Abstract
The distribution of reproductive success within societies is a key determinant of the outcomes of social evolution. Attempts to explain social diversity, therefore, require that we quantify reproductive skews and identify the mechanisms that generate them. Here, we address this priority using life history and genotypic data from >600 individuals in 40 wild groups of the cooperatively breeding white-browed sparrow weaver, Plocepasser mahali. We show that groups comprise up to six males and seven females, but within-group reproduction is completely monopolised by a single dominant male and female, while extra-group males sire 12–18 % of offspring. Strong within-group kin structure could frequently explain these monopolies, as subordinates had typically delayed dispersal from their natal groups and so frequently (1) lacked within-group outbreeding partners, and/or (2) stood to gain little from contesting dominant reproduction, being almost as related to the dominant’s young as they would have been to their own. Kin structure alone cannot account entirely for these monopolies, however, as they remained complete following the immigration of unrelated males and females. That subordinate females remain reproductively quiescent despite also showing comparable body condition to dominants, overlapping them substantially in age, and showing no evidence of elevated stress hormone levels raises the possibility that they exercise reproductive restraint due instead to a threat of action by dominants and/or deficits in offspring fitness that might arise if subordinates bred. Our findings highlight the complexity of the mechanisms that generate reproductive disparities in animal societies and the challenge of identifying them when skews are complete.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The distribution of reproductive success among same-sex individuals in society (reproductive skew) is a key determinant of the outcomes of social evolution. More extreme reproductive skews towards dominants are expected to promote the evolution of specialisations to breeding and non-breeding roles (Bourke 1999; O’Riain et al. 2000; Young and Bennett 2010), yield stronger intra-sexual selection for traits that facilitate dominance acquisition (Clutton-Brock et al. 2006; Rubenstein and Lovette 2009), and elevate relatedness among offspring, facilitating the evolution of cooperation (Hamilton 1964; Griffin and West 2002). Attempts to explain the diversity of social systems therefore demand that we explain the marked variation across and within species in reproductive skew (Keller and Reeve 1994). While reproductive skew theory has provided valuable insights into how certain processes are likely to impact skew, the sensitivity of their predictions to model assumptions has led to widespread recognition of the need to prioritise empirical research into the mechanisms that generate skew in natural populations (Magrath et al. 2004; Hodge 2009; Koenig et al. 2009).
The assumption that reproductive skew arises from intra-sexual conflict over reproduction is central to skew models (Keller and Reeve 1994; Johnstone 2000). However, it is now clear that other mechanisms frequently play a key role in generating skew (Wasser and Barash 1983; Koenig and Haydock 2004; Young 2009), and that the relevance of current theory to natural populations will therefore depend on the extent to which this is the case (Magrath et al. 2004; Hodge 2009; Koenig et al. 2009; Young 2009). A range of factors other than the actions of the dominant may reduce a subordinate’s expected fitness payoff from attempting to breed within the group, and may thereby favour the evolution of reproductive restraint among subordinates (Young 2009). While such factors are many and varied (e.g. subordinates being younger or in poorer body condition than dominants; Wasser and Barash 1983, or their young simply being outcompeted by those of the dominant; Hodge 2009; Young 2009), key among them are likely to be two consequences of the high within-group relatedness typical of societies in which offspring delay dispersal from their natal groups. First, in many such species subordinates of both sexes may lack access to unrelated breeding partners within their groups (e.g. Koenig et al. 1998; Cooney and Bennett 2000). Under these circumstances, inbreeding depression would be expected to devalue their expected payoff from attempting to breed (e.g. McRae 1996; see Koenig and Haydock 2004 for a review), potentially favouring reproductive restraint regardless of the actions of the dominant. Indeed, a large body of evidence now supports a key role for such inbreeding avoidance in generating skew in vertebrate societies (reviewed in Koenig and Haydock 2004).
A second general mechanism through which kin structure may (further) favour the expression of restraint is less frequently considered in empirical studies of vertebrates. Where subordinates are closely related to the dominant’s young, the subordinate’s expected inclusive fitness pay off from breeding within the group may be markedly reduced by indirect fitness costs arising from (1) the need to kill the dominant’s young to replace them with their own (e.g. Mumme et al. 1983; Young and Clutton-Brock 2006) or (2) costs to the dominants’ young arising from competition with the subordinate’s (Hodge 2009; Clutton-Brock et al. 2010). Such indirect costs of reproduction will be at their greatest in the many monogamous species where subordinates help to rear future generations of their parents’ young, as these subordinates may be as closely related to the dominant’s young as they would be to their own (Reeve and Keller 1996). As cooperative vertebrates appear to have evolved most readily from monogamous ancestors (Cornwallis et al. 2010; Lukas and Clutton-Brock 2012), these indirect fitness costs could ultimately prove of widespread importance in generating reproductive restraint among subordinates in vertebrate societies. Attempts to examine the mechanisms that yield reproductive skew in vertebrate societies should therefore examine the impact of kin structure on the subordinates’ (1) access to unrelated breeding partners, and (2) relatedness to the dominant’s young, as both factors could act in concert to favour the evolution of reproductive restraint regardless of any disruptive actions of dominants.
Intra-sexual competition between dominants and subordinates often clearly does play a key role in generating reproductive skew, though the mechanisms through which it acts remain the subject of considerable debate (Keller and Reeve 1994; Clutton-Brock et al. 2001; Magrath et al. 2004; Cant and Johnstone 2009; Koenig et al. 2009; Young 2009). In many species dominants employ a variety of tactics to actively disrupt subordinate reproduction, including subjecting them to chronic stress (e.g. Young et al. 2006), mutilation (e.g. Peeters and Higashi 1989), and destroying their eggs or young (e.g. Mumme et al. 1983; Young and Clutton-Brock 2006). However, where dominants are sufficiently capable of disrupting subordinate reproduction and/or subjecting breeding subordinates to additional costs (e.g. through attacks or evictions), selection may favour the evolution of complete reproductive restraint among subordinates, given the threat of such action from dominants (Johnstone and Cant 1999; Cant and Johnstone 2009; Young 2009). While there is little direct evidence to date that reproductive restraint among subordinate vertebrates is imposed by threats of dominant interference (Young 2009; Cant 2011; but see Wong et al. 2007; Young et al. 2008; Saltzman et al. 2009), this is doubtless due in part to the difficulty of testing this hypothesis directly, which would require forcing subordinates to breed at super-normal levels so as to elicit the contingent responses of dominants (MA Cant and AJ Young, unpublished data). That subordinates frequently show complete reproductive quiescence in the absence of overt interference from dominants (e.g. via stress induction or infanticide; Creel 2001) nevertheless provides compelling support for the view that subordinates frequently do exercise restraint for some reason. Where this is the case, a key priority now is to tease apart the relative importance of processes that may diminish the subordinate’s expected payoff from attempting to breed (thereby favouring restraint) (1) regardless of the likely actions of dominants (e.g. a lack of access to unrelated breeding partners or subordinates being young or in poor body condition) and (2) due to a threat of action by dominants.
Here, we investigate the patterns of reproductive skew in both sexes of the cooperatively breeding white-browed sparrow weaver, Plocepasser mahali, and the mechanisms that generate them. The sparrow weavers (genus Plocepasser) are a relatively little studied group of cooperatively breeding birds whose four species inhabit arid and semi-arid regions throughout sub-Saharan Africa (Sinclair and Ryan 2010). Unlike their better studied relatives, the cooperative but colonial sociable weaver (Philetairus socius, Covas et al. 2004), the sparrow weavers live in year-round territorial cooperatively breeding groups (Collias and Collias 1978; Lewis 1981). While their breeding systems have yet to be characterised using molecular techniques, early work across the geographical range of the white-browed sparrow weaver has revealed that they live in multi-male multi-female groups of up to 14 birds in which a single female appears to incubate the eggs and a dominant male closely guards her (Collias and Collias 1978; Lewis 1981; Ferguson 1988; Wingfield et al. 1991). Endocrine research has also shown that any reduced reproductive rate among subordinates could not be readily attributed to stress imposed by dominants, as subordinates do not show elevated circulating stress hormone levels (Wingfield et al. 1991). White-browed sparrow weavers also show well-developed cooperation, with group members contributing to the care of young, sentinelling, territory defence, and the weaving of their nest and roost chambers (Collias and Collias 1978; Lewis 1981). Advancing our understanding of the resolution of reproductive conflict in this species, and the context in which cooperation has evolved, now demands the application of genetic methods to characterise the breeding system. In this study, we therefore use genotypic and life history data from our longitudinal field study of >600 individuals from >40 cooperative groups of white-browed sparrow weavers in the South African Kalahari desert to: (1) quantify the extent of reproductive skew and extra-group parentage in both sexes; (2) investigate the extent to which within-group kin structure alone might account for the reproductive skews observed (by constraining subordinate access to unrelated breeding partners or yielding high subordinate relatedness to the dominant’s young); (3) investigate whether the low reproductive rates of subordinates may arise simply from them being younger or in poorer body condition than dominants; and finally (4) consider whether skew among females also appears to arise from active interference by dominants and the extent to which it is necessary to invoke a role for subordinate restraint in response to threat of dominant action. We then consider the applicability of current skew theory to this novel model system in the light of our findings.
Methods
Study population and field methods
Our study population comprises 40 cooperative groups of white-browed sparrow weavers, which defend small year round territories in an area of approximately 1.5 km2, west of the Korranaberg mountains in Tswalu Kalahari Reserve in the Northern Cape province of South Africa (27°16′S, 22°25′E). The study site lies in a shallow basin of Kalahari bushveld, sparsely vegetated with grasses (Eragrostis, Stipagrostis, Aristade, and Schmidtia species), bushes (principally Acacia mellifera, Rhigozum trichotomum, Grewia flava, and Lycium cinereu), and trees (principally Acacia erioloba, with occasional Acacia haematoxylon and Boscia albitrunca). Sparrow weaver social groups could be distinguished from one another as all group members typically foraged together each day, engaged in cooperative sentinelling, offspring care, weaving and territorial defence against neighbouring groups, and roosted together each night in individually woven chambers in a single tree or cluster of trees near the centre of their territory. All birds were fitted with a single metal ring and three colour rings for identification (under SAFRING license 1444), and records of group compositions were maintained with at least weekly visits. Adult males and females could be readily distinguished as males have dark brown beaks and females have paler flesh-coloured beaks, the distinction becoming apparent at around three to 6 months of age (Voigt et al. 2007; personal observation). Fledglings could be readily distinguished in the field by their more rounded beaks of intermediate colour (which also had a light area at the base of the tip) and fleshy yellow gape. The dominant bird of each sex was determined by weekly monitoring of key dominance-related behaviours as described in previous studies of this species (Collias and Collias 1978; Lewis 1981; Wingfield et al. 1991; Voigt et al. 2007). Specifically, dominants of either sex more frequently demonstrated within-group aggression such as the chasing and displacing of other individuals, the dominant pair regularly produced synchronised duet song together and the dominant male consistently produced solo song at dawn throughout the breeding season. The lay dates of all eggs and hatch dates of chicks were determined via nest checks every 1 to 2 days.
During the study period (October 2007 to April 2011), our 40 focal groups laid zero to six clutches (mode 3) per group per year, resulting in zero to eight (mode 2) fledglings per group per year, all during extended breeding seasons coinciding with the southern summer (October to April), in which clutch initiation rates peaked after heavy rainfall. Clutches typically comprised just two eggs (154 of 223 clutches of known size at completion), but ranged in size from one to (very rarely) four eggs. Incubation lasted 14–19 days from the laying of the first egg and the degree of synchrony of hatching varied greatly, with all chicks hatching 0–3 days apart. Once the chicks were large enough (>5 days after hatching), a small nick was made in the wing vein using a 26-g needle, to release a small (<25 μl) whole blood sample that was drawn into a capillary tube and diluted in 96 % ethanol for long-term storage for genetic analysis. To ensure complete genetic coverage of adults, any unringed birds in the population were caught as soon as possible for the fitting of rings and the drawing of a small blood sample (using the same method) for genotyping. Blood samples were stored at a minimum dilution of 1 part blood to 20 parts ethanol. Adults were caught individually at night from their roost chambers, using a pair of custom-built capture bags on the end of poles. After processing, the birds were returned to their roost chambers. All protocols were approved by the University of Pretoria ethics committee and complied with regulations stipulated in the Guidelines for the use of Animals in Research.
Microsatellite markers and genotyping
We genotyped 607 white-browed sparrow weavers at 13 microsatellite loci (Table 1). The markers comprised seven developed for a Namibian population of this species (McRae and Amos 1999), three grey-capped sociable weaver (Pseudonigrita arnaudi) markers (McRae et al. 2005), and three EST-derived markers from Dawson et al. (2010). Observed and expected heterozygosities and predicted null allele frequencies were calculated using CERVUS v3.0.3 (Kalinowski et al. 2007) and are presented in Table 1. Tests for departures from Hardy–Weinberg Equilibrium (HWE) and for linkage disequilibrium were performed in GENEPOP v3.4 (Rousset 2008). No markers were found to deviate from HWE (all p > 0.14), nor did any pair of markers show patterns consistent with linkage disequilibrium (all p > 0.05) when tested on 24 presumed unrelated individuals from the population (breeding pairs from the first year of the study). CERVUS identified a high frequency of null alleles at locus GCSW8 (18 %) and GCSW31 (11 %) and so these markers were dropped from the marker set ultimately used for parentage assignment, along with GCSW57 which showed a disproportionately high genotyping error rate due to inconsistent amplification of alleles and scoring difficulty. We used the genotypic data from the remaining ten markers in the work outlined below (Table 1). Multiplexes were designed using Multiplex Manager v1.0 (Holleley and Geerts 2009) to check for potential incompatibilities (dimerisation, hairpins, etc.) between primer sets. DNA was extracted using ammonium acetate precipitation as detailed in Nicholls et al. (2000). PCRs were carried out in 10-μl reactions using Qiagen PCR Mastermix (Qiagen Inc.) at the following temperature profile: 95 °C for 5 min, followed by 35 cycles of 94 °C for 30s, 52 °C for 90s, and 72 °C for 60 s, with a final step of 72 °C for 10 min. Samples were genotyped on an ABI 3130xl Capillary Sequencer (Applied Biosystems, USA) and allele sizes scored using Genemapper v3.7. Genotyping error rates were calculated by the repeat genotyping of 20 % of samples (from DNA extraction) and comparing their genotypes to the original samples. Genotyping repeatability was estimated to be 99 %.
Quantification of extra-group parentage and reproductive skew
To quantify the extent of extra-group parentage and reproductive skew, we used insights drawn from two different approaches: (1) parentage assignments made using the program COLONY (see below), and (2) simple exclusion analyses (in which certain candidate parents can be excluded as true parents of an offspring on the basis of their genotype mismatching that of the offspring at a threshold number of loci). Complementing COLONY-based parentage assignments data with information from exclusion analyses is a valuable approach, as it can allow clear inferences to still be drawn from offspring that COLONY cannot assign parents with high confidence. Precisely how we utilised the COLONY and exclusion data in each of the analyses is explained in the “Results” section alongside the presentation of the relevant findings.
Of the 422 offspring that hatched in the 241 breeding attempts conducted by our core 36 study groups, we were able to successfully genotype 329 (78 %). Occasionally offspring could not be genotyped because they died before reaching the age at which sampling was possible; the vast majority of them died in the first half of the nestling period and so will not have contributed to reproductive success anyway. Of the offspring that successfully fledged from these breeding attempts, we had genotyped fully 96 % (192 of 200).
Parentage assignment using COLONY
We used the program COLONY v2.0 (Wang and Santure 2009) to assign parentage to 385 genotyped offspring. COLONY assigns parents using a likelihood method to identify full and half sibships from within the sample of candidate offspring, which subsequently allows the parental haplotypes compatible with those sibships to be constructed. Parentage is assigned to sampled individuals whose haplotypes are consistent with the corresponding sibship (Wang and Santure 2009). This sample of 385 comprised 329 offspring of known natal origin (from 201 clutches laid in 36 groups) that had hatched from eggs in a monitored breeding attempt, in addition to 56 offspring whose origin was unknown because they were first discovered as fledglings after a break in monitoring. These latter individuals could have been either natal to the group in which they were first observed, or recent immigrants from elsewhere. We included these additional 56 because many are likely to have been siblings of the other 329 offspring, and larger sibships in the sample will improve the power that COLONY has to assign parentage (Wang and Santure 2009).
To create the list of candidate mothers for each offspring, for the 329 offspring of known natal origin we included only females within their social group at the time of egg laying, and for the 56 offspring of unknown natal origin we included all sampled females in the population that could have been alive during the window in which they could have been laid. Extra-group maternity in cooperative breeders is rare (e.g. Richardson et al. 2001), and so for offspring of known natal origin this was considered the most parsimonious manner in which to parameterize the COLONY simulations. We found no evidence that this was inappropriate, with the majority of offspring being confidently assigned to a within-group female, and the small proportion of unassigned offspring being compatible with a within-group female by exclusion (see below). To create the list of candidate fathers for each offspring, we included all sampled males in the population that could have been alive at the time the offspring was laid. For a full description of the methods used to create the candidate parent sets see Electronic supplementary material (ESM).
To assign parentage, we performed three replicate runs with the following COLONY parameters: medium run length, full-likelihood estimation with high precision, and allowing for both male and female polygamy. Each run was performed with a different random number seed, with the expectation that true relationships would be reliably recovered in multiple runs, irrespective of the starting configuration of the search algorithm (see Wang 2004). Conversely, spurious assignments may only occur in single runs and can thus be identified as such by their lack of consistency across runs. We specified a 1 % error rate per locus for both the allelic dropout and stochastic (e.g. mutation, false alleles, mis-typing) error types, as parameterized from our repeat genotyping/error rate calculations. Allele frequencies for the COLONY run were calculated from the sample, and updated based on the inferred relationships.
Assignments for both paternity and maternity were only accepted if they met one of the following criteria: (1) assignments where all three runs produced the same parent with a probability of 95 % or more; (2) those where the same parent was recovered three times, with two of the runs assigned above the 95 % probability threshold; (3) assignments where two runs produced a consensus assignment, both above the 95 % threshold, but where the third run failed to converge on any candidate parent. The latter can occur due to the choice of random number seed for a run, which determines the path of the random search algorithm seeking the best parental configuration, and can thus occasionally fail to find the best (correct) parent. We refer to assignments meeting any of these criteria as being assigned at the ‘95 % consensus threshold’. In any case where there was a conflict in assignment across runs (i.e. >1 individual listed as the most likely candidate), that relationship was considered unassigned, irrespective of the probabilities associated with the relationships.
Analyses of kin structure
We used the program COANCESTRY v1.0 (Wang 2011) to calculate pairwise moment estimators of relatedness between all pairwise combinations of individuals for use in the analyses of kin structure. The statistical methods for each of the tests conducted are outlined in the relevant figure legends.
Spatial analysis
Distance between territories was calculated as the Euclidean distance between territory centres of social groups, where territory centres are defined as the GPS co-ordinates of the roost trees. Where >1 roost tree was regularly used, the average co-ordinates for these trees were taken and used in subsequent calculations.
Statistical analysis
Models for the egg survival, clutch size, and body condition analyses (see below) were fitted using the package ‘lme4’ (Bates et al. 2012) in the statistical programme R v2.15.3 (R Development Core Team 2013). Support for models by the data was evaluated using an information theoretic approach where models were ranked by AICc. Models within Δ6 units AICc of the top model were considered to be equally well supported (Richards 2008, Richards et al. 2011). However, we also applied the ‘nesting rule’ (Richards 2008) which removes models from the Δ6 AICc set that are more complex versions of nested models higher up in the Δ6 set with lower AICc values (i.e. better support). Application of this rule serves to avoid the retention of unnecessarily complex models (Richards 2008, Richards et al. 2011, see also Arnold 2010). For each analysis, model data and AICc ranking are provided in ESM Tables S1–S4.
Egg survival rate and clutch size analyses
To investigate whether there was any evidence of reproductive conflict when subordinate females did have within-group outbreeding partners, we investigated whether, under these circumstances, clutches differed in size or eggs showed a reduced probability of surviving to hatching. To test for clutch size effects, we used data from 87 clutches from 23 groups for which we knew both the clutch size and whether all subordinate females within the group had access to within-group outbreeding opportunities, and for hatching probability we used the subset of 83 clutches (n = 23 groups) for which we knew the exact proportion of successfully hatched eggs. To investigate the effect of within-group outbreeding opportunities for subordinate females on clutch size, we used a generalized linear mixed model (GzLMM) with a Poisson error structure and log link, with number of eggs fitted as a response. To then investigate the effect of subordinate outbreeding opportunities on the probability of eggs surviving to hatching, we used a GzLMM with a binomial error structure and logit link, with a two-column vector as a response specifying the counts of the number of eggs that did, and did not, hatch, respectively. For both models, we also fitted group size as a covariate (to control its potential effects on each), and specified a random intercept term for social group to control for repeated measures within groups.
Body condition analyses
Finally, we investigated whether reproductive quiescence among subordinate females could be due to them having poorer body condition than their dominant females. To do this, we calculated the Scaled Mass Index (SMI) of Peig and Green (2009) to compare body condition among dominant and subordinate females. The SMI scales the mass of all individuals in a sample to that expected if they were all of identical skeletal size, making condition measures comparable among all individuals.
Females: We used masses and tarsus sizes taken from 596 capture records of 155 females from 38 social groups between the months of October and March inclusive (the peak breeding season). We scaled the masses of all females to the mean tarsus length of our sample (24.5 mm), using an SMA slope of 2.57 calculated as the ratio between the slope estimate from a Type I regression of log-transformed mass on tarsus, and the correlation coefficient between mass and tarsus (details in Peig and Green 2009). We then used this scaled mass index as a proxy measure for body condition. Males: We repeated the analysis as above for females, using 809 records from 156 males from 40 social groups captured between October and March. All individuals were scaled to the mean male tarsus length of 24.77 mm using an SMA slope of 2.4 (details above). To then test for differences in body condition between dominants and subordinates, for each sex we used a mixed model with body condition as the response term, and dominance status (dominant or subordinate), a two-level factor specifying whether the measurement was taken in the first or second half of the breeding season, and their interaction as fixed effects. We specified individual ID and social group ID as random intercept terms in the model to control for repeated measures of each.
Results
Group compositions
Social groups varied markedly in size from 2 to 12 birds and comprised approximately equal numbers of males and females (Fig. 1a). Groups were headed by a single female who was behaviourally dominant to all other females (the ‘dominant female’), and a single male who was behaviourally dominant to all other males, closely associated with the dominant female, initiated and joined duets with the dominant female, consistently sang dawn song during the breeding season and was typically the last bird to enter the roost chambers at dusk (the ‘dominant male’). New groups were founded by mixed-sex pairs or trios (n = 16 new groups were founded during the study period, invariably by males and females from different source groups), and grew in size via the delayed dispersal of offspring of both sexes into subordinate positions within the group and, less frequently, the immigration of birds of both sexes. Over the course of this study, 38 % (13/34) of groups had an immigrant subordinate present for at least one breeding attempt. The mean number of breeding attempts containing an immigrant subordinate was 17.5 %, expressed as the grand mean of each group’s percentage of breeding attempts where an immigrant subordinate was present (n = 34 groups, range 0–57 %).
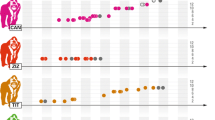
a Frequency distribution of the numbers of males and females present in the group at the time of laying of the 180 genotyped breeding attempts from 34 groups. b Reproductive skew among males and females within groups and the proportion of extra-group paternity. The estimate for the incidence of EGP presented on the paternity bar is the mean of the upper (18.2 % EGP) and lower (11.6 % EGP) estimates derived from our COLONY analyses (see “Results” section)
Observations of copulations were rare. Of 16 observed full copulations in which we were certain of the identities of both parties, the majority (10; 62.5 %) involved the dominant male, who was only ever seen mating with his dominant female (n = 6 groups). The remaining six copulations (37.5 %) involved subordinate immigrant males; three were matings with dominant females (n = 3 groups), and three were matings with the same single subordinate female in one particular season. This female was still associated with her natal group at the time, but was attempting to found a new group with one of the males that she mated with (as evidenced by weaving in discrete trees on the edge of her natal group’s territory and frequently ranging separately in that area). She was not known to lay eggs during this period, and every clutch within her natal group could be attributed to the dominant female. All of these documented interactions were between birds that were likely to have been unrelated.
The distribution of reproductive success among females
For 144 breeding attempts by 39 groups we were able to identify the female who either incubated the clutch or brooded the chicks, either because she was the only female in the group or because her incubation or brooding was confirmed visually or by elimination (during the day, the incubating bird could be identified by elimination of those seen elsewhere, and during the night, the incubating or brooding bird could be identified by elimination of those caught from other roost chambers). Each breeding attempt was consistently incubated and/or brooded by the same female, and this one female consistently incubated and/or brooded every successive breeding attempt within her group for which information was available, without interruption, until the end of monitoring or her permanent disappearance from the group (at which point another female took over this apparent breeding position). Invariably, when female dominance status was confirmed with behavioural observations, this single breeding female was also found to be the dominant female (confirmed for 35 females across 34 groups).
To calculate genetic metrics of maternity, we restricted our attention to the 270 genotyped offspring (from 168 clutches in 35 groups) for whom every female present in the focal group was individually identifiable (colour ringed) at the time of laying and had since been genotyped. COLONY assigned maternity at the 95 % consensus threshold for the vast majority of these to a single within-group female (260 offspring, 96.3 %), and the remaining 10 offspring that couldn’t be assigned at this high level of stringency also had genotypes that perfectly matched the haplotype of just one within-group female, to whom we therefore manually assigned maternity. As such, there was no evidence of egg dumping by extra-group females. Of these 270 offspring with assigned maternity, 263 were laid during the tenure of a known breeding female (invariably the group’s dominant female; see above). Without exception, maternity for these 263 offspring had been genetically assigned to their group’s dominant breeding female. The remaining seven offspring hatched during a time where behavioural data on dominance status of within-group females were incomplete, and therefore, these offspring were excluded from our analysis of within-group skew.
The distribution of reproductive success among males
To calculate genetic metrics of paternity, we restricted our attention to the 292 offspring for whom every male present in the focal group was individually identifiable (colour ringed) at the time of laying and had since been genotyped.
Extra-group paternity
Of the 292 offspring considered, COLONY assigned 264 a father at the 95 % consensus threshold, from a candidate set that included all potential fathers in the entire study population (see methods). Of these 264 offspring with assigned fathers, 38 (14.4 %) were assigned an extra-group father. Of the remaining 28 unassigned offspring, 15 had genotypes that mismatched all within-group males at one or more loci and so are likely to have been sired by extra-group males (this exclusion approach could overestimate EGP as single locus genotyping errors or mutations could be yielding the false exclusion of within-group fathers). Combining these two figures yields an estimate of 18.2 % EGP (38 + 15/292).
To yield the most conservative COLONY-based estimate of the EGP rate, we then checked whether the genotypes of any of the 38 offspring that COLONY had assigned to extra-group fathers (see above) were also consistent with paternity by a within-group male. Indeed, the within-group dominant male offered a perfect haplotype match to 9 of these 38 offspring (once the maternal genotype had been accounted for), and so we conservatively assigned these manually as cases of within-group paternity. In one other case, a within-group subordinate male was found to offer a perfect paternity match for an offspring, but as no subordinate was ever unambiguously found to gain within-group paternity (see below), we maintained the COLONY assignment of an extra-group father in this case. This left a total of 29 offspring assigned to extra-group males from COLONY’s 264 assignments. Of the 28 offspring unassigned by COLONY, just 5 mismatched every within-group male at more than one locus (now a conservative estimate of the incidence of EGP by exclusion as some of these permitted single mismatches will actually reflect extra-group paternity). Combining these figures then yields a conservative estimate of 11.6 % EGP (29 + 5/292). Using our COLONY assignments in concert with the exclusion approach therefore suggests that the true rate of EGP is between 11.6 and 18.2 % of offspring (13.3 to 20 % of clutches). These estimates lie within the range of EGP estimates yielded by the application of exclusion methods alone (without the use of COLONY-based assignment methods; see “ESM”).
Within-group reproductive skew among males
To assess the extent of within-group skew among males, we focused our attention on the parentage of the 204 offspring that COLONY had assigned to within-group males at the 95 % consensus level, whose clutches were laid at a time when (1) all within-group males were ringed and genotyped; (2) we were certain from behavioural observations of the identity of the dominant male (n = 30 dominant males across 27 groups; in three groups we had observed dominance turnover). Exclusion analyses: Of these 204 offspring, 172 had genotypes that were consistent only with paternity by the within-group dominant male (after accounting for the maternal genotype). Of the remaining 32 offspring, 29 had genotypes consistent with paternity by more than one within-group male, though this always included the dominant male. The remaining three offspring had genotypes consistent with paternity by a subordinate male, but they only mismatched the genotype of the dominant male at one locus, and so could reflect dominant male paternities with a single locus genotyping error or mutation. COLONY analyses: All 204 offspring were assigned by COLONY at the 95 % consensus level to the resident dominant male. This suggests that the three cases of possible subordinate paternity identified by exclusion were the result of genotyping errors that COLONY corrected using sibship information.
The role of kin structure in generating skew
Inbreeding avoidance
While dominant breeding pairs are typically unrelated to each other (Fig. 2), the pairwise relatedness values for all potential mixed-sex within-group pairings by subordinates were markedly higher (paired t = −5.5, df = 67.2, p < 0.0001, n = 31 groups; Fig. 2). Indeed, our life history and genetic data suggest that in 75 % of breeding attempts, all adult subordinate females lacked unrelated males within their group (Fig. 3a), and that in 67 % of breeding attempts, all adult subordinate males were closely related to the dominant female (their only possible within-group breeding partner, given the skew among females; Fig. 4a). However, in 25 % of breeding attempts our life history data suggest that one or more subordinate females did have unrelated males within their groups (Fig. 3a), and our genetic data confirm that they were just as (un)related to these males as dominant breeding pairs were to each other (Figs. 2 and 3b). Similarly, in 33 % of breeding attempts our life history data suggest that one or more subordinate males were unrelated to the dominant female (Fig. 4a), and our genetic data confirm that they were just as (un)related to her as dominant breeding pairs were to each other (Figs. 4b and 2). In 5 % of cases, subordinate males were unrelated to one or more immigrant subordinate females. Subordinates of both sexes could also have circumvented any absence of within-group outbreeding partners simply by mating indiscriminately with opposite sex birds in their nearest neighbouring groups. Such pairings would have been just as outbred as those among dominant breeding pairs (t = −0.06, df = 64.9, p = 0.95, n = 36 groups; Fig. 2). Neighbouring territories abutted each other, and the centres of nearest neighbouring territories were just 117.36 ± 7.52 m apart (n = 36 territories).
The levels of relatedness between different combinations of potential mating partnerships (dominant breeding pairs, subordinates mating within group, and subordinates mating with members of their nearest neighbouring group). The bars present data for our 36 study groups on 1, January 2011. From left to right they present: (1) mean relatedness between the groups’ dominant breeding pairs; (2) mean relatedness between adult (birds in at least their second season of life) subordinates and all opposite sex adults within their group (an average value was calculated for each adult subordinate in the group on this date, and these were all averaged per group to yield a data set of single values for each of the n = 31 groups that contained subordinate adults); (3) the mean levels of relatedness between adult subordinates and all of the opposite sex birds in their nearest neighbouring group (again, an average value was calculated per subordinate and these were averaged across all subordinates per group to yield a data set of single values for each of n = 31 groups). Bars present means ± standard errors (SE), and the p values reflect the outcome of Tukey’s HSD pairwise tests between groups
a The frequency with which life history data suggests that subordinate females (SFs) had unrelated within-group males (WGMs). The bars reflect the number of breeding attempts (of the 112 monitored in which the group contained one or more SFs at the time of laying) in which our life history data suggests that (1) all adult SFs in the group lacked access to unrelated WGMs (they were all still within their natal group and no males had immigrated since any of them had fledged), or (2) one or more adult SFs in the group had access to unrelated WGMs (one or more of the SFs was an immigrant and/or one or more males had immigrated since one or more of the SFs had fledged). b The relatedness between SFs and their WGMs in scenarios in which they were classified (see Fig. 3a) as either (1) lacking unrelated WGMs (for each such breeding attempt, the average relatedness of all adult SFs to all WGMs was calculated, and then these values were averaged per group to yield single values for the n = 21 groups in which this context arose), or (2) having unrelated WGMs [for each such breeding attempt, the average relatedness of those SFs (with unrelated WGMs) to their putative unrelated WGMs was calculated, and then these values were averaged per group to yield single values for the n = 9 groups in which this context arose]. The bars present means ± SE
a The frequency with which life history data suggests that subordinate males (SMs) were unrelated to their dominant female (DF; their only possible breeding partner, given the skew among females). The bars reflect the number of breeding attempts (of the 116 monitored in which the group contained one or more SMs at the time of laying) in which our life history data suggests that (1) all adult SMs in the group were closely related to their DF (because they were all still within their natal group and the DF had not changed since their fledging), or (2) one or more adult SMs in the group were unrelated to their DF (because one or more of the SMs was an immigrant and/or the DF had changed since one or more of the SMs had fledged). b The relatedness between SMs and their DFs in scenarios in which they were classified (see Fig. 4a) as either (1) being closely related to her (for each such breeding attempt, the average relatedness of all SMs to their DF was calculated, and then these values were averaged per group to yield single values for the n = 21 groups in which this context arose), or (2) being unrelated to their DF [for each such breeding attempt, the average relatedness of those SMs (deemed unrelated to their DF) to their DF was calculated, and then these values were averaged per group to yield single values for the n = 15 groups in which this context arose]. The bars present means ± SE
Relatedness to the dominant’s young
In 88 (67 %) of the 132 breeding attempts in which one or more adult subordinates (of either sex) were present and of known origin, all such adult subordinates were within their natal group and were helping to rear the offspring of the dominants that reared them (Fig. 5a). These subordinates were therefore closely related to the dominant’s young (Fig. 5b; r = 0.415 ± 0.024), though significantly less so than parents are to their own young in our population (paired t test, t = −3.35, df = 21, p = 0.005). However, in 12 (9 %) of these 132 breeding attempts, all adult subordinates were still within their natal group, but one or more of the dominants that had reared them had been replaced since the fledging of one or more of the subordinates (Fig. 5a). These subordinates that had experienced dominant turnover since fledging were therefore significantly less related to the young that they were rearing than those that had not (unpaired t test, t = 4.85, df = 7.5, n = 22,5 groups, p = 0.001; Fig. 5b). In the remaining 32 (24 %) of these 132 breeding attempts, one or more subordinates had immigrated in to the group (Fig. 5a), and these immigrants were significantly less related to the dominants’ young than those natal subordinates who had experienced dominance turnover (unpaired t test, t = −3.1, df = 10.5, n = 13,5 groups, p = 0.01; Fig. 5b).
Subs subordinates, Imm immigrant, DT dominance turnover since one or more of the subordinates fledged. a The frequency distribution of breeding attempts (from a total of 132 breeding attempts in 34 groups in which all adults subordinates were of known origin), in which (1) [‘No DT’] all adult subordinates were within their natal groups and helping to rear the offspring of the dominants that reared them (their putative parents, subject to EGP), (2) [‘DT’] all adult subordinates were within their natal groups, but one or more had experienced one or more dominant replacements since fledging (turnover of one or more of their putative parents), and (3) [‘1+ Subs Imm’] one or more adult subordinates had themselves immigrated in to the group. b The corresponding levels of relatedness between the focal adult subordinates in question and the brood that they were rearing in that breeding attempt for each of the classes presented in Fig. 4a. In each case, the average relatedness of the subordinate in question to the brood being rearing was calculated, all such values for a given breeding attempt were averaged, and the values for all breeding attempts per group were averaged to yield a data set of group means for each bar. The sample size for each bar (n = social groups) is displayed above the bar. The bars present means ± SE
Alternative explanations for reproductive restraint among subordinates
There was no support in the data for an effect of dominance status on male mass (October to March, mean (±SE) dominant male scaled mass = 45.26 g (±0.32), subordinate scaled mass = 45.12 g (±0.27). The best supported model contained only an effect of time, where males increase in condition as the season progresses, but do so independent of status. All other models within the Δ6 AICc model set were more complex versions of the top model and therefore were not retained under the nesting rule (Richards 2008; ESM Table S1).
For females, the best supported model contained an interaction between dominance status and time (ESM Table S2). Dominant females were in superior body condition than their subordinates in the first half of the breeding season (October to December, mean (±SE) dominant scaled mass = 44.84 g (±0.55), subordinate scaled mass = 42.77 g (±0.32)), but this difference had disappeared by the latter half of the breeding season (dominant scaled mass = 43.54 g (±0.5); subordinate scaled mass = 43.56 g (±0.31)) when the majority (63 %; 113/180) of clutches were laid. All other models had ΔAICc >6 (ESM Table S2), and therefore, the top model was considered to be the only model with support in the data, with an Akaike weight of 0.99.
Where all birds within a group were of known age, the dominants were typically the oldest birds of their sex within the group. However, across the population, dominants and subordinates overlapped substantially in age: the youngest known dominant female was 1.0 year old, while subordinate females of known age ranged up to 3.7 years old; the youngest known dominant male was 1.3 years old, while subordinate males of known age ranged up to 4.2 years old. Finally, it is unlikely that subordinates did not breed alongside their dominants simply because this species is incapable of breeding without helpers, as most new groups are founded by pairs and of 25 breeding attempts by pairs on territories of their own 16 (64 %) successfully reared offspring to fledging.
Do subordinate females contest the dominant’s monopoly?
In the 144 breeding attempts in 39 groups for which information was available, no subordinate female was ever seen (or determined via elimination) to be incubating eggs or brooding chicks overnight. The incubation periods of two successive clutches also never overlapped, and in the case of the shortest re-clutching interval observed following a successful breeding attempt, both clutches were laid by the same dominant female. There was no evidence to suggest that subordinate females ever added eggs to, or substituted eggs in to, the clutches of dominants: of the 141 clutches from 40 groups for which we know the lay date of every egg, without exception each egg was laid 1 day after the last and (as we mark eggs found on their day of laying) we can confirm that eggs were never substituted in to clutches or added to them following clutch completion. There was also no evidence suggestive of undetected egg destruction (by either dominants or subordinates) during periods when subordinate females might have stood to benefit most from attempting to breed.
There was no evidence that clutch size at completion differed when subordinate females did have access to within-group outbreeding partners (ESM Table S3; n = 87 breeding attempts). The best-supported model contained only the term for group size, and although a model containing an additional term for unrelated males being present was in the Δ6 AICc candidate set (ΔAICc 2.1, Table S3), it was not retained under the nesting rule, being a more complex version of the top model with weaker AICc support. Furthermore, there was also no evidence that eggs were less likely to survive from laying to hatching when unrelated male breeding partners were present (ESM Table S4, n = 83 breeding attempts). As with the clutch size analysis, the best supported model contained only the group size term and therefore the model also containing a term for unrelated males (ΔAICc 1.33, Table S4) with weaker support was not retained (Table S4).
Discussion
Our life history and genetic data indicate that white-browed sparrow weavers live in extended mixed-sex family groups in which within-group reproduction is completely monopolized by a single dominant breeding pair. There was no evidence of brood parasitism, joint-nesting, or cooperative polyandry. These complete skews do not reflect genetic monogamy, however, as they are accompanied by relatively high levels of extra-group paternity, with 12–18 % of offspring arising from extra-group matings by the dominant female. Subordinates of both sexes had typically delayed dispersal from their natal groups, yielding strong within-group kin structure, which is likely to have favoured the evolution of both cooperation and reproductive restraint among subordinates. The observed reproductive monopolies of dominants cannot be attributed solely to effects of kin structure on a subordinate’s expected payoff from breeding, however, as skew remained complete even after the immigration of unrelated birds had (1) yielded within-group outbreeding opportunities for subordinates of both sexes and/or (2) left some subordinates markedly less related to the dominants’ young than they would have been to their own. The reproductive quiescence of subordinates cannot be readily attributed to stress imposed by dominants either, as previous research has demonstrated that neither natal nor immigrant subordinates of either sex show elevated circulating stress hormone levels (Wingfield et al. 1991). As subordinate males have been observed attempting to copulate with the dominant female, the dominant male’s monopoly is most likely maintained in large part through his intensive mate guarding. Subordinate females, by contrast, appear never to attempt to lay eggs, despite frequently having unrelated males within their group, being less related to the dominants’ young than they would have been to their own, showing comparable body condition to dominants and overlapping dominants substantially in age. We therefore suggest, below, that subordinate females may exercise complete reproductive restraint due instead to a prevailing threat of action by the dominant female (e.g. a threat of eviction or infanticide) and/or reductions in offspring fitness were subordinates to breed. We discuss the relative importance of these mechanisms for the generation of reproductive skew in this species in more detail below before considering the extent to which their likely roles are captured by current skew theory.
The lack of subordinate reproduction within sparrow weaver groups could be driven in large part by negative effects of high within-group relatedness on their expected payoffs from breeding, which may thereby favour reproductive restraint (Koenig and Haydock 2004; Koenig et al. 2009; Young 2009). Subordinates of both sexes often lacked unrelated breeding partners within their groups, in which context inbreeding depression may markedly reduce their expected payoffs from breeding (e.g. McRae 1996; reviewed in Koenig and Haydock 2004). It seems unlikely that subordinate female sparrow weavers in our population ever entirely lack access to unrelated males, however, given the evident potential to mate extra-group (as dominant females do). Indeed, our findings indicate that indiscriminate matings with opposite sex birds in neighbouring groups would have yielded pairings that were just as outbred as those among dominant breeding pairs. For many subordinate females, strong within-group kin structure may have further reduced their expected payoff from breeding for a second reason, regardless of their access to unrelated males. In the majority of breeding attempts, all subordinates were helping to rear the offspring of the dominants that had reared them (their putative parents, subject to extra-group paternity), leaving them almost as related to the dominants’ young as they would have been to their own. As such, even subordinates with outbreeding opportunities may frequently have stood to gain little or no net fitness payoff from contesting dominant reproduction.
Reproductive restraint arising from high within-group relatedness cannot, however, account for reproductive skew remaining complete in both sexes even after the immigration of unrelated birds. Indeed, similar patterns have been observed in other co-operatively breeding species (e.g. Florida-Scrub Jays (Aphelocoma coerulescens), Quinn et al. 1999; pied babblers (Turdoides bicolor), Nelson-Flower et al. 2011) where subordinate reproduction is limited despite their being unrelated to the opposite-sex within-group dominant. Among male sparrow weavers, the reproductive monopolies of dominants are likely maintained at least in part by active interference in the reproductive attempts of subordinates via mate guarding (as per Komdeur et al. 1999); rarely during the breeding season is the dominant male more than a few metres from the dominant female. Indeed, evidence that subordinate male white-browed sparrow weavers frequently have testes that are as well developed as dominants’ (Wingfield et al. 1991), coupled with our three observations of a subordinate immigrant male mating with the dominant female, suggests that such guarding is necessary. Sperm competition could also facilitate the dominant male’s monopolisation of fertilisations despite these matings, either because the dominant’s sperm may be more likely to fertilise the eggs if he is of superior quality to the extra-pair male (Kempenaers and Dhondt 1993) or because he is able to displace his rival’s sperm (e.g. last sperm precedence; Birkhead and Moller 1992). Female choice could also play a key role in yielding skew among males, via assessments of the complex song that males sing at dawn (Voigt et al. 2007); while subordinate males do frequently sing dawn song they are invariably out-sung by their dominant (York 2012). A role for female choice could also explain why dominant males are capable of completely monopolizing within-group reproduction but are nevertheless frequently cuckolded by extra-group males. While such a pattern could reflect dominant males being briefly overpowered by incoming extra-group males, it might more plausibly reflect mating preferences on the part of the dominant female, who may conduct extra-group forays of her own (Double and Cockburn 2000). The drivers of extra-group mating in this population are now a key focus of our ongoing work.
Our evidence suggests that subordinate females never attempt to lay eggs of their own. Subordinate females were never known to have incubated eggs, brooded chicks, or produced surviving offspring, and there was no evidence to suggest that they ever added to, or replaced eggs in, the clutches of dominants. Nor was there evidence of enhanced clutch failure during times of potential conflict. As their reproductive quiescence cannot be readily attributed to chronic stress imposed by dominants (Wingfield et al. 1991), it seems likely that subordinate females are instead exercising complete reproductive restraint. Such restraint cannot be attributed solely to subordinate females showing inferior body condition to dominants, as, while this was the case for the first part of the breeding season, our data showed them to be in similar condition to dominants during the latter part when the majority of clutches were laid. It is also unlikely that subordinate females simply aren’t old enough to breed successfully (e.g. perhaps they have yet to develop the foraging skills needed to support reproduction; Wasser and Barash 1983), as dominant and subordinate females overlapped substantially in age across the population. That subordinates didn’t simply pair up and breed alongside their dominants cannot be attributed to an inability of this species to breed without helpers either, as our findings reveal that 64 % of breeding attempts by pairs on independent territories yielded fledglings.
At least two mechanisms exist that could plausibly be reducing subordinate females’ expected fitness payoffs from breeding to the point of favouring complete reproductive restraint. First, selection may favour restraint due in part to a credible threat that dominants would disrupt and/or punish subordinate reproduction were it to be attempted (Cant and Johnstone 2009; Young 2009; Cant 2011). While such threats could ultimately prove of widespread importance in generating skew in vertebrate societies, there remains little direct evidence to date that they play such a role, doubtless due in part to the challenges entailed in unequivocally demonstrating their importance (MA Cant and AJ Young unpublished data). Second, even if dominants lacked the ability to detect subordinate reproduction, subordinates might simply stand to gain no inclusive fitness payoff from breeding alongside a dominant if doing so compromised the fitness of their own offspring and/or those of a dominant relative (Hodge 2009; Young 2009). Such offspring fitness deficits could arise (1) if the addition of young to a communal brood (or breeding in a parallel nest) generated a super-optimal brood size (e.g. Clutton-Brock et al. 2010; Cant et al. 2010), (2) if the replacement of one or more of the dominant’s eggs with the subordinate’s disrupted within-brood maternal effects that might otherwise optimise brood level fitness, or (3) if the dominant’s young were superior competitors, such that the subordinate’s young suffered differentially when reared alongside them (Hodge et al. 2009). While such costs to offspring of plural/communal breeding might conceivably favour restraint even when dominants are unable to detect, disrupt, or punish subordinate reproduction (Young 2009; MA Cant and AJ Young unpublished data), the role that they play in maintaining reproductive skews remains largely unexplored.
As attempts to explain the marked variation across species in the extent of reproductive skew may be well served by working towards a single integrated theoretical approach to skew (Johnstone 2000), it is useful to consider the extent to which current theory captures the mechanisms that may generate reproductive skew in this system (see also Magrath et al. 2004; Koenig et al. 2009). Most conspicuously perhaps, as is the case in many other cooperative vertebrates, the concession of reproduction to subordinate sparrow weavers is evidently not required to either retain them within the group or maintain their cooperation, contrary to the scenario envisaged by early concession models of reproductive sharing (Keller and Reeve 1994). Our findings are more consistent with the view that the delayed dispersal of subordinates arises from ecological constraints on independent breeding (Emlen 1991), which may thereby leave dominants free to attempt to monopolise reproduction without the threat of subordinate departure, as envisaged by restraint models and encompassed by synthetic approaches (Johnstone 2000; Magrath et al. 2004; Cant 2011). While skew models typically consider skew a product solely of intra-sexual conflict over reproduction (see Cant and Reeve 2002 for an exception), it is likely that in this species, as in many others (Koenig and Haydock 2004), a lack of within-group outbreeding partners may play a key role in generating skew. Perhaps surprisingly, the widespread role that inbreeding avoidance may play has yet to be formally integrated in to skew models. This could presumably be addressed by extending existing restraint models to incorporate a new term, to account for the suite of other factors that may act in concert with the threat of action by dominants to reduce a subordinate’s expected payoff from attempting to breed (see Magrath et al. 2004; Koenig et al. 2009 and Young 2009 for discussion). Such a synthetic approach might presumably highlight that even a threat of weak action by dominants may be sufficient to yield complete subordinate restraint when these other factors are simultaneously reducing a subordinate’s expected fitness payoff from breeding (MA Cant and AJ Young unpublished data).
In summary, the application of genetic methods to the breeding system of a cooperative sparrow weaver (genus Plocepasser) has revealed extended family groups in which high levels of within-group kinship (diminished in part by a relatively high incidence of extra-group paternity) are likely to have favoured the evolution of both cooperation and reproductive suppression among subordinates. Reproductive restraint in response to factors independent of the presence and/or actions of same-sex dominants cannot, however, readily account for the complete reproductive skew seen in both sexes. While active interference by dominants may play a key role in the maintenance of skew among males (via mate-guarding), our findings raise the possibility that subordinate females exercise complete reproductive restraint, due, at least in part, to a threat of action by dominants and/or fitness costs to offspring that would otherwise arise were subordinates to breed. As in the many other cooperative vertebrates in which subordinates now appear to exercise complete reproductive restraint, it remains to be established precisely how, and indeed whether, dominants would disrupt and/or punish subordinate reproduction were it to be attempted. Empirical research to address this outstanding challenge to our wider understanding of the causes of reproductive skew in vertebrates should therefore be prioritised.
References
Arnold TW (2010) Uninformative parameters and model selection using Akaike’s information criterion. J Wildl Manag 74:1175–1178
Bates D, Maechler M, Bolker B (2012) lme4: Linear mixed-effects models using S4 classes. R package version 0.999999-0, http://cran.r-project.org/web/packages/lme4/index.html. Accessed 12/02/2013
Birkhead TR, Moller AP (1992) Sperm competition in birds: evolutionary causes and consequences. Academic Press, London
Bourke AFG (1999) Colony size, social complexity and reproductive conflict in social insects. J Evol Biol 12:245–257
Cant MA (2011) The role of threats in animal cooperation. Proc R Soc Lond B 278:170–178
Cant MA, Johnstone RA (2009) How threats influence the evolutionary resolution of within-group conflict. Am Nat 173:759–771
Cant MA, Reeve HK (2002) Female control of the distribution of paternity in cooperative breeders. Am Nat 160:602–611
Cant MA, Hodge SJ, Bell MBV, Gilchrist JS, Nichols HJ (2010) Reproductive control via eviction (but not the threat of eviction) in banded mongooses. Proc R Soc Lond B 277:2219–2226
Clutton-Brock TH, Brotherton PNM, Russell AF, O’Riain MJ, Gaynor D, Kansky R, Griffin A, Manser M, Sharpe L, McIlrath GM, Small T, Moss A, Monfort S (2001) Cooperation, control, and concession in meerkat groups. Science 291:478–481
Clutton-Brock TH, Hodge SJ, Spong G, Russell AF, Jordan NR, Bennett NC, Sharpe LL, Manser MB (2006) Intrasexual competition and sexual selection in cooperative mammals. Nature 444:1065–1068
Clutton-Brock TH, Hodge SJ, Flower TP, Spong GF, Young AJ (2010) Adaptive suppression of subordinate reproduction in cooperative mammals. Am Nat 176:664–673
Collias NE, Collias EC (1978) Cooperative breeding-behavior in white-browed sparrow weaver. Auk 95:472–484
Cooney R, Bennett NC (2000) Inbreeding avoidance and reproductive skew in a cooperative mammal. Proc R Soc Lond B 267:801–806
Cornwallis CK, West SA, Davis KE, Griffin AS (2010) Promiscuity and the evolutionary transition to complex societies. Nature 466:969–972
Covas R, Doutrelant C, du Plessis MA (2004) Experimental evidence of a link between breeding conditions and the decision to breed or to help in a colonial cooperative bird. Proc R Soc Lond B 271:827–832
Creel S (2001) Social dominance and stress hormones. Trends Ecol Evol 16:491–497
Dawson DA, Horsburgh GJ, Kupper C, Stewart IRK, Ball AD, Durrant KL, Hansson B, Bacon I, Bird S, Klein A, Krupa AP, Lee JW, Martin-Galvez D, Simeoni M, Smith G, Spurgin LG, Burke T (2010) New methods to identify conserved microsatellite loci and develop primer sets of high cross-species utility—as demonstrated for birds. Mol Ecol Res 10:475–494
Double M, Cockburn A (2000) Pre-dawn infidelity: females control extra-pair mating in superb fairy-wrens. Proc R Soc Lond B 267:465–470
Emlen ST (1991) Evolution of cooperative breeding in birds and mammals. In: Krebs JR, Davies NB (eds) Behavioural ecology: an evolutionary approach. Blackwell Science, Oxford, pp 301–337
Ferguson JWH (1988) Geographic variation in social behavior of white browed sparrow weavers, Plocepasser mahali. S Afr J Zool 23:1–6
Griffin AS, West SA (2002) Kin selection: fact and fiction. Trends Ecol Evol 17:15–21
Hamilton WD (1964) The genetical evolution of social behaviour. Parts I and II. J Theor Biol 7:1–52
Hodge SJ (2009) Understanding variation in reproductive skew: directions for future empirical research. In: Hager R, Jones CB (eds) Reproductive skew in vertebrates: proximate and ultimate causes. Cambridge University Press, Cambridge, pp 439–466
Hodge SJ, Bell MBV, Mwanguhya F, Kyabulima S, Waldick RC, Russell AF (2009) Maternal weight, offspring competitive ability, and the evolution of communal breeding. Behav Ecol 20:729–735
Holleley CE, Geerts PG (2009) Multiplex Manager 1.0: a cross-platform computer program that plans and optimizes multiplex PCR. Biotechniques 46:511–517
Johnstone RA (2000) Models of reproductive skew: a review and synthesis (invited article). Ethology 106:5–26
Johnstone RA, Cant MA (1999) Reproductive skew and the threat of eviction: a new perspective. Proc R Soc Lond B 266:275–279
Kalinowski ST, Taper ML, Marshall TC (2007) Revising how the computer program CERVUS accommodates genotyping error increases success in paternity assignment. Mol Ecol 16:1099–1106
Keller L, Reeve HK (1994) Partitioning of reproduction in animal societies. Trends Ecol Evol 9:98–102
Kempenaers B, Dhondt AA (1993) Why do females engage in extra-pair copulations? A review of hypotheses and their predictions. Belg J Zool 123:93–103
Koenig WD, Haydock J (2004) Incest and incest avoidance. In: Koenig WD, Dickinson JL (eds) Ecology and evolution of cooperative breeding in birds. Cambridge University Press, Cambridge, pp 142–156
Koenig WD, Haydock J, Stanback MT (1998) Reproductive roles in the cooperatively breeding acorn woodpecker: incest avoidance versus reproductive competition. Am Nat 151:243–255
Koenig WD, Shen SF, Krakauer AH, Haydock J (2009) Reproductive skew in avian societies. In: Hager R, Jones CB (eds) Reproductive skew in vertebrate societies: proximate and ultimate causes. Cambridge University Press, Cambridge, pp 227–264
Komdeur J, Kraaijeveld-Smit F, Kraaijeveld K, Edelaar P (1999) Explicit experimental evidence for the role of mate guarding in minimizing loss of paternity in the Seychelles warbler. Proc R Soc Lond B 266:2075–2081
Lewis DM (1981) Determinants of reproductive success of the white browed sparrow weaver, Plocepasser mahali. Behav Ecol Sociobiol 9:83–93
Lukas D, Clutton-Brock T (2012) Cooperative breeding and monogamy in mammalian societies. Proc R Soc Lond B 279:2151–2156
Magrath RD, Johnstone RA, Heinsohn RG (2004) Reproductive skew. In: Koenig WD, Dickinson JL (eds) Ecology and evolution of cooperative breeding in birds. Cambridge University Press, Cambridge, pp 157–176
McRae SB (1996) Family values: costs and benefits of communal nesting in the moorhen. Anim Behav 52:225–245
McRae SB, Amos W (1999) Characterization of hypervariable microsatellites in the cooperatively breeding white-browed sparrow weaver Plocepasser mahali. Mol Ecol 8:903–904
McRae SB, Emlen ST, Rubenstein DR, Bogdanowicz SM (2005) Polymorphic mircosatellite loci in a plural breeder, the grey-capped social weaver (Pseudonigrita arnaudi), isolated with an improved enrichment protocol using fragment size-selection. Mol Ecol Notes 5:16–20
Mumme RL, Koenig WD, Pitelka FA (1983) Reproductive competition in the communal acorn woodpecker—sisters destroy each others eggs. Nature 306:583–584
Nelson-Flower MJ, Hockey PAR, O’Ryan C, Raihani NJ, du Plessis MJ, Ridley AR (2011) Monogamous dominant pairs monopolize reproduction in the cooperatively breeding pied babbler. Behav Ecol 22:559–565
Nicholls JA, Double MC, Rowell DM, Magrath RD (2000) The evolution of cooperative pair breeding in thornbills Acanthiza (Pardalotidae). J Avian Biol 31:165–176
O’Riain MJ, Jarvis JUM, Alexander R, Buffenstein R, Peeters C (2000) Morphological castes in a vertebrate. Proc Natl Acad Sci USA 97:13194–13197
Peeters C, Higashi S (1989) Reproductive dominance controlled by mutilation in the queenless ant Diacamma australe. Naturwissenschaften 76:177–180
Peig J, Green AJ (2009) New perspectives for estimating body condition from mass/length data: the scaled mass index as an alternative method. Oikos 118:1883–1891
Quinn JS, Woolfenden GE, Fitzpatrick JW, White BN (1999) Multi-locus DNA fingerprinting supports genetic monogamy in Florida scrub-jays. Behav Ecol Sociobiol 45:1–10
R Development Core Team (2013) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Reeve HK, Keller L (1996) Relatedness asymmetry and reproductive sharing in animal societies. Am Nat 148:764–769
Richards SA (2008) Dealing with overdispersed count data in applied ecology. J Appl Ecol 45:218–227
Richards SA, Whittingham MJ, Stephens PA (2011) Model selection and model averaging in behavioural ecology: the utility of the IT-AIC framework. Behav Ecol Sociobiol 65:77–89
Richardson DS, Jury FL, Blaakmeer K, Komdeur J, Burke T (2001) Parentage assignment and extra‐group paternity in a cooperative breeder: The Seychelles warbler (Acrocephalus sechellensis). Mol Ecol 10:2263–2273
Rousset F (2008) GENEPOP’ 007: a complete re-implementation of the GENEPOP software for Windows and Linux. Mol Ecol Res 8:103–106
Rubenstein DR, Lovette IJ (2009) Reproductive skew and selection on female ornamentation in social species. Nature 462:786–789
Saltzman W, Digby LJ, Abbott DH (2009) Reproductive skew in female common marmosets: what can proximate mechanisms tell us about ultimate causes? Proc R Soc Lond B 276:389–399
Sinclair I, Ryan P (2010) Birds of Africa: South of the Sahara. Struik Publishers, Johannesburg
Voigt C, Leitner S, Gahr M (2007) Socially induced brain differentiation in a cooperatively breeding songbird. Proc R Soc Lond B 274:2645–2652
Wang J (2004) Sibship reconstruction from genetic data with typing errors. Genetics 166:1963–1979
Wang JL (2011) COANCESTRY: a program for simulating, estimating and analysing relatedness and inbreeding coefficients. Mol Ecol Res 11:141–145
Wang J, Santure AW (2009) Parentage and sibship inference from multilocus genotype data under polygamy. Genetics 181:1579–1594
Wasser SK, Barash DP (1983) Reproductive suppression among female mammals—implications for biomedicine and sexual selection theory. Q Rev Biol 58:513–538
Wingfield JC, Hegner RE, Lewis DM (1991) Circulating levels of luteinizing hormone and steroid hormones in relation to social status in the cooperatively breeding white-browed sparrow weaver, Plocepasser mahali. J Zool 225:43–58
Wong MYL, Buston PM, Munday PL, Jones GP (2007) The threat of punishment enforces peaceful cooperation and stabilizes queues in a coral reef fish. Proc R Soc Lond B 274:1093–1099
York JE (2012) The evolution and regulation of dawn chorus performance in white-browed sparrow weaver society. PhD Thesis, University of Bristol
Young AJ (2009) The causes of physiological suppression in vertebrate societies: a synthesis. In: Hager R, Jones CB (eds) Reproductive skew in vertebrate societies: proximate and ultimate causes. Cambridge University Press, Cambridge, pp 397–437
Young AJ, Bennett NC (2010) Morphological divergence of breeders and helpers in wild Damaraland mole-rat societies. Evolution 64:3190–3197
Young AJ, Clutton-Brock TH (2006) Infanticide by subordinates influences reproductive sharing in cooperatively breeding meerkats. Biol Lett 2:385–387
Young AJ, Carlson AA, Monfort SL, Russell AF, Bennett NC, Clutton-Brock TH (2006) Stress and the suppression of subordinate reproduction in cooperatively breeding meerkats. Proc Natl Acad Sci 103:12005–12010
Young AJ, Monfort SL, Clutton-Brock TH (2008) The causes of physiological suppression among female meerkats: a role for subordinate restraint due to the threat of infanticide? Horm Behav 53:131–139
Acknowledgments
We are grateful to our many field assistants during this period (Tom Reed, Brenna Boyle, Andy Mason, Francesca Jarero, Lexy Russell, Padraig Cregg, Lynda Casson, Sam Farrell, Lynda Donaldson, Jenny Sturgeon, and Robyn Silcock) for their tireless efforts under the Kalahari sun, to Northern Cape Conservation for permission to conduct the research, and to E.O. and Son and all at Tswalu Kalahari Reserve for their exceptional logistical support. We thank two anonymous reviewers for helpful comments that improved an earlier draft of this manuscript. This study was funded by a BBSRC David Phillips research fellowship to AY and a NERC blue skies research fellowship to AY.
Ethical standards
All protocols were approved by the University of Pretoria ethics committee and complied with regulations stipulated in the Guidelines for the use of Animals in Research and complied with the current laws of the Republic of South Africa.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by J. A. Graves
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 121 kb)
Rights and permissions
About this article
Cite this article
Harrison, X.A., York, J.E., Cram, D.L. et al. Complete reproductive skew within white-browed sparrow weaver groups despite outbreeding opportunities for subordinates of both sexes. Behav Ecol Sociobiol 67, 1915–1929 (2013). https://doi.org/10.1007/s00265-013-1599-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-013-1599-1