Abstract
Introduction
Atypical teratoid/rhabdoid tumors (ATRT) are rare aggressive neoplasms of the CNS affecting predominantly very young children.
Methods
We retrospectively reviewed the imaging findings of 9 CT and 32 MR examinations of the brain and spine of 33 children.
Results
Of the 33 tumors, 11 were located in the infratentorial compartment, 16 in the supratentorial compartment, 5 in both cranial compartments, and 1 in the lower thoracic spinal cord. The mean age of the children with infratentorial or infra- and supratentorial tumors was significantly lower than the mean age of the children with purely supratentorial tumors. Heterogeneity on imaging, large size and high tumor stages are striking features reflecting the aggressive nature of this histopathological entity. Although not present in the majority of children, a distinct and unusual pattern of a wavy band-like enhancement surrounding a central hypointensity was present in 12 of 32 children (38%) in whom contrast medium was used.
Conclusion
To the best of our knowledge this is the largest number of imaging examinations of ATRTs so far reported. A rather unusual pattern of contrast enhancement may be typical of ATRTs.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Rhabdoid tumors of the kidney are a subgroup of renal tumors in children defined by distinct pathological features and associated with a bad prognosis. Extrarenal locations of rhabdoid tumors including the CNS have been described increasingly frequent since the first neuropathological definition of this entity in 1987 [1]. Because of a rhabdoid component within these tumors, the term atypical teratoid/rhabdoid tumor (ATRT) has been coined. Although formerly often misdiagnosed as primitive neuroectodermal tumors (PNET), a distinct immunohistochemical, histopathological and molecular biological pattern allows the differential diagnosis.
ATRTs are found in young children with a mean age of about 2 years. Their location has been reported more often to be infratentorial, although several studies have reported a more frequent supratentorial origin. Dissemination at the time of diagnosis is frequent and prognosis in spite of intensive treatment is usually unfavorable. Several reports on the results of imaging procedures have not defined a typical CT or MRI picture of these tumors which have mostly been reported to resemble medulloblastomas (infratentorial PNET) [2–7].
Patients and methods
We retrospectively reviewed all available preoperative imaging examinations (9 CT and 32 MR examinations) of 33 children referred to the National Trial Centers for Malignant Pediatric Brain Tumors at the University of Würzburg (Dr. S. Rutkowski) and for Radiooncology of Pediatric Brain Tumors at the University of Tübingen (Dr. E. Dannemann-Stern and Prof. Dr. R. Kortmann) in whom a central review of neuropathology by the German Reference Centre for Neuropathology at the University of Bonn (Prof. Dr. T. Pietsch) had confirmed an ATRT of the CNS.
The review consisted of the location, MR signal and CT density compared to gray matter, contrast behavior, and the presence of leptomeningeal dissemination. In one child only a preoperative CT scan was available. In 32 children MRI had been performed, but in one child only MRI without contrast medium could be reviewed. The MRI examinations were not performed according to a defined protocol, but included at least a T2- and T1-weighted sequence. Information on the dosage of contrast medium was not available. In 20 patients a spinal MRI scan could be reviewed. A diffusion-weighted sequence without an apparent diffusion coefficient (ADC) map had been performed in four children.
Results
The group consisted of 21 boys and 12 girls between 1 day and 14 years of age (median age 22 months). The median age of the girls (12 months) was considerably lower than that of the boys (23 months), but the difference was not statistically significant (P = 0.34, Mann-Whitney test). The clinical and imaging characteristics of the 33 children are summarized in Table 1.
Location
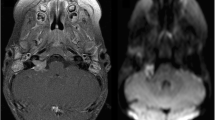
Of 15 tumors located in the supratentorial region, 12 were in the frontal lobe, 2 in the temporal lobe, and 1 in the parietal lobe. Of 11 tumors located in the infratentorial compartment, 6 were in the cerebellar hemispheres, and 4 were in the vermis. Of the tumors in the hemispheres and vermis, five showed extension to the brainstem or the pineal gland. One tumor was located purely within the pons. Six tumors were located in the supratentorial as well as in the infratentorial compartment, and one tumor was located in the lower thoracic spine (Fig. 1).
Dissemination
In five patients (15%) a visible meningeal dissemination was present at the time of diagnosis (Fig. 2). There was a statistically significant difference between patients with and those without meningeal dissemination with lower age associated with dissemination (median age with dissemination 134 days, median age without dissemination 800 days; P = 0.037). Survival time was not significantly different between these groups, but the median survival time was of course longer in the group without meningeal dissemination (331 days vs. 231 days). In one patient two different and isolated tumors with the same morphology in the suprasellar and trigeminal regions were seen without any sign of further meningeal dissemination (Fig. 3). In one patient the frontal tumor had invaded and destroyed the skull and extended into the galea.
Tumor size
The mean tumor volume, calculated according to the formula a×b×c/2, for all tumors was 66 cm3 (median 43 cm3). Infratentorial tumors were significantly smaller than supratentorial tumors (mean 82 vs. 35 cm3, median 62 vs. 32 cm3; P = 0.042). Tumors affecting both compartments were significantly larger than infratentorial tumors (mean 82 vs. 35 cm3, median 72 vs. 32 cm3; P = 0.026), but were not significantly different in size from supratentorial tumors.
CT density
All nine tumors with a CT scan available showed hyperdense attenuation values compared to gray matter. Three tumors showed fine calcifications and six were without calcifications.
MR signal
T2 signal intensity compared to gray matter was evaluable in 30 tumors. In two patients only FLAIR images had been acquired. In 17 tumors mixed intensities with hypointense values predominating were found. In the 13 predominantly homogeneous tumors hypo-, iso- and hyperintensity were evenly distributed (five were hypointense, four were isointense, and four were hyperintense). T1 signal compared to gray matter was evaluable in 27 tumors of which 12 were quite homogeneous mainly hypointense, and 15 were inhomogeneous mainly hypo- and hyperintense. Of the 33 tumors, 15 (45%) showed signs of bleeding mainly with a diffuse pattern. Only in four children could a circumscribed area of hemorrhage be identified (Fig. 4).
On diffusion-weighted images all four tumors showed an inhomogeneous pattern of restricted diffusion. ADC maps or values were not provided for review.
Edema
In 17 (52%) of the 33 children the tumor was surrounded by edema of varying degree.
Contrast enhancement
No enhancement was visible in 5 of 32 tumors (16%). Medium enhancement was seen in 10 (37%) of the remaining 27 tumors and strong enhancement in 17 (63%). Of the 27 tumors, 3 (11%) showed some enhancement in less than 50% of the tumor volume, while the majority (24) showed enhancement in more than 50% of the tumor volume.
Cysts or necrosis
Only eight tumors showed no cystic or necrotic parts. In 18 tumors possible necrotic areas with density or intensity values different from those of cerebrospinal fluid (CSF) were seen, and in 5 tumors possible cysts with density or intensity values comparable to those of CSF were seen. One patient showed cysts as well as necrosis. A peripheral position of a cystic part of the tumor separating the solid part from the normal brain was seen in 13 tumors (9 supratentorial and 4 infratentorial). All peripheral cysts showed rim enhancement (Fig. 5).
“Typical” enhancement pattern
In six patients an enhancement pattern consisting of a band-like rim of strong enhancement completely surrounding a central cystic or necrotic area (Figs. 2 and 6) was seen and another six patients showed at least in some part of the tumor a partially closed band-like enhancing rim (total 12 out of 32, 38%).
Discussion
An increasing number of reports on ATRTs have been published since the original neuropathological definition of this tumor entity in 1987 [1] and its further definition [8]. Imaging has always been described as nonspecific [3, 5, 6, 9, 10], so that only a usually large and frequently infratentorial tumor with signs of bleeding and the presence of meningeal dissemination already at the time of diagnosis in a patient of young age has been defined as suspicious for an ATRT.
Within a multicenter group we have been able to collect preoperative imaging examinations of 33 patients with ATRT. In accordance with other reports [2, 4, 11–13] among our patients boys also predominated (64%). There was a tendency for a lower age in the female patients (medians: all 22 months, boys 23 months, girls 12 months).
In our patients supratentorial tumors were more frequent than infratentorial tumors, in contrast to most reports of the results of imaging examinations [4, 5, 9, 11] but in accordance with the findings of the largest series evaluating treatment and outcome in this rare entity [12, 13]. It is not surprising that the supratentorial tumors and those affecting both compartments were significantly larger than those in the infratentorial area. As a sign of the well-known bad prognosis, 15% of our patients showed a meningeal dissemination of the tumor at the time of diagnosis and this was significantly correlated with a younger age. As also reported by Gandhi et al. [14], two separated tumors, one along the trigeminal nerve and the other in the suprasellar region, without signs of general dissemination were seen in one patient as a rare location [2, 15, 16], and also a single ATRT in the lower thoracic cord was seen in one patient. One frontal tumor had destroyed the skull and extended into the galea, a feature also reported by Arslanoglu et al. [9] and Evans et al. [10].
CT density values in the solid parts of the tumors prior to enhancement were uniformly hyperdense, probably representing a high cellular density. Signal intensity values on T1- and T2-weighted MR images varied widely, and obvious signs of mainly disseminated bleeding were found in a large proportion of tumors (45%). In four patients diffusion-weighted images were available for review, and all showed an inhomogeneously restricted diffusion.
A considerable number (16%) of tumors did not enhance after intravenous administration of contrast medium while most tumors showed enhancement of medium to strong intensity. In contrast to the typical appearance of infra- and supratentorial PNETs most of the tumors (52%) in our patient group were surrounded by some edema. Cysts or necroses were not present in only 25% of tumors. Cysts in a peripheral position as described by Arslanoglu et al. [9] between the solid part of the tumor and the normal brain were seen in 39% of our patients, with an even distribution between the infra- and supratentorial compartments. Although not as frequent as reported by Arslanoglu et al., this feature seems to be a regular finding in ATRTs, and we found peripheral cysts twice as frequently as Parmar et al. [5].
Most previous reports describe the imaging features of ATRT as nonspecific. Besides the typical features of frequent dissemination and bleeding in usually large tumors, in 38% of our patients we found a pattern of enhancement that seems to us quite unusual. This unusual pattern consisted of a band-like wavy rim of strong and quite uniform enhancement completely or only partly surrounding a central cystic or necrotic area. We also found this pattern in the images published in several recent reports on ATRT [4, 7, 9, 17]. However, this “typical” pattern does not seem to be specific, as we found it only in some of our patients, but at least if it is present it could help in the differential diagnosis of tumors of early childhood.
References
Lefkowitz IB, Rorke LB, Packer RJ (1987) Atypical teratoid tumor of infancy: definition of an entity (abstract). Ann Neurol 22:448–449
Bambakidis NC, Robinson S, Cohen M, Cohen AR (2002) Atypical teratoid/rhabdoid tumors of the central nervous system: clinical, radiographic and pathological features. Pediatr Neurosurg 37:64–70
Fenton LZ, Foreman NK (2003) Atypical teratoid/rhabdoid tumor of the central nervous system in children: an atypical series and review. Pediatr Radiol 33:554–558
Meyers SP, Khademian ZP, Biegel JA, Chuang SH, Korones DN, Zimmerman RA (2006) Primary intracranial atypical teratoid/rhabdoid tumors of infancy and childhood: MRI features and patient outcomes. AJNR Am J Neuroradiol 27:962–971
Parmar H, Hawkins C, Bouffet E, Rutka J, Shroff M (2006) Imaging findings in primary intracranial atypical teratoid/rhabdoid tumors. Pediatr Radiol 36:126–132
Reddy AT (2005) Atypical teratoid/rhabdoid tumors of the central nervous system. J Neurooncol 75:309–313
Zuccoli G, Izzi G, Bacchini E, Tondelli MT, Ferrozzi F, Bellomi M (1999) Central nervous system atypical teratoid/rhabdoid tumour of infancy. CT and MR findings. Clin Imaging 23:356–360
Rorke LB, Biegl JA (2000) Atypical teratoid/rhabdoid tumor. In: Kleihues P, Cavenee WK (eds) World Health Organization classification of tumors. Pathology and genetics of tumors of the nervous system. IARC Press, Lyon, pp 145–148
Arslanoglu A, Aygun N, Tekhtani D, Aronson L, Cohen K, Burger P, Yousem DM (2004) Imaging findings of CNS atypical teratoid/rhabdoid tumors. AJNR Am J Neuroradiol 25:476–480
Evans A, Ganatra R, Morris SJ (2001) Imaging features of primary malignant rhabdoid tumour of the brain. Pediatr Radiol 31:631–633
Cheng YC, Lirng JF, Chang FC, Guo WY, Teng MM, Chang CY, Wong TT, Ho DM (2005) Neuroradiological findings in atypical teratoid/rhabdoid tumor of the central nervous system. Acta Radiol 46:89–96
Hilden JM, Meerbaum S, Burger P, Finlay J, Janss A, Scheithauer BW, Walter AW, Rorke LB, Biegel JA (2004) Central nervous system atypical teratoid/rhabdoid tumor: results of therapy in children enrolled in a registry. J Clin Oncol 22:2877–2884
Tekautz TM, Fuller CE, Blaney S, Fouladi M, Bronciser A, Merchant TE, Krasin M, Dalton J, Hale G, Kun LE, Wallace D, Gilbertson RJ, Gajjar A (2005) Atypical teratoid/rhabdoid tumors (ATRT): improved survival in children 3 years of age and older with radiation therapy and high dose alkylator-based chemotherapy. J Clin Oncol 23:1491–1499
Gandhi CD, Krieger MD, McComb JG (2004) Atypical teratoid/rhabdoid tumor: an unusual presentation. Neuroradiology 46:834–837
Tamiya T, Nakashima H, Ono Y, Kawada S, Hamazaki S, Furuta T, Matsumoto K, Ohmoto T (2000) Spinal atypical teratoid/rhabdoid tumor in an infant. Pediatr Neurosurg 32:145–149
Tanizaki Y, Oka H, Utsuki S, Shimizu S, Suzuki S, Fujii K (2006) Atypical teratoid/rhabdoid tumor arising from the spinal cord – case report and review of the literature. Clin Neuropathol 25:81–85
Lee YK, Choi CG, Lee JH (2004) Atypical teratoid/rhabdoid tumor of the cerebellum: report of two infantile cases. AJNR Am J Neuroradiol 25:481–483
Conflict of interest statement
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was funded by the German Children’s Cancer Foundation (Deutsche Kinderkrebsstiftung).
Rights and permissions
About this article
Cite this article
Warmuth-Metz, M., Bison, B., Dannemann-Stern, E. et al. CT and MR imaging in atypical teratoid/rhabdoid tumors of the central nervous system. Neuroradiology 50, 447–452 (2008). https://doi.org/10.1007/s00234-008-0369-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-008-0369-7