Abstract
Introduction
Experimental models of osteoporosis in rabbits are useful to investigate anabolic agents because this animal has a fast bone turnover with predominant remodelling over the modelling processes. For that purpose, it is necessary to characterize the densitometric values of each type of bony tissue.
Objective
To determine areal bone mass measurement in the spine and in trabecular, cortical and subchondral bone of the knee in healthy and osteoporotic rabbits.
Design
Bone mineral content and bone mineral density were measured in lumbar spine, global knee, and subchondral and cortical bone of the knee with dual energy X-ray absorptiometry using a Hologic QDR-1000/W densitometer in 29 skeletally mature female healthy New Zealand rabbits. Ten rabbits underwent triplicate scans for evaluation of the effect of repositioning. Osteoporosis was experimentally induced in 15 rabbits by bilateral ovariectomy and postoperative corticosteroid treatment for 4 weeks. Identical dual energy X-ray absorptiometry (DXA) studies were performed thereafter.
Results
Mean values of bone mineral content at the lumbar spine, global knee, subchondral bone and cortical tibial metaphysis were: 1934±217 mg, 878±83 mg, 149±14 mg and 29±7.0 mg, respectively. The mean values of bone mineral density at the same regions were: 298±24 mg/cm2, 455±32 mg/cm2, 617±60 mg/cm2 and 678±163 mg/cm2, respectively. Bone mineral content and bone density of healthy rabbits followed a normal distribution at the four skeletal regions studied. Precision after triplicate repositioning yielded a coefficient of variation ranging from 2.6% to 3.8%. The least significant change ranged between 7.3% and 10.7%. Bone mineral density measured at the four different skeletal regions correlated significantly. Bone mineral density in osteoporotic rabbits was significantly lower in the four regions studied than that in controls, rendering a T-score of, respectively, −2.0±1.1 in the lumbar spine, −2.2±2.1 in the global knee, −1.9±0.6 in the subchondral bone, and −5.7±3.1 in the cortical tibia (P<0.05).
Conclusions
DXA is a reliable and precise method to evaluate the bone mass in rabbits. Our results also suggest that subchondral bone is a bone of mixed densitometric characteristics with marked cortical bone predominance.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoporosis is a skeletal disease characterized by loss of bone strength, leading to an increased susceptibility to fractures. Since bone mass plays a major role in bone strength, it is essential to measure it precisely to diagnose and follow the treatment of osteoporosis both in humans and laboratory animals. Dual-energy X-ray absorptiometry (DXA) is a non-invasive method of measuring bone mineral content (BMC) and bone mineral density (BMD) in vivo. The measurements of BMC and BMD in animal models have been carried out with instruments designed for human clinical use.
The development of animal models has been essential to studies of bone mass as well as for the development of new drugs for osteoporosis [1–4]. Although the ovariectomized rat represents the “gold standard” in experimental animal models for postmenopausal osteoporosis studies [5–10], it has several disadvantages that may not provide the ideal model for the human condition, such as the failure to achieve true skeletal maturity and the low rate of intracortical remodelling [11, 12]. Rabbits achieve skeletal maturity at approximately 6 months of age, show significant intracortical remodelling [4, 13–15], have faster bone turnover than other rodents, even than primates, and a significant bone loss can be induced within a short period of time [13, 14, 16]. These aspects turn this animal into a promising model to study osteoporosis and to investigate anabolic drugs.
DXA is the currently utilized technique to assess sequential studies of BMC and BMD in both humans and laboratory animals. This technique has the necessary precision when used in small laboratory animals [17–20]. Rabbits have been employed to study bone ingrowth around implants in osteoporotic models and for glucocorticoid-induced osteopenia [4, 21–24]. However, rabbit osteoporosis has not been fully characterized and, to our knowledge, systematic DXA measurements at different skeletal regions have not been published [25, 26].
The interest in the relationship between osteoporosis and osteoarthritis has recently increased [27–29]. Evidence of the role played by subchondral bone in the pathogenesis of osteoarthritis, as well as in the progression of cartilage damage, has been demonstrated [30, 31], and it is therefore necessary to characterize DXA measurements fully in this specific anatomical area.
The aim of our study was to determine bone mass values of normality in a healthy and osteoporotic population of mature female rabbits and to characterize the densitometric measurements in this population in trabecular, cortical and subchondral bone.
Materials and methods
Animals
Twenty-nine, 8-month-old (3.8–4.8 kg body weight), skeletally mature, female New Zealand rabbits were included in the study (Granja Universal, Pamplona, Spain). The animals became acclimatized after 2 weeks and were housed individually in stainless-steel cages and maintained on a 12-h light/12-h dark cycle at room temperature. The animals had free access to water and standard rabbit chow (Panlab, Barcelona, Spain). Bone mineral determination at different anatomical sites was performed in all 29 rabbits at baseline to characterize densitometric values, and 20 rabbits were reserved for the study of experimentally induced osteoporosis in this species.
Bone mineral measurements and definition of regions of interest
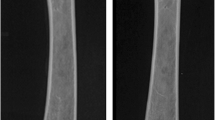
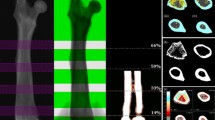
DXA analyses were carried out with a Hologic QDR-1000/W densitometer based on pencil-beam technology (Hologic, Waltham, Mass., USA) with a 1 mm diameter collimator on the X-ray output. The densitometer was calibrated daily. Specific software for small sample analysis, which increases the spatial resolution, was used (space per scanning line of 0.0254 cm and resolution for each point of 0.0127 cm; version 6.2). Bone area (BA), BMC and BMD were measured in the lumbar spine (L3 and L4 vertebrae) and in the left knee. Measurements were carried out in vivo with the animals placed in the supine decubitus position under general anaesthesia [32]. The rabbits were placed on a methacrylate bed, specifically designed for the study, with 30° of lateral inclination so that a true posterior–anterior joint view could be obtained (Fig. 1). After digital radiological screening, a point 3 cm below the navel was considered to be the external guide to focus the DXA beam about L3–L4. DXA of the left knee was carried out with the leg in full extension and internal rotation and with the beam focused immediately distal to the joint line [33]. Images with condylar overlapping were excluded (Fig. 2).
Measurements were carried out in vivo with animals placed in the supine decubitus position under general anaesthesia. A point 3 cm below the navel was used as an external guide to focus the DXA beam (A). DXA of the knees was obtained by the placement of the rabbits on a methacrylate bed, specifically designed for the study, with 30° of lateral tilt to obtain a true posterior–anterior joint view (B)
DXA analysis of a rabbit knee showing the anatomical areas evaluated. The global knee was selected to evaluate trabecular and cortical bone (large rectangle; A). Four squared areas adjacent to the joint line were used to assess subchondral bone in each femoral condyle and tibial plateaux (B). To study cortical bone, we outlined a metaphyseal area immediately distal to the tibio-fibular joint (C)
DXA analysis of various skeletal regions with different degrees of trabecular and cortical remodelling were performed. The means of the absorptiometric values of the third and fourth vertebrae were calculated for the lumbar spine. Regarding the knee, three different anatomical areas were selected: (a) the global knee, considering a ROI of 266 lines of width and 22 lines above and under the joint space. This zone was considered as an index of combined trabecular and cortical bone (Fig. 2). (b) Four differentiated subregions corresponding to medial and lateral femoral condyles and tibial plateaux (17×11 pixels, 0.06 cm2 each), located, respectively, 1 mm above and 1 mm below the joint line at the areas of maximum contact between the femoral condyles and the tibial plateaux. The mean values of BMC and BMD at these four zones were considered to be representative of subchondral bone. (c) Finally, we analysed an area of 15×9 pixels (0.04 cm2) at the lateral cortex of the proximal metaphysis of the tibia immediately distal to the tibio-fibular joint, as the main reference of cortical bone.
Assessment of precision
In order to evaluate the effect of repositioning, we performed scans of the knee and lumbar spine three times in the same week in ten healthy rabbits. We estimated the lower statistically significant change for these three repeated measurements, the so-called least significant change (LSC), which was estimated in accordance with Glueer et al. [34], calculating the BMD and BMC changes (as percentages) exceeding 2√2 times the precision error of the technique (with 95% confidence interval). The reproducibility of the measurements was controlled by scanning the s/n Q-239 Hologic spine phantom of known composition in each session, according to the manufacturer’s recommendations. The in vitro coefficient of variation (CV) during the designed period of study was 0.49% for BMD and 0.60% for BMC.
Rabbit model of osteoporosis
After baseline BMC and BMD had been determined, 15 animals underwent bilateral ovariectomy and, 2 weeks later, they were given daily intramuscular (i.m.) injections of methylprednisolone hemisuccinate to induce osteoporosis. The injection of 1 mg/kg per day (2.5 μmol/kg per day) lasted for 4 consecutive weeks [35]. Bilateral ovariectomy was carried out under general anaesthesia by a standard technique [36]. Antibiotic prophylaxis with cefonicid (100 mg/kg) (Monocid; Smith and Beecham, Madrid, Spain) was administered prior to surgery and for the following 5 days. Five additional rabbits were employed as healthy controls. All the rabbits underwent a second DXA analysis 6 weeks after ovariectomy, or after the first DXA in the control group, and were killed thereafter. These experiments were approved by the local ethics committee, and guidelines for the care and use of small animals were followed.
Statistical analysis
The descriptive analysis of densitometric values included the baseline BA, BMC and BMD, as well as the mean and standard deviation (SD), which were used to calculate the T-score in our population of rabbits. In addition, the coefficient of variation (CV), defined as SD/mean×100, was determined to estimate interindividual variability. Normality hypothesis of data distribution was defined through the Kolmogorov–Smirnov test. Correlation analysis between different skeletal regions was assessed by Pearson’s correlation coefficient (r) with their respective hypothesis test. The in vivo reproducibility of the measurements was evaluated through the CV of three repeated measurements performed over the same week at the lumbar spine, global knee, tibial metaphysis at cortical level and subchondral bone in each rabbit, as explained before. Variations in BMD and in T-score were calculated as the difference between baseline and final bone mineral density and as the reduction in the number of SDs over the reference mean values obtained in our population of rabbits prior to any experimental intervention. Student’s t-test was used to compare the bone mass variations after the study period. A statistically significant value was chosen at P<0.05. In order to reduce the number of animals as much as possible in future studies using this experimental model of osteoporosis, we calculated the least number of rabbits needed to detect statistical significances, following the proceedings described by Dupont and Plummer [37]. Statistical analysis was performed through the SPSS program, version 10.0 (SPSS, Chicago, Ill., USA).
Results
Descriptive analysis of densitometric values
BA, BMC and BMD measurements for the different anatomical locations studied in the population of healthy rabbits are shown in Table 1. When the statistical distribution of data was evaluated, the Kolmogorov–Smirnov test revealed that BMC and BMD resembled a normal distribution, and changes in these paramenters followed a parallel distribution in the four anatomical locations assessed (Table 1). It is interesting to notice that BMD increased progressively from lumbar spine to global knee, subchondral bone, and to proximal tibial metaphysis, which showed the highest density. These variations reflected the different ratios of trabecular and cortical bone existing in the areas studied.
With regard to the correlation shown by BMD measurements at the different anatomical sites, values of BMD at the spine, global knee and subchondral bone correlated significantly (Fig. 3), whereas BMD of the metaphyseal cortex of the tibia did not correlate with any of the other locations.
Precision of vertebrae and knee DXA measurements. Effect of repositioning
After triple repositioning of the animals, the CV ranged from 2.6% to 3.8% in the anatomical regions considered. These data were supported by the value of the least significant change of BMD, that oscillated from 7.3% to 10.7% at the same areas (Table 2).
DXA analysis in osteoporotic rabbits
Table 3 shows the variations in BMD and T-score in osteoporotic and control animals at the lumbar spine, global knee, subchondral bone of the knee and tibial metaphysis. When these variations were compared, there were no significant differences in BMD between controls and osteoporotic animals at baseline at any skeletal site. However, 6 weeks after ovariectomy, a statistically significant reduction of BMD in ovariectomized rabbits was observed in comparison to controls, clearly exceeding the LSC mentioned before and being maximal in the cortical region of the tibial metaphysis. According to these data, the smallest number of rabbits that were required to demonstrate statistically significant differences in BMD before and after the experimental intervention was eight for the lumbar spine and six or fewer for the three remaining sites.
Discussion
Animal models of osteoporosis can be classified into two main categories: those where increased bone resorption is the dominant mechanism (i.e. ovariectomy) and models where the formation deficit is prevailing (i.e. senile rats or corticosteroid administration) [3, 15, 35, 38]. Corticosteroid administration and ovariectomy are the main models of experimental osteoporosis described in rabbits [21, 35, 39], but the induction of the disease by any of these methods alone has been controversial. For this reason we hypothesized that the combination of both models would reproduce the disease in a more consistent manner. The results of this study demonstrate that the combination of ovariectomy and corticosteroid treatment induces rapid trabecular and cortical osteoporosis, reducing the time to achieve osteoporosis and sparing the final dose of corticosteroids. The efficiency of this combined experimental model is confirmed by the low number of animals necessary to detect significant variations in BMD. Although there is not consensus in the literature about the ideal doses of corticosteroids to induce osteoporosis, mean doses of methylprednisolone were used because they have an effect limited to bone turnover without the development of inflammation, necrosis or vascular changes in subchondral bone, one of the target tissues to be analysed in this experiment [35].
DXA with ultra-high resolution software is a tool of high precision superior to other non-invasive procedures based on the attenuation of a beam of γ-rays, X-rays or quantitative ultrasound [40–45]. However, and until newer methods capable of reconstructing a three-dimensional data set of bone—such as quantitative computed tomography or microscopic computed tomography—are widely available, more studies on densitometric validation in animal models are needed, especially when complex anatomical structures or new anatomical areas such as subchondral or cortical bone are to be evaluated [2, 46–48]. The low values of LSC found in our study, and the fact that the intra-individual CVs were similar to those reported in investigations carried out in other animal species, demonstrate that high-precision densitometric measurements can be performed with this technique [17, 18, 40, 49–51].
The lumbar spine is the usual anatomical location where BMD variations are monitored in experimental models of osteoporosis. This model shows that the global knee might constitute an excellent site for that purpose, according to values of LSC and the low number of animals needed to detect significant changes. A Hologic QDR-1000/W densitometer based on pencil-beam technology was used in this study instead of newer devices based on fan-beam technology, but it does not seem to constitute a major drawback, since comparative studies have demonstrated an excellent correlation in the measurements of in vitro and in vivo bone mass between two generations of DXA systems [52–54].
It has been suggested that subchondral bone mineral density may play a key role in the aetiology and development of joint diseases, such as osteoarthritis and rheumatoid arthritis [27, 55]. Since these entities frequently coexist with osteoporosis, and the relationship between osteoporosis and joint diseases has yet to be established, it is relevant to characterize the densitometric values in this specific area. To our knowledge, there are no experimental studies evaluating the bone mineral density and bone mineral content of subchondral bone, and DXA could be used for this purpose. This investigation proves that DXA is also an effective and precise technique to measure mineral density and to detect bone mass variations in subchondral bone. Global and subchondral knee bone obtained intermediate densitometric values between cortical and trabecular bone that were quantitatively superior at the subchondral bone, suggesting that this area might have a transitional structure between trabecular and cortical bone, with a higher content in cortical tissue. In fact, BMD values in subchondral bone obtained through DXA were quite similar to those in the proximal tibial metaphysis, and it could be speculated that both may have a parallel behaviour in osteoporosis.
Finally, cortical bone showed a high inter-individual variation coefficient in BMC and BMD, a finding which has also been described in human cadaveric tibiae [48]. Those variations could be explained by difficulties in repositioning the animal in a reproducible manner or by the fact that bone at this level could be more metaphyseal than diaphyseal and, therefore, have a higher composition of trabecular bone than in the mid-shaft, which is the area usually evaluated for cortical bone in humans [56, 57]. However, histomorphometric studies have shown that bone at the level assessed here is predominantly cortical [58], and the greater BMC and BMD values in this area when compared with lumbar vertebrae, suggest that it represents a tissue with densitometric characteristics of compact bone. Further investigations using volumetric techniques based on microscopic computed tomography will probably help to clarify the bony structure of the knee.
In summary, experimental models of osteoporosis in rabbits can be very helpful, especially to investigate agents with bone anabolic properties, because this animal has a fast bone turnover with predominant remodelling over the modelling processes in comparison with others species. Osteoporosis can be induced in the rabbit by a combination of ovariectomy and corticosteroid treatment in a reproducible manner, and significant trabecular and cortical bone loss is obtained within a short period of time. DXA is a precise technique to evaluate BMC and BMD in trabecular and subchondral bone in this animal. Densitometric values of subchondral bone are closer to those of cortical tissue than trabecular tissue, suggesting that subchondral and cortical bone might follow similar changes in osteoporosis. This constitutes a key finding when the relationship between osteoporosis and other joint diseases, where subchondral bone may play an important role, is under study.
References
Turner AS. Animal models of osteoporosis—necessity and limitations. Eur Cell Mater 2001;1:66–81
Turner RT, Maran A, Lotinun S, et al. Animal models for osteoporosis. Rev Endocr Metab Disord 2001;2:117–27
Bellino FL. Non-primate animal models of menopause: workshop report. Menopause 2000;7:14–24
Kimmel DB. Animal models for in vivo experimentation in osteoporosis research. In: Marcus R, Feldman D, Kelsey J, editors. Osteoporosis, vol 2. San Diego, California: Academic Press; 2001. pp. 29–47
Thompson DD, Simmons HA, Pirie CM, Ke HZ. FDA Guidelines and animal models for osteoporosis. Bone 1995;17 Suppl 4:125S–33S
Wronski TJ, Lowry PL, Walsh CC, Ignaszewski LA. Skeletal alterations in ovariectomized rats. Calcif Tissue Int 1985;37:324–8
Wronski TJ, Walsh CC, Ignaszewski LA. Histologic evidence for osteopenia and increased bone turnover in ovariectomized rats. Bone 1986;7:119–23
Wronski TJ, Yen CF. The ovariectomized rat as an animal model for postmenopausal bone loss. Cell Mater 1992;1 Suppl 1:69–74
Kalu DN. The ovariectomized rat as a model of postmenopausal osteopenia. Bone Miner 1991;15:175–91
Frost HM, Jee WSS. On the rat model of human osteopenia and osteoporoses. Bone Miner 1992;18:227–36
Rodgers JB, Mounier-Faugere MC, Malluche H. Animal models for the study of bone loss after cessation of ovarian functions. Bone 1993;14:369––77
Mosekilde Li. Assessing bone quality—animal models in preclinical osteoporosis research. Bone 1995;17 Suppl 4:343S–52S
Gilsanz V, Roe TF, Gibbens DT, et al. Effect of sex steroids on peak bone density of growing rabbits. Am J Physiol 1988;255:E416–E21
Wu DD, Boyd RD, Fix TJ, Burr DB. Regional patterns of bone loss and altered bone remodelling in response to calcium deprivation in laboratory rabbits. Calcif Tissue Int 1990;47:18–23
Newman E, Turner AS, Wark JD. The potential of sheep for the study of osteopenia: current status and comparison with other animal models. Bone 1995;16:277S–84S
Lindgren JU, DeLuca HF, Mazess RB. Effects of 1,25(OH)2D3 on bone tissue in the rabbit: studies on fracture healing, disuse osteoporosis, and prednisone osteoporosis. Calcif Tissue Int 1984;35:591––5
Griffin MG, Kimble R, Hopfer W, Pacifici R. Dual-energy X-ray absorptiometry of the rat: accuracy, precision and measurement of bone loss. J Bone Min Res 1993;8:795–800
Casez JP, Muehlbauer RC, Lippuner K, Kelly T, Fleisch H, Jaeger P. Dual-energy X-ray absorptiometry for measuring total bone mineral content in the rat: study of accuracy and precision. Bone Miner 1994;26:61–8
Yamauchi H, Kushida K, Yamazaki K, Inoue T. Assessment of spine bone mineral density in ovariectomized rats using DXA. J Bone Miner Res 1995;10:1033–9
Petersen MM, Nielsen PK, Lewin E, Olgaard K. Dual energy X-ray absorptiometry in small rats with low bone mineral density. Calcif Tissue Int 2000;67:455–9
Southard TE, Southard KA, Krizan KE, et al. Mandibular bone density and fractal dimension in rabbits with induced osteoporosis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2000;89:244–9
Cao T, Shirota T, Yamazaki M, Ohno K, Michi KI. Bone mineral density in mandibles of ovariectomized rabbits. Clin Oral Implants Res 2001;12:604–8
Mori H, Manabe M, Kurachi Y, Nagumo M. Osteointegration of dental implants in rabbit bone with low mineral density. J Oral Maxillofac Surg 1997;55:351–61
Cao T, Shirota T, Ohno K, Michi KI. Mineralized bone loss in partially edentulous trabeculae of ovariectomized rabbit mandibles. J Periodontal Res 2004;39:37–41
Dequeker J. The relationship between osteoporosis and osteoarthritis. Clin Rheum Dis 1985;11:271–96
Norris SA, Pettifor JM, Gray DA, Biscardi A, Buffenstein R. Validation and application of dual-energy X-ray absorptiometry to measure bone mineral density in rabbit vertebrae. J Clin Densitom 2000;3:49–55
Pogrund H, Rutenberg M, Makin M, Robin GC, Steinberg R, Bloom R. Osteoarthritis of the hand and osteoporosis. Clin Orthop Relat Res 1986;203:239–43
Hart DJ, Mootoosamy I, Doyle DV, Spector TD. The relationship between osteoarthritis and osteoporosis in the general population: the Chingford Study. Ann Rheum Dis 1994;53:158–62
Sambrook P, Naganathan V. What is the relationship between osteoarthritis and osteoporosis? Bailliere’s Clin Rheumatol 1997;11:695–710
Verstraeten A, van Ermen H, Haghebaert G, Nijs J, Geusens P, Dequeker J. Osteoarthrosis retards the development of osteoporosis. Clin Orthop Relat Res 1991;264:169–77
Grynpas MD, Alpert B, Katz I, Lieberman I, Pritzker KP. Subchondral bone in osteoarthritis. Calcif Tissue Int 1991;49:20–6
Benito MJ, Sánchez-Pernaute O, López Armada MJ, et al. Cyclosporin A prevents the histologic damage of antigen arthritis without inducing fibrosis. Arthritis Rheum 2000;43:311–9
Messner K, Fahlgren A, Ross I, Andersson B. Simultaneous changes in bone mineral density and articular cartilage in a rabbit meniscectomy model of knee osteoarthrosis. Osteoarthritis Cartilage 2000;8:197–206
Glueer CC, Blake G, Lu Y, Blunt BA, Jergas M, Genant HK. Accurate assessment of precision errors: how to measure the reproducibility of bone densitometry techniques. Osteoporos Int 1995;5:262–70
Eberhardt AW, Yeager-Jones A, Blair HC. Regional trabecular bone matrix degeneration and osteocyte death in femora of glucocorticoid-treated rabbits. Endocrinology 2001;142:1333–40
Kaplan HM, Timmons EH. The rabbit. A model for the principles of mammalian physiology and surgery. New York: Academic Press; 1979
Dupont WD, Plummer WD. Power and sample size calculations: a review and computer program. Control Clin Trials 1990;11:116–28
Simon MR. The rat as an animal for the study of senile idiopathic osteoporosis. Acta Anat 1984;119:248–50
Grardel B, Sutter B, Flautre B, Viguier E, Lavaste F, Hardouin P. Effects of corticosteroids on skeletal growth in rabbits evaluated by dual-photon absorptiometry, microscopic connectivity and vertebral compressive strength. Osteoporos Int 1994;4:204–10
Gala-Paniagua J, Díaz Curiel M, De la Piedra Gordo C, Castilla Reparaz C, Torralbo García M. Bone mass assessment in rats by dual energy X-ray absorptiometry. Br J Radiol 1998;71:754–8
Shipp CC, Berger PS, Deehr MS, Dawson-Hughes B. Precision of dual photon absorptiometry. Calcif Tissue Int 1988;42:287–92
Grier SJ, Turner AS, Alvis MR. The use of dual-energy x-ray absorptiometry in animals. Invest Radiol 1996;31:50–62
Sartoris DJ, Resnick D. Current and innovative methods for noninvasive bone densitometry. Radiol Clin North Am 1990;28:257–78
Lang P, Steiger P, Faulkner K, Glueer CC, Genant HK. Osteoporosis. Current techniques and recent developments in quantitative bone densitometry. Radiol Clin North Am 1991;29:49–76
Guglielmi G, Glueer CC, Majumdar S, Blunt BA, Genant HK. Current methods and advances in bone densitometry. Eur Radiol 1995;5:129–39
Hartke JR. Non-primate models of osteoporosis. Lab Anim Sci 1998;48:623–9
Schmidt C, Priemel M, Kohler T, et al. Precision and accuracy of peripheral quantitative computed tomography (pQCT) in the mouse skeleton compared with histology and microcomputed tomography (μCT). J Bone Miner Res 2003;18:1486–96
Veitch SW, Findlay SC, Ingle BM, et al. Accuracy and precision of peripheral quantitative computed tomography measurements at the tibial metaphysis. J Clin Densitom 2004;7:209–17
Rozenberg S, Vandromme J, Neve J, et al. Precision and accuracy of in-vivo bone mineral measurement in rats using dual-energy X-ray absorptiometry. Osteoporos Int 1995;5:47–53
Jayo MJ, Rankin SE, Weaver DS, Carlson CS, Clarkson TB. Accuracy and precision of lumbar bone mineral content by dual-energy X-ray absorptiometry in live female monkeys. Calcif Tissue Int 1991;49:438–40
Turner AS, Mallinckrodt CH, Alvis MR, Bryant HU. Dual-energy X-ray absorptiometry in sheep: experiences with in vivo and ex vivo studies. Bone 1995;17 Suppl 4:381S–7S
Faulkner KG, Glueer CC, Estilo M, Genant HK. Cross-calibration of DXA equipment: upgrading from Hologic QDR 1000/W to QDR 2000. Calcif Tissue Int 1993;52:79–89
Barthe N, Braillon P, Ducassou D, Basse-Cathalinat B. Comparison of two Hologic DXA systems (QDR 1000 and QDR 4500/A). Br J Radiol 1997;70:728–39
Dal Mas I, Biscardi A, Ripamonti U. Comparison of two Hologic DXA systems (QDR 1000 and QDR 4500/A for in vivo bone mass measurement in the baboon (Papio ursinus). J Clin Densitom 2002;5:313–8
Burr DB. Anatomy and physiology of the mineralized tissues: role in the pathogenesis of osteoarthritis. Osteoarthritis Cartilage 2004;12:S20–S30
Sievanen H, Oja P, Vuori I. Precision of dual-energy X-ray absorptiometry in determining bone mineral density and content of various skeletal sites. J Nucl Med 1992;33:1137–42
Casez JP, Troendle A, Lippuner K, Jaeger P. Bone mineral density at the tibia using dual-energy X-ray absorptiometry in normal women and in patients with vertebral osteoporosis or primary hyperparathyroidism. J Bone Miner Res 1994;9:1851–7
Kaartinen E. A histomorphometric study of rabbit tibiofibular bones after rigid intramedullary nailing. Ann Chir Gynaecol 1993;82:189–93
Acknowledgements
The authors wish to thank Dr. C. de la Piedra Gordo, from the Bone Metabolism Research Unit, Dr. O. Sánchez-Pernaute, from the Inflammation Research Unit, and M. Torralbo García, from the Bone Densitometry Unit (Fundación Jiménez Díaz) for their valuable advice and technical assistance in this study. This work was partly funded by grants from the Comunidad Autónoma de Madrid (CAM, 08.7/0007.1/2003), the Spanish Ministerio de Ciencia y Tecnología (SAF 2003/08379) and Fondo de Investigaciones Sanitarias (FIS CP03/0011). M.E.M. is a fellow of the Fundación Conchita Rábago.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Castañeda, S., Largo, R., Calvo, E. et al. Bone mineral measurements of subchondral and trabecular bone in healthy and osteoporotic rabbits. Skeletal Radiol 35, 34–41 (2006). https://doi.org/10.1007/s00256-005-0022-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-005-0022-z