Abstract
Objective: To determine factors affecting the population pharmacokinetics of oral cyclosporin (CsA) in cardiac allograft recipients during the first 3 weeks after surgery.
Methods: Data were obtained from routine trough monitoring and from two extra samples drawn during a dosing interval on a randomly selected day. Whole blood CsA concentrations were assayed using high-performance liquid chromatography (HPLC). Approximately equal numbers of patients were prescribed Sandimmun (SAN) or Neoral (NEO) CsA formulations. Parameter values of a one-compartment kinetic model with first-order absorption and elimination were sought together with the inter-patient and intra-patient variances using the NONMEM program.
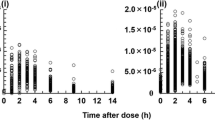
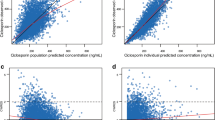
Results: Improved fits resulted from using the following expression in the model to adjust apparent bioavailability as a function of post-operative day (POD): f=0.2 + 10 × ABS (POD−5)/[(POD + 7) × 60]. The CsA clearance (CL/f ) was found to be influenced by current body weight (WT). There was an absorption lag time of about 35 min with SAN, but zero lag time with NEO. Oral bioavailability (f ) was increased by about 35% with concomitant diltiazem and about 18% with NEO. The CL/f was10% higher during the daytime than at night. The final pharmacokinetic model was validated using 200 bootstrap samples of the original data.
Conclusions: Using a validated population modelling approach, it was found that a number of factors influence the pharmacokinetics of CsA during the early post-operative period in cardiac transplant patients. These influences affecting oral bioavailability and clearance may need to be taken into account for maintaining appropriate concentrations of CsA in the bloodstream.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 29 June 1999 / Accepted in revised form: 28 April 2000
Rights and permissions
About this article
Cite this article
Parke, J., Charles, B. Factors affecting oral cyclosporin disposition after heart transplantation: bootstrap validation of a population pharmacokinetic model. E J Clin Pharmacol 56, 481–487 (2000). https://doi.org/10.1007/s002280000164
Issue Date:
DOI: https://doi.org/10.1007/s002280000164