Abstract
Purpose
Concomitant unloading procedures, such as high tibial osteotomy (HTO), are increasingly recognized as an important part of cartilage repair. This study presents survival rate, functional outcome, complication rate, and return to work following combined single-stage autologous chondrocyte implantation (ACI) and HTO.
Methods
Forty patients with a mean follow-up of 60 months with isolated full thickness cartilage defects of the medial femoral condyle (MFC) and concomitant varus deformity were included in this retrospective case series. All patients were treated with a single-stage combined ACI and HTO between January 2004 and December 2010. Functional outcome was evaluated prior to surgery and at follow-up using standard scores (Lysholm, VAS, KOOS). Treatment failure was defined as the need for re-operation. Return to work was evaluated using the REFA score.
Results
With all patients (mean age 36.8 SD ± 8.1 years; varus deformity 4.9 ± 1.8 °; mean defect size 4.6 ± 2.1 cm²) a clinical investigation was performed a mean of 60.5 months (SD ± 2.5) postoperatively. Four patients required reintervention (failure rate 10 %). VAS decreased significantly from 6.7 ± 1.9 points preoperatively to 2.2 ± 1.3 points postoperatively. The mean Lysholm score at follow-up was 76.2 ± 19.8 points. The mean KOOS subscales were 81.4 ± 18.0 for pain, 81.3 ± 14.0 for symptoms, 87.6 ± 16.2 for activity in daily living, 66.7 ± 22.8 for function in sport and recreation, and 55.5 ± 22.0 for knee-related quality of living. Mean duration of incapacity from work was 94.5 ± 77 days. Absenteeism from work depended on work load (return to work REFA 0: 68.9 ± 61.4 days vs. REFA 4: 155.0 ± 111.0 days).
Conclusion
Single-stage autologous chondrocyte implantation and concomitant high tibial osteotomy is a reliable and safe treatment with satisfying clinical outcome and improved functional outcome. However, we found a remarkable stay at work rate, which depended on the work load.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Isolated cartilage defects of the medial compartment of the knee joint are common orthopaedic problems in middle-aged patients that are associated with pain, loss of function [38], and impaired quality of life comparable to severe osteoarthritis [17]. Furthermore, isolated cartilage defects tend to progress to osteoarthritis over time and can therefore be considered a precondition for joint degeneration [8, 32]. Treatment of focal cartilage defects should therefore focus on both reduction of clinical symptoms and avoidance of further joint degeneration.
Autologous chondrocyte implantation (ACI) is a standard therapy for full-thickness cartilage defects of the knee [4, 9, 31]. Functional outcome depends on several factors such as defect size and localization, number of defects, patient’s age, alignment, cell quality, work load and sports activity [27, 28]. Although ACI has been initially introduced for traumatic cartilage defects, results in degenerative cartilage lesion are also promising [25, 21]. Since degenerative cartilage lesions are usually associated with concomitant pathologies such as malalignment or instability, surgical treatment of these pathologies is of importance to increase the success rate of ACI. Although underlying pathologies are variegated, cartilage defects of the medial femoral condyle are frequently associated with varus malalignment [24]. Uncorrected malalignment may result in premature failure of the additive knee reconstructive procedure because of high stress concentration in and around the cartilage defect. Concomitant alignment therapies, such as high tibial osteotomy (HTO), may therefore be valuable in order to optimize the biomechanical conditions for healing and survival of the transplanted chondrocytes and may improve the functional outcome [13, 15, 22, 40].
Scientific evidence regarding combination of realignment procedures and cartilage repair is weak and the amount of studies published in this context is low. A recent meta analysis observed higher survival rates for combined HTO and biological reconstruction in contrast to isolated HTO [15]. Few studies have evaluated the results following HTO and different cartilage therapies, including ACI. Nevertheless the timing of ACI and concomitant HTO is still under discussion as these surgeries can be performed in a one- or two-stage fashion [39]. Concomitant single-stage HTO and ACI avoid a third surgery for the patient, decrease recovery time and may facilitate return to work and daily living. On the other hand, a combined procedure may increase the complication rate because of a more demanding operation and a prolonged operation time [2, 39]. While only few publications present clinical and functional outcome after combined ACI and HTO data regarding incapacity of work have not been published so far.
The present study analyses survival rate, clinical outcome, and return to work data after combined single-stage ACI and HTO. The hypothesis of the authors was that a single-stage fashion leads to good functional outcome and incapacity to work comparable to isolated HTO or ACI.
Material and methods
Patients
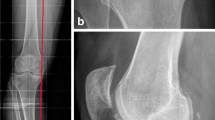
This retrospective case series was designed to verify the safety and effectiveness of combined single-stage ACI and HTO in patients with cartilage defects of the medial femoral condyle and accompanying varus malalignment (Figs. 1, 2, 3, and 4).
Inclusion criteria were circumscribed full-thickness cartilage defects of the MFC grade III or IV according to the International Cartilage Repair Society (ICRS) [12] which fulfilled the criteria for ACI [9], and a minimum varus deformity of 2°. Exclusion criteria were corresponding full-thickness cartilage defects of the medial compartment (“kissing lesions”), full-thickness cartilage defects of the lateral femorotibial or patellofemoral compartment, absence or extensive loss of the lateral meniscus, obesity (defined as a BMI ≥ 35), active knee flexion below 120° or an extension deficiency exceeding 10°, high-grade ligamentous instabilities, a history of fractures of the affected lower limb, active local or systemic infections, and inflammatory arthropathy.
Between January 2004 and December 2010, 40 patients were treated with combined single-stage ACI and HTO. The detailed patient characteristics are presented in Table 1. Before surgery, radiographs of the knee joint (a.p. view and true lateral view at 30° of knee flexion) and a.p. long-leg weight-bearing radiographs were obtained. Limb alignment was assessed by the technique of Pauwels [14].
All operations were performed under general anaesthesia. Intravenous antibiotics and standard thromboembolic prophylaxis were used. In all 40 patients, a routine arthroscopy of the affected knee joint was performed. Indication for ACI was performed by the recommendations given by the German Society of Orthopaedic Surgeons [15]. Chondrocytes were harvested with a standardized cartilage biopsy tool (Storz, Tuttlingen, Germany) from the intercondylar notch [16]. After expansion of autologous chondrocytes, ACI (product Cartigro™, Stryker, Duisburg, Germany) was performed as described in detail elsewhere [2]. Between one and two billion chondrocytes were applied per cm² cartilage defect. HTO was performed according to the technique recommended by the AO International knee expert group [17]. The TomoFixTM system (TomofixTM , Solothurn, Synthes, Switzerland) was used to stabilize the osteotomy, which was done in a biplanar fashion. All osteotomies aimed to result in a mild overcorrection between 50 and 70 % of the tibial plateau (mean postoperative femorotibial angle 7.5° ± 2.6) [18]. Patients were mobilized on the first postoperative day. Continuous passive motion (CPM) was recommended from day 1 postoperatively for six weeks for up to four hours per day. Limited weight bearing was recommended for six weeks. Individual limits of flexion were recommended depending on the exact defect location to avoid early exposure to axial compression and shear forces. After full weight bearing was achieved, full-leg radiographs were taken to analyse postoperative weight bearing axis.
Assessment of clinical outcome and work load
Revision surgery during the follow-up period was defined as failure. Revision surgery was indicated in patients with postoperative complications or persistent pain related to the implant site in combination with signs of graft failure or graft complication on MRI. Diagnostic arthroscopy was performed on a regular basis during hardware removal and was therefore not defined as revision surgery.
Lysholm, visual analog scale (VAS) and knee osteoarthritis outcome scores (KOOS) were used for clinical assessment [19]. VAS was assigned pre-operatively and at follow-up while Lysholm and KOOS were determined at follow-up only. The duration of incapacity to work was achieved by interrogation. Workload was evaluated according the REFA score. We evaluated data at a median follow-up of 60 months (±2.5) by calling patients.
Statistical analysis
SPSS for Windows (Version 21.0; SPSS, Chicago, IL) was used for statistical analyses designed to examine the data ascertained in this study. Quantitative variables at baseline were expressed as mean ± SD. For statistical evaluation of clinical data a nonparametric Mann–Whitney test was used. P < 0.05 was considered significant.
Results
Survival
After a mean follow-up of 60.5 ± 2.5 months, 40 patients were available for examination (follow-up rate 100 %). The mean defect size was 4.6 ± 2.7 cm² with a mean varus deformity of 4.70 ± 2.0° preoperatively. After HTO, the mean valgus angle was 2.6° ± 1.5.
Revision surgery was necessary in four cases (failure rate 10 %). Reasons for revision surgery were: persisting pain and failure of the ACI on MRI in two cases, overcorretion with postoperative valgization >70 % of the tibial plateau in one case, and nonunion of the osteotomy gap in one case. Both patients with failed ACI received additional microfracturing in combination with implant removal. In the case of overcorrection, re-varization was performed on day 12 after the initial surgery. The patient with nonunion of the osteotomy gap was treated with autologous cancellous bone transplantation from the iliac crest. In the further course the same patient suffered a wound infection and was treated with repeated surgical revision and intravenous antibiotics. Thirty-four patients received implant removal (82.9 %) at 17.4 ± 6.7 months postoperatively. No further complications due to implant removal were observed. No patient required partial or total knee replacement during the follow-up period. Thus, survival rate in terms of prosthetic knee-replacement was 100 % at five years postoperatively.
Functional outcome
VAS for pain significantly improved from 6.7 ± 1.9 preoperatively to 2.2 ± 1.4 points postoperatively (p = 0.00). The Lysholm score showed a mean increase of 22 points (p = 0.00) from pre-operatively 54.4 ± 18.9 to 76.2 ± 19.8 points postoperatively. The mean KOOS subscales at follow-up were: 81.4 ± 18.0 for pain, 81.3 ± 14.0 for symptoms, 87.6 ± 16.2 for activity in daily living, 66.7 ± 22.8 for function in sport and recreation, and 55.5 ± 22.0 for knee-related quality of living. As it is not recommended to calculate a total KOOS score we also calculated a KOOS4 Score (KOOS4 = (KOOSPain + KOOSSympt + KOOS Sports rec + KOOSQOL) which received a mean of 71.2 ± 16.1 points.
Return to work
The mean duration of incapacity to work was 94.5 ± 77.0 days. In addition, the prior performed arthroscopic cartilage biopsy caused 14.1 ± 16.1 days of incapacity of work and implant removal resulted in another 26.7 ± 48.3 days of work incapacity.
According to the REFA score, 11 patients were involved in work without special physical strain (28.2 %), nine patients were involved in work with small physical strain (23.1 %), five patients in work with moderate physical strain (12.8 %), and 14 patients in work with hard or mostly heavy physical strain (35.8 %). Absenteeism from work strongly correlated with the physical workload. Patients REFA grade I returned to work after a mean of 68.1 ± 61.4 days while patients grade IV performing work with most heavily physical strain stayed off work for a mean of 155.0 ± 111 days (p = 0.023).
Discussion
The main findings of the present study were that combined single-stage ACI and HTO results in a relatively low complication rate, good to excellent functional outcome and absence from work comparable to HTO or ACT performed as a single procedure.
The technique of HTO has much improved in recent years and consideration of the biomechanical axis, angular stable implants and intraoperative navigation lead to reliable mid-term results [3, 6, 12, 37]. Recently, HTO is of growing interest as a concomitant surgery in patients suffering from full-thickness cartilage defects of the medial femoral condyle and varus deformity [5, 15, 22]. There is consensus that concomitant malalignment needs to be addressed in cartilage therapies [14, 5, 22] in order to optimize the biomechanical environment at the healing site. Even without significant medialization of the weight-bearing axis, approximately 75 % of the weight is transferred to the medial compartment increasing to 80–90 % even in mild varus deformities of only 3–5° [26]. In a recent biomechanical study, Mina et al. measured pressure distribution around focal cartilage defects in varying tibio-femoral alignments. They pointed out that contact pressure in cartilage lesions concentrates around the rim, which is also a vulnerable area in ACT. They found complete unloading of the medial compartment between 6 and 10° valgus and an equal distribution between 0 to 4° of valgus. As a certain amount of loading is substantial for chondrocyte biology [7] they concluded that correction to 0–4° valgus favours cartilage repair [24].
Even though the biomechanical background for unloading procedures during cartilage repair is proven, the timing of both surgeries is still under debate as they can be performed in a one- or two-stage fashion [39]. Combined ACI and HTO eliminates a third surgery (cartilage biopsy included) and anaesthesia decreases overall recovery time and result in shorter rehabilitation periods [2].
Rehabilitation following ACI in the early phase (weeks one to six) follows a complex protocol as chondrocytes require passive motion and do not tolerate shear forces for the first six weeks.
Thus continuous passive motion combined with partial weight bearing with 20–25 % of body weight on crutches is essential [10, 18]. Medial open wedge HTO with angular stable implants does not require partial weight bearing at all as there is no loss of correction from the biomechanical point of view [1]. Thus combination of both techniques does not result in longer rehabilitation. On the other hand a single-stage fashion results in longer operative times and larger exploration at once, which might be associated with higher complication rates, which is why several authors recommend a two-stage fashion [20, 29].
In 2010, Willey et al. published data of 35 patients following one-stage HTO or distal femur osteotomy (DFO) in combination with significant additional knee reconstruction like cartilage repair, meniscal transplantation or ligament reconstruction. They observed 20 % of major complications and concluded that the rate of complications in a single-stage fashion is similar to that seen in a two-stage fashion. However, their study presented data of a heterogenous cohort including several different techniques and a follow-up period limited to 12 months; furthermore, only ten patients received cartilage repair (two cases of cartilage resurfacing, eight cases of microfracturing).
Concerning safety of the procedure, the overall complication rate in the present study was 10 %, which is significant but still comparably low. Miller et al. reported complication rates of 36.9 % for medial open wedge-osteotomy [23]. Spahn et al. monitored overall complication rates of 43.6 % when performing HTO with the Puddu plate, while he found a significantly lower complication rate of 16.7 % when using C-plates. These results underline the importance of implant choice in OW-HTO [34]. Very low implant associated complication rates were observed when an internal plate fixator was used for OW-HTO [11, 30]. Regarding ACI, a recent systematic review by Harris et al. reported re-operation rates of 33 % following ACI in general, whereas the re-operation was 18 % for patients treated with second generation ACI, as used in the present study [16]. In summary, complication rates following combined single-stage ACI and HTO in the present study are comparable to those of the singular surgeries while no data after a two-stage procedure is available.
With regard to the functional outcome, we found that patients experienced a significant pain relief as measured with the VAS and a significant increase of knee function as measured with the Lysholm score even after a mean follow up of more than five years. In addition, good to excellent results for the KOOS subscales pain, symptoms and activities of daily living were evident. However, inferior results were seen in the subscales function in sport and recreation and knee-related quality of living. Thus, further studies are needed to investigate return to sports and quality of life following combined single-stage ACT and HTO. Direct comparison of our data with other studies reporting functional outcome following simultaneous ACT and HTO is difficult as only few studies are available [15]. Among those, various different techniques for cartilage repair, implants and scoring systems were used. In a recently published review comparing clinical outcomes of 11 studies (366 subjects), Trinh et. al observed significantly improved functional outcomes in patients undergoing combined ACI and patellofemoral realignment as compared to patients undergoing isolated ACI for patellofemoral cartilage defects without differences of complication rates [36]. Concerning the medial femorotibial compartment, Sterett et. al reported improved Lysholm scores with 67 points after nine years and 97 % survival rates after five years following microfracture and HTO in 106 patients with a mean age of 52 years suffering from cartilage degenerations of the MFC [35]. Nonetheless, patients in the present study suffered from severe full thickness cartilage defects, which is why ACI was indicated. Minzlaff et. al evaluated long-term results and survival rates of 74 patients (mean age 38 years, follow-up 7.5 years) after combined osteochondral autologous transfer (OAT) and closed or open-wedge HTO. In their case series, significantly improved Lysholm scores with mean increases of 33 points were seen. Mean survival rate in terms of delay of TKA was 90.1 % at 8.5 years.
There are only two studies presenting data of combined ACI and medial open-wedge HTO. First of all, higher survival rates for patients undergoing combined ACI and HTO compared to ACI alone even in mild varus deformities have been reported [5]. Additionally, in 2008 Franceschi et al. performed arthroscopic ACI and medial open-wedge HTO in eight patients with chondral defects of the MFC (mean age 50 years). After a follow up of 28 months they found significantly improved functional scores concluding that combined HTO and ACI is a viable treatment option for this highly selected patient group [13]. Results are comparable to the results of our study while both cohorts included rather small patient cohorts.
Apart from clinical outcome, patient compliance and motivation for the rehabilitation program are crucial. In contrast to isolated HTO, a six-week rehabilitation protocol with only 20 kg of partial weight bearing is necessary after ACI. Thus, absence from work is an important topic for each patient facing combined ACI and HTO. Unfortunately, only two studies report return to work following HTO while no data concerning ACI or combined surgeries are available. Hoell et al. measured a mean incapacity to work of 13.9 weeks after closed wedge osteotomy [19]. Schroeter et al. found a mean duration of incapacity to work of 87 days (range 14–450) [33]. These results are comparable to the mean return to work after 94.5 ± 77.0 days in the present study. In addition, the basic arthroscopy and cartilage biopsy caused 14.1 ± 16.1 days of work and finally implant removal resulted again in 26.7 ± 48.3 of work incapacity. Nonetheless absence from work strongly depends on the physical work strain. Patients performing work with hard or mostly heavy work strain need to be informed about probable longer periods off work than desk workers. While data describing return to work after ACI are not available, comparable inactive periods due to ACI can be estimated. Thus combined alignment and cartilage therapy may reduce incapacity of work in comparison to the isolated surgeries. This important question needs to be addressed in further clinical trials presenting return to work data after ACI and two-stage combinations of HTO and ACI.
Concerning limitations of the present study, a control group undergoing two-stage HTO and ACI is missing as all patients underwent single-stage surgery. Therefore it is difficult to directly compare results. Furthermore, a 60-month follow-up only represents short- to mid-term results and patients in the present cohort were rather young. Nevertheless, patients with focal cartilage defects of the MFC and concomitant varus deformity represent a selected subgroup within cartilage repair patients. Therefore patient selection and study design seem appropriate and reasonable.
Conclusion
In conclusion, combined single-stage HTO and ACI results in risk for complication and absence from work comparable to the singular surgeries while clinical and functional outcome improves significantly.
References
Agneskirchner JD, Freiling D, Hurschler C et al. (2006) Primary stability of four different implants for opening wedge high tibial osteotomy. Knee Surg Sports Traumatol Arthrosc 14(3):291–300
Amendola A (2007) Knee osteotomy and meniscal transplantation: indications, technical considerations, and results. Sports Med Arthrosc 15(1):32–38
Amendola A, Bonasia DE (2010) Results of high tibial osteotomy: review of the literature. Int Orthop 34(2):155–160
Behrens P, Bosch U, Bruns J et al (2004) [Indications and implementation of recommendations of the working group “tissue regeneration and tissue substitutes” for autologous chondrocyte transplantation (ACT)]. Z Orthop Ihre Grenzgeb 142(5):529–539
Bode G, Schmal H, Pestka JM et al (2013) A non-randomized controlled clinical trial on autologous chondrocyte implantation (ACI) in cartilage defects of the medial femoral condyle with or without high tibial osteotomy in patients with varus deformity of less than 5 degrees. Arch Orthop Trauma Surg 133(1):43–49
Bode G, von Heyden J, Pestka J et al (2013) Prospective 5-year survival rate data following open-wedge valgus high tibial osteotomy. Knee Surg Sports Traumatol Arthrosc. doi:10.1007/s00167-013-2762-y
Buckwalter JAMJ, Brown TD (2006) Perspectives on chondrocyte mechanobiology and osteoarthritis. Biorheology 43(3–4):603–609
Cicuttini F, Ding C, Wluka A et al (2005) Association of cartilage defects with loss of knee cartilage in healthy, middle-age adults: a prospective study. Arthritis Rheum 52(7):2033–2039
Cole BJ, Pascual-Garrido C, Grumet RC (2009) Surgical management of articular cartilage defects in the knee. J Bone Joint Surg Am 91(7):1778–1790
Edwards PK, Ackland TR, Ebert JR (2013) Accelerated weightbearing rehabilitation after matrix-induced autologous chondrocyte implantation in the tibiofemoral joint: early clinical and radiological outcomes. Am J Sports Med 41(10):2314–2324
Floerkemeier S, Staubli AE, Schroeter S et al (2012) Outcome after high tibial open-wedge osteotomy: a retrospective evaluation of 533 patients. Knee Surg Sports Traumatol Arthrosc 21(1):170–180
Floerkemeier S, Staubli AE, Schroeter S et al (2013) Outcome after high tibial open-wedge osteotomy: a retrospective evaluation of 533 patients. Knee Surg Sports Traumatol Arthrosc 21(1):170–180
Franceschi F, Longo UG, Ruzzini L et al (2008) Simultaneous arthroscopic implantation of autologous chondrocytes and high tibial osteotomy for tibial chondral defects in the varus knee. Knee 15(4):309–313
Hangody LFP (2003) Autologous osteochondral mosaicplasty for the treatment of full-thickness defects of weight-bearing axis joints: ten year of experimental and clinical experience. Injury 39(Suppl1):32–39
Harris JD, McNeilan R, Siston RA et al (2013) Survival and clinical outcome of isolated high tibial osteotomy and combined biological knee reconstruction. Knee. doi:10.1016/j.knee.2012.12.012
Harris JD, Siston RA, Brophy RH et al (2011) Failures, re-operations, and complications after autologous chondrocyte implantation–a systematic review. Osteoarthr Cartil 19(7):779–791
Heir S, Nerhus TK, Rotterud JH et al (2010) Focal cartilage defects in the knee impair quality of life as much as severe osteoarthritis: a comparison of knee injury and osteoarthritis outcome score in 4 patient categories scheduled for knee surgery. Am J Sports Med 38(2):231–237
Hirschmuller A, Baur H, Braun S et al (2011) Rehabilitation after autologous chondrocyte implantation for isolated cartilage defects of the knee. Am J Sports Med 39(12):2686–2696
Hoell S, Suttmoeller J, Stoll V et al (2005) The high tibial osteotomy, open versus closed wedge, a comparison of methods in 108 patients. Arch Orthop Trauma Surg 125(9):638–643
Lattermann CJR (1996) High tibial osteotomy alone or combined with ligament reconstruction in anterior cruciate ligament-deficient knees. Knee Surg Sports Traumatol Arthrosc 4(1):32–38
Macmull S, Jaiswal PK, Bentley G et al (2012) The role of autologous chondrocyte implantation in the treatment of symptomatic chondromalacia patellae. Int Orthop 36(7):1371–1377
McNamara I, Birmingham TB, Fowler PJ et al (2013) High tibial osteotomy: evolution of research and clinical applications–a Canadian experience. Knee Surg Sports Traumatol Arthrosc 21(1):23–31
Miller BS, Downie B, McDonough EB et al (2009) Complications after medial opening wedge high tibial osteotomy. Arthroscopy 25(6):639–646
Mina C, Garrett WE Jr, Pietrobon R et al (2008) High tibial osteotomy for unloading osteochondral defects in the medial compartment of the knee. Am J Sports Med 36(5):949–955
Minas T, Gomoll AH, Solhpour S et al (2010) Autologous chondrocyte implantation for joint preservation in patients with early osteoarthritis. Clin Orthop Relat Res 468(1):147–157
Muller M, Strecker W (2008) Arthroscopy prior to osteotomy around the knee? Arch Orthop Trauma Surg 128(11):1217–1221
Niemeyer P, Lenz P, Kreuz PC et al (2010) Chondrocyte-seeded type I/III collagen membrane for autologous chondrocyte transplantation: prospective 2-year results in patients with cartilage defects of the knee joint. Arthroscopy 26(8):1074–1082
Niemeyer P, Salzmann G, Feucht M et al (2014) First-generation versus second-generation autologous chondrocyte implantation for treatment of cartilage defects of the knee: a matched-pair analysis on long-term clinical outcome. Int Orthop. doi:10.1007/s00264-014-2368-0
Noyes FR, Barber-Westin SD, Hewett TE (2000) High tibial osteotomy and ligament reconstruction for varus angulated anterior cruciate ligament-deficient knees. Am J Sports Med 28(3):282–296
Pape D, Kohn D, van Giffen N et al (2013) Differences in fixation stability between spacer plate and plate fixator following high tibial osteotomy. Knee Surg Sports Traumatol Arthrosc 21(1):82–89
Rogers BA, David LA, Briggs TW (2010) Sequential outcome following autologous chondrocyte implantation of the knee: a six-year follow-up. Int Orthop 34(7):959–964
Schinhan M, Gruber M, Vavken P et al (2012) Critical-size defect induces unicompartmental osteoarthritis in a stable ovine knee. J Orthop Res 30(2):214–220
Schroter S, Mueller J, van Heerwaarden R et al (2013) Return to work and clinical outcome after open wedge HTO. Knee Surg Sports Traumatol Arthrosc 21(1):213–219
Spahn G, Wittig R (2002) Primary stability of various implants in tibial opening wedge osteotomy: a biomechanical study. J Orthop Sci 7(6):683–687
Sterett WI, Steadman JR, Huang MJ et al (2010) Chondral resurfacing and high tibial osteotomy in the varus knee: survivorship analysis. Am J Sports Med 38(7):1420–1424
Trinh TQ, Harris JD, Siston RA et al (2013) Improved outcomes with combined autologous chondrocyte implantation and patellofemoral osteotomy versus isolated autologous chondrocyte implantation. Arthroscopy 29(3):566–574
Tunggal JA, Higgins GA, Waddell JP Complications of closing wedge high tibial osteotomy. Int Orthop 34(2):255–261
W-Dahl A, Robertsson O, Lidgren L (2010) Surgery for knee osteoarthritis in younger patients. Acta Orthop 81(2):161–164
Willey M, Wolf BR, Kocaglu B et al (2010) Complications associated with realignment osteotomy of the knee performed simultaneously with additional reconstructive procedures. Iowa Orthop J 30:55–60
Wolcott M, Traub S, Efird C (2010) High tibial osteotomies in the young active patient. Int Orthop 34(2):161–166
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bode, G., Ogon, P., Pestka, J. et al. Clinical outcome and return to work following single-stage combined autologous chondrocyte implantation and high tibial osteotomy. International Orthopaedics (SICOT) 39, 689–696 (2015). https://doi.org/10.1007/s00264-014-2547-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-014-2547-z