Abstract
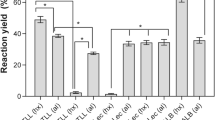
The substrate selectivity of numerous commercially available lipases from microorganisms, plants and animal tissue towards 9-octadecenoic acids with respect to the cis/trans configuration of the CC double bond was examined by the esterification of cis- and trans-9-octadecanoic acid (oleic and elaidic acid respectively) with n-butanol in n-hexane. A great number of lipases studied, e.g. those from Pseudomonas sp., porcine pancreas or Carica papaya, were unable to discriminate between the isomeric 9-octadecenoic acids. However, lipases from Candida cylindracea and Mucor miehei catalysed the esterification of oleic acid 3–4 times faster than the corresponding reaction of elaidic acid and therefore have a high preference for the cis isomer. Of all biocatalysts examined, only recombinant lipases from Candidaantarctica favoured elaidic acid as substrate. While the preference of Candida antarctica lipase B for the trans isomer was quite low, Candida antarctica lipase A had an extraordinary substrate selectivity and its immobilized enzyme preparation [Chirazyme L-5 (3) from Boehringer] esterified elaidic acid about 15 times faster than oleic acid.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 29 October 1998 / Received revision: 18 December 1998 / Accepted: 21 December 1998
Rights and permissions
About this article
Cite this article
Borgdorf, R., Warwel, S. Substrate selectivity of various lipases in the esterification of cis- and trans-9-octadecenoic acid. Appl Microbiol Biotechnol 51, 480–485 (1999). https://doi.org/10.1007/s002530051420
Issue Date:
DOI: https://doi.org/10.1007/s002530051420