10):2923–2925. doi:10.1002/art.24804
Criado G, Simelyte E, Inglis JJ, Essex D, Williams RO (2009) Indoleamine 2,3dioxygenase-mediated tryptophan catabolism regulates accumulation of Th1/Th17 cells in the joint in collagen-induced arthritis. Arthritis Rheum 60(5):1342–1351. doi:10.1002/art.24446
Tuettenberg A, Huter E, Hubo M, Horn J, Knop J, Grimbacher B, Kroczek RA, Stoll S, Jonuleit H (2009) The role of ICOS in directing T cell responses: ICOS-dependent induction of T cell anergy by tolerogenic dendritic cells. J Immunol 182(6):3349–3356. doi:10.4049/jimmunol.0802733
Ip WK, Lau YL (2004) Distinct maturation of, but not migration between, human monocyte-derived dendritic cells upon ingestion of apoptotic cells of early or late phases. J Immunol 173(1):189–196
Skoberne M, Beignon AS, Larsson M, Bhardwaj N (2005) Apoptotic cells at the crossroads of tolerance and immunity. Curr Top Microbiol 289:259–292
Kushwah R, Wu J, Oliver JR, Jiang G, Zhang JY, Siminovitch KA, Hu J (2010) Uptake of apoptotic DC converts immature DC into tolerogenic DC that induce differentiation of Foxp3(+) Treg. Eur J Immunol 40(4):1022–1035. doi:10.1002/eji.200939782
Kushwah R, Oliver JR, Zhang JY, Siminovitch KA, Hu J (2009) Apoptotic dendritic cells induce tolerance in mice through suppression of dendritic cell maturation and induction of antigen-specific regulatory T cells. J Immunol 183(11):7104–7118. doi:10.4049/jimmunol.0900824
Perone MJ, Larregina AT, Shufesky WJ, Papworth GD, Sullivan ML, Zahorchak AF, Stolz DB, Baum LG, Watkins SC, Thomson AW, Morelli AE (2006) Transgenic galectin-1 induces maturation of dendritic cells that elicit contrasting responses in naive and activated T cells. J Immunol 176(12):7207–7220
Zheng X, Suzuki M, Zhang X, Ichim TE, Zhu F, Ling H, Shunnar A, Wang MH, Garcia B, Inman RD, Min WP (2010) RNAi-mediated CD40-CD154 interruption promotes tolerance in autoimmune arthritis. Arthritis Res Ther 12(1):R13. doi:10.1186/ar2914
Morelli AE, Thomson AW (2007) Tolerogenic dendritic cells and the quest for transplant tolerance. Nat Rev Immunol 7(8):610–621. doi:10.1038/nri2132
Korn T, Bettelli E, Oukka M, Kuchroo VK (2009) IL-17 and Th17 cells. Annu Rev Immunol 27:485–517. doi:10.1146/annurev.immunol.021908.132710
Dong C (2008) TH17 cells in development: an updated view of their molecular identity and genetic programming. Nat Rev Immunol 8(5):337–348. doi:10.1038/nri2295
Holoshitz J, De Almeida DE, Ling S (2010) A role for calreticulin in the pathogenesis of rheumatoid arthritis. Ann N Y Acad Sci 1209:91–98. doi:10.1111/j.1749-6632.2010.05745.x
Miossec P (2007) Interleukin-17 in fashion, at last: ten years after its description, its cellular source has been identified. Arthritis Rheum 56(7):2111–2115. doi:10.1002/art.22733
Lubberts E (2008) IL-17/Th17 targeting: on the road to prevent chronic destructive arthritis? Cytokine 41(2):84–91. doi:10.1016/j.cyto.2007.09.014
Harrington LE, Hatton RD, Mangan PR, Turner H, Murphy TL, Murphy KM, Weaver CT (2005) Interleukin 17-producing CD4+ effector T cells develop via a lineage distinct from the T helper type 1 and 2 lineages. Nat Immunol 6(11):1123–1132. doi:10.1038/ni1254
Sato K, Kawamura T, Abo T (2010) “Senobi” stretch ameliorates asthma symptoms by restoring autonomic nervous system balance. J Investig Med 58(8):968–970. doi:10.231/JIM.0b013e3181f9167b
Takayanagi H (2005) Inflammatory bone destruction and osteoimmunology. J Periodontal Res 40(4):287–293. doi:10.1111/j.1600-0765.2005.00814.x
Paradowska-Gorycka A, Grzybowska-Kowalczyk A, Wojtecka-Lukasik E, Maslinski S (2010) IL-23 in the pathogenesis of rheumatoid arthritis. Scand J Immunol 71(3):134–145. doi:10.1111/j.1365-3083.2009.02361.x
Torres-Aguilar H, Aguilar-Ruiz SR, Gonzalez-Perez G, Munguia R, Bajana S, Meraz-Rios MA, Sanchez-Torres C (2010) Tolerogenic dendritic cells generated with different immunosuppressive cytokines induce antigen-specific anergy and regulatory properties in memory CD4+ T cells. J Immunol 184(4):1765–1775. doi:10.4049/jimmunol.0902133
Stoop JN, Harry RA, von Delwig A, Isaacs JD, Robinson JH, Hilkens CM (2010) Therapeutic effect of tolerogenic dendritic cells in established collagen-induced arthritis is associated with a reduction in Th17 responses. Arthritis Rheum 62(12):3656–3665. doi:10.1002/art.27756
Turnquist HR, Raimondi G, Zahorchak AF, Fischer RT, Wang Z, Thomson AW (2007) Rapamycin-conditioned dendritic cells are poor stimulators of allogeneic CD4+ T cells, but enrich for antigen-specific Foxp3+ T regulatory cells and promote organ transplant tolerance. J Immunol 178(11):7018–7031
Fischer R, Turnquist HR, Taner T, Thomson AW (2009) Use of rapamycin in the induction of tolerogenic dendritic cells. Handb Exp Pharmacol 188(III):215–232. doi:10.1007/978-3-540-71029-5_10
Miller RH, Fyffe-Maricich SL (2010) Restoring the balance between disease and repair in multiple sclerosis: insights from mouse models. Dis Model Mech 3(9–10):535–539. doi:10.1242/dmm.001958
Birch NW, Zeleznik-Le NJ (2010) Glycogen synthase kinase-3 and leukemia: restoring the balance. Cancer Cell 17(6):529–531. doi:10.1016/j.ccr.2010.05.017
Xu X, Guo Z, Jiang X, Yao Y, Gao Q, Ding Y, Cao X (2011) Regulatory dendritic cells program generation of interleukin-4-producing alternative memory CD4 T cells with suppressive activity. Blood 117(4):1218–1227. doi:10.1182/blood-2010-05-285494
Boissier MC, Assier E, Falgarone G, Bessis N (2008) Shifting the imbalance from Th1/Th2 to Th17/treg: the changing rheumatoid arthritis paradigm. Joint Bone Spine 75(4):373–375. doi:10.1016/j.jbspin.2008.04.005
Zhai Y, Ghobrial RM, Busuttil RW, Kupiec-Weglinski JW (1999) Th1 and Th2 cytokines in organ transplantation: paradigm lost? Crit Rev Immunol 19(2):155–172
Chabaud M, Aarvak T, Garnero P, Natvig JB, Miossec P (2001) Potential contribution of IL-17-producing Th(1)cells to defective repair activity in joint inflammation: partial correction with Th(2)-promoting conditions. Cytokine 13(2):113–118. doi:10.1006/cyto.2000.0811
van Duivenvoorde LM, Han WG, Bakker AM, Louis-Plence P, Charbonnier LM, Apparailly F, van der Voort EI, Jorgensen C, Huizinga TW, Toes RE (2007) Immunomodulatory dendritic cells inhibit Th1 responses and arthritis via different mechanisms. J Immunol 179(3):1506–1515
Nadkarni S, Mauri C, Ehrenstein MR (2007) Anti-TNF-alpha therapy induces a distinct regulatory T cell population in patients with rheumatoid arthritis via TGF-beta. J Exp Med 204(1):33–39. doi:10.1084/jem.20061531
Notley CA, Ehrenstein MR (2010) The yin and yang of regulatory T cells and inflammation in RA. Nat Rev Rheumatol 6(10):572–577. doi:10.1038/nrrheum.2010.143
Chen W, Liang X, Peterson AJ, Munn DH, Blazar BR (2008) The indoleamine 2,3-dioxygenase pathway is essential for human plasmacytoid dendritic cell-induced adaptive T regulatory cell generation. J Immunol 181(8):5396–5404
Bianco NR, Kim SH, Ruffner MA, Robbins PD (2009) Therapeutic effect of exosomes from indoleamine 2, 3-dioxygenase-positive dendritic cells in collagen-induced arthritis and delayed-type hypersensitivity disease models. Arthritis Rheum 60(2):380–389. doi:10.1002/Art.24229
Matsui JI, Ryals BM (2005) Hair cell regeneration: an exciting phenomenon…but will restoring hearing and balance be possible? J Rehabil Res Dev 42(4 Suppl 2):187–198
Sharma MD, Baban B, Chandler P, Hou DY, Singh N, Yagita H, Azuma M, Blazar BR, Mellor AL, Munn DH (2007) Plasmacytoid dendritic cells from mouse tumor-draining lymph nodes directly activate mature Tregs via indoleamine 2, 3-dioxygenase. J Clin Invest 117(9):2570–2582. doi:10.1172/JCI31911
Tisch R (2010) Immunogenic versus tolerogenic dendritic cells: a matter of maturation. Int Rev Immunol 29(2):111–118. doi:10.3109/08830181003602515
Chorny A, Gonzalez-Rey E, Fernandez-Martin A, Pozo D, Ganea D, Delgado M (2005) Vasoactive intestinal peptide induces regulatory dendritic cells with therapeutic effects on autoimmune disorders. Proc Natl Acad Sci USA 102(38):13562–13567. doi:10.1073/pnas.0504484102
Kim SH, Kim S, Evans CH, Ghivizzani SC, Oligino T, Robbins PD (2001) Effective treatment of established murine collagen-induced arthritis by systemic administration of dendritic cells genetically modified to express IL-4. J Immunol 166(5):3499–3505
Kim SH, Kim S, Oligino TJ, Robbins PD (2002) Effective treatment of established mouse collagen-induced arthritis by systemic administration of dendritic cells genetically modified to express FasL. Mol Ther 6(5):584–590
Martin E, Capini C, Duggan E, Lutzky VP, Stumbles P, Pettit AR, O’Sullivan B, Thomas R (2007) Antigen-specific suppression of established arthritis in mice by dendritic cells deficient in NF-kappaB. Arthritis Rheum 56(7):2255–2266. doi:10.1002/art.22655
Balanescu A, Nat R, Regalia T, Radu E, Bojinca V, Ionescu R, Predescu V, Popescu E, Predeteanu D (2003) Correlation between the immunophenotypical presentation of dendritic cells and the clinical response to anti-rheumatic treatment in rheumatoid arthritis. Rom J Intern Med 41(3):255–267
Hilkens CM, Isaacs JD, Thomson AW (2010) Development of dendritic cell-based immunotherapy for autoimmunity. Int Rev Immunol 29(2):156–183. doi:10.3109/08830180903281193
Sidhu M, Griffiths MM, Bradley DS (2009) Vaccination with collagen-pulsed dendritic cells prevents the onset and reduces the disease severity in the mouse model of spontaneous polychondritis. Clin Exp Immunol 157(3):350–358. doi:10.1111/j.1365-2249.2009.03968.x
Pouniotis DS, Proudfoot O, Bogdanoska V, Apostolopoulos V, Fifis T, Plebanski M (2004) Dendritic cells induce immunity and long-lasting protection against blood-stage malaria despite an in vitro parasite-induced maturation defect. Infect Immun 72(9):5331–5339. doi:10.1128/IAI.72.9.5331-5339.2004
Pandha HS, John RJ, Hutchinson J, James N, Whelan M, Corbishley C, Dalgleish AG (2004) Dendritic cell immunotherapy for urological cancers using cryopreserved allogeneic tumour lysate-pulsed cells: a phase I/II study. BJU Int 94(3):412–418. doi:10.1111/j.1464-410X.2004.04922.x
Akbar SM, Furukawa S, Hasebe A, Horiike N, Michitaka K, Onji M (2004) Production and efficacy of a dendritic cell-based therapeutic vaccine for murine chronic hepatitis B virus carrier. Int J Mol Med 14(2):295–299
Aguillon JC, Cruzat A, Contreras-Levicoy J, Dotte A, Pesce B, Aravena O, Salazar L, Catalan D, Abello P, Aguirre A, Llanos C, Cuchacovich M (2005) Emergent therapies for rheumatoid arthritis. Rev Med Chil 133(8):969–976
Popov I, Li M, Zheng X, San H, Zhang X, Ichim TE, Suzuki M, Feng B, Vladau C, Zhong R, Garcia B, Strejan G, Inman RD, Min WP (2006) Preventing autoimmune arthritis using antigen-specific immature dendritic cells: a novel tolerogenic vaccine. Arthritis Res Ther 8(5):R141. doi:10.1186/ar2031
Figdor CG, de Vries IJ, Lesterhuis WJ, Melief CJ (2004) Dendritic cell immunotherapy: mapping the way. Nat Med 10(5):475–480. doi:10.1038/nm1039
Nestle FO, Banchereau J, Hart D (2001) Dendritic cells: on the move from bench to bedside. Nat Med 7(7):761–765. doi:10.1038/89863
Acknowledgments
The authors wish to thank Dr. Fu Jinqiu, Tian Zhoubin and Zhao Kai for their review of the manuscript. This work was supported by grant from the National Natural Science Foundation of China (No. 30801258 M.H.) and The Star of Jinan Youth Science and Technology Project (No. 20100114).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, Y., Zhang, A., Du, H. et al. Tolerogenic dendritic cells and rheumatoid arthritis: current status and perspectives. Rheumatol Int 32, 837–844 (2012). https://doi.org/10.1007/s00296-011-2133-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-011-2133-2