Abstract
To evaluate protective effects of Tutukon®, a plant derived herbal product, on the development of rat urolithiasis model. A total of 45 rats were divided into three groups namely; Group 1 (control group; drinking water + zinc disk), Group 2 (0.5 % ethylene glycol [EG] to drinking water + zinc disk) and Group 3 (study group—0.5 % EG + Tutukon + zinc disk). Moreover, zinc disks were placed into bladder of rats to serve as a nidus for stone development. Five rats from each group were killed at the end of the 1st, 2nd and 4th week. The level of bladder inflammation, the disk weights and the urine oxalate, calcium and pH values and were evaluated and compared. The inflammation scores of the pathological evaluation were not significantly different among three groups. At the end of the 28th day, weights of the zinc disks were significantly higher in Group 2 (394.4 ± 41.2) when compared to Group 1 (1517.5 ± 367.3) and Group 3 (386.2 ± 26.9) (p = 0.016). The disk weights increased gradually at 7th, 14th and 28th days in Group 1 (p = 0.018) and Group 2 (p = 0.009) while remained stable in Group 3 (p = 0.275). Urine calcium levels were not affected among three groups throughout the study period. At the end of the 28th day, while the urine oxalate levels of rats in Group 1 was lower than that of both Group 2 (p = 0.046) and Group 3 (p = 0.008); Group 2 and Group 3 had similar oxalate excretion levels (p = 0.701). However, the difference was not significant. Tutukon seems to decrease stone deposition on zinc disks implanted in the bladder of rats. The exact mechanism of this preventive effect is, however, not well understood.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Urinary stone disease, both as the first episode or recurrences, is a common public health problem with an increasing economical burden [1]. Moreover, the stone recurrences together with the repetitive treatments performed may results in functional deterioration [2]. Despite dramatic improvements in the endourological treatment modalities, advancements in the medical treatment and metaphylaxis of stone disease are limited. Potassium citrate, allopurinol, thiazide diuretics are currently available medications that are recommended in medical treatment and metaphylaxis of urinary stone disease. However, in most of the cases patients ignore long-term usage of these agents due to complications or inconsistent data on the duration of the treatment.
The evaluation of the metaphylactic or chemolytic activities of the newly developed drugs for urolithiasis are usually carried out in experimental models that cover all the processes involved in the urinary stone disease including nucleation, supersaturation and crystal growth [3–6]. Although in vitro models provide most of these steps, the effects of endogenous stone promoters and inhibitors cannot be simulated [7]. Therefore, animal models and mostly the rats are preferred to mimic the stone disease and to evaluate the effects of test drugs.
Herbal agents have long been evaluated for their long-term usage in both the treatment and prophylaxis of urinary stone disease [5, 7–14].
Calcium is the most common component of the urinary tract stones while it is seen as calcium oxalate stones in almost 60 % of the cases [15]. It has been shown that calcium and oxalate are equally important in the development of calcium oxalate stones [16]. Ethylene glycol (EG) has long been used to induce hyperoxaluria in rats [17] and is a perfect model for evaluating the metabolic effects of any given treatment. Implantation of calcium oxalate crystals, plastic disks, non-absorbable surgical stich pieces or zinc disks into the animal bladder to induce urolithiasis are the currently available stone models in animals [17]. Although zinc disk implantation model is the most widely used model and has been more extensively reported in the scientific literature, the resultant stone is a magnesium ammonium phosphate stone [18, 19]. However, if EG is added to the diet of rats, the final stone composition becomes calcium-oxalate [17].
Tutukon® (Laboratorio Miguel&Garriga, S.A. Barcelona, Spain) is an herbal agent known for its antioxidant, anti-apoptotic and nephron-protective agent [20]. The present study is designed to evaluate the effects of Tutukon on the development of bladder stones when added to the drinking water of the rats. To induce hyperoxaluria 0.75 % EG was added to the drinking water and zinc disks were implanted into their bladders to serve as a stone nidus.
Materials and methods
Herbal agent
Tutukon is a fixed dose plant based agent herbal agent compromised of essential fatty acids, flavonoid quercatin, polysaccharides, rosamarinic acid, boldin and flavonglicozides. It has been used as Resolutivo Regium for its antioxidant, anti-inflammatory, diuretic, spasmolytic, antibacterial and nephron-protective effects. The drug is available as hidrolate form in bottles of 600 ml. The recommended adult dose is 45 ml three times daily. Every 100 ml of the drug is composed aqueous distillate of the dried parts of 570 mg Enguisetumarvensis stem, 330 mg of whole plant of Spergularia rubra, 280 mg of Peumus boldus leaves, 170 mg of flowers of Opuntia ficusindica, 170 mg of flowers of Sideritisangustifolia, 170 mg of Rozmarinus officinales leaves, 170 mg of Cynodon dactylon rhizomes and 170 mg of Melissa officinalis leaves.
Study design
All procedures were performed after approval by the Institutional Animal Care and Usage Committee of the Bagcilar Laboratory Animal Center. A total of 45 female Wistar rats of 10–12 weeks old and weighing 250–300 g were included in this study. All rats were housed in polypropylene cages. Each cage contained five rats. Other conditions including 12 h of dark and light cycle, room temperature of 22 ± 2 °C and humidity of 40–60 % together with free access to standard food was maintained throughout the study.
The rats were divided into nine groups (Table 1). After implantation of zinc disk, treatment with EG and Tutukon was initiated on the second postoperative day. Ethylene glycol (0.75 %) was added to drinking water of rats in Group 2 and 0.75 % EG plus Tutukon 15 ml/day in Group 3.
At the end of the predefined follow-up period, rats were killed by cervical dislocation after a single intramuscular injection of 60 mg/kg ketamine hydrochloride (Ketalar, Eczacibasi Inc., Istanbul, Turkey) 10 mg/kg xylazine (Rompun, Bayer Turk Inc., Istanbul, Turkey). After killing, urine samples from the bladder were obtained for biochemical analysis and complete bladders were harvested for histopathological evaluation. The weights of the zinc-disks were also noted and compared.
Operative technique
After 6 h of fasting, all rats were anesthetized by a single intramuscular injection of 60 mg/kg ketamine and 10 mg/kg xylazine. When the reflexes to pinching of paws abated the abdominal wall of each rat was shaved with electric clippers and the skin was cleansed with 10 % povidine iodine (Poviodine, Dioagnokim Inc., Istanbul, Turkey). A 2 cm lower abdominal wall incision was made and the muscles were separated using blunt dissection. The urinary bladders were exposed and a small incision was performed to open the lumen of the bladder. A zinc-disk weighting 20 ± 2 mg were inserted into the bladder and the incisions were closed using 4/0 absorbable polyglactine (Vicryl, Ethicon Inc., Somerville, NJ, USA). After replacement of bladder into its original place, the muscles were approximated with 3/0 absorbable polyglactine and the skin was closed using 2/0 silk (Perma-hand Silk, Ethicon Inc., Somerville, NJ, Istanbul). All wounds were cleansed with 10 % povidine iodine at the end of the operation and the rats were left for recovery.
Histopathological evaluation
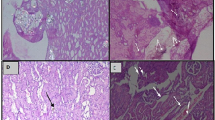
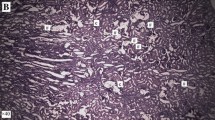
After killing of rats, the urinary bladders were exposed from a midline incision. Before the removal of the bladders, urine samples were obtained. The bladders were fixed within 10 % formalin solution for 24 h prior to paraffin embedding. The wax blocks were then incised for 4–6 μm pieces and stained with hematoxylin-eosin. A genitourinary pathologist experienced in animal models examined the slices under light-microscopy and recorded the level of edema (none, mild, moderate and severe), vascular congestion (none, mild, moderate and severe), level of inflammation (none, mild, moderate and severe), changes in transitional epithelium (fibrosis, calcification, mitosis, dysplasia), thickness of the epithelium (in millimeters) and epithelial cell layer (≤7 or >7).
Laboratory analysis
Regarding the assessment of urinary stone forming risk factors; while calcium levels were assessed using photometric arsenazo method (Olympus AU2700 analyzer—Beckman coulter Inc, CA, USA), citrate was assessed using HPLC (high performance liquid chromatography) method (Shimadzu SIL20AHT—Shimadzu Corporation, Kyoto, Japan) and lastly urinary pH was assessed using universal indicator strips (Merck KGaA, Darmstadt, Germany). The collected stones were analyzed using infrared spectroscopy (Spectrum Two IR, Perkin Elmer, Waltham, MA, USA).
Statistical analysis
Statistical analyses were performed using Number Cruncher Statistical System 2007 (NCSS, LLC, Kaysville, UT) statistical software package program. In addition to descriptive statistics (mean and standard deviation), comparison of groups was performed using Kruskal–Wallis test. Moreover, subgroup comparisons were performed using Dunn’s multiple comparisons test and quantitative comparisons using Chi-square and Fischer exact tests. Statistical significance was set as p < 0.05.
Results
Effects on stone growth
The weight of the zinc disks increased in rats under normal diet at the end of 7th and 14th days (p = 0.018) (Table 2). However, Dunn’s multiple comparison test revealed that the weights almost stabilized after 2 weeks (p = 0.016 and p = 0.754 for comparison of 7th and 14th days and 14th and 28th days, respectively) (Table 5). The results were also similar for rats fed with tap water and 0.75 % EG, the weights gradually increased early in the study and then stabilized (p = 0.014 and p = 0.149, for comparison of 7th and 14th days and 14th and 28th days, respectively). On the other hand, the weights did not increase significantly in rats fed with 0.75 % EG and Tutukon during the study period (p = 0.275, Table 2). Moreover, although at the end of first week the disks in Group 2 and 3 were heavier than disks in Group 1 (p = 0.032 and p = 0.014, respectively) this difference was not significant between Group 1 and Group 3 at days 14 and 28 (p = 0.251 and p = 0.530, respectively) (Table 3).
Effects on urine parameters
The urinary oxalate levels within Group 1 (p = 0.167), Group 2 (p = 0.120) and Group 3 (p = 0.754) were stable throughout the study duration (Table 4). At the end of first week, the mean urinary oxalate level of Group 1 was significantly lower than that of Group 2 (p = 0.015) and Group 3 (p = 0.48) while it was similar between Group 2 and Group 3 (p = 0.347). Similarly, although oxalate levels were lower in Group 1 when compared to Group 2 (p = 0.046) and Group 3 (p = 0.08) at 28th day, rats from Group 2 and Group 3 had similar urinary oxalate levels (p = 0.701).
Urinary calcium levels of rats throughout the study are provided in Table 4. The calcium levels of rats in Group 1 (0.418), Group 2 (0.789) and Group 3 (0.114) were not different at the 7th, 14th and 28th days of the study.
Urinary pH levels of rats were also recorded (Table 4). In Group 3, mean pH values were different at 7th, 14th and 28th day measurements (p = 0.039). pH levels at 7th day was lower than that of 14th (p = 0.016) and 28th days (p = 0.142).
Stone analysis was also performed. The stone crystals deposited around the zinc disks were composed of calcium oxalate in 38 and magnesium ammonium phosphate in six rats (two rats developed mixed stone crystals).
Effects on bladder inflammation
At the end of 7th day, the level of vascular congestion seen in rats of Group 1 was significantly milder than Group 2 and Group 3 (p = 0.003). Although the difference was not significant, the congestion was also milder in Group 3 than Group 2 (p = 0.206). Edema was more prominent in Group 1 at 14th when compared to Group 2 (p = 0.048) and Group 3 (p = 0.041). Otherwise, the level of edema was similar within each group and between different groups. Level of inflammation was not different among different groups at 7th, 14th and 28th days (p = 0.514, p = 0.341 and p = 0.592, respectively). Presence of changes in transitional epithelium (fibrosis, calcification, mitosis, dysplasia), thickness of the epithelium and number epithelial cell layer were also similar between groups.
Discussion
Prevention of stone recurrences is an important step in the treatment of a stone patient. However, contrary to minimally invasive treatment options, little improvements occurred in metaphylaxis of urolithiasis during the last 30 years. Currently available drugs are limited to potassium citrate, allopurinol, diuretics and thiopronine depending on the stone composition and urinary pH. The efficacy of these agents and the duration that should be used are doubtful. Herbal agents, collectively known as phytotherapy, are used as an alternative to primary medical treatment of various diseases in many countries.
As hyperoxaluria is the main risk factor for calcium oxalate stone formation, EG is widely used in experimental studies to develop calcium oxalate crystals by inducing hyperoxaluria [17]. Although widespread usage to induce hyperoxaluria, this model is being criticized because of its side effects including metabolic acidosis, cellular injury and necrosis in tubular epithelial cells [21, 22]. However, Tutukon, has been shown to limit the EG induced apoptosis and crystal deposition in renal tubular cells [20]. There are no general rules that predict the effects of urinary pH on the biological processes as any pH changes alter numerous variables in the urine. Cohen stated that urinary pH levels of normal rats range from 5.5 to 7.5 while this range may be extended to 5.0–9.0 with modifications in diet [23]. The pH levels measured in this study were between 6.5 and 8.4 with slightly increased levels reported in Group 1 and Group 3 (Table 4). Though not significant, this decrease may explain the development of smaller stones around the zinc disks in the current study.
Concentrations of calcium and oxalate in the urine play an important role in the formation of stone crystals. Drugs that decrease the urinary excretion of these stone promoting ions may prevent development and deposition of stone crystals. However, the administration of Tutukon in this study did not alter the urinary calcium and oxalate levels indicating that the inhibitory effect on stone development is independent from the urinary concentrations of these ions. Collecting urine samples using metabolic cages may further clarify this uncertainty. Although calcium and oxalate concentrations of urine play an important role in the formation of crystals, the supersaturated status of the urine with these ions suggests that factors other than calcium and oxalate also play a role in the pathogenesis of urolithiasis.
On the other hand, in addition to the use of these medical agents; recent data clearly demonstrated that as a promising and effective alternative, herbal medicine has gained an increasing importance in the prevention of underestimated toxic effects of certain medications causing morphologic as well as functional changes in different organ systems [24]. Phytotherapy on this aspect may be applied in a complementary fashion to ameliorate the toxic effects of these drugs. Evaluation of the available data in the literature so far did show that majority of these phytotherapeutic agents have diuretic, anti-inflammatory, antioxidant, vasodilator and spasmolytic effects [25–28]. The active ingredients responsible for such specific effects have been found to be essential oils, flavonoids, saponins, xanthine derivatives and glycosides [20]. Numerous herbal agents including rubus idaeus [12], phyllanthus niruri [25], herniaria hirsute [26], alisma orientalis [27] and costus spiralis roscoe [28] were evaluated for their antiurolithiatic activities in animal models. Most of these agents showed protective effects through unknown mechanisms. Ghalayini et al. demonstrated a relationship between urinary calcium and oxalate levels and malondialdehyde in animals with calcium oxalate stones [12]. Rubus idaeus (also known as European raspberry) seems to prevent hyperoxaluria induced renal injury and the development of calcium oxalate crystals by decreasing the generation of malondialdehyde. Phyllanthus niruri, a widespread tropical plant commonly found in coastal areas, seems to prevent growth of calcium crystals prevents crystal development without altering the urine citrate or magnesium levels. Herniara hirsute, a species of flowering plants, on the other hand, seems to act by dissolving pre-existing deposits [26]. Atmani et al. showed that the plant extract has an impressive prophylactic effect without significantly changing the urine output, pH or chemistry. The authors speculated that the plant extract inhibits the calcium oxalate monohydrate crystal binding to the renal epithelial cells in a concentration dependent manner [29]. Alisma orientalis, acts by suppressing every step of the stone development process including crystal formation, growth and aggregation of calcium oxalate crystals [27]. Moreover, the extract significantly decreases the renal calcium levels. All these studies point out that extracts of various herbs may be effectively used for prevention of stone growth and recurrences although we do not completely understand the exact mechanism in most of the cases. Tutukon, an herbal extract is derived from various parts of eight different plants. Of these ingredients, extracts of Opuntia ficus indica [7], Rozmarinus officinales [30] and Cynodon dactylon [31] have been evaluated for their effects on stone development. However, as a combination therapy, to our knowledge, this is the first study that demonstrates the beneficial effects of Tutukon on development of stones in rats with hyperoxaluria and implanted zinc disks. As in previous studies, our findings are insufficient to explain the mechanisms. We think that it is not possible to justify this effect solely depending on the slight increase in urine pH.
This study has several limitations. First of all, the study group constituted only from female rats. As a result the effects of Tutukon in male and female rats were not compared. It has been shown that in zinc disk model, the stones developed in female rats are somehow smaller than the stones developed in male rats [19]. However, despite the differences in size of the stone developed, the effects of herbal medicals evaluated in different genders are usually similar [28].Moreover, the stone models used in this study resulted in the formation of stones sufficient for statistical significance. Therefore, to not increase the number of animals used in the experiment, only female rats were used within the study. Also, the urine analysis was performed using the spot urine collected at the time of killing. Although the urine collected for a longer time using a metabolic cage yields better results, human studies demonstrated that despite a minor inaccuracy by relating parameters to creatinine, spot urine is sufficient in the metabolic evaluation [32]. Finally, the study protocol does not have a treatment arm (initiation of Tutukon after formation of stones). However, the study aims to evaluate the effects of Tutukon when used as metaphylaxis, not as chemolysis.
In conclusion, we successfully developed bladder stones in rats after implantation of zinc disks and hyperoxaluria with EG. Moreover, supplementation of drinking water with Tutukon successfully decreased the size of bladder stones. The mechanisms underlying this effect are still unknown, but are apparently unrelated to urinary excretion of calcium and oxalate. Further studies are necessary to demonstrate the beneficial effects of Tutukon on prevention of urolithiasis in humans.
References
Kirkali Z, Rasooly R, Star RA, Rodgers GP (2015) Urinary stone disease: progress, status, and needs. Urology 86(4):651–653. doi:10.1016/j.urology.2015.07.006
Sairam K, Scoffone CM, Alken P, Turna B, Sodha HS, Rioja J, Wolf JS Jr, de la Rosette JJ (2012) Percutaneous nephrolithotomy and chronic kidney disease: results from the CROES PCNL global study. J Urol 188(4):1195–1200. doi:10.1016/j.juro.2012.06.027
Hirayama H, Wang Z, Nishi K, Ogawa A, Ishimatu T, Ueda S, Kubo T, Nohara T (1993) Effect of Desmodium styracifolium-triterpenoid on calcium oxalate renal stones. Br J Urol 71(2):143–147
Huang P, Yang SW, Huang WH, Kong FZ, Lou YJ (2006) Effect of rongshi granule on renal stone formation and osteopontin expression in rat urolithiasis model. Zhongguo Zhong Yao Za Zhi 31(14):1188–1191
Mi J, Duan J, Zhang J, Lu J, Wang H, Wang Z (2012) Evaluation of antiurolithic effect and the possible mechanisms of Desmodium styracifolium and Pyrrosiae petiolosa in rats. Urol Res 40(2):151–161. doi:10.1007/s00240-011-0401-y
Naghii MR, Jafari M, Mofid M, Eskandari E, Hedayati M, Khalagie K (2015) The efficacy of antioxidant therapy against oxidative stress and androgen rise in ethylene glycol induced nephrolithiasis in Wistar rats. Hum Exp Toxicol 34(7):744–754. doi:10.1177/0960327114558889
Meiouet F, El Kabbaj S, Daudon M (2011) In vitro study of the litholytic effects of herbal extracts on cystine urinary calculi. Prog Urol 21(1):40–47. doi:10.1016/j.purol.2010.05.009
Nishihata M, Kohjimoto Y, Hara I (2013) Effect of kampo extracts on urinary stone formation: an experimental investigation. Int J Urol 20(10):1032–1036. doi:10.1111/iju.12098
Lin E, Ho L, Lin MS, Huang MH, Chen WC (2013) Wu-Ling-San formula prophylaxis against recurrent calcium oxalate nephrolithiasis—a prospective randomized controlled trial. Afr J Tradit Complement Altern Med 10(5):199–209
Xiang M, Zhang S, Lu J, Li L, Hou W, Xie M, Zeng Y (2011) Antilithic effects of extracts from Urtica dentata hand on calcium oxalate urinary stones in rats. J Huazhong Univ Sci Technolog Med Sci 31(5):673–677. doi:10.1007/s11596-011-0580-3
Nagata M, Takayama T, Mugiya S, Ohzono S (2011) Pharmacotherapy for preventing calcium containing stone formation. Clin Calcium 21(10):1530–1534 (doi:CliCa111015301534)
Ghalayini IF, Al-Ghazo MA, Harfeil MN (2011) Prophylaxis and therapeutic effects of raspberry (Rubus idaeus) on renal stone formation in Balb/c mice. Int Braz J Urol 37(2):259–266 (discussion 267)
Erickson SB, Vrtiska TJ, Lieske JC (2011) Effect of cystone(R) on urinary composition and stone formation over a one year period. Phytomedicine 18(10):863–867. doi:10.1016/j.phymed.2011.01.018
Miyaoka R, Monga M (2009) Use of traditional Chinese medicine in the management of urinary stone disease. Int Braz J Urol 35(4):396–405
Wilson DM (1989) Clinical and laboratory approaches for evaluation of nephrolithiasis. J Urol 141(3 Pt 2):770–774
Pak CY, Adams-Huet B, Poindexter JR, Pearle MS, Peterson RD, Moe OW (2004) Rapid communication: relative effect of urinary calcium and oxalate on saturation of calcium oxalate. Kidney Int 66(5):2032–2037. doi:10.1111/j.1523-1755.2004.00975.x
Khan SR, Shevock PN, Hackett RL (1989) Urinary enzymes and calcium oxalate urolithiasis. J Urol 142(3):846–849
Olson ME, Nickel JC, Costerton JW (1989) Infection-induced struvite urolithiasis in rats. Am J Pathol 135(3):581–583
Prasad KV, Bharathi K, Srinivasan KK (1994) Evaluation of Ammannia baccifera Linn. for antiurolithic activity in albino rats. Indian J Exp Biol 32(5):311–313
Sahin C, Sarikaya S, Basak K, Cetinel CA, Narter F, Eryildirim B, Saglam E, Sarica K (2015) Limitation of apoptotic changes and crystal deposition by Tutukon following hyperoxaluria-induced tubular cell injury in rat model. Urolithiasis 43(4):313–322. doi:10.1007/s00240-015-0777-1
Khan SR (1995) Calcium oxalate crystal interaction with renal tubular epithelium, mechanism of crystal adhesion and its impact on stone development. Urol Res 23(2):71–79
Lieske JC, Norris R, Swift H, Toback FG (1997) Adhesion, internalization and metabolism of calcium oxalate monohydrate crystals by renal epithelial cells. Kidney Int 52(5):1291–1301
Cohen SM (1995) Role of urinary physiology and chemistry in bladder carcinogenesis. Food Chem Toxicol 33(9):715–730
Mukherjee S, Mukherjee N, Saini P, Roy P, Babu SP (2015) Ginger extract ameliorates phosphamidon induced hepatotoxicity. Indian J Exp Biol 53(9):574–584
Freitas AM, Schor N, Boim MA (2002) The effect of Phyllanthus niruri on urinary inhibitors of calcium oxalate crystallization and other factors associated with renal stone formation. BJU Int 89(9):829–834
Atmani F, Slimani Y, Mimouni M, Hacht B (2003) Prophylaxis of calcium oxalate stones by Herniaria hirsuta on experimentally induced nephrolithiasis in rats. BJU Int 92(1):137–140
Tsai CH, Chen YC, Chen LD, Pan TC, Ho CY, Lai MT, Tsai FJ, Chen WC (2008) A traditional Chinese herbal antilithic formula, Wulingsan, effectively prevents the renal deposition of calcium oxalate crystal in ethylene glycol-fed rats. Urol Res 36(1):17–24. doi:10.1007/s00240-007-0122-4
Araujo Viel T, Diogo Domingos C, da Silva Monteiro AP, Riggio Lima-Landman MT, Lapa AJ, Souccar C (1999) Evaluation of the antiurolithiatic activity of the extract of Costus spiralis roscoe in rats. J Ethnopharmacol 66(2):193–198
Atmani F, Farell G, Lieske JC (2004) Extract from Herniaria hirsuta coats calcium oxalate monohydrate crystals and blocks their adhesion to renal epithelial cells. J Urol 172(4 Pt 1):1510–1514
Naber KG (2013) Efficacy and safety of the phytotherapeutic drug Canephron(R) N in prevention and treatment of urogenital and gestational disease: review of clinical experience in Eastern Europe and Central Asia. Res Rep Urol 5:39–46. doi:10.2147/RRU.S39288
Khajavi Rad A, Hadjzadeh MA, Rajaei Z, Mohammadian N, Valiollahi S, Sonei M (2011) The beneficial effect of cynodon dactylon fractions on ethylene glycol-induced kidney calculi in rats. Urol J 8(3):179–184
Strohmaier WL, Hoelz KJ, Bichler KH (1997) Spot urine samples for the metabolic evaluation of urolithiasis patients. Eur Urol 32(3):294–300
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
All procedures involving animals were in accordance with the ethical standards of the institution.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Yuruk, E., Tuken, M., Sahin, C. et al. The protective effects of an herbal agent tutukon on ethylene glycol and zinc disk induced urolithiasis model in a rat model. Urolithiasis 44, 501–507 (2016). https://doi.org/10.1007/s00240-016-0889-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00240-016-0889-2