Abstract
Background
Doppler US to measure abdominal blood flow velocities (ABFV) is increasingly used to investigate intestinal haemodynamics in several clinical conditions in neonates. Studies that provide reference values of ABFV during the entire neonatal period are currently lacking.
Objective
To make available normal reference values of ABFV and Doppler indices in the coeliac trunk and superior mesenteric artery during the first month of life in term and healthy preterm infants.
Materials and methods
ABFV were obtained with colour Doppler US in 69 neonates (12 term, 57 preterm) divided into four gestational age groups (25–28 weeks, 29–32 weeks, 33–36 weeks, and 37–41 weeks).
Results
ABFV increased with increasing gestational and postnatal age. We also provide normal reference values of ABFV and Doppler indices to compare with measurements of abdominal blood flow changes during the neonatal period for diagnostic, therapeutic and prognostic purposes.
Conclusion
These longitudinal reference values provide a useful tool for assessing possible alteration in ABFV secondary to neonatal pathologies.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Abdominal US is a powerful diagnostic tool in neonatal intensive care units. It is used to provide both morphological information and quantitative blood flow velocity data, exploiting the Doppler effect. In particular, colour Doppler flow imaging allows clear and rapid visualization of the splanchnic vascular anatomy. The study of abdominal blood flow velocities (ABFV) in adults provides useful information about the mesenteric circulation in physiological and pathological situations [1, 2]. Doppler US examination of the abdominal circulation in neonates has recently become a recognized method in the detection of impaired intestinal function [3].
Several studies have examined abdominal flowmetry in order to establish standards of normality, but have not reported normal values of ABFV and Doppler indices (DI) in preterm infants of differing gestational age (GA) and beyond the first week of life [4–6]. The aims of this study were to establish normal values of ABFV and DI for the coeliac trunk (CT) and the superior mesenteric artery (SMA) in healthy term and preterm infants during the first month of life, and to verify the presence of ABFV changes in the CT and SMA at different GAs.
Materials and methods
Subjects
Informed consent was obtained and the study protocol and consent forms were approved by the Ethics Committee of the Department of Paediatrics. The study included neonates with the following characteristics: absence of fetal distress and prenatal flowmetric alterations; absence of maternal pathology; absence of asphyxia at birth; birth weight appropriate for GA; absence of congenital malformations; no evidence of septicaemia during the study; normal cranial ultrasound scan; and first Doppler imaging performed after 12 h of age [7–15]. We also considered and excluded all known conditions that could influence ABFV.
Screening methods
Peak systolic velocity (PSV), end-diastolic velocity (EDV), mean velocity (MV), time-averaged MV (TAMV) and DI (resistive index, RI, and pulsatility index, PI) were measured in the CT and the SMA of 402 preterm infants and 20 term infants. Examinations were performed on days 1, 3 and 7 for term infants and on days 1, 3, 7, 14, 21 and 28 for preterm infants.
The neonates were exposed to the following:
-
1.
Prophylaxis for apnoea of prematurity (for infants with a GA <32 weeks) with caffeine citrate: bolus of 10 mg/kg on the first day of life (DOL) and 2.5 mg/kg per day every 24 h from the second DOL until 34 weeks’ postconceptional age. The measurements were made before the administration of each dose [16–19].
-
2.
Pharmacological prophylaxis for ductus arteriosus for infants with a GA ≤30 weeks with 10 mg/kg ibuprofen during the first DOL and 5 mg/kg on the second and third days. All infants showed a patent ductus arteriosus with no haemodynamically significant left-to-right shunt on the first DOL, which was closed on the third DOL. Flowmetric measurements were obtained before the first dose of ibuprofen and 12 h after the third dose [20, 21].
-
3.
Blue light phototherapy in neonates with hyperbilirubinaemia. ABFV was measured before commencement of and about 12 h after phototherapy [22–24].
-
4.
Administration of fluids in term infants. According to our protocol, fluid intake was 60 ml/kg per day on the first day, increasing by 10 ml/kg in the first 3 days, and 20 ml/kg from the fourth to the sixth day to reach 160 ml/kg per day from the seventh day onwards. During phototherapy the intake was increased by 20%.
Fluid was administered through a venous umbilical catheter during the first three DOL, followed by a percutaneous catheter. Umbilical arterial lines were never placed.
None of the neonates suffered from hypotension or was exposed to drug therapies that could have changed arterial blood pressure or ABFV. During the study period all neonates were normoxic (transcutaneous pO2 60–80 mmHg), normocapnic (transcutaneous pCO2 40–50 mmHg) with a normal oxygen arterial saturation (SatO2 94–98%) and without metabolic acidosis. All studied neonates showed haematocrit levels of 40–55% and examinations were performed remote from any blood transfusion. They were normothermic and never suffered from any metabolic pathology [25]. None of these infants showed significant apnoea or bradycardia during the 24-h prior to the flowmetric study. In total, 756 measurements (378 in the CT and 378 in the SMA) were obtained.
Study group
After excluding all known conditions able to influence ABFV, 69 of 422 screened infants (12 term, 57 preterm) were selected and enrolled in this study. We considered as “healthy” both term and preterm babies. The GA of the study group ranged from 25 to 42 weeks and birth weight ranged from 550 to 4,200 g. The neonates were divided into four groups: group 1 was composed of 12 neonates with GA 25–28 weeks; group 2 included 20 neonates with GA 29–32 weeks; group 3 was composed of 25 neonates with GA 33–36 weeks; and group 4 included 12 neonates with GA 37–41 weeks. The characteristics of the study groups are detailed in Table 1.
-
Group 1:
Infants showed respiratory insufficiency and/or respiratory distress syndrome for which endotracheal intubation and respiratory support was necessary for several days (range 1–7 days, mean 4 days). We used synchronized mechanical ventilation to avoid phases of asynchronous ventilation. Blood flow velocities were measured when the infant was in a stable condition. Enteral feeding with breast milk was started on the third DOL and was increased daily by 10 ml/kg per day. Infants showed neither feeding problems nor signs of abdominal pathology. Enteral feeding was administered as a continuous nasogastric infusion [26]. Breast milk was fortified with supplemental protein intake after the 15th DOL.
-
Group 2:
None of the neonates in this group suffered from respiratory or metabolic disease. Enteral feeding with breast milk was started on the third DOL, continuing with daily increases of 10–20 ml/kg per day. No infant showed feeding disorders or intestinal pathology. The infants were initially fed by intermittent nasogastric infusion, and later a bottle was used [26]. Breast milk was fortified with a supplemental protein intake after the 15th DOL.
-
Group 3:
None of these infants suffered from respiratory, metabolic or gastrointestinal pathology. Enteral feeding was started on the first DOL with daily increases of 10–20 ml/kg. Feeds were administered through a nasogastric tube or bottle [26].
-
Group 4:
This group included healthy full-term neonates, without fetal distress or birth asphyxia. Breast feeding was started on the first DOL.
Measurement of variables
During the examinations neonates were placed in the supine position. Rarely, in the presence of bowel gas, they were placed on the right side. Heart rate, respiratory rate and oxygen saturation were measured using a Hewlett-Packard HP monitor (HP M1205A OmniCare; Hewlett-Packard, Andover, MA); transcutaneous O2 and CO2 were measured using a Tina radiometer (Radiometer Medical, Copenhagen, Denmark); and blood pressure was measured using a noninvasive oscillometric Dinamap (Critikon, Tampa, FL). In all groups ABFV was measured 30 min before breast feeding.
Flow imaging and blood flow velocity were obtained using a colour Doppler unit (Hewlett-Packard Doppler Image Point; Andover, MA) with a short-range 7.5-MHz curved linear array probe. The transmitted Doppler frequency was 5 MHz with a 50-Hz filter. The intensity emitted by the transducer was 15.5 mW/cm2 spatial peak temporal average (SPTA). During abdominal imaging the US power was reduced to 50%. The percentage of US emission (‘duty factor’) was 0.1%. The Doppler gain was 80±5%. The mean time of each measurement was 6 min (range 5–9 min) and exposure time was less than 1 s. The system simultaneously displayed two-dimensional images, the colour flow image and blood flow velocity waves.
The CT and SMA were visualized in the sagittal plane using a probe placed under the xiphoid apophysis. The mean volume was placed about 0.5 cm from the next portion of the vessel to avoid aortic blood flow. In the CT there was no need for angle correction, while in the SMA (about 50% of the studied subjects) a correction of insonation angle by 4–7° was necessary. To maintain accuracy, ABFV measurements were obtained from homogeneous blood flow wave patterns over ten cardiac cycles.
PSV and EDV were calculated from the Doppler display of velocities obtained with vessel insonation. MV was obtained by integrating the area under the velocity curve over one cardiac cycle and TAMV was obtained by gathering all velocities from the mid-point of the area under the Doppler curve (mean of all detectable velocities). The RI or Pourcelot index and the PI or Gosling index, representing the ratio between different flow velocities, were calculated on the basis of the following formulas: RI=PSV−EDV/PSV; PI=PSV−EDV/MV [27, 28]. All SMA and CT blood flow velocities were measured by a single operator (P.P.).
Statistical analysis
The results are presented as means±standard deviation. One-way analysis of variance (ANOVA) was used for comparisons of blood flow velocities and DI between the groups of neonates, and repeated measures ANOVA was used to compare quantitative variables on different days in the same group. Bonferroni's correction was used for multiple comparisons. STATA/IC 10.0 software (STATACORP LP, College Station, TX) was used for statistical analysis. P values <0.05 were considered statistically significant.
Results
Superior mesenteric artery
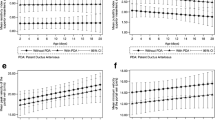
Table 2 shows ABFV values in the SMA in groups of different GA during the study period. The study revealed two interesting findings: 1) flow velocities increased in all groups, but each group maintained a different trend in the SMA during the first month of life; 2) ABFV in Groups 2 and 3 at the 28th DOL were nearly the same as those of term babies.
Peak systolic velocity
A significant increase in PSV was evident from birth to the seventh DOL in group 4 and from birth through the first month of life in the other groups. Comparison of daily data among the groups showed that term infants had significantly higher PSV values than preterm infants. Among preterm infants, group 1 had significantly lower values than groups 2 and 3 during the entire first month of life. Group 3 had significantly higher values than group 2 on DOL 3, 21, and 28.
End-diastolic velocity
EDV also significantly increased in all groups on the different days analysed. EDV of term neonates was significantly higher than that of preterm infants in the first week. Among preterm infants, group 1 had lower values than group 2, but the difference was significant only on the first DOL. Group 1 had significantly lower EDV values than group 3 in the first 2 weeks of life. Group 2 had lower EDV values than group 3, but the difference was significant only on DOL 14.
Mean velocity
MV values in the SMA showed significant daily increases in the three preterm groups, while they remained stable in term infants. Term neonates had significantly higher MV values than all preterm groups in the first week of life. Comparison among the three preterm groups showed lower EDV values in group 1 than in group 2, but the difference was significant only on DOL 1, 7, and 21. Group 3 showed higher EDV values than group 1 in the first month of life, but statistical significance was reached only in the first 2 weeks of life. Similarly, group 3 had higher EDV values than group 2, but the difference was significant only on DOL 7 and 14.
Time-averaged mean velocity
Like MV, TAMV values in the SMA also showed a progressive significant daily increase in the three preterm groups, while they remained stable in term infants. Term neonates had significantly higher TAMV values than all preterm groups in the first week of life. Group 1 had lower TAMV values than group 2, but the difference was significant only on DOL 14. Group 1 TAMV values were significantly lower than those of group 3 in the first 2 weeks of life. Group 3 had significantly higher TAMV values than group 2 on DOL 3, 7, and 14.
Doppler indices
There were no significant changes in RI and PI in this study.
Coeliac trunk
Table 3 shows ABFV values in the CT in groups of different GA. Key observations include: 1) PSV increased in all groups, but each group maintained a different trend during the first month of life; 2) EDV progressively decreased in preterm infants during the first month of life; and 3) PSV values of preterm infants were about the same as those of term neonates on DOL 28.
Peak systolic velocity
PSV showed significant daily increases in the four groups, as in the SMA. PSV values in term infants were significantly higher than in the three preterm groups. Among preterm infants, group 1 had slightly lower values than group 2, and significantly lower values than group 3. Group 3 had significantly higher values than group 2 on DOL 1, 14, 21, and 28.
End-diastolic velocity
During the first week of life, EDV values remained stable in term and preterm infants. Later, a significant daily decrease was observed in preterm infants; data for term infants were lacking. Term neonates had significantly higher EDV values than groups 1 and 2, whereas the values were significantly higher than those of group 3 only on DOL 1 and 7. No significant differences were found among the three groups of preterm infants.
Mean velocity
MV values were stable in term neonates in the first week of life. Among preterm infants, MV increased significantly in group 1 and remained stable in the first month of life in groups 2 and 3. During the first week of life term infants showed significantly higher MV values than each group of preterm neonates. No significant differences were observed among the preterm infant groups.
Time-averaged mean velocity
Like MV, TAMV was stable in term neonates in the first week of life. Among preterm infants, TAMV increased significantly in groups 1 and 3 but remained stable in group 2 during the first month of life. Comparison of daily values among the groups showed significantly higher MV values in group 4 than in other groups. No significant differences were identified among the preterm groups except a significantly lower TAMV in group 1 than in groups 2 and 3 on the first DOL.
Doppler indices
Unlike the SMA results, RI and PI in the CT showed significant increases in all groups during the first month of life. No differences in daily values were observed among the groups.
Discussion
The Doppler technique was first used in adults for noninvasive measurement of intestinal blood flow [29]. The first study of abdominal Doppler flowmetry in infants was performed by Leidig in 1989, and the use of the colour Doppler flow imaging technique to study neonatal abdominal haemodynamics was validated during the mid 1990s [4, 30]. Intestinal blood flow is regulated by dynamic (arterial blood pressure, circulating blood volume, cardiac output and haematocrit level) and metabolic mechanisms (variations of blood pO2, pCO2, pH and some metabolites). Autoregulation of intestinal blood flow in neonates and especially in premature infants is more limited than in adults, with major predispositions toward ischaemia and haemorrhage [31]. Many physiological, pathological and pharmacological conditions can induce changes in intestinal blood flow, and consequently in ABFV. As in adults, feeding induces an increase in blood flow in both the SMA and the CT in neonates, and reduction or cessation of ABFV increases with feeding can be predictive of feeding-related problems [32–35].
ABFV determination has allowed identification of intestinal perfusion alterations in small-for-GA infants and in babies with intrauterine growth retardation, and has facilitated the identification of ductus arteriosus, especially in neonates at risk of necrotizing enterocolitis [36–41]. Changes in ABFV are induced by indomethacin, caffeine, dopamine and dobutamine, but also by medical interventions such as umbilical arterial catheterization, phototherapy and nasal continuous positive airway pressure [42–55].
This study showed that a progressive increase in ABFV occurs in the SMA from birth and throughout the first month of life in preterm babies. A daily increase in PSV was also evident in the CT in all age groups, but unlike in the SMA, EDV progressively decreased in preterm infants. We cannot exclude the possibility that this trend is the same in term infants. Under normovolaemic conditions the increases in PSV, EDV and MV in the SMA are caused by a redistribution of abdominal systemic blood flow, because of the progressive opening of vascular beds due to a decrease in peripheral resistance. Physiological factors playing a role in the postnatal increase in blood velocities in the SMA include increasing blood pressure, increasing stroke volume and closure of the ductus arteriosus [56]. Nitric oxide, produced by intestinal vascular endothelial cells, could play a key role in the dilatation of the microcirculation. This role decreases with postnatal age. Furthermore, the increase in blood flow velocity should be associated with a further rise of enzymatic activities that increases nitric oxide activity, an age-dependent mechanism in the postnatal period [57]. Feeding could also play a significant role in the increase in ABFV with the rise of metabolic demand that controls the production of vascular endothelial growth factors responsible for anatomic growth of the intestinal vascular bed [58].
Our results showed that the mechanisms regulating the increase in ABFV act in a uniform manner in all babies, not only in the first week but also during the first month of life. This phenomenon appeared about the same in the four study groups, but the results highlight a particular GA-dependent trend. In preterm babies, who at 28 days were nearing the postconceptional term, ABFV were similar to ABFV of term babies during their first week of life. Premature babies with a lower GA (groups 1 and 2), however, maintained lower ABFV values during the first month. The dissimilar absolute trends of the groups could stem from differences in developmental stage of the intestinal vascular network.
There are few published studies investigating colour Doppler flow imaging of the CT, and none examining CT afferent vascular systems in preterm and term neonates. Postnatal increases in PSV in the CT could have the same causes as ABFV increases in the SMA. The EDV reduction that we observed in preterm infants could have been influenced by the extremely complex tributary system of the CT. We hypothesize that gastric, hepatic and splenic vascular resistance changes may affect the trend in the EDV. We cannot exclude the occurrence of the same changes in term neonates given that we only have data for the first seven DOL.
Our results suggest that since ABFV values in each group were lower than postconceptional values, monitoring of intestinal blood flow in neonates must be related not only to GA but also to postnatal age. Our data show the existence of ABFV changes not only in the first week of life, but also during the entire first month of life.
Availability of normal flowmetric references in both the SMA and the CT could be useful in monitoring splanchnic flowmetry in all conditions with an increased necrotizing enterocolitis risk (low PSV in the SMA and high PSV in the CT) and in the evaluation of the ischaemia-reperfusion of established necrotizing enterocolitis (high PSV in the SMA) [38]. A further clinical use of the normal reference for splanchnic flowmetry may be better timing of enteral feeding introduction; this would reduce the risks of feed intolerance, especially for very-low-GA neonates.
Conclusion
This study provided normal reference values of ABFV and DI in the SMA and the CT in term infants during the first week and in preterm infants during the first month of life. These data highlight the differences in ABFV, both in absolute measurement and in chronological trends, between term and preterm neonates of different GA. This difference may be due, if we assume similar developmental stimuli, to different anatomical conditions considered to be GA-dependent. Our data may serve as a reference in neonatal physiology and pathology, allowing the correct interpretation of ABFV changes in the neonatal period. Further studies of the abdominal circulation in preterm babies, especially those of lower GA, and in the tributary vascular systems of the CT are required. These would contribute to the goals of improving the ability to prevent necrotizing enterocolitis and improving approaches to enteral feeding of neonates.
References
Dietrich CF, Jedrzejczyk M, Ignee A (2007) Sonographic assessment of splanchnic arteries and the bowel wall. Eur J Radiol 64:204–212
Perko MJ (2001) Duplex ultrasound for assessment of superior mesenteric artery blood flow. Eur J Vasc Endovasc Surg 21:106–117
Murdoch EM, Sinha AK, Shanmugalingam ST et al (2006) Doppler flow velocimetry in the superior mesenteric artery on the first day of life in preterm infants and the risk of neonatal necrotizing enterocolitis. Pediatrics 118:1999–2003
Leidig E (1989) Pulsed Doppler ultrasound blood flow measurements in the superior mesenteric artery of the newborn. Pediatr Radiol 19:169–172
Van Bel F, Van Zwieten PH, Guit GL et al (1990) Superior mesenteric artery blood flow velocity and estimated volume flow: duplex Doppler US study of preterm and term neonates. Radiology 174:165–169
Maruyama K, Koizumi T, Tomomasa T et al (1999) Intestinal blood-flow velocity in uncomplicated preterm infants during the early neonatal period. Pediatr Radiol 29:472–477
Shanklin DR, Cooke RJ (1993) Effects of intrauterine growth on intestinal length in the human fetus. Biol Neonate 64:76–81
Martinussen M, Brubakk AM, Vik T et al (1997) Relationship between intrauterine growth retardation and early postnatal superior mesenteric artery blood flow velocity. Biol Neonate 71:22–30
Robel-Tillig E, Vogtmann C, Faber R (2000) Postnatal intestinal disturbances in small-for-gestational-age premature infants after prenatal haemodynamic disturbances. Acta Paediatr 89:324–330
Korszun P, Dubiel M, Breborowicz G et al (2002) Fetal superior mesenteric artery blood flow velocimetry in normal and high-risk pregnancy. J Perinat Med 30:235–241
Weir FJ, Ohlsson A, Fong K et al (1999) Does endothelin-1 reduce superior mesenteric artery blood flow velocity in preterm neonates? Arch Dis Child Fetal Neonatal Ed 80:F123–127
Akinbi H, Abbasi S, Hilpert PL et al (1994) Gastrointestinal and renal blood flow velocity profile in neonates with birth asphyxia. J Pediatr 125:625–627
Robel-Tillig E, Vogtmann C, Bennek J (2002) Prenatal hemodynamic disturbances – pathophysiological background of intestinal motility disturbances in small for gestational age infants. Eur J Pediatr Surg 12:175–179
Kempley ST, Murdoch E (2000) Splanchnic haemodynamic disturbances in perinatal sepsis. Arch Dis Child Fetal Neonatal Ed 83:F139–142
Martinussen M, Brubakk AM, Linker DT et al (1994) Mesenteric blood flow velocity and its relation to circulatory adaptation during the first week of life in healthy term infants. Pediatr Res 36:334–339
Lane AJ, Coombs RC, Evans DH et al (1999) Effect of caffeine on neonatal splanchnic blood flow. Arch Dis Child Fetal Neonatal Ed 80:F128–129
Hoecker C, Nelle M, Poeschl J et al (2002) Caffeine impairs cerebral and intestinal blood flow velocity in preterm infants. Pediatrics 109:784–787
Hoecker C, Nelle M, Beedgen B et al (2006) Effects of a divided high loading dose of caffeine on circulatory variables in preterm infants. Arch Dis Child Fetal Neonatal Ed 91:F61–64
Soraisham AS, Elliott D, Amin H (2008) Effect of single loading dose of intravenous caffeine infusion on superior mesenteric artery blood flow velocities in preterm infants. J Paediatr Child Health 44:119–121
Feigen LP, King LW, Ray J et al (1981) Differential effects of ibuprofen and indomethacin in the regional circulation of the dog. J Pharmacol Exp Ther 219:679–684
Grosfeld JL, Kamman K, Gross K et al (1983) Comparative effects of indomethacin, prostaglandin E1, and ibuprofen on bowel ischemia. J Pediatr Surg 18:738–742
Yao AC, Martinussen M, Johansen OJ et al (1994) Phototherapy-associated changes in mesenteric blood flow response to feeding in term neonates. J Pediatr 124:309–312
Pezzati M, Biagiotti R, Vangi V et al (2000) Changes in mesenteric blood flow response to feeding: conventional versus fiber-optic phototherapy. Pediatrics 105:350–353
Kadalraja R, Patole SK, Muller R et al (2004) Is mesenteric blood flow compromised during phototherapy in preterm neonates? Arch Dis Child Fetal Neonatal Ed 89:F564
Nelle M, Hocker C, Zilow EP et al (1994) Effects of red cell transfusion on cardiac output and blood flow velocities in cerebral and gastrointestinal arteries in premature infants. Arch Dis Child Fetal Neonatal Ed 71:F45–48
Lane AJ, Coombs RC, Evans DH et al (1998) Effect of feed interval and feed type on splanchnic haemodynamics. Arch Dis Child Fetal Neonatal Ed 79:F49–53
Pourcelot L (1976) Diagnostic ultrasound for cerebral vascular disease. In: Donald I, Levis N (eds) Present and future of diagnostic ultrasound. Krooyker Science Publishers, Rotterdam, pp 141–171
Gosling RG, King DH (1974) Arterial assessment by Doppler-shift ultrasound. Proc R Soc Med 67:447–449
Jager K, Bollinger A, Valli C et al (1986) Measurement of mesenteric blood flow by duplex scanning. J Vasc Surg 3:462–469
Weir FJ, Fong K, Ryan ML et al (1995) Superior mesenteric artery and renal artery blood flow velocity measurements in neonates: technique and interobserver reliability. Pediatr Radiol 25:145–148
Reber KM, Nankervis CA, Nowicki PT (2002) Newborn intestinal circulation. Physiology and pathophysiology. Clin Perinatol 29:23–39
Gladman G, Sims DG, Chiswick ML (1991) Gastrointestinal blood flow velocity after the first feed. Arch Dis Child 66:17–20
Fang S, Kempley ST, Gamsu HR (2001) Prediction of early tolerance to enteral feeding in preterm infants by measurement of superior mesenteric artery blood flow velocity. Arch Dis Child Fetal Neonatal Ed 85:F42–45
Pezzati M, Dani C, Tronchin M et al (2004) Prediction of early tolerance to enteral feeling by measurement of superior mesenteric artery blood flow velocity: appropriate versus small-for-gestational-age preterm infants. Acta Pediatr 93:797–802
Robel-Tillig E, Knupfer M, Pulzer F et al (2004) Blood flow parameters of the superior mesenteric artery as an early predictor of intestinal dysmotility in preterm infants. Pediatr Radiol 34:958–962
Maruyama K, Koizumi T (2001) Superior mesenteric artery blood flow velocity in small for age infants of very low birth weight during the early neonatal period. J Perinat Med 29:64–70
Wong SN, Lo RN, Hui PW (1990) Abnormal renal and splanchnic arterial Doppler pattern in premature babies with symptomatic patent ductus arteriosus. J Ultrasound Med 9:125–130
Coombs RC, Morgan ME, Durbin GM et al (1992) Abnormal gut blood flow velocities in neonates at risk of necrotising enterocolitis. J Pediatr Gastroenterol Nutr 15:13–19
Kempley ST, Gamsu HR (1992) Superior mesenteric artery blood flow velocity in necrotising enterocolitis. Arch Dis Child 67:793–796
Deeg KH, Rupprecht T, Schmid E (1993) Doppler sonographic detection of increased flow velocities in the celiac trunk and superior mesenteric artery in infants with necrotizing enterocolitis. Pediatr Radiol 23:578–582
Nowicki PT, Nankervis CA (1994) The role of the circulation in the pathogenesis of necrotizing enterocolitis. Clin Perinatol 21:219–234
Grosfeld JL, Chaet M, Molinari F et al (1996) Increased risk of necrotizing enterocolitis in premature infants with patent ductus arteriosus treated with indomethacin. Ann Surg 224:350–355
Coombs RC, Morgan ME, Durbin GM et al (1990) Gut blood flow velocities in the newborn: effects of patent ductus arteriosus and parenteral indomethacin. Arch Dis Child 65:1067–1071
Yanowitz TD, Yao AC, Werner JC et al (1998) Effects of prophylactic low-dose indomethacin on hemodynamics in very low birth weight infants. J Pediatr 132:28–34
Christmann V, Liem KD, Semmekrot BA et al (2002) Changes in cerebral, renal and mesenteric blood flow velocity during continuous and bolus infusion of indomethacin. Acta Paediatr 91:440–446
Hentschel R, Hensel D, Brune T et al (1995) Impact on blood pressure and intestinal perfusion of dobutamine or dopamine in hypotensive preterm infants. Biol Neonate 68:318–324
Seri I, Abbasi S, Wood DC et al (2002) Regional hemodynamic effects of dopamine in the indomethacin-treated preterm infant. J Perinatol 22:300–305
Kempley ST, Gamsu HR (1992) Randomised trial of umbilical arterial catheter position: Doppler ultrasound findings. Arch Dis Child 67:855–859
Davey AM, Wagner CL, Cox C et al (1994) Feeding premature infants while low umbilical artery catheters are in place: a prospective, multicenter trial. J Pediatr 124:795–799
Rand T, Weninger M, Kohlhauser C et al (1996) Effects of umbilical arterial catheterisation on mesenteric hemodynamics. Pediatr Radiol 26:435–438
Roll C, Hanssler L (1998) Effect of umbilical arterial catheters on intestinal blood supply. Acta Paediatr 87:955–959
Shah JB, Bracero LA, Gewitz MH et al (1998) Umbilical artery catheters and blood flow velocities in the superior mesenteric artery: effect of insertion, removal, aspiration, and bolus infusion. J Clin Ultrasound 26:73–77
Havranek T, Johanboeke P, Madramootoo C et al (2007) Umbilical artery catheters do not affect intestinal blood flow responses to minimal enteral feedings. J Perinatol 27:375–379
Kadalraja R, Patole SK, Muller R et al (2004) Is mesenteric blood flow compromised during phototherapy in preterm neonates? Arch Dis Child Fetal Neonatal Ed 89:F564
Havranek T, Madramootoo C, Carver JD (2007) Nasal continuous positive airway pressure affects pre- and postprandial intestinal blood flow velocity in preterm infants. J Perinatol 27:704–708
Havranek T, Thompson Z, Carver JD (2006) Factors that influence mesenteric artery blood flow velocity in newborn preterm infants. J Perinatol 26:493–497
Reber KM, Mager GM, Miller CE et al (2001) Relationship between flow rate and NO production in postnatal mesenteric arteries. J Physiol Gastrointest Liver Physiol 280:G43–G50
Shigeru I, Brawley L, Wheeler T et al (2002) Vasodilatation to vascular endothelial growth factor in the uterine artery of the pregnant rat is blunted by low dietary protein intake. Pediatr Res 51:485–491
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Papacci, P., Giannantonio, C., Cota, F. et al. Neonatal colour Doppler ultrasound study: normal values of abdominal blood flow velocities in the neonate during the first month of life. Pediatr Radiol 39, 328–335 (2009). https://doi.org/10.1007/s00247-008-1112-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-008-1112-6