Abstract
Low-grade inflammation is recognized as an important factor in the development and progression of a multitude of diseases including type 2 diabetes mellitus and cardiovascular disease. The potential of using antibody-based therapies that neutralize key players of low-grade inflammation has gained scientific momentum as a novel therapeutic strategy in metabolic diseases. As interleukin-6 (IL-6) is traditionally considered a key pro-inflammatory factor, the potential of expanding the use of anti-IL-6 therapies to metabolic diseases is intriguing. However, IL-6 is a molecule of a very pleiotropic nature that regulates many aspects of not only inflammation but also metabolism. In this review, we give a brief overview of the pro- and anti-inflammatory aspects of IL-6 and provide an update on its role in metabolic regulation, with a specific focus on glucose homeostasis and adipose tissue metabolism. Finally, we shall discuss the metabolic implications and clinical potential of blocking IL-6 signaling, focusing on glucose homeostasis and lipid metabolism.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
During the past two decades, a growing body of evidence shows that low-grade inflammation is a crucial factor in the development and progression of a cluster of diseases, e.g., type 2 diabetes mellitus (T2DM) and cardiovascular disease [1]. In patients with low-grade inflammation, circulating levels of interleukin-6 (IL-6) are often increased and have been associated with detrimental metabolic actions [2]. However, a large number of contrasting studies indicate that chronically elevated IL-6 may have beneficial metabolic effects [3]. These conflicting opinions highlight the complexity of the IL-6 molecule, and it is still debated whether IL-6 is “a bad or a good guy” in the regulation of metabolism.
Moreover, a great deal of our knowledge regarding the metabolic effects of IL-6 is based on correlational studies, in vitro cell culture studies of supraphysiological concentrations of IL-6, and studies performed in rodents. Bearing in mind that there seem to be important species-specific differences, particularly of the glucose regulatory actions of IL-6, caution should be taken when conclusions are made based on data from rodent studies.
In this review, we will introduce the pleotropic molecule IL-6 and provide a brief overview of its role in different contexts where it is increased (acute and chronic inflammation and exercise). Moreover, we will discuss the complex role of IL-6 in the regulation of metabolism, with a specific focus on glucose homeostasis and adipose tissue metabolism. Finally, we shall discuss the metabolic implications of blocking IL-6 signaling and the clinical potential of IL-6 receptor blockade, focusing on glucose homeostasis and lipid metabolism.
Interleukin-6 signaling
IL-6 was identified in 1989 and is part of the IL-6 family of cytokines that include IL-11, oncostatin M, leukemia inhibitory factor (LIF), ciliary neurotrophic factor (CNTF), cardiotrophin-1, and cardiotrophin-like cytokine [4]. IL-6 is produced by immune cells, chondrocytes, osteoblasts, endothelial cells, skeletal muscle cells, smooth muscle cells, pancreatic islet β-cells, among several other cell types [4]. Of note, IL-6 is furthermore a myokine and an adipokine as it is secreted from skeletal muscle in response to exercise and from adipose tissue, respectively [5, 6].
The upstream stimuli for IL-6 secretion differ according to cell type. In immune cells, tumor necrosis factor-alpha (TNF-α) induces IL-6 secretion upon activation of the nuclear factor-κβ signaling pathway. Conversely, in skeletal muscle, IL-6 secretion is not induced by TNF-α-mediated nuclear factor-κβ activation, but is suggested to result from increased cytosolic Ca2+ and activation of P38 mitogen-activated protein kinase or calcineurin during skeletal muscle contraction [7]. Moreover, a recent study identified lactate production as a mediator of IL-6 release from muscle fibers [8].
Upon secretion, IL-6 mediates its biological effects via two distinct pathways, the classic signaling pathway and the trans-signaling pathway. The classic signaling occurs when IL-6 binds to its transmembrane IL-6 receptor (IL-6R) on target cells. The IL-6/IL-6R complex associates with the transmembrane signal-transducing glycoprotein 130 (gp130) and thereby activates downstream signaling cascades [4]. IL-6 can also signal via an alternative route called “trans-signaling” by binding to its soluble receptor [9, 10]. When IL-6 binds to the soluble IL-6 receptor (sIL-6R), a soluble complex is formed. This soluble complex then binds to gp130, and as gp130 is ubiquitously expressed on cells in the entire body, the IL-6/sIL-6 complex is able to signal in most tissues [11, 12]. The downstream signal transduction of IL-6 involves different intracellular pathways that include the JAK-STAT3, JAK-SHP-2-MAPK, and the PI3K pathway [11,12,13,14].
Taken together, IL-6 is not only secreted by a broad range of cells but can also target cells universally, and due to its various upstream and downstream signaling pathways, the final outcome of IL-6 signaling depends on the interplay of the type of target cell, its intracellular environment, and concomitant external stimuli of the cell [4]. Again, this highlights the tremendous pleotropic nature of IL-6. Therefore, it is not surprising that IL-6 is implicated in the regulation of the immune system (having both pro- and anti-inflammatory effects), bone metabolism, the nervous system, the hemopoietic system, the endocrine system, energy metabolism, glucose homeostasis, and lipid metabolism [4].
Interleukin-6 elevation during acute and chronic inflammation
Due to acute elevations of IL-6 in the initial phase of an infection or in response to noninfectious stimuli such as a burn or traumatic injuries, early studies identified IL-6 as a pro-inflammatory cytokine [15]. In these contexts, macrophages and monocytes secrete IL-6, leading to concentrations that are up to 1000 times higher than baseline circulating levels [16]. Increased concentrations of pro-inflammatory cytokines such as interleukin-1 (IL-1) and TNF-α precede and are the main inducers of the IL-6 elevation [17,18,19,20]. IL-6 then stimulates the secretion of various acute phase proteins, e.g., C-reactive protein (CRP) [21] that acts as warning signals, supporting the immune system in eliminating the source of stress from the system [22]. Thus, in the context of acute inflammation, elevation of IL-6 is a protective and beneficial response.
The role of IL-6 during chronic inflammation is still not well understood. Chronic low-grade inflammation is characterized by a two- to threefold elevation in systemic concentrations of cytokines (IL-1, TNF-α, and IL-6) and acute phase proteins, e.g., CRP [23, 24]. Chronic low-grade inflammation is found in states of the so-called metaflammation, observed in the metabolic syndrome, obesity, and type 2 diabetes mellitus (T2DM) [25]. A central source of cytokines in “metaflammation” is the adipose tissue, infiltrated by macrophages [5, 25, 26]. The inflamed adipose tissue is believed to arise due to hypertrophic adipocyte expansion that leads to compression of capillaries and tissue ischemia. This, in turn, leads to necrosis of the adipocytes and attraction of macrophages, which trigger the secretion of IL-6, TNF-α, IL-1β, and other pro-inflammatory cytokines [27]. While low-grade inflammatory factors such as TNF-α and IL-1β are implicated in insulin resistance and development of T2DM [28], the contributing role of IL-6 in metabolic disease is less established (reviewed in the subsequent sections).
Interleukin-6 elevation during exercise
Today, it is well known that during exercise muscle contractions lead to increased systemic concentrations of various peptides and cytokines, including IL-6, interleukin-1 receptor antagonist (IL-1ra), and interleukin-10 (IL-10) [29,30,31]. In 2003, it was proposed that a cytokine or a peptide produced, expressed, and released by muscle fibers in response to exercise, exerting autocrine, paracrine, or endocrine effects, should be named a “myokine” [32].
Thus, IL-6 was the first myokine to be discovered and has since been studied most extensively [33, 34]. IL-6 is the first detectable cytokine in the circulation [7] and it increases exponentially (up to 100-fold) [35,36,37], proportional to the duration of exercise and the amount of muscle mass engaged in the exercise [35]. Exercise-induced IL-6 occurs without a preceding increase in the pro-inflammatory cytokines TNF-a and IL-1 [38, 39] and initiates an anti-inflammatory response, as it inhibits the expression of TNF-α and IL-1 [20, 29, 40] and stimulates the production of the anti-inflammatory molecule IL-1ra [41], soluble TNF-α-receptors (sTNF-αR) [42], and IL-10 [43] among others.
The role of IL-6 in glucose homeostasis
The interest in IL-6 in energy metabolism arose from studies in IL-6-deficient mice showing that mice lacking IL-6 develop mature-onset obesity along with glucose intolerance [44, 45]. Today, it is well established that IL-6 regulates glucose homeostasis; however, whether its role is beneficial or detrimental is debated [46].
The generally accepted view of IL-6 as a “bad guy” with regard to glucose homeostasis is primarily based on epidemiological studies showing a correlational relationship, in vitro cell culture studies of supraphysiological concentrations of IL-6, and some animal studies [47]. Epidemiological studies have revealed that IL-6 is implicated in the chronic inflammation that accompanies conditions such as obesity and T2DM [47].
In support of this idea, infusions of IL-6 have been shown to impair insulin action in mice [48], whereas blocking of IL-6 signaling improves hepatic insulin sensitivity [49]. These findings suggest that IL-6 may be involved in hepatic insulin resistance, which may derive from increased phosphorylation of SOCS3 that binds insulin receptor substrates (IRS) and targets them for proteasomal degradation, thus impeding insulin-mediated glucose uptake [50]. However, the negative effects of IL-6 in the liver appear to arise only when IL-6 is secreted from the adipose tissue [51].
In contrast to the view of IL-6 as a “bad guy” in the regulation of glucose homeostasis, a growing body of evidence indicates that chronically elevated IL-6 is beneficial, as increased levels might serve as an adaptive mechanism aiming at improving glycemic control. In line with this, a study by Ellingsgaard et al. showed that IL-6 knockout mice on a high-fat diet were unable to expand the pancreatic alpha-cell mass, resulting in reduced glucose-stimulated insulin secretion (a beta-cell defect). In this context, increased IL-6 was seen as an adaptive response, necessary to maintain proper insulin secretion and glycemic control in response to a high-fat diet [52]. Subsequent studies by the same group demonstrated that IL-6 mediates cross-talk between insulin-sensitive tissues and pancreatic beta-cells via the incretin hormone glucagon-like peptide-1 (GLP-1). More specifically, IL-6 was shown to promote enhancement of GLP-1 production and consequently increased secretion of insulin [53]. Another study confirmed that IL-6 regulates the incretin axis, as glucose regulatory action of the other incretin hormone glucose insulinotropic polypeptide (GIP) was found to be mediated via IL-6 [54].
the Brüning group recently identified IL-6 as an important determinant of the alternative activation of macrophages, as IL-6 counterbalance a shift of macrophage populations toward a pro-inflammatory phenotype, indicating a role of IL-6 in limiting systemic inflammation [55]. Later, the same group showed that IL-6, trans-signaling in the central nervous system (CNS), suppresses feeding and improves glycemic control, an effect that seems to be enhanced in obese mice [56]. In addition, Mauer et al. showed that IL-6 signaling limits systemic inflammation and improves glycemic control in macrophages and hepatocytes of lean and obese mice [55, 57]. Correspondingly, another group proposed that increased IL-6 mRNA in insulin-resistant tissues may be an attempt to overcome the metabolic dysfunction [58].
Taken together, elevation of IL-6 during low-grade inflammation may serve as an adaptive mechanism in an attempt to increase insulin production and improve glucose homeostasis at least in mouse models. However, there seem to be differences between the actions of IL-6 in mice and in humans, and some of the effects of IL-6 may be species-specific [59]. In support of this, mouse IL-6 is only 42% identical to the human IL-6 molecule [60, 61].
A beneficial role of acutely elevated IL-6 in glucose homeostasis is suggested from exercise studies, where an acute bout of exercise with elevations of plasma IL-6 increases glucose uptake in the periphery [62]. It would indeed be a counterintuitive physiological response to release a factor that promotes insulin resistance, in a condition where increased insulin sensitivity is needed. In cell culture studies, increased glucose uptake was accompanied by translocation of the glucose transporter GLUT4 from intracellular compartments to the plasma membrane in skeletal myotubes [63]. Moreover, IL-6-induced IL-1ra during exercise is speculated to result in limited IL-1-induced pancreas damage and thus improved insulin secretion [62, 64]. However, whether IL-6 plays a role for the adaptations in glucose homeostasis occurring with exercise in humans remains to be clarified.
In humans, a huge increase in circulating IL-6 (by means of an infusion) induces fever, release of catecholamines, and elevated plasma glucose levels [65, 66]. In contrast, a modest elevation of plasma IL-6, comparable to the IL-6 elevation induced by exercise, seems to induce an anti-inflammatory and mostly beneficial effect in the maintenance of glucose homeostasis in humans. Despite the overall positive effects of short-term IL-6 exposure, the effects seem to be context dependent [43, 67,68,69]. An infusion of recombinant IL-6 in physiological concentrations improves insulin sensitivity during a hyperinsulinemic, euglycaemic clamp in healthy individuals [63], but not in patients with T2DM [70]. In support of this context dependency, an infusion of IL-6 was found to increase endogenous hepatic glucose production (revealing a direct muscle-liver cross-talk) [69]; however, this was observed exclusively during exercise, whereas no effect was found during resting conditions [43, 67, 68].
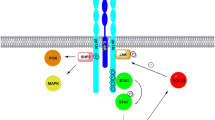
In line with the above-described beneficial effects of an acute increase in IL-6, we recently showed that infusing IL-6 prior to a meal improves postprandial glucose homeostasis in humans (Fig. 1). More specifically, we came across a previously unidentified role of acutely increased IL-6 (by an infusion or an acute bout of exercise) in delaying the rate of gastric emptying. Our studies demonstrated that an infusion of IL-6 delay gastric emptying rate, leading to a reduction in postprandial glucose. Moreover, independent of gastric emptying rate, IL-6 reduces postprandial insulin secretion. The effects of IL-6 on gastric emptying and insulin secretion were maintained in patients with T2DM [59].
In a series of human studies performed at the Centre for Physical Activity Research, an acute increase in IL-6 (by an infusion or an acute bout of exercise) delayed gastric emptying rate. Following an IL-6 infusion, the deceleration in gastric emptying rate reduced postprandial glucose. In addition, IL-6 reduced postprandial insulin. Thus, an acute increase in IL-6 delays gastric emptying with direct effects on glucose homeostasis in humans. Adapted with permission from Lang Lehrskov et al. [59]
In conclusion, IL-6 regulation of glucose homeostasis is complex and still not fully understood. The conflicting observations regarding the metabolic role of IL-6 stimulate the ongoing debate as to whether IL-6 is good or bad in the context of low-grade inflammation in humans.
Given the various glucose regulatory effects of IL-6, we need to be aware of the metabolic consequences of IL-6 blockade in auto-immune/inflammatory disease (reviewed in the subsequent sections).
Furthermore, it is tempting to pose the question whether the effects of acute IL-6 administration might be exploited for therapy to improve glycemia. To be able to answer this, further studies are needed to address how IL-6 exerts its actions, and whether the potential beneficial effects of IL-6 are preserved in different metabolic states (obesity, metabolic syndrome, and type 2 diabetes).
The role of IL-6 in lipid metabolism
IL-6 is suggested to participate in mediating the obesity-associated insulin resistance preceding T2DM [71]. The rationale for this is based on studies showing an association between elevated circulating levels of IL-6 due to obesity and insulin resistance in liver and adipose tissue [71].
Conversely, mounting evidence has consolidated a central role of IL-6 in the regulation of lipid metabolism. IL-6 has been identified as a lipolytic factor from studies in mice, where IL-6 knockout mice developed mature-onset obesity that was partly reversed after repetitive infusions of IL-6 [44]. A subsequent study showed that chronic administration of IL-6 results in reductions in rodent mesenteric and retroperitoneal fat depots [44]. Transgenic mice overexpressing IL-6 have been shown to stay lean and be protected from the adverse effects of a high-fat diet [72]. Based on these findings, an anti-obesity effect of IL-6 has been suggested [73]. Furthermore, we know from studies in humans that a single infusion of IL-6 stimulates lipolysis and β oxidation at the whole-body level [74], both when administered at high and low doses [75]. The lack of a concomitant increase in plasma adrenaline or insulin indicates that the effect is driven by IL-6 per se. Another study demonstrated an increased glycerol release from subcutaneous adipose tissue in response to an acute IL-6 infusion [76]. Of note, patients with Castleman’s disease (abnormal production of IL-6 by germinal center B cells) are characterized by hypolipidemia [77]. These findings of IL-6 as a critical regulator of lipolysis and β oxidation have been confirmed by multiple in vitro studies demonstrating that IL-6 treatment increases lipolysis and β oxidation both in myotubes and adipocytes [63, 74, 78, 79]. Lipolysis has been shown to be stimulated by IL-6 in several adipose tissue depots, including human subcutaneous and visceral depots [80]; however, the specific molecular mechanism conveying lipolysis in adipose tissue remains unknown, but may involve activation of AMP-activated kinase (AMPK) [63, 79, 81]. In contrast, it is well known that IL-6 induces β oxidation via AMPK [7, 63, 79, 81]. AMPK phosphorylates and inactivates acetyl CoA carboxylase β (ACCβ), which decreases malonyl-CoA content and thus relieves its allosteric inhibition of carnitine-palmitoyl transferase 1 (CPT-1) [82, 83]. CPT-1 catalyzes the formation of carnitine-acyl and transports into the mitochondrion where β oxidation takes place [84].
Other roles for IL-6 in lipid metabolism include IL-6-stimulated leptin secretion from human omental and subcutaneous adipose tissues as demonstrated by Trujillo et al. [80], which may affect appetite and caloric intake. They also found that IL-6 decreased lipoprotein lipase (LPL) activity by 56% in omental and by 68% in subcutaneous adipose tissue. These findings indicate that IL-6 may act to diminish lipid uptake and deposition in adipose tissue and suggest the concept of IL-6 as an “adipostat” which controls adipocyte size [80].
Overall, solid evidence exists that IL-6 plays an essential physiological role in the regulation of adipose tissue and lipid metabolism. As such, the notion of IL-6 as an anti-obesity agent should be “weighed” against its importance as a causal driver of the development and progression of obesity-associated diseases such as T2DM.
The implication of IL-6 receptor blockade in inflammatory diseases
IL-6 is the major mediator of inflammatory joint destruction in rheumatoid arthritis (RA) [85] and thus, in 2010, the IL-6 receptor antibody, tocilizumab, the only available medical treatment targeting IL-6 signaling, was approved for the treatment of moderate to severe rheumatoid arthritis [86]. Today, tocilizumab is also approved for the treatment of other inflammation-related diseases such as Castleman’s disease and systemic juvenile idiopathic arthritis [87]. Moreover, it is debated whether tocilizumab can be used also in the treatment of other diseases with elevated IL-6 levels (other auto-immune diseases, malignant diseases, cardiovascular disease, and T2DM) [86]. Even though the potential of targeting inflammation by tocilizumab in the treatment of T2DM is intriguing, the recognition of IL-6 as a pleiotropic factor with important effects on metabolism is paramount and the effects of IL-6 blockade on metabolism need first to be clarified.
The implication of IL-6 receptor blockade on glucose homeostasis
Given that IL-6 is speculated to be a co-inducer of the development of insulin resistance associated with obesity, the idea of blocking IL-6 in order to prevent and treat hyperglycemia has gained attention [88, 89]. Several studies point toward a beneficial effect of IL-6 receptor blockade on glycemic control in patients treated with tocilizumab with and without T2DM [90,91,92,93]. In patients with RA, hba1c levels were decreased after the initiation of tocilizumab treatment [94], and a review concluded that tocilizumab improves insulin sensitivity in inflammatory arthritis [92].
However, as discussed above, the role of IL-6 in the regulation of insulin sensitivity and overall glucose homeostasis is rather complex. Thus, even though treatment with IL-6 blockade to prevent or treat T2DM may seem appealing, it still remains unexplored and further research is needed.
Along this line, anti-inflammatory treatment with IL-1 blockade and other anti-inflammatory agents have recently been suggested as potential candidates to treat or prevent diabetes [89, 95].
The implication of IL-6 receptor blockade on lipid metabolism
Based on the solid evidence of IL-6 being a lipolytic factor, it has been speculated whether IL-6 receptor blockade in the treatment of inflammatory disorders may lead to the unwanted side effects of obesity, i.e., insulin resistance/T2DM, and cardiometabolic morbidity and mortality, thus opposing its potential as a treatment for immunometabolic diseases.
Addressing this concern, studies in humans receiving tocilizumab have consistently shown increased systemic levels of cholesterol, low-density lipoproteins (LDL), high-density lipoproteins (HDL), and triglycerides, which are well-known adverse effects in patients receiving tocilizumab, as reviewed by Choy et al. [96]. Recently, our research group has performed a randomized placebo-controlled trial including abdominally obese and physically inactive, but otherwise healthy, people for a 12-week exercise intervention with concomitant blockade of IL-6 signaling by tocilizumab [97]. The study demonstrated that blocking IL-6 signaling during aerobic bike training completely abolishes the exercise-induced reduction of visceral adipose tissue (Fig. 2). This finding reveals that IL-6 is required to obtain the adipose tissue-reducing effects of exercise and consolidates the role of IL-6 in the regulation of lipid metabolism. Moreover, LDL and total cholesterol were increased by tocilizumab, and this increase was not reversed by exercise.
A recent randomized placebo-controlled trial including abdominally obese, inactive participants, performed at the Centre for Physical Activity Research, demonstrated that IL-6 receptor blockade abolishes the fat-reducing effects of aerobic bike exercise. Thus, IL-6 signaling is a requirement for reduction in visceral adipose tissue following exercise. Adapted with permission from Wedell-Neergaard et al. [97]
Clinical considerations of IL-6 receptor blockade in immunometabolic diseases
Several studies have confirmed the safety and efficacy of tocilizumab in reducing inflammation in patients with pro-inflammatory diseases [98, 99] and have sparked an interest to expand the application of tocilizumab for the treatment of immunometabolic diseases. To this end, emerging evidence, as reviewed above, suggests that blocking IL-6 signaling may improve glucose homeostasis in a context where IL-6 is chronically elevated. However, caution should be taken as the role of IL-6 in glucose homeostasis is complex and context-dependent as described above. In contrast to the potentially beneficial role of IL-6 receptor blockade on glucose homeostasis, IL-6 receptor blockade leads to weight gain and hyperlipidemia, which may oppose the idea of using IL-6 receptor blockade to treat metabolic disease. Interestingly, the adverse effects on lipid metabolism may not necessarily translate into an increased cardiometabolic risk, as implied by follow-up studies of patients treated with tocilizumab [100]. Therefore, the long-term importance of the beneficial and adverse metabolic consequences of IL-6 receptor antagonism needs to be further addressed to fully grasp the clinical potential of this drug in the treatment of metabolic diseases.
Conclusion
In conclusion, the pleotropic molecule IL-6 has both pro- and anti-inflammatory characteristics and the outcome of IL-6 signaling depends on the context. During acute inflammation, IL-6 is a key player in orchestrating the acute immune response to control an infectious stimulus, which involves both IL-6-mediated induction of anti-inflammatory cytokines (IL-1ra, IL-8, IL-10, etc.) and acute phase reactants, including CRP. Chronic low-grade inflammation is a defining feature of metabolic diseases, where circulating levels of IL-6, TNF-α, IL-1β, and other cytokines are increased. Recently, chronic low-grade inflammation has been recognized as a driver of adverse metabolic effects. Therefore, it is intriguing to suggest the low-grade inflammatory cytokines as targets for antibody-based treatment strategies for metabolic diseases. Yet, while research shows promising results of antibodies directed at IL-1β in the treatment of metabolic disease, the potential of anti-IL-6-based therapies may be more challenging due to the paramount role of this myokine in metabolism: IL-6 is an important regulator of glucose metabolism; however, its role is complex and context- and species-dependent, so that in some situations, it improves while in other, it aggravates insulin sensitivity and glucose homeostasis. In regard to lipid metabolism, solid evidence has identified IL-6 as a lipolytic factor. IL-6 stimulates lipolysis and β oxidation, increases leptin, and reduces lipoprotein lipase, which has led to the suggestion that IL-6 is an “adipostat” that attempts to control and prevent adipocyte hypertrophy. Overall, this complex role of IL-6 in glucose homeostasis and the anti-obesity effect of IL-6 warrant further research and apparently “weigh” against the potential of treating metabolic diseases with antibody-based therapies that neutralize IL-6.
References
Pedersen BK (2009) The diseasome of physical inactivity--and the role of myokines in muscle--fat cross talk. J Physiol 587:5559–5568
Pedersen BK, Akerström TCA, Nielsen AR, Fischer CP (2007) Role of myokines in exercise and metabolism. J Appl Physiol 103:1093–1098
Pedersen BK (2019) Physical activity and muscle–brain crosstalk. Nat Rev Endocrinol. https://doi.org/10.1038/s41574-019-0174-x
Kamimura D, Ishihara K, Hirano T (2003) IL-6 signal transduction and its physiological roles: the signal orchestration model. Rev Physiol Biochem Pharmacol 149:1–38
Mohamed-Ali V, Goodrick S, Rawesh A, Katz DR, Miles JM, Yudkin JS, Klein S, Coppack SW (1997) Subcutaneous adipose tissue releases interleukin-6, but not tumor necrosis factor-α, in vivo. J Clin Endocrinol Metab 82:4196–4200
De Rossi M, Bernasconi P, Baggi F, de Waal Malefyt R, Mantegazza R (2000) Cytokines and chemokines are both expressed by human myoblasts: possible relevance for the immune pathogenesis of muscle inflammation. Int Immunol 12:1329–1335
Pedersen BK, Febbraio MA (2008) Muscle as an endocrine organ: focus on muscle-derived interleukin-6. Physiol Rev 88:1379–1406
Hojman P, Brolin C, Nørgaard-Christensen N, Dethlefsen C, Lauenborg B, Olsen CK, Åbom MM, Krag TO, Gehl J, Pedersen BK (2019) IL-6 release from muscles during exercise is stimulated by lactate-dependent protease activity. Am J Physiol Endocrinol Metab 19. https://doi.org/10.1152/ajpendo.00414.2018
Rose-John S (2012) IL-6 trans-signaling via the soluble IL-6 receptor: importance for the proinflammatory activities of IL-6. Int J Biol Sci 8:1237–1247
Rose-John S, Heinrich PC (1994) Soluble receptors for cytokines and growth factors: generation and biological function. Biochem J 300:281–290
Hibi M, Murakami M, Saito M, Hirano T, Taga T, Kishimoto T (1990) Molecular cloning and expression of an IL-6 signal transducer, gp130. Cell 63:1149–1157
Heinrich PC, Behrmann I, Haan S, Hermanns HM, Uller-newen GM (2003) Principles of IL 6 type cytokine signaling and its regulating. Biochem J 20:1–20
Yang L, Wang L, Lin HK, Kan PY, Xie S, Tsai MY, Wang PH, Chen YT, Chang C (2003) Interleukin-6 differentially regulates androgen receptor transactivation via PI3K-Akt, STAT3, and MAPK, three distinct signal pathways in prostate cancer cells. Biochem Biophys Res Commun 305:462–469
Zhong Z, Wen Z, Darnell JE (1994) Stat3: a STAT family member activated by tyrosine phosphorylation in response to epidermal growth factor and interleukin-6 author. Science. 264:95–98
Barton BE (1997) IL-6: insights into novel biological activities. Clin Immunol Immunopathol 85:16–20
Damas P et al (1992) Cytokine serum level during severe sepsis in human IL-6 as a marker of severity. Ann Surg 215:356–362
Libermann TA, Baltimore D (1990) Activation of interleukin-6 gene expression through the NF-kappa B transcription factor. Mol Cell Biol 10:2327–2334
Zhang YH, Lin JX, Vilcek J (1990) Interleukin-6 induction by tumor necrosis factor and interleukin-1 in human fibroblasts involves activation of a nuclear factor binding to a kappa B-like sequence. Mol Cell Biol 10:3818–3823
Shimizu H, Mitomo K, Watanabe T, Okamoto S, Yamamoto K (1990) Involvement of a NF-kappa B-like transcription factor in the activation of the interleukin-6 gene by inflammatory lymphokines. Mol Cell Biol 10:561–568
Benatti FB, Pedersen BK (2015) Exercise as an anti-inflammatory therapy for rheumatic diseases - myokine regulation. Nat Rev Rheumatol 11:86–97
Unver N, McAllister F (2018) IL-6 family cytokines: key inflammatory mediators as biomarkers and potential therapeutic targets. Cytokine Growth Factor Rev 41:10–17
T.W., D. C (2000) Function of C-reactive protein. Ann Med 32:274–278
Wilund KR (2007) Is the anti-inflammatory effect of regular exercise responsible for reduced cardiovascular disease? Clin Sci (Lond) 112:543–555
Bruunsgaard H (2005) Physical activity and modulation of systemic low-level inflammation. J Leukoc Biol 78:819–835
Hotamisligil GS (2017) Inflammation, metaflammation and immunometabolic disorders. Nature 542:177–185
Hotamisligil GS, Erbay E (2008) Nutrient sensing and inflammation in metabolic diseases. Nat Rev Immunol 8:923–934
Sun K, Kusminski CM, Scherer PE (2011) Adipose tissue remodeling and obesity. J Clin Invest 121:2094–2101
Donath MY (2014) Targeting inflammation in the treatment of type 2 diabetes: time to start. Nat Rev Drug Discov 13:465–476
Steensberg A, Fischer CP, Keller C, Møller K, Pedersen BK (2015) IL-6 enhances plasma IL-1ra, IL-10, and cortisol in humans. Am J Physiol Metab 285:E433–E437
Steensberg A, Febbraio MA, Osada T, Schjerling P, van Hall G, Saltin B, Pedersen BK (2001) Interleukin-6 production in contracting human skeletal muscle is influenced by pre-exercise muscle glycogen content. J Physiol 537:633–639
Pedersen BK, Steensberg A, Fischer C, Keller C, Ostrowski K, Schjerling P (2001) Exercise and cytokines with particular focus on muscle derived IL-6. Exerc Immunol Rev 7:18–31
Pedersen BK, Steensberg A, Fischer C, Keller C, Keller P, Plomgaard P, Febbraio M, Saltin B (2003) Searching for the exercise factor: is IL-6 a candidate? J Muscle Res Cell Motil 24:113–119
Ostrowski K, Rohde T, Zacho M, Asp S, Pedersen B (1998) Evidence that IL-6 is produced in skeletal muscle during intense long-term muscle activity. J Physiol 508:949–953
Steensberg A, van Hall G, Osada T, Sacchetti M, Saltin B, Pedersen BK (2000) Production of interleukin-6 in contracting human skeletal muscles can account for the exercise-induced increase in plasma interleukin-6. J Physiol 529:237–242
Fischer C (2006) Interleukin-6 in acute exercise and training: what is the biological relevance. Exerc Immunol Rev 12:6–33
Helge JW, Stallknecht B, Pedersen BK, Galbo H, Kiens B, Richter EA (2003) The effect of graded exercise on IL-6 release and glucose uptake in human skeletal muscle. J Physiol 546:299–305
Pedersen BK (2013) Muscle as a secretory organ. Compr Physiol 3:1337–1362
Ullum H et al (1994) Bicycle exercise enhances plasma IL-6 but does not change IL-1α, IL-1β, IL-6, or TNF-α pre-mRNA in BMNC. Cytokines:93–97
Ostrowski K, Hermann C, Bangash A, Schjerling P, Nielsen JN, Pedersen BK (1998) A trauma-like elevation of plasma cytokines in humans in response to treadmill running. J Physiol 513:889–894
Starkie R, Ostrowski SR, Jauffred S, Febbraio M, Pedersen BK (2003) Exercise and IL-6 infusion inhibit endotoxin-induced TNF-alpha production in humans. FASEB J 17:884–886
Gershenwald JE, Fong YM, Fahey TJ, Calvano SE, Chizzonite R, Kilian PL, Lowry SF, Moldawer LL (1990) Interleukin 1 receptor blockade attenuates the host inflammatory response. Proc Natl Acad Sci U S A 87:4966–4970
Petersen AMW, Pedersen BK (2006) The role of IL-6 in mediating the anti-inflammatory effects of exercise. J Physiol Pharmacol 57:43–51
Steensberg A, Fischer CP, Keller C, Møller K, Pedersen BK (2003) IL-6 enhances plasma IL-1ra, IL-10, and cortisol in humans. Am J Physiol Endocrinol Metab 285:E433–E437
Wallenius V, Wallenius K, Ahrén B, Rudling M, Carlsten H, Dickson SL, Ohlsson C, Jansson JO (2002) Interleukin-6-deficient mice develop mature-onset obesity. Nat Med 8:75–79
Matthews VB, Allen TL, Risis S, Chan MHS, Henstridge DC, Watson N, Zaffino LA, Babb JR, Boon J, Meikle PJ, Jowett JB, Watt MJ, Jansson JO, Bruce CR, Febbraio MA (2010) Interleukin-6-deficient mice develop hepatic inflammation and systemic insulin resistance. Diabetologia 53:2431–2441
Kristiansen OP, Mandrup-poulsen T (2005) The good, the bad, or the indifferent? Diabetes 54:114–124
Pedersen BK, Febbraio M a (2012) Muscles, exercise and obesity: skeletal muscle as a secretory organ. Nat Rev Endocrinol 8:457–465
Kim H-J, Higashimori T, Park SY, Choi H, Dong J, Kim YJ, Noh HL, Cho YR, Cline G, Kim YB, Kim JK (2004) Differential effects of interleukin-6 and -10 on skeletal muscle and liver insulin action in vivo. Diabetes 53:1060–1067
Cai D, Yuan M, Frantz DF, Melendez PA, Hansen L, Lee J, Shoelson SE (2005) Local and systemic insulin resistance resulting from hepatic activation of IKK-β and NF-κB. Nat Med 11:183–190
Senn JJ, Klover PJ, Nowak IA, Zimmers TA, Koniaris LG, Furlanetto RW, Mooney RA (2003) Suppressor of cytokine signaling-3 (SOCS-3), a potential mediator of interleukin-6-dependent insulin resistance in hepatocytes. J Biol Chem 278:13740–13746
Kraakman MJ, Allen TL, Whitham M, Iliades P, Kammoun HL, Estevez E, Lancaster GI, Febbraio MA (2013) Targeting gp130 to prevent inflammation and promote insulin action. Diabetes Obes Metab 15:170–175
Ellingsgaard H, Ehses JA, Hammar EB, van Lommel L, Quintens R, Martens G, Kerr-Conte J, Pattou F, Berney T, Pipeleers D, Halban PA, Schuit FC, Donath MY (2008) Interleukin-6 regulates pancreatic α-cell mass expansion. Proc Natl Acad Sci 105:13163–13168
Ellingsgaard H, Hauselmann I, Schuler B, Habib AM, Baggio LL, Meier DT, Eppler E, Bouzakri K, Wueest S, Muller YD, Hansen AMK, Reinecke M, Konrad D, Gassmann M, Reimann F, Halban PA, Gromada J, Drucker DJ, Gribble FM, Ehses JA, Donath MY (2011) Interleukin-6 enhances insulin secretion by increasing glucagon-like peptide-1 secretion from L cells and alpha cells. Nat Med 17:1481–1489
Timper K, Dalmas E, Dror E, Rütti S, Thienel C, Sauter NS, Bouzakri K, Bédat B, Pattou F, Kerr-Conte J, Böni-Schnetzler M, Donath MY (2016) Glucose-dependent insulinotropic peptide stimulates glucagon-like peptide 1 production by pancreatic islets via interleukin 6, produced by α cells. Gastroenterology 151:165–179
Mauer J, Chaurasia B, Goldau J, Vogt MC, Ruud J, Nguyen KD, Theurich S, Hausen AC, Schmitz J, Brönneke HS, Estevez E, Allen TL, Mesaros A, Partridge L, Febbraio MA, Chawla A, Wunderlich FT, Brüning JC (2014) Signaling by IL-6 promotes alternative activation of macrophages to limit endotoxemia and obesity-associated resistance to insulin. Nat Immunol 15:423–430
Timper K, Denson JL, Steculorum SM, Heilinger C, Engström-Ruud L, Wunderlich CM, Rose-John S, Wunderlich FT, Brüning JC (2017) IL-6 improves energy and glucose homeostasis in obesity via enhanced central IL-6 trans-signaling. Cell Rep 19:267–280
Wunderlich FT, Ströhle P, Könner AC, Gruber S, Tovar S, Brönneke HS, Juntti-Berggren L, Li LS, van Rooijen N, Libert C, Berggren PO, Brüning JC (2010) Interleukin-6 signaling in liver-parenchymal cells suppresses hepatic inflammation and improves systemic insulin action. Cell Metab 12:237–249
Carey AL, Bruce CR, Sacchetti M, Anderson MJ, Olsen DB, Saltin B, Hawley JA, Febbraio MA (2004) Interleukin-6 and tumor necrosis factor-α are not increased in patients with type 2 diabetes: evidence that plasma interleukin-6 is related to fat mass and not insulin responsiveness. Diabetologia 47:1029–1037
Lang Lehrskov L, Lyngbaek MP, Soederlund L, Legaard GE, Ehses JA, Heywood SE, Wewer Albrechtsen NJ, Holst JJ, Karstoft K, Pedersen BK, Ellingsgaard H (2018) Interleukin-6 delays gastric emptying in humans with direct effects on glycemic control. Cell Metab 27:1201–1211.e3
Hirano T, Yasukawa K, Harada H, Taga T, Watanabe Y, Matsuda T, Kashiwamura SI, Nakajima K, Koyama K, Iwamatsu A, Tsunasawa S, Sakiyama F, Matsui H, Takahara Y, Taniguchi T, Kishimoto T (1986) Complementary DNA for a novel human interleukin (BSF-2) that induces B lymphocytes to produce immunoglobulin. Nature 324:73–76
Van Snick J et al (1988) cDNA cloning of murine interleukin-HP1: homology with human interleukin 6. Eur J Immunol 18:193–197
Knudsen SH, Pedersen BK (2015) Targeting inflammation through a physical active lifestyle and pharmaceuticals for the treatment of type 2 diabetes. Curr Diab Rep 15(1–9):82
Carey AL, Steinberg GR, Macaulay SL, Thomas WG, Holmes AG, Ramm G, Prelovsek O, Hohnen-Behrens C, Watt MJ, James DE, Kemp BE, Pedersen BK, Febbraio MA (2006) Interleukin-6 increases insulin-stimulated glucose disposal in humans and glucose uptake and fatty acid oxidation in vitro via AMP-activated protein kinase. Diabetes 55:2688–2697
Karstoft K, Pedersen BK (2016) Exercise and type 2 diabetes: focus on metabolism and inflammation. Immunol Cell Biol 94:146–150
Tsigos C, Papanicolaou DA, Kyrou I, Defensor R, Mitsiadis CS, Chrousos GP (1997) Dose-dependent effects of recombinant human interleukin-6 on glucose regulation. J.Clin.Endocrinol.Metab 82:4167–4170
Stouthard J et al (1995) Endocrinologic and metabolic effects of interleukin-6 in humans. Am J Phys 268:E813–E819
Lyngsø D, Simonsen L, Bülow J (2002) Interleukin-6 production in human subcutaneous abdominal adipose tissue: the effect of exercise. J Physiol 543:373–378
Petersen a MW, Pedersen BK (2005) The anti-inflammatory effect of exercise. J Appl Physiol 98:1154–1162
Febbraio MA, Hiscock N, Sacchetti M, Fischer CP, Pedersen BK (2004) Interleukin-6 is a novel factor mediating glucose homeostasis during skeletal muscle contraction. Diabetes 53:1643–1648
Harder-Lauridsen NM et al (2014) Effect of IL-6 on the insulin sensitivity in patients with type 2 diabetes. Am J Physiol Metab 306:E769–E778
Fève B, Bastard J-P (2009) The role of interleukins in insulin resistance and type 2 diabetes mellitus. Nat Rev Endocrinol 5:305–311
Sadagurski M, Norquay L, Farhang J, D’Aquino K, Copps K, White MF (2010) Human IL6 enhances leptin action in mice. Diabetologia 53:525–535
Xu Y, Zhang Y, Ye J (2018) IL-6: a potential role in cardiac metabolic homeostasis. Int J Mol Sci 19. https://doi.org/10.3390/ijms19092474
Petersen EW et al (2005) Acute IL-6 treatment increases fatty acid turnover in elderly humans in vivo and in tissue culture in vitro. Circulation 102:E155–E162
Van Hall G et al (2003) Interleukin-6 stimulates lipolysis and fat oxidation in humans. J Clin Endocrinol Metab 88:3005–3010
Lyngsø D, Simonsen L, Bülow J (2002) Metabolic effects of interleukin-6 in human splanchnic and adipose tissue. J Physiol 543:379–386
Nishimoto N et al (2005) Humanized anti – interleukin-6 receptor antibody treatment of multicentric Castleman disease. Blood 106:2627–2633
Päth G et al (2007) Human breast adipocytes express interleukin-6 (IL-6) and its receptor system: increased IL-6 production by beta-adrenergic activation and effects of IL-6 on adipocyte function. J Clin Endocrinol Metab 86:0–7
Al-Khalili L et al (2006) Signaling specificity of interleukin-6 action on glucose and lipid metabolism in skeletal muscle. Mol Endocrinol 20:3364–3375
Trujillo ME, Sullivan S, Harten I, Schneider SH, Greenberg AS, Fried SK (2004) Interleukin-6 regulates human adipose tissue lipid metabolism and leptin production in vitro. J Clin Endocrinol Metab 89:5577–5582
Ruderman NB, Keller C, Richard AM, Saha AK, Luo Z, Xiang X, Giralt M, Ritov VB, Menshikova EV, Kelley DE, Hidalgo J, Pedersen BK, Kelly M (2006) Interleukin-6 regulation of AMP-activated protein kinase: potential role in the systemic response to exercise and prevention of the metabolic syndrome. Diabetes 55:S48–S54
Hardie DG, Ross FA, Hawley SA (2017) AMPK - a nutrient and energy sensor that maintains energy homeostasis. Nat Rev Mol Cell Biol 13:251–262
Jeon SM (2016) Regulation and function of AMPK in physiology and diseases. Exp Mol Med 48:e245
Longo N, Frigeni M, Pasquali M (2016) Carnitine transport and fatty acid oxidation. Biochim Biophys Acta 1863:2422–2435
Tanaka T, Hishitani Y, Ogata A (2014) Monoclonal antibodies in rheumatoid arthritis: comparative effectiveness of tocilizumab with tumor necrosis factor inhibitors. Biologics 8:141-53. https://doi.org/10.2147/BTT.S37509
Shetty A et al (2014) Tocilizumab in the treatment of rheumatoid arthritis and beyond. Drug Des Devel Ther 8:349–364
Iking-Konert C et al (2014) Interleukin-6 inhibition as a potential therapeutic target in rheumatic diseases. Rheumatol. 73:269–276
Esser N, Legrand-Poels S, Piette J, Scheen AJ, Paquot N (2014) Inflammation as a link between obesity, metabolic syndrome and type 2 diabetes. Diabetes Res Clin Pract 105:141–150
Esser N, Paquot N, Scheen AJ (2014) Anti-inflammatory agents to treat or prevent type 2 diabetes, metabolic syndrome and cardiovascular disease. Expert Opin Investig Drugs 24:283–307
Ogata A, Morishima A, Hirano T, Hishitani Y, Hagihara K, Shima Y, Narazaki M, Tanaka T (2011) Improvement of HbA1c during treatment with humanised anti-interleukin 6 receptor antibody, tocilizumab. Ann Rheum Dis 70:1164–1165
Schultz O, Oberhauser F, Saech J, Rubbert-Roth A, Hahn M, Krone W, Laudes M (2010) Effects of inhibition of interleukin-6 signalling on insulin sensitivity and lipoprotein (A) levels in human subjects with rheumatoid diseases. PLoS One 5:e14328
Ursini F, Russo E, Ruscitti P, Giacomelli R, De Sarro G (2018) The effect of non–TNF-targeted biologics and small molecules on insulin resistance in inflammatory arthritis. Autoimmun Rev 17:399–404
Castañeda S, Remuzgo-Martínez S, López-Mejías R, Genre F, Calvo-Alén J, Llorente I, Aurrecoechea E, Ortiz AM, Triguero A, Blanco R, Llorca J, González-Gay MA (2018) Rapid beneficial effect of the IL-6 receptor blockade on insulin resistance and insulin sensitivity in non-diabetic patients with rheumatoid arthritis. Clin Exp Rheumatol
Otsuka Y, Kiyohara C, Kashiwado Y, Sawabe T, Nagano S, Kimoto Y, Ayano M, Mitoma H, Akahoshi M, Arinobu Y, Niiro H, Akashi K, Horiuchi T (2018) Effects of tumor necrosis factor inhibitors and tocilizumab on the glycosylated hemoglobin levels in patients with rheumatoid arthritis; an observational study. PLoS One 13:e0196368
Everett BM, Donath MY, Pradhan AD, Thuren T, Pais P, Nicolau JC, Glynn RJ, Libby P, Ridker PM (2018) Anti-inflammatory therapy with canakinumab for the prevention and management of diabetes. J Am Coll Cardiol 71:2392–2401
Choy E, Sattar N (2009) Interpreting lipid levels in the context of high-grade inflammatory states with a focus on rheumatoid arthritis: a challenge to conventional cardiovascular risk actions. Ann Rheum Dis 68:460–469
Wedell-Neergaard A-S et al (2018) Exercise-induced changes in visceral adipose tissue mass are regulated by IL-6 signaling: a randomized controlled trial. Cell Metab:1–12
Smolen JS, Beaulieu A, Rubbert-Roth A, Ramos-Remus C, Rovensky J, Alecock E, Woodworth T, Alten R (2008) Effect of interleukin-6 receptor inhibition with tocilizumab in patients with rheumatoid arthritis (OPTION study): a double-blind, placebo-controlled, randomised trial. Lancet 371:987–997
Genovese MC, McKay JD, Nasonov EL, Mysler EF, da Silva NA, Alecock E, Woodworth T, Gomez-Reino JJ (2008) Interleukin-6 receptor inhibition with tocilizumab reduces disease activity in rheumatoid arthritis with inadequate response to disease-modifying antirheumatic drugs: the tocilizumab in combination with traditional disease-modifying antirheumatic drug therapy study. Arthritis Rheum 58:2968–2980
Rao VU, Pavlov A, Klearman M, Musselman D, Giles JT, Bathon JM, Sattar N, Lee JS (2015) An evaluation of risk factors for major adverse cardiovascular events during tocilizumab therapy. Arthritis Rheum 67:372–380
Acknowledgments
We are grateful to Helga Ellingsgaard for providing us the opportunity to write this review. The Centre for Physical Activity Research (CFAS) is supported by a grant from TrygFonden.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
This article is a contribution to the special issue on Inflammation and Type 2 Diabetes - Guest Editor: Marc Y. Donath
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lehrskov, L.L., Christensen, R.H. The role of interleukin-6 in glucose homeostasis and lipid metabolism. Semin Immunopathol 41, 491–499 (2019). https://doi.org/10.1007/s00281-019-00747-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00281-019-00747-2