Abstract
Background
Among the types of pancreatic anastomosis used after pancreatoduodenectomy (PD), Blumgart type reconstruction has rapidly been distributed for its theoretical reasonableness, including secure tight adaptation of jejunal wall and pancreatic parenchyma without cause of parenchymal laceration. The clinical appropriateness of our modified Blumgart method was demonstrated by comparing to that of Kakita method.
Methods
Retrospective analysis of 156 patients underwent elective open PD, reconstructed former 78 patients with the Kakita method, utilizing a full-thickness penetrating suture for tight stump adhesion. The later 78 patients were treated with the modified Blumgart method, which involved clamping the pancreatic parenchymal stump by the jejunal seromuscular layers with horizontal mattress-type penetration sutures. Evaluated variables were the rate of pancreatic fistula (PF) and the length of postoperative hospital stay (POHS).
Results
The rate of ISGPF grade B + C PF was 29/78 (37.2 %) in the Kakita group and 16/78 (20.5 %) in the Blumgart group (P = 0.033). The median POHS for the Kakita group was 23 days, whereas that for the Blumgart group was 16 days (P < 0.001), one of the shortest value among Japanese high-volume centers. There was no perioperative intensive hemorrhage or deaths in either group.
Conclusion
A unique concept of Blumgart pancreatic anastomosis, i.e., utilizing the jejunum as an interstitial cushion to prevent pancreatic laceration at the knot site, has become realistic through a simple “one step” modification. This technique, also providing flexible handling space at main pancreatic duct anastomosis, should contribute to the improved PF prevention and shortening the POHS.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pancreatic fistula (PF) after pancreaticoduodenectomy (PD) is an unsolved problem [1–5], associating with high mortalities as nationwide survey from United State (including 68,643 cases) and Japan (17,634 cases), report 7.6 and 2.9 %, respectively [4, 6]. Recent trend in technical discussion among worldwide pancreatic surgeons seems to focus in two issues, that is the site of anastomosis (i.e., pancreaticojejunostomy vs. pancreaticogastrostomy [7–9]) and the way of anastomosis (i.e., two layer anastomosis with duct-to-mucosa anastomosis vs. Invagination method [10–12]). In addition to those methods, Japanese pancreatic surgeons have uniquely progressed in their technique since 1996 when Dr. Kakita invented an original tight adhesion anastomosis using the sutures that directly penetrate the full thickness of pancreatic parenchyma [13, 14]. Many Japanese institutes have adopted this Kakita method in the 2000s and this has been one of the major reconstruction methods after PD in Japan to date [15]. However, all conventional procedures including Kakita method possess a weak point that the ligation of the pancreatic parenchymal suture gives shear forces to pancreatic glands laceration during knot-tying.
The emerging technique of pancreatic anastomosis since 2010 s may be Blumgart method, the unique concept of which is utilizing the jejunum as an interstitial cushion to prevent the pancreatic laceration at the knot site [16, 17]. This method includes stitching the seromuscular layer of the jejunum in a horizontal mattress manner at both the anterior and posterior wall sides. Although the techniques in original method were rather complicated, the concept has been refined with simple and secure modifications by German group (by Kleespies et al. [18]) and Japanese group (by Fujii et al. [19]), and rapidly accepted and distributed to worldwide pancreatic surgeons.
We herein describe the technical details of our modified Blumgart pancreatic anastomoses, integrating both German and Japanese modifications. We also present the results of our series of 78 modified Blumgart pancreatic anastomoses by comparing with 78 control cases with Kakita method.
Methods
A total of 156 patients underwent elective open pancreaticoduodenectomy at the Department of Surgery, University Hospital of Tsukuba, Japan. The first half of the series includes 78 patients who were operated on before June 2010 using the Kakita method. The latter 78 cases were operated on after June 2010 and for whom the modified Blumgart pancreatic anastomosis technique was used. This case–control study was approved by the Ethics committee of University of Tsukuba Hospital (institutional reference number; H26-132).
Operative procedure
Pancreatic transections
Pancreatic transections were performed using sharp scalpels. Any bleeding and the stumps of small branches of the pancreatic ducts were ligated with 5–0 monofilament stitches using magnification glasses when needed.
Mode of reconstruction
The reconstruction was performed according to the modified Child’s method, where a single jejunal limb was brought up retrocolically to the right of the middle colic vessels. Pancreatic anastomosis was made 10–15 cm from the jejunal limb end, and hepaticojejunostomy [using continuous sutures with double-armed 4-0 PDS-II, (polydioxanone, Ethicon, Inc., Somerville, NJ)] was performed 5–15 cm distal to the pancreatic anastomosis. Gastrojejunostomy (using interrupted Gambee sutures with 4-0 PDS-II) was performed 40 cm away from the site of hepaticojejunostomy. Gastrojejunostomy was performed retrocolic to the left of the middle colic vessels, and the gastric antrum was fixed to the mesocolon, with the anastomosis located in an inframesocolic position.
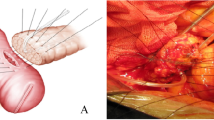
Kakita pancreatic anastomosis (Fig. 1)
A total of four penetrating sutures were placed in the seromuscular layer using double-armed absorbable sutures (3-0 PDS-II). The widths of penetrating sutures were 0.5 cm away from the pancreatic stump contour at both the anterior and the posterior sides, in order to securely cover the cut surface of the pancreas. The needles of posterior wall were then penetrated through the full thickness of the pancreatic parenchyma from posterior to anterior 1 cm from the cut margin. The main pancreatic duct (MPD) was anastomosed to the full thickness of the jejunum using 4 running sutures (double-armed 4-0 PDS-II), each takes responsibility for 90° of the circumference. A 6-Fr external stent tube (Akita Sumitomo Bake, Akita, Japan) was inserted only for cases of soft pancreatic tissue.
The technical flow of the Kakita’s tight adhesion pancreatic anastomosis. a A total of four vertical seromuscular penetrating sutures were placed at the antimesenteric site of the jejunal limb using double-armed 3-0 PDS-II. Needles from the posterior wall were then pierced through the full thickness of the pancreatic parenchyma from back to front 1 cm from the cut margin. b After completion of the MPD-jejunum anastomosis, Kakita sutures were tied for tight adhesion. As a matter of fact, sutures were tied using gentle force in order to avoid laceration of the pancreatic parenchyma, resulting in loose adhesion especially in the cases with soft pancreas
Subsequent to the completion of the MPD-jejunum anastomosis, the Kakita sutures were tied using gentle force in order to avoid laceration of the pancreatic parenchyma.
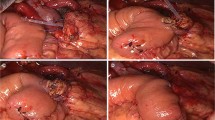
The modified Blumgart method (Fig. 2)
A total of three seromuscular horizontal mattress sutures with double-armed 3-0 PDS-II were placed at the posterior half, 1 cm away from the marked pancreatic contour. Both needles of the double-arm sutures are then pierced through the full thickness of the pancreatic parenchyma from posterior to anterior 1 cm from the cut margin. In this method, six penetrations were made using three double-armed 3-0 PDS-II. Needles from the central suture were placed cranial and caudal to the MPD in order to bind the MPD.
The modified Blumgart one-step pancreatic anastomosis. a A total of three seromuscular horizontal mattress sutures using double-armed 3-0 PDS-II were placed at the posterior semicircle. Both needles of the double arm were then pierced through the full thickness of the pancreatic parenchyma from back to front 1 cm from the cut margin. When tying a knot in the MPD anastomosis, towing the penetrating 3-0 PDS-II allowed approximation of the pancreas and jejunum conferring great help in the avoidance of MPD lacerations. b After completion of the MPD-jejunum anastomosis, needles of both horizontal mattress sutures were pierced through the anterior wall of the jejunum 1 cm from the pancreatic margin longitudinally to cover the seromuscular layer of the anterior semicircle. c Completion of the anastomosis by tying three firm sutures on the anterior serosal wall of the jejunum, without a fear of parenchymal laceration
The MPD anastomosis to the full thickness of the jejunal wall was made via the “quarter by four method,” an identical manner described in the Kakita method section. When tying the knot for the MPD anastomosis, towing the penetrating 3-0 PDS-II will approximate the pancreas and jejunum, conferring great help in avoiding MPD lacerations. This support could never be obtained using the conventional Kakita method.
After completion of the MPD-jejunum anastomosis, the needles of both Blumgart sutures were pierced through the anterior wall of the jejunum 1 cm away from the pancreatic margin longitudinally to cover the seromuscular layer of the anterior half. The anastomosis was completed by tying three firm sutures on the anterior serosal wall of the jejunum, without a fear of parenchymal laceration.
In both the Kakita and modified Blumgart methods, two prophylactic drainage tubes were placed at the superior and inferior sites of the pancreaticojejunostomy.
Postoperative management
The nasogastric tube was almost invariably removed the next morning. A central venous catheter or enteral nutrition was unnecessary, as the majority of the patients accepted liquid oral intake on POD 1 followed by liquid diet starting on POD 2–3, and solid food beginning on POD 4–7.
The volumes of the drained fluid were measured every day, and their amylase levels were monitored on POD 1, 3, and 5. The drainage tubes were removed on POD 4–5 for patients who were judged as ISGPF grade None or A. In possible PF grade B or C patients, regardless of the low concentration of drainage amylase levels, peritoneal tubes were replaced to new 18–22-Fr silicon tube (Fuji systems Co., Tokyo, Japan) by our own surgical team. The new drainage tubes were maintained in an open manner, and dirty fluids, if present, were intermittently lavaged with normal saline. We performed those in-hospital lavages for 3 weeks on average, for the patients with ISGPF grade B PF. Patients were discharged without transitional medical facilities when their activities had recovered to daily living.
Outcome measurements
The primary outcomes were the occurrence rate of PF according to the ISGPF criteria [3] and the length of postoperative hospital stay (POHS). The MPD size and tissue texture were focused to see the correlation with primary outcomes. Secondary outcome measurements were in-hospital mortality and overall postoperative morbidity. Complications other than pancreatic fistula were recorded according to the Clavien–Dindo categorization [20].
Statistical analysis
Differences between groups for categorical variables were examined using a χ 2 test or Fisher’s exact test, as appropriate, and a Student t test or Mann–Whitney test were used for continuous variables. All P values were considered statistically significant at 0.05 or less. All statistical analyses were performed using SPSS statistical analysis software package v. 21 (IBM SPSS Statistics, USA).
Results
Patients in both the Kakita and modified Blumgart groups had similar demographical backgrounds (Table 1). For operative variables, the surgical times were shorter in the modified Blumgart group (490 min) compared to the Kakita group (537 min, P = 0.013). Other variables, especially pancreatic texture and MPD size, were comparable, accounting that 56–58 % of the patients had a soft pancreatic texture, and 46–50 % of the patients were with small MPD size of ≤3 mm. The 58 % of the cases were operated by two staff surgeons (T.O. and S.H) under an agreed-upon standardized operating procedure, and the rest 42 % of the cases were operated by 26 residencies under the direction of the two staff surgeons.
Pancreatic fistula rate and POHS days (Table 2)
Pancreatic fistula of ISGPF Grades A, B, and C occurred in 13 (16.7 %), 26 (33.3 %), and 3 (3.8 %), respectively, in the Kakita group, and in 18 (23.1 %), 15 (19.2 %) and 1 (1.3 %), respectively, in the Blumgart group. Clinically relevant Grade B + C pancreatic fistula rate was 37.2 % in the Kakita group and 20.5 % in the modified Blumgart group (P ≤ 0.05). No patients experienced postoperative hemorrhage. Complications other than pancreatic fistula occurred 27/78 (34.6 %) in the Kakita group and 23/78 (29.5 %) in the modified Blumgart group. In total, the morbidity rate in the Kakita and the modified Blumgart groups was 65.4 and 60.3 %, respectively. There were no postoperative in-hospital deaths in either the Kakita or the modified Blumgart groups. The median and mean POHS values for the Kakita group were 23 and 26.4 days, respectively. These values were significantly shorter in the modified Blumgart group, 16 (P < 0.001) in Blumgart group and 19.6 days (P < 0.001) in Kakita group.
The effect of pancreatic condition on PF and POHS (Table 3)
PF (ISGPF B + C) in soft pancreatic texture patients were significantly higher than those of hard texture in both Kakita and Blumgart group. The advantage of modified Blumgart method was consistent even when the cases were restricted to soft pancreatic texture, demonstrating PF rate improvement from 25/45 (55.6 %) to 14/44 (31.8 %, P = 0.024). Other subgroups analysis did not reached the significant difference.
Discussion
The Blumgart type pancreatic anastomosis is rapidly distributed to worldwide pancreatic surgeons, since the long-lasting topic in pancreatic anastomosis that the secure adaptation of jejunal wall and pancreatic parenchyma without shear forces causing parenchymal laceration has been achieved by the jejunal clamping concept. The evidences of improved PF preventing power by this simple Blumgart type procedure have been accumulating.
The original Blumgart method, outlined in Fig. 3, may be regarded as a “two-step Blumgart procedure” because two sutures are used for completion of the jejunal clamping concept. The first suture is placed between the posterior wall of the jejunum and the anterior surface of the pancreatic parenchyma, and the second suture is placed using a different needle to catch the loop of the first suture and a bite of the anterior jejunal seromuscular layer. It should be noted, however, that the anterior wall is still exposed to shear force at the first knot-tying.
The original Blumgart two-step pancreatic anastomosis. a Four to six sutures penetrating full thickness of the pancreatic were started on the anterior wall of the pancreas, continuing with a seromuscular bite of the jejunum and then back through the pancreas from the posterior to anterior aspects. b After completion of the main pancreatic duct anastomosis, sutures were tied on the anterior wall of the pancreas. The anterior seromuscular jejunal sutures were made using a second suture, taking a bite of the pancreatic capsule, and passing the needle under the previously tied suture. c Completion of the anastomosis by tying second sutures on the anterior serosal wall of the jejunum
The modified Blumgart method adopted here may be called a “one-step Blumgart method,” as it uses one suture loop for anterior and posterior wall clamping and the concept is applied for both walls (Fig. 2). Although we have contrived this method without knowing previous publications, the principle of our modification is almost identical to that reported by German [18] and Japanese [19] groups. We utilize only three sutures with wider bites, and single tying of three Blumgart suture provides tight adaption of pancreatic parenchyma and jejunum. We are satisfied with the performance of modified Blumgart method since we can make tight tying with strong force, without a fear of parenchymal laceration.
The additional great merit of Blumgart method is its flexible support for secure MPD anastomosis. During the hand stitch process, an enough space between MPD and jejunum of each surgeons’s demand could be provided because the Blumgart sutures are not tied and free at this stage. This flexibility is especially useful when the MPD positioned very dorsal margin of the pancreas, which will be a problem in Cattell-Warren two layer method. When tying a knot of the MPD anastomosis, towing the penetrating Blumgart sutures allowed approximation of the pancreas and jejunum conferring great help in the avoidance of MPD lacerations. This support never has been acquired by Kakita method. An additional appealing point of our modification is in placing neighboring Blumgart sutures as close as possible, tracing the circular outlining at 1–1.5 cm wider than the pancreas cut shape (Fig. 2). This consideration will minimize the space between knots, which possibly be a source of parenchymal leakage.
The evidence supporting Blumgart pancreatic anastomosis has been accumulating (Table 4). The report from the original U.S.A. group that included 187 patients demonstrated a 6.9 % PF rate [17], which should be a good representation among the United States specialities, reporting 17.8–18.5 % [21, 22]. A German and Japanese groups have also reported decreased PF from 13 to 4 % [18] and from 36 to 2.5 % [19], respectively. The favorable tendency was also confirmed in our study, with the PF rate decreasing from 37.2 to 20.5 %. Moreover, when the cases were limited to the risky soft pancreatic textures, the modified Blumgart group showed lower PF rate (31.8 %) in comparison with Kakita group (55.6 %, P = 0.024). One may assume that our ISGPF B + C rate is relatively high in both the Kakita and the modified Blumgart groups. We think it may be owing to our postoperative care policy to prophylactically exchange drainage tubes without hesitation intending to minimize patient’s mortality [23]. As a result, we have been providing continuous 250 PD patients with zero mortality to date and low reoperation rate (6/156 = 3.8 %).
The POHS may be an alternative more objective parameter, which gives appropriate evaluations of surgical qualities. Although our 20.5 % PF ratio in modified Blumgart group is quite high, POHS of 16 days is one of the shortest among Japanese pancreatic specialities, reporting 21–27 days [6, 15, 19, 24, 25]. Although our POHS of 16 days seems still longer than those in the NY (10 days [17] ) and Munich (15 days [18] ) groups (Table 4), it should be mentioned that our POHS is a “go home discharge,” during which patients do not transit any medical facilities. The limitation of our study is being a historic control study; therefore, a better result in Blumgart group may attribute to the gaining experience. In any event, POHS of 16 days may be a benchmark after PD in the present day, since earlier discharge less than 14 days will run a risk of post-discharge severe complications including reoperation and mortality [26, 27].
Although the Blumgart method was originally published in Surgery of the Liver and Biliary Tract (3rd edn) in 2000 [16], the actual distribution has started with the publication by Kleespies et al. [18]. Accumulated modifications such as utilizing one suture for the anterior and posterior wall [18], and a wide U-shape suture that minimizes the space between the knots [19], make the concept of the Blumgart method more realistic. Although the debate regarding the benefits of pancreatic anastomosis between the duct-mucosa anastomosis vs. the invagination procedure is still on going [10, 21, 28], Blumgart type pancreatic anastomosis should be considered as an alternative arm to be compared, since this emerging spread technique is simple and supported by accumulating good PF preventing value.
References
Yeo CJ, Cameron JL, Sohn TA et al (1997) Six hundred fifty consecutive pancreaticoduodenectomies in the 1990 s: pathology, complications, and outcomes. Ann Surg 226:248–257
Buchler MW, Wagner M, Schmied BM et al (2003) Changes in morbidity after pancreatic resection: toward the end of completion pancreatectomy. Arch Surg 138:1310–1314
Bassi C, Dervenis C, Butturini G et al (2005) Postoperative pancreatic fistula: an international study group (ISGPF) definition. Surgery 138:8–13
Teh SH, Diggs BS, Deveney CW et al (2009) Patient and hospital characteristics on the variance of perioperative outcomes for pancreatic resection in the United States: a plea for outcome-based and not volume-based referral guidelines. Arch Surg 144:713–721
Harnoss JC, Ulrich AB, Harnoss JM et al (2014) Use and results of consensus definitions in pancreatic surgery: a systematic review. Surgery 155:47–57
Kimura W, Miyata H, Gotoh M et al (2014) A pancreaticoduodenectomy risk model derived from 8575 cases from a national single-race population (Japanese) using a web-based data entry system: the 30-day and in-hospital mortality rates for pancreaticoduodenectomy. Ann Surg 259:773–780
Poon RTP, Lo SH, Fong D et al (2002) Prevention of pancreatic anastomotic leakage after pancreaticoduodenectomy. Am J Surg 183:42–52
Duffas JP, Suc B, Msika S et al (2005) A controlled randomized multicenter trial of pancreatogastrostomy or pancreatojejunostomy after pancreatoduodenectomy. Am J Surg 189:720–729
Topal B, Fieuws S, Aerts R et al (2013) Pancreaticojejunostomy versus pancreaticogastrostomy reconstruction after pancreaticoduodenectomy for pancreatic or periampullary tumours: a multicentre randomised trial. Lancet Oncol 14:655–662
Kennedy EP, Yeo CJ (2011) Dunking pancreaticojejunostomy versus duct-to-mucosa anastomosis. J Hepatobiliary Pancreat Sci 18:769–774
Haane C, Mardin WA, Schmitz B et al (2013) Pancreatoduodenectomy–current status of surgical and perioperative techniques in Germany. Langenbecks Arch Surg 398:1097–1105
Peng SY, Wang JW, Lau WY et al (2007) Conventional versus binding pancreaticojejunostomy after pancreaticoduodenectomy: a prospective randomized trial. Ann Surg 245:692–698
Kakita A, Takahashi T, Yoshida M et al (1996) A simpler and more reliable technique of pancreatojejunal anastomosis. Surg Today 26:532–535
Kakita A, Yoshida M, Takahashi T (2001) History of pancreaticojejunostomy in pancreaticoduodenectomy: development of a more reliable anastomosis technique. J Hepatobilary Pancreat Surg 8:230–237
Satoi S, Toyokawa H, Yanagimoto H et al (2010) Is a nonstented duct-to-mucosa anastomosis using the modified Kakita method a safe procedure? Pancreas 39:165–170
Brennan M (2000) Pancreaticojejunostomy. In: Blumgart LH, Fong Y (eds) Surgery of the liver and biliary tract, 3rd edn. Saunders, New York, pp 1073–1089
Grobmyer SR, Kooby D, Blumgart LH et al (2010) Novel pancreaticojejunostomy with a low rate of anastomotic failure-related complications. J Am Coll Surg 210:54–59
Kleespies A, Rentsch M, Seeliger H et al (2009) Blumgart anastomosis for pancreaticojejunostomy minimizes severe complications after pancreatic head resection. Br J Surg 96:741–750
Fujii T, Sugimoto H, Yamada S et al (2014) Modified blumgart anastomosis for pancreaticojejunostomy: technical improvement in matched historical control study. J Gastrointest Surg 18:1108–1115
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240:205–213
Berger AC, Howard TJ, Kennedy EP et al (2009) Does type of pancreaticojejunostomy after pancreaticoduodenectomy decrease rate of pancreatic fistula? A randomized, prospective, dual-institution trial. J Am Coll Surg 208:738–747; discussion 747-739
Fong ZV, Ferrone CR, Thayer SP et al (2014) Understanding hospital readmissions after pancreaticoduodenectomy: can we prevent them?: a 10-year contemporary experience with 1,173 patients at the Massachusetts General Hospital. J Gastrointest Surg 18:137–144; discussion 144-135
Sugimoto M, Takahashi S, Gotohda N et al (2013) Schematic pancreatic configuration: a risk assessment for postoperative pancreatic fistula after pancreaticoduodenectomy. J Gastrointest Surg 17:1744–1751
Sakamoto Y, Kajiwara T, Esaki M et al (2009) Roux-en-Y reconstruction using staplers during pancreaticoduodenectomy: results of a prospective preliminary study. Surg Today 39:32–37
Tani M, Kawai M, Hirono S et al (2010) A prospective randomized controlled trial of internal versus external drainage with pancreaticojejunostomy for pancreaticoduodenectomy. Am J Surg 199:759–764
Kazaure HS, Roman SA, Sosa JA (2012) Association of postdischarge complications with reoperation and mortality in general surgery. Arch Surg 147:1000–1007
Hyder O, Dodson RM, Nathan H et al (2013) Influence of patient, physician, and hospital factors on 30-day readmission following pancreatoduodenectomy in the United States. JAMA Surg 148:1095–1102
Bassi C, Falconi M, Molinari E et al (2003) Duct-to-mucosa versus end-to-side pancreaticojejunostomy reconstruction after pancreaticoduodenectomy: results of a prospective randomized trial. Surgery 134:766–771
Acknowledgments
The authors thank Prof. Sata, from the Jichi medical University, Japan, for the useful discussion on the technique of modified Blumgart method.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Oda, T., Hashimoto, S., Miyamoto, R. et al. The Tight Adaptation at Pancreatic Anastomosis Without Parenchymal Laceration: An Institutional Experience in Introducing and Modifying the New Procedure. World J Surg 39, 2014–2022 (2015). https://doi.org/10.1007/s00268-015-3075-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-015-3075-8