Abstract
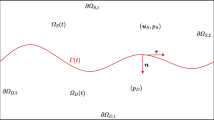
A theoretical framework is presented for describing blood flow through the irregular vasculature of a solid tumor. The tumor capillary bed is modeled as a capillary tree of bifurcating segments whose geometrical construction involves deterministic and random parameters. Blood flow along the individual capillaries accounts for plasma leakage through the capillary walls due to the transmural pressure according to Sterling’s law. The extravasation flow into the interstitium is described by Darcy’s law for a biological porous medium. The pressure field developing in the interstitium is computed by solving Laplace’s equation subject to derived boundary conditions at the capillary vessel walls. Given the arterial, venous, and tumor surface pressures, the problem is formulated as a coupled system of integral and differential equations arising from the interstitium and capillary flow transport equations. Numerical discretization yields a system of linear algebraic equations for the interstitial and capillary segment pressures whose solution is found by iterative methods. Results of numerical computations document the effect of the interstitial hydraulic and vascular permeability on the fractional plasma leakage. Given the material properties, the fractional leakage reaches a maximum at a particular grade of the bifurcating vascular tree.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Baish JW, Jain RK (2000) Fractals and cancer. Cancer Res 60: 3683–3688
Baish JW, Netti PA, Jain RK (1997) Transmural coupling of fluid flow in microcirculatory network and interstitium in tumors. Microvasc Res 53: 128–141
Baxter LT, Jain RK (1989) Transport of fluid and macromolecules in tumors I. Role of interstitial pressure and convection. Microvasc Res 37: 77–104
Baxter LT, Jain RK (1990) Transport of fluid and macromolecules in tumors II. Role of heterogeneous perfusion and lymphatics. Microvasc Res 40: 246–263
Chaplain MAJ, McDougall SR, Anderson ARA (2006) Mathematical modeling of tumor-induced angiogenesis. Annu Rev Biomed Eng 8: 233–257
Chung H-W, Chung H-J (2001) Correspondence re: J. W. Baish and R. K. Jain, Fractals and Cancer. Cancer Res 60:3683–3688 (2000). Cancer Res 61: 8347–8351
Dreher MR, Liu W, Michelich CR, Dewhirst MW, Yuan F, Chilkoti A (2006) Tumor vascular permeability, accumulation, and penetration of macromolecular drug carriers. J Nat Cancer Inst 98(5): 335–344
El-Kareh AW, Secomb TW (1995) Effect of increasing vascular hydraulic conductivity on delivery of macromolecular drugs to tumor cells. Int J Rad Onc Biol Phys 32: 1419–1423
Fleischman GJ, Secomb TW, Gross JF (1986a) Effect of extravascular pressure gradients on capillary fluid exchange. Math Biosc 81: 145–164
Fleischman GJ, Secomb TW, Gross JF (1986b) The interaction of extravascular pressure fields and fluid exchange in capillary networks. Math Biosc 82: 141–151
Gazit Y, Baish JW, Safabakhsh N, Leunig M, Baxter LT, Jain RK (1997) Fractal characteristics of tumor vascular architecture during tumor growth and regression. Microcirculation 4: 395–402
Jain RK (1987) Transport of molecules in the tumor interstitium: a review. Cancer Res 47: 3039–3051
Jain RK, Baxter LT (1988) Mechanisms of heterogeneous distribution of monoclonal antibodies and other molecules in tumors: Significance of interstitial pressure. Cancer Res 48: 7022–7032
Karshafian R, Burns PN, Henkelman MR (2003) Transit time kinetics in ordered and disordered vascular trees. Phys Med Biol 48: 3225–3237
Less JR, Skalak TC, Sevick EM, Jain RK (1991) Microvascular architecture in a mammary carcinoma: branching patterns and vessel dimensions. Cancer Res 51: 265–273
Milosevic MF, Fyles AW, Hill RP (1999) The relationship between elevated interstitial fluid pressure and blood flow in tumors: A bioengineering analysis. Int J Rad Oncol Biol Phys 43: 1111–1123
Netti PA, Roberge A, Boucher Y, Baxter LT, Jain RK (1996) Effect of transvascular fluid exchange on pressure-flow relationship in tumors: A proposed mechanism for tumor blood flow heterogeneity. Microvasc Res 52: 27–46
Pindera MZ, Ding H, Chen Z (2008) Convected element method for simulation of angiogenesis. J Math Biol 57: 467–495
Pozrikidis C (1997) Introduction to theoretical and computational fluid dynamics. Oxford University Press, New York
Pozrikidis C (2008) Numerical computation in science and engineering, 2nd edn. Oxford University Press, New York
Pozrikidis C, Farrow DA (2003) A model of fluid flow in solid tumors. Ann Biomed Eng 31: 181–194
Sands H, Jones PL, Shah SA, Palme D, Vessella RL, Gallagher BM (1988) Correlation of vascular permeability and blood flow with monoclonal antibody uptake by human clouser and renal cell xenografts. Cancer Res 48: 188–193
Sevick EM, Jain RK (1989a) Geometric resistance to blood flow in solid tumors perfused ex vivo: effects of tumor size and perfusion pressure. Cancer Res 49: 3506–3512
Sevick EM, Jain RK (1989b) Geometric resistance to blood flow in solid tumors perfused ex vivo: effect of hematocrit on intratumor blood viscosity. Cancer Res 49: 3513–3519
Tsafnat N, Tsafnat G, Lambert TD (2004) A three-dimensional fractal model of tumour vasculature. In: Proc. 26th Ann. Int. Conf. IEEE EMBS, pp 683–686
Vaughan ATM, Anderson PW, Dykes P, Chapman CE, Bradwell AR (1987) Limitations to the killing of tumours using radiolabelled antibodies. Br J Radiol 60: 567–578
Zhang X-Y, Lick J, Dewhirst MW, Yuan F (2000) Interstitial hydraulic conductivity in a fibrosarcoma. Am J Physiol Heart Circ Physiol 279: H2726–H2734
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pozrikidis, C. Numerical simulation of blood and interstitial flow through a solid tumor. J. Math. Biol. 60, 75–94 (2010). https://doi.org/10.1007/s00285-009-0259-6
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00285-009-0259-6