Abstract
Echocardiography is the mainstay of screening and disease surveillance in isolated left ventricular non-compaction (iLVNC). The aim of our study is to determine the early regional and global myocardial functional changes and whether the myocardial changes that cannot be detected by conventional echocardiography could be detected by tissue Doppler imaging (TDI) or two-dimensional speckle-tracking echocardiography (STE) in iLVNC cases without symptoms. Longitudinal and circumferential strain (S) and strain rates (SR) as determined by STE in 20 children aged 12.1 ± 3.3 years was compared with those in 20 controls. All children underwent echocardiographic assessment using two-dimensional, tissue Doppler and speckle-tracking echocardiography. iLVNC patients who had normal systolic function by ejection and shortening fractions were included in this study. According to the TDI in all three segments [the non-compacted (NC), neighboring NC (NNC) and compacted (C) segments], isovolumic contraction time, isovolumic relaxation time and myocardial performance index values were significantly higher, while ejection time were significantly lower in the iLVNC group. According to STE in two segments (NC and NNC-segments) longitudinal S and SR values and also circumferential S and SR values were significantly lower in the iLVNC group compared with the control group; whereas, in the global measurements both longitudinal and circumferential S and SR values in all three segments were significantly lower in the iLVNC group compared with the control group. We believe that TDI and STE that evaluates myocardial deformation can be used for the detection of early myocardial dysfunction in the iLVNC patients who are subclinical and whose left ventricular functions were detected as normal by conventional methods with normal ejection and shortening fractions.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
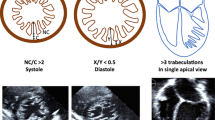
Isolated left ventricular non-compaction (iLVNC) is a rare genetic form of cardiomyopathy. It is thought to be caused by arrest in normal endomyocardial embryogenesis and consists of multiple trabeculations and deep intertrabecular recesses of the myocardium [20, 26]. The characteristic features of iLVNC are a two-layered ventricular wall, which has two parts: a thinner compacted epicardial layer and an inner non-compacted layer. This wall has prominent trabeculations associated with deep, intertrabecular recesses communicating only with the ventricular cavity but not with the coronary circulation [5, 11]. In LVNC, typical prominent trabeculations and deep recesses may be detected by echocardiography, and the ratio of non-compacted (NC) to compacted (C) layers is often used to diagnose this lesion.
The recently introduced strain (S) and strain rate (SR) echocardiography enables angle independent assessment of myocardial deformation in both the longitudinal and circumferential dimensions [8]. The purpose of this study was to determine the potential role of tissue Doppler imaging (TDI) and speckle-tracking echocardiography (STE) for identifying LV dysfunction in patients with iLVNC who had no evidence of cardiac impairment on conventional echocardiography with normal systolic function by ejection fraction (EF) and shortening fraction (FS).
Materials and Methods
Twenty asymptomatic patients who were followed in our pediatric cardiology clinic with the diagnosis of iLVNC were included in this study. iLVNC was diagnosed incidentally in the patients who were consulted to pediatric cardiology clinic because of chest pain or murmur. None of the patients had LV dysfunction by conventional echocardiographic methods (normal EF and FS). A control group included 20 sex- and age-matched healthy children who were evaluated to investigate the etiology of the cardiac murmur and have normal cardiac findings. This prospective study was approved by the hospital’s ethics committee.
The diagnosis of iLVNC by transthoracic echocardiography was made in accordance with suggested criteria. iLVNC patients strictly fulfilled all five echocardiographic diagnostic criteria for iLVNC: (1) absence of co-existing cardiac abnormalities [16]; (2) a two-layered structure of the LV wall, with the end-diastolic ratio of non-compacted to compacted layer ≥2 [14, 25, 28]; (3) finding this structure predominantly in the apical, mid-lateral and mid-inferior regions of the left ventricle [16]; (4) blood flow from the ventricular cavity into more than three deep intertrabecular recesses as assessed by colour Doppler imaging [16, 27, 28]; and (5) same echogenicity of the trabeculaes with myocardial tissue [27].
Different echocardiographic diagnostic criteria have been developed by researchers for the diagnosis of LVNC [7, 16, 24, 27, 28]. In this study, we assessed the wall thickness at the end of the diastole in order to minimize the possibility of false positive results. Similarly, NC/C ratio was evaluated during diastole in the cardiac magnetic resonance-based studies [14, 25, 33].
Two-dimensional grayscale harmonic images at a frame rate of 60–80 frames/s were obtained in the left lateral decubitus position using a commercially available ultrasound system (iE33, Philips, The Netherlands), equipped with a broadband (1–5 MHz) S5-1 transducer (frequency transmitted 1.7 MHz, received 3.4 MHz). Measurements of LV dimensions, FS, and EF were obtained in accordance with the recommendations of the American Society of Echocardiography [19]. Analysis of the datasets was performed using QLAB Advanced Quantification Software (version 6.0, Philips, The Netherlands).
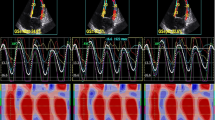
Apical four-chamber views were obtained and longitudinal peak annular velocities during systole (S m), early (E m), and late (A m) diastole, ejection time (ET), isovolumic contraction time (IVCT; the time from the end of A m to the beginning of S m) and isovolumic relaxation time (IVRT; the time from the end of S m to the beginning of E m) were measured throughout a cardiac cycle at apical anterolateral wall, at basal anterolateral wall and at basal interventricular septum [2, 12]. The myocardial performance index (MPI) was calculated at these three segments as follows: (ICT + IRT)/ET [29].
According to the recommendations of the American Society of Echocardiography and European Society of Echocardiography, a 17-segment model was used for assessment of regional LV wall motion [6, 19]. The segmental and global longitudinal myocardial deformation was calculated on seven segments (apical lateral, mid-anterolateral, basal anterolateral, apical septum, mid-inferoseptum, basal inferoseptum and apical cap) based on the entire traced contour of the left ventricles. Similarly, from the short-axis view, the left ventricle was divided into six segments (mid-anteroseptum, mid-anterior, mid-anterolateral, mid-inferolateral, mid-inferior, and mid-inferoseptum) for quantification of segmental and global circumferential myocardial deformation. The calculations were made on these six segments based on the entire short-axis contour at papillary muscle level [6, 19] (Fig. 1).
We entitled the segments according to the involvement of disease as NC, NNC and C. For longitudinal S and SR, apical lateral wall, basal anterolateral wall and basal inferoseptum were accepted as NC segment, NNC segment and C-segment, respectively. In all patients non-compaction was seen in mid-anterior and/or mid-anterolateral walls in the circumferential section. The mid-inferolateral segment adjacent to the mid-anterolateral wall was accepted as the NNC segment and the mid-inferoseptal segment was accepted as the C segment.
Statistical Analysis
The statistical analysis was performed by SPSS (Statistical Package for the Social Sciences, Chicago, IL, version 16.0). Numeric variables are expressed as mean ± SD and categorical ones are expressed as percentages (%). Normal distribution of samples was detected by Shapiro–Wilk test. Comparisons of demographic data and echocardiographic parameters between patients and controls were performed using two independent samples t test for normally distributed variables and by Mann–Whitney U test for non-normally distributed ones. Statistical significance for all analyses was assumed at p < 0.05.
Results
The median age of patients at presentation was 12.1 ± 3.3 years. Non-compaction involved the LV apex, apical lateral and mid-anterolateral wall in all patients. The mean ratio of non-compacted to compacted layers was 2.4 ± 0.4. None of the patients had right ventricular involvement. The body weight, heart rate and body mass index were similar between patients and controls. EF, FS and LV end-diastolic diameter were within normal limits in both patient and control groups by conventional echocardiography. Similarly, there was no statistically significant difference between the patient and the control groups in terms of mitral wave peak-E, peak-A velocities and E/A ratio (Table 1).
Electrocardiographic (ECG) findings of 7 of 20 patients (35 %) with iLVNC showed left axis deviation. Left bundle branch block was observed in four of these patients. The patient group had no specific ECG findings except for increase in the frequency of left axis deviation. Measures of global and regional deformation were not significantly different between these four patients and other iLVNC patients.
Table 2 summarizes the parameters of NC, NNC and C tissue Doppler imaging analysis in patients and controls. IVCT, IVRT and MPI values were statistically significantly longer at NC (p = 0.01, p = 0.01 and p < 0.001, respectively), NNC (p = 0.001, p = 0.005, p < 0. 001, respectively) and C segments (p = 0.01, p = 0.011, p = 0.027, respectively) in the patient group compared to the control group. ET values were significantly shorter at NC (p < 0.001), NNC (p < 0.001) and C segments (p = 0.027) in the patient group compared to the control group. Although S m, E m and A m values were low in all the studied areas, only C-segment E m values were significantly lower in the patient group (p = 0.037).
NC, NNC and C-segment longitudinal S and SR values are shown in Table 3. All values were significantly lower in the patient group than control group except C-segment S and SR values. Global longitudinal S values were −18 ± 2.76 and −23.8 ± 3.27 % (p < 0.001) and global longitudinal SR values were −0.56 ± 0.43 and −0.84 ± 0.41 s−1 (p = 0.04) in the patient and control groups, respectively (Figs. 2 and 3).
NC, NNC and C-segment circumferential S and SR values are shown in Table 4. All values were significantly lower in the patient group than control group except C-segment S and SR values. Global circumferential S values were −24.6 ± 4 and −27.5 ± 3.34 % (p = 0.019); and global circumferential SR values were −0.63 ± 0.52 and −0.96 ± 0.29 s−1 (p < 0.001) in the patient and control groups, respectively (Figs. 2, 3).
Discussion
Myocardial involvement is often asymptomatic in iLVNC patients. Patients often become symptomatic in adulthood. Conventional echocardiographic measurements are often normal in young and asymptomatic patients [22]. Thus, detecting early cardiac involvement in subclinical iLVNC patients is not possible by these methods. For these reasons, there is a need for advanced echocardiography methods [3, 23]. TDI has been used for this purpose for many years but recently a new method, called the STE method, was started to be used by children. TDI is a more sensitive method than conventional echocardiography and both global and regional left ventricular systolic and diastolic functions can be assessed by it [1, 9, 17]. Some studies reported that iLVNC patients’ TDI values are decreased only in patients with low ejection fractions [3, 21]. In contrast, Tufekcioglu et al. [30] showed impaired TDI values in iLVNC patients even if EF is normal. In our study, IVCT, IVRT and MPI values were significantly higher, and ET value was significantly lower in the patient group compared with the control group in all segments.
MPI is a parameter that measures LV systolic and diastolic functions, and the values below 0.40 are accepted as normal [29]. In our study, mean MPI value was significantly higher in the patient group, and this indicates impaired LV systolic and diastolic functions in the patient group. Unlike our study, McMahon et al. [21] examined pediatric patients with low EF. Mitral valve lateral and septal annulus S a, E a and A a values were significantly lower; on the other hand, MPI values were significantly higher in these children. Low EF is a rare finding for pediatric iLVNC patients. We made our TDI studies on myocardial tissue. We found low S m, E m and A m values in NC, NNC and C-segments in our iLVNC patients. However, these values were significantly decreased in the patient group only for the E m value in the C segment.
It has been reported that mitral valve lateral annulus E a value is very important for the prognosis of patients with iLVNC. The value under 7.8 cm/s is a decisive indicator for congestive heart failure requiring hospitalization, heart transplantation and even death [21]. In our study, the mean values for E m by TDI evaluation were 8.9 cm/s in the NC segment. It was noteworthy that E m values were significantly lower in the NC segment and its neighborhood.
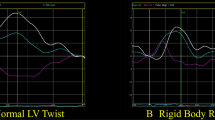
To overcome the limitations of TDI, a new technique called STE was developed. The regional and global myocardial functions can be evaluated quantitatively by using S and SR with the STE technique [8, 10]. STE has beneficial effects to contribute to the prognosis and disease management [23]. There are very few studies in the literature that assess myocardial involvement by S and SR techniques in patients with LVNC [18, 31, 32]. Van Dalen et al. [31, 32] reported impaired twist motion of heart by STE technique in LVNC patients. According to this study, rotation of the base of the heart was clockwise, but rotation of the apex of the heart was counter clockwise in the dilated cardiomyopathy patients and the control group. However, rotation of both base and apex of the heart were in the same direction in the LVNC patients.
Koh et al. [18] studied nine children with LVNC and reported that segmental and global longitudinal S values were significantly lower in the iLVNC patients, as in our study. Also, they found that circumferential S values of apical, mid and basal segments were significantly lower in the patient group. Similarly, S and SR values in NC and NNC segments in circumferential sections were significantly lower in the patient group in our study. For the C segment, although values were lower in the patient group, the difference was not significant. Global circumferential S and SR values were also significantly lower in the patient group in our study. The lower global S and SR values in the iLVNC patient group of our study suggested that longitudinal and circumferential muscle fibers in these patients can be affected even in the subclinical period.
Bellavia et al. [3] divided their adult iLVNC patients into two groups, EF less than 50 % and more than 50 %. Even the normal EF and TDI values, global and apical longitudinal S values were significantly lower than the control group in patients with iLVNC. Similar to the study by Bellavia et al. [3], global longitudinal S values were significantly lower in iLVNC patients compared with the control group in our study. This indicates the impaired systolic function and deformation of LV in the longitudinal axis. We observed the decreased deformation in the longitudinal axis, especially in NC and NNC segments, and this decrease was significant. These results showed that LV systolic functions were affected in iLVNC patients.
In the study by Koh et al. [18], the presence of heart failure in nearly all patients may explain the decrease in S and SR values in these patients. In our study, none of the patients revealed myocardial dysfunction by conventional methods. Myocardial function deteriorates with advancing age in iLVNC patients; this increases the likelihood of patients to become symptomatic. So, as shown by Bellavia et al. [3], myocardial deformation and abnormal TDI values are not surprising findings in iLVNC patients at older ages. The key point for iLVNC patients is detecting the deterioration of myocardial functions before the symptoms started. In our study more patients were evaluated, and none of our patients had myocardial dysfunction compared to the study of Koh et al. We think that the demonstration of impaired myocardial deformation at NC and NNC segments in iLVNC patients with normal LV functions by conventional methods, and impaired myocardial functions at C segment by TDI, is valuable from this perspective by our study.
In a recent study, mortality rate of patients with iLVNC was reported 6 % in the 5-year follow-up. Also, hypertrophic cardiomyopathy and restrictive cardiomyopathy cases were reported in the 2-year follow-up in the patients with iLVNC [15]. Early detection of ventricular dysfunction and arrhythmias can reduce the risk of sudden death [4]. If there is LV dysfunction by conventional echocardiography in patients with iLVNC, medical treatment for heart failure is recommended. If left ventricular dysfunction is detected early by TDI and STE and medical therapy is started at an early stage, the risk of sudden death may be reduced. However, further studies are required to support this opinion.
Limitations
We should address several limitations with regard to the present study. This was a single-center, single-ethnicity study. We did not examine intra- and interobserver variability rates. The TDI method has some limitations such that it can be affected by active and passive tissue movements, and it cannot distinguish translational movements from tethering [13, 23, 25]. During STE, frame rate of 60–80 frames/s was an unavoidable limitation in this study. Maybe these patients will never get sick, therefore a long-term follow up is essential. Despite these limitations, we feel that STE offers additional benefit to clinicians for screening and ongoing surveillance of patients with iLVNC.
Conclusion
As a result, follow up of myocardial functions with TDI and STE that has been detected as normal by conventional echocardiography will enable early detection of subclinical myocardial dysfunction of the iLVNC patients. In our study, while only NC and NNC segments S and SR values were significantly lower with STE, all of the NC, NCC and C-segments showed significant impairments in patients by TDI.
References
Abraham TP, Dimaano VL, Liang HY (2007) Role of tissue Doppler and strain echocardiography in current clinical practise. Circulation 116:2597–2609
Ayabakan C, Özkutlu S (2004) Left ventricular myocardial velocities in healthy children: quantitative assessment by tissue Doppler echocardiography and relation to the characteristics of filling of the left ventricle. Cardiol Young 14:156–163
Bellavia D, Michelena H, Martinez M, Pellikka PA, Bruce CJ, Connolly HM et al (2010) Speckle myocardial imaging modalities for early detection of myocardial impairment in isolated left ventricular non-compaction. Heart 96:440–447
Brescia ST, Rossano JW, Pignatelli R, Jefferies JL, Price JF, Decker JA et al (2013) Mortality and sudden death in pediatric left ventricular noncompaction in a tertiary referral center. Circulation 127:2202–2208
Burke A, Mont E, Kutys R, Virmani R (2005) Left ventricular noncompaction: a pathological study of 14 cases. Hum Pathol 36(4):403–411
Cerqueira MD, Weissman NJ, Dilsizian V, Jacobs AK, Kaul S, Laskey WK et al (2002) Standardized myocardial segmentation and nomenclature for tomographic imaging of the heart: a statement for healthcare professionals from the cardiac imaging committee of the council on clinical cardiology of the American Heart Association. Circulation 105:539–542
Chin T, Perloff J, Williams R, Jue K, Mohrmann R (1990) Isolated noncompaction of left ventricular myocardium. A study of eight cases. Circulation 82:507–513
D’hooge J, Heimdal A, Jamal F, Kukulski T, Bijnens B, Rademakers F et al (2000) Regional strain and strain rate measurements by cardiac ultrasound: principles, implementation and limitations. Eur J Echocardiogr 1:154–170
Gorcsan J 3rd, Deswal A, Mankad S, Mandarino WA, Mahler CM, Yamazaki N et al (1998) Quantification of the myocardial response to low-dose dobutamine using tissue Doppler echocardiographic measures of velocity and velocity gradient. Am J Cardiol 81(5):615–623
Greenberg NL, Firstenberg MS, Castro PL, Main M, Travaglini A, Odabashian JA et al (2002) Doppler-derived myocardial systolic strain rate is a strong index of left ventricular contractility. Circulation 105:99–105
Hamamichi Y, Ichıda F, Hasihimoto I, Uese KHK, Miyawaki T, Tsukano S (2001) Isolated noncompaction of the ventricular myocardium: ultrafast computed tomography and magnetic resonance imaging. Int J Cardiovasc Imaging 17(4):305–314
Harada K, Orino T, Yasuoka K, Tamura M, Takada G (2000) Tissue Doppler imaging of left and right ventricles in normal children. Tohoku J Exp Med 191:21–29
Heimdal A, Stoylen A, Torp H, Skjaerpe T (1998) Real-time strain rate imaging of the left ventricle by ultrasound. J Am Soc Echocardiogr 11:1013–1019
Jacquier A, Thuny F, Jop B, Giorgi R, Cohen F, Gaubert JY et al (2010) Measurement of trabeculated left ventricular mass using cardiac magnetic resonance imaging in the diagnosis of left ventricular non-compaction. Eur Heart J 31:1098–1104
Jefferies JL, Wilkinson JD, Sleeper LA, Colan SD, Lu M, Pahl E et al (2015) Cardiomyopathy phenotypes and outcomes for children with left ventricular myocardial noncompaction: results from the pediatric cardiomyopathy registry. J Card Fail S1071-9164(15)00580-1. doi:10.1016/j.cardfail.2015.06.381
Jenni R, Oechslin E, Schneider J, Attenhofer Jost C, Kaufmann P (2001) Echocardiographic and pathoanatomical characteristics of isolated left ventricular non-compaction: a step towards classification as a distinct cardiomyopathy. Heart 86:666–671
Khraiche D, Pellerin D (2009) Tissue Doppler, Doppler strain, and non-Doppler strain: tips, limitations, and applications. In: Nihoyannopoulos P, Kisslo J (eds) Echocardiography. Springer, London, pp 79–100
Koh C, Hong WJ, Wong SJ, Cheung YF (2010) Systolic-diastolic coupling of myocardial deformation of the left ventricle in children with left ventricular noncompaction. Heart Vessels 25:493–499
Lang RM, Bierig M, Devereux RB, Flachscampf FA, Foster E, Pellikka PA et al (2005) Recommendations for chamber quantification: a report from the American Society of Echocardiography’s Guidelines and Standards Committee and the Chamber Quantification Writing Group, developed in conjunction with the European Association of Echocardiography, a branch of the European Society of Cardiology. J Am Soc Echocardiogr 18:1440–1463
Maron BJ, Towbin JA, Thiene G, Antzelevitch C, Corrado D, Arnett D et al (2006) Contemporary definitions and classification of the cardiomyopathies: an American heart association scientific statement from the council on clinical cardiology, heart failure and transplantation committee; quality of care and outcomes research and functional genomics and translational biology interdisciplinary working groups; and council on epidemiology and prevention. Circulation 113(14):1807–1816
McMahon CJ, Pignatelli RH, Nagueh SF, Lee VV, Vaughn W, Valdes SO et al (2007) Left ventricular non-compaction cardiomyopathy in children: characterisation of clinical status using tissue Doppler-derived indices of left ventricular diastolic relaxation. Heart 93:676–681
Murphy RT, Thaman R, Blanes JG, Ward D, Sevdalis E, Papra E et al (2005) Natural history and familial characteristics of isolated left ventricular non-compaction. Eur Heart J 26(2):187–192
Nesbitt GC, Mankad S, Oh JK (2009) Strain imaging in echocardiography: methods and clinical applications. Int J Cardiovasc Imaging 25:9–22
Paterick TE, Jamil Tajik A (2014) Left ventricular noncompaction cardiomyopathy: lessons from the past to explain a diagnostic conundrum. Letters to the editor. J Am Soc Echocardiogr 27(10):1128–1130
Petersen SE, Selvanayagam JB, Wiesmann F, Robson MD, Francis JM, Anderson RH et al (2005) Left ventricular non-compaction: insights from cardiovascular magnetic resonance imaging. J Am Coll Cardiol 46:101–105
Samsa LA, Yang B, Lıu J (2013) Embryonic cardiac chamber maturation: trabeculation, conduction, and cardiomyocyte proliferation. Am J Med Genet C Semin Med Genet 163C:157–168
Stollberger C, Finsterer J, Blazek G (2002) Left ventricular hypertrabeculation/noncompaction and association with additional cardiac abnormalities and neuromuscular disorders. Am J Cardiol 90:899–902
Stollberger C, Finsterer J (2004) Left ventricular hypertrabeculation/noncompaction. J Am Soc Echocardiogr 17(1):91–100
Tei C, Ling LH, Hodge DO, Bailey KR, Oh JK, Rodeheffer RJ et al (1995) New index of combined systolic and diastolic myocardial performance: a simple and reproducible measure of cardiac function—a study in normals and dilated cardiomyopathy. J Cardiol 26(6):357–366
Tufekçioglu O, Aras D, Yıldız A, Topaloglu S, Maden O (2008) Myocardial contraction properties along the long and short axes of the left ventricle in isolated left ventricular non-compaction: pulsed tissue Doppler echocardiography. Eur J Echocardiogr 9:344–350
Van Dalen B, Caliskan K, Soliman O, Nemes A, Vletter W, ten Cate F et al (2008) Left ventricular solid body rotation in non-compaction cardiomyopathy: a potential new objective and quantitative functional diagnostic criterion? Eur J Heart Failure 10:1088–1093
Van Dalen B, Caliskan K, Soliman O, Kauer F, van der Zwaan H, Vletter W et al (2011) Diagnostic value of rigid body rotation in noncompaction cardiomyopathy. J Am Soc Echocardiogr 24:548–555
Willemsen HM, van den Berg MP (2011) A few more pieces in the puzzle of noncompaction cardiomyopathy. Eur J Heart Fail 13:127–129
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ari, M.E., Cetin, I.I., Kocabas, A. et al. Decreased Deformation in Asymptomatic Children with Isolated Left Ventricular Non-compaction and Normal Ejection Fraction. Pediatr Cardiol 37, 201–207 (2016). https://doi.org/10.1007/s00246-015-1266-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00246-015-1266-6