Abstract
Using data from the Peking Vertebral Fracture Study, we conducted a longitudinal cohort study to investigate the association between type 2 diabetes mellitus (T2DM) and the risk of incident fractures, especially of vertebral fractures (VFs), and we also examined the modifying effect of body mass index (BMI) on this association and the effect of bone mineral density (BMD) T-score as a risk factor for incident fractures in T2DM. Chinese postmenopausal women were enrolled (n = 982), among whom 186 had T2DM. Incident VFs were confirmed by lateral radiographs of the thoracolumbar spine (T4–L5), while incident clinical non-VFs were self-reported. BMDs at the lumbar spine (LS) and femoral neck (FN) were measured by dual-energy X-ray absorptiometry. T2DM and non-DM women were at similar risk for VFs (OR 0.74, 95% CI 0.32–1.74), even adjusting for age, BMI, BMD, and previous fractures. Meanwhile, T2DM women had nearly twice the risk for non-VFs (HR 1.95, 95% CI 1.11–3.35) compared with non-DM women. After stratifying by BMI, the risk of VFs remained similar between diabetics and non-diabetics despite their BMI status (p for interaction = 0.470), and the risk of non-VFs was positively associated with T2DM only in women with BMI ≥ 25 kg/m2 (HR 3.59, 95% CI 1.68–7.65) (p for interaction = 0.065). Although LS BMD T-score was similarly and negatively associated with incident VFs both in T2DM (OR 0.34, 95% CI 0.12–0.88) and non-DM women (OR 0.60, 95% CI 0.44–0.82) (p for interaction = 0.430), the FN BMD T-score was not found to be significantly associated with either non-VFs or VFs among T2DM women. Comparing T2DM and non-DM women with similar fracture risks, the mean difference in LS T-score was − 0.36 (95% CI − 1.77 to 1.04) for VF, and difference in FN T-score was 1.61 (95% CI − 0.11 to 3.34) for non-VF. In conclusion, Chinese postmenopausal women with T2DM had a similar risk of incident VFs, but a significantly higher risk of incident non-VF, compared to women without DM. Higher BMI did not modify the effect of T2DM on risk of VFs, but it increased the association between T2DM and risk of non-VFs. LS BMD T-score was similarly and negatively associated with VF risk in T2DM and non-DM women and appear to be useful for clinical evaluation of VF risk.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Numerous epidemiological studies have confirmed that type 2 diabetes mellitus (T2DM) is associated with an increased risk of fractures [1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17]. Nevertheless, studies conducted among the Chinese population are rare [6,7,8]. Chinese people with T2DM have different clinical features from Caucasians, with most having normal weight, a lower prevalence of abdominal obesity, and lower insulin secretion and sensitivity [18,19,20]. Therefore, it is possible that the association between T2DM and fractures in the Chinese population is different from that in the Caucasian population. However, the few previous studies performed using Chinese subjects are limited by several factors, including reporting only a single type of fracture [8], cross-sectional study design [6], ignoring important confounders, such as bone mineral density (BMD) and history of previous fractures [7, 8], and self-reporting of fractures unconfirmed by radiographs [7, 8]. In addition, no longitudinal study has been conducted for diabetic subjects in mainland China until now.
There is also less information available on vertebral fractures (VF) in subjects with T2DM. This is most likely due to the difficulty in identifying VFs, most of which are clinically silent [21, 22]. Moreover, studies investigating the risk of incident VFs in individuals with T2DM have reached controversial results, with risk being increased in studies using clinical VFs as the outcome [5, 9, 11], but not different from non-diabetic counterparts in studies based on morphometric VFs [12, 13, 23, 24].
Previous studies have shown that T2DM is usually associated with a 5–10% higher areal BMD than in healthy subjects [6, 25,26,27,28,29,30]. Thus, it is difficult for clinicians to identify and treat diabetic patients at high risk of fractures as BMD may underestimate fracture risks in these patients. For this reason, other factors are needed to predict fracture risk in patients with T2DM. Suppressed bone turnover is clearly associated with T2DM [6, 25, 31, 32]. However, cross-sectional studies examining the role of bone turnover markers (BTMs) in predicting fracture risk have been conducted with different outcomes [25, 32, 33]. Therefore, a longitudinal study is needed to clarify whether low BTMs contributes to the increased risk of incident fractures in patients with T2DM.
High body mass index (BMI) or obesity was previously considered as a protective factor for factures, especially for hip fractures [9, 17, 34]. However, higher BMI is normally associated with more severe T2DM, which is speculated to be related to a higher risk of fractures. New emerging cohort studies have shown that the association between T2DM and increased risk of fractures remains unchanged after adjusting for BMI [9, 13, 16, 24], and Holmberg et al. [9] reported that the effect of BMI on diabetes-associated facture risk differs according to the site.
To clarify these issues, we used data from the Peking Vertebral Fracture Study (PK-VF), a large-scale community-based epidemiologic study designed to determine the prevalence and incidence of morphometric vertebral fractures among Chinese postmenopausal women in Beijing, China, from January 2008 through June 2014. We conducted this longitudinal cohort study mainly to examine whether the risk of incident fractures, especially of morphometric VFs, was higher among Chinese postmenopausal women with T2DM than among women without diabetes. We also sought to determine whether BMI has a modifying effect on the association between T2DM and incident fractures. Finally, we investigated whether BMD T-score or BTM were significantly associated with incident fractures in T2DM women.
Subjects and Methods
Subjects
The PK-VF has already conducted two surveys from January 2008 through January 2009, and from March 2013 through June 2014. The cohort of the first survey (baseline study) was constituted by randomly sampling postmenopausal women from seven districts throughout Beijing, namely Dong Cheng, Chao Yang, Xi Cheng, Hai Dian, Feng Tai, Shi Jing Shan, and Fang Shan. The Coordinating Center was located at the Peking Union Medical College Hospital (PUMCH). The populations of the seven districts in our study ranged from 350,000 to 1,920,000. One or two communities in each district were randomly selected and represented at least 5% of the total population of the district. A community in our study was defined as a group of interacting people characterized by similar culture and lifestyle, living at a specific geographic area, and sharing common spaces. Postmenopausal women were stratified by age and randomly sampled according to the age composition of those communities. Ahead of sampling, written notices explaining the nature of the proposed study were posted on the bulletin board of each community center. Then telephone calls were made to invite the randomly selected participants to join the study. In the end, a total of 2070 postmenopausal women aged 47–108 years without any known metabolic bone diseases were enrolled (Fig. 1). Every participant completed a standardized questionnaire concerning items such as demographic information, years since menopause (YSM), clinical fracture history before the baseline, medical history including diabetes, and usage of different drugs including anti-diabetic medication. Weight and height for each participant were measured by standard methods, and BMI was calculated according to the standard formula: BMI = weight/height2. Women with BMI ≥ 24 kg/m2 were defined as overweight or obese [35]. Measurements of serum bone turnover markers (BTMs) and BMDs, as well as assessment of morphometric VFs were also performed for each participant by certified staff. The full details of the baseline study were previously published [6, 36, 37]. During the second survey (follow-up study), we attempted to call and persuade every woman to participate in a second visit. However, 493 (23.82%) women refused to participate, 418 (20.19%) women had relocated out of Beijing or never responded to a phone call, and another 59 (2.85%) women had died. At last, 1100 (53.14%) postmenopausal women aged 50 to 93 years were successfully recruited in the follow-up study, with a median duration of 5.2 ± 1.0 years in follow-up (Fig. 1). The same aforementioned questionnaire and measurements were performed for each participant again, and incident clinical fractures having occurred during the follow-up period were additionally recorded. In this study, because we mainly aimed to investigate how T2DM affects the risk of incident fractures among women with natural menopause, we excluded women who met one of the following criteria (Fig. 1): 1) menstruation stopped after a hysterectomy; 2) diagnosis of diabetes before 40 years old indicating possible type 1 diabetes; 3) the presence of diseases that likely affect bone metabolism, including chronic liver disease, chronic renal disease, rheumatoid arthritis, or other connective tissue disease and significant gastrointestinal disease; and 4) taking drugs that affect bone metabolism, including corticosteroid, anticonvulsant drugs, bisphosphonates, calcitonin, activated vitamin D, or estrogen and progesterone. Eventually, a total of 982 subjects were included. This study was approved by the Department of Scientific Research, and the ethics committee of PUMCH. All of the subjects agreed to participate in this study and signed informed consent forms.
Biochemical Measurements
In the baseline study, a fasting blood sample was collected from each woman in the morning (7–9 am). After blood sampling, the serum was separated and cryopreserved at − 80 °C. All of the biochemical measurements were conducted within 3–6 months after sampling. The concentration of fasting plasma glucose (FPG) was assessed by standard methods in the central laboratory of PUMCH. Serum levels of BTMs, including C-terminal telopeptide of type I collagen (β-CTX) and N-aminoterminal prepeptide of type I procollagen (P1NP), were measured by an automated Roche electrochemiluminescence system (E170; Roche Diagnostics, Basel, Switzerland). The measuring ranges of β-CTX and P1NP were 0.01–6 ng/ml and 5–1200 ng/ml, respectively. The intra-assay and inter-assay coefficients of variation were 2.0% and 3.2% for β-CTX, 2.3% and 1.7% for P1NP.
Ascertainment of Type 2 Diabetes Mellitus
Baseline diabetes status was determined by self-reported diagnosis of diabetes or FPG ≥ 7.0 mmol/L. Self-reported diagnosis of diabetes was confirmed on the basis of an affirmative answer to the questions asked at baseline: “Were you ever diagnosed with diabetes by a doctor when you were not pregnant, and how old were you when you were diagnosed with diabetes? Or did you ever use an anti-diabetic medication such as insulin, metformin, acarbose, sulfonylureas, thiazolidinedione and the like?” In the end, 186 (18.9%) women with probable type 2 diabetes mellitus were included in our longitudinal study, and 161 (16.4%) women were diagnosed based on self-reporting, while 25 (2.5%) women were diagnosed by high FPG only. Based on the diabetes status, subjects were classified into a T2DM group and non-DM group (Fig. 1 and Table 1). Hemoglobin A1c (HbA1c) was not measured in PK-VF.
BMD Measurements and Ascertainment of Osteoporosis
In the baseline study, 954 (97.1%) women took BMD measurements at the lumbar spine (LS, including L1 to L4), femoral neck (FN), total hip and greater trochanter, using dual-energy X-ray absorptiometry (DXA) with either Lunar DPX or Norland equipment, from which T-scores were generated. The manufacturer’s phantom was calibrated every day before the measurement started. The coefficients of variation were 0.75–1.7% for LS and 0.56–1.0% for FN. Cross-calibration equations between the two kinds of machines are listed below [38].
According to the World Health Organization definition, osteoporosis was defined as T-score ≤ − 2.5. A woman was defined with osteoporosis if her T-score of at least one bone site was not over − 2.5. Accordingly, a woman with FN T-score ≤ − 2.5 or LS T-score ≤ − 2.5 was defined with FN osteoporosis or LS osteoporosis, respectively.
Ascertainment of Fractures
Morphometric VFs were assessed by the lateral radiographs of the thoracolumbar spine (T4–L5) in 886 (90.2%) women at first visit and in 868 (88.4%) women at both visits. Using Genant’s semiquantitative (SQ) visual criteria [39], two experienced radiologists independently evaluated the radiographs to diagnose VF. Previous VF with SQ ≥ 1 was confirmed by baseline X-ray. Incident VF was defined as an increase of at least 1 SQ score from baseline.
Self-reports of previous fractures before the first visit and self-reports of incident fractures from 2008 through 2014 were obtained using the aforementioned questionnaire. Fractures at the spine, rib, proximal humerus, distal forearm, hand, pelvis, hip, distal femur, tibia, fibula, and foot were recorded. The specific time and cause of each fracture were inquired. Only fractures occurring during routine activities or due to mild trauma were analyzed in our study. If a woman had multiple incident fractures, only the first one was used for our analysis. If a woman had a self-reported VF that was not verified by lateral radiographs of spine, we did not consider that she really had a clinically diagnosed VF. As a consequence, 23 women with self-reported previous VFs and 12 women with self-reported incident VFs were identified; meanwhile, 214 women with previous morphometric VFs and 43 women with incident morphometric VFs were identified. Only 11 (47.8%) self-reported previous VFs and 5 (41.7%) self-reported incident VFs were confirmed by X-rays. In summary, previous and incident clinical fractures were defined as self-reported fractures occurring during routine activities or due to mild trauma before the first visit and during the 2008–2014 period, respectively, with vertebral fractures confirmed by the lateral radiographs of the spine. A woman was then defined with any incident fracture if she had an incident VF or an incident clinical non-VF. A similar definition was also applied to any previous fracture.
Statistical Analysis
All of the statistical analyses were conducted using SPSS for Windows version 20.0 (SPSS Inc., Chicago, IL) or Stata (version 14.2; Stata Corporation, Inc., College Station, TX, USA). Data were presented as frequencies (percentages), mean (standard deviation [SD]), median (interquartile ranges [IQR]), correlation coefficient (r), hazard ratio (HR), odds ratio (OR), and 95% confidence interval (95% CI). Kolmogorov–Smirnov tests were used to verify the normal or skewed distributions of baseline continuous variables. Student’s t test was conducted to compare normally distributed continuous variables between the T2DM group and non-DM group, while Mann-Whitney U tests were conducted to compare non-parametric continuous variables between groups. Comparisons of categorical variables were performed by Pearson’s χ2 test. General linear model analysis of variance (GLM-ANOVA) was performed to further compare the BTMs and BMDs between groups, adjusting for age, YSM, and BMI. Because BTMs and FN BMD were normally distributed only after logarithmic transformation with base 10, transformed values were used in GLM-ANOVA. Logistic regression analyses were performed to compare the odds ratio for any incident fractures and incident VFs between groups, while Cox proportional hazards regression analyses were used to compare the hazard ratio for incident clinical non-VFs between groups, with or without adjustment for age, YSM, BMI, BMDs, and any previous fractures. All the ratios were further calculated among population stratified by BMI status (BMI ≥ 24 kg/m2 and BMI < 24 kg/m2) with adjustment for age, YSM, BMI, FN BMD, and any previous fractures. The association was tested for interactions between T2DM and BMI status. The effects of baseline BMD T-scores and BTMs on the fracture incidence in women with and without T2DM were examined by logistic regression analysis for incident VFs and by Cox proportional hazards regression analysis for incident clinical non-VFs. Those associations were tested for interactions between BMD T-scores and diabetes status, and between BTMs and diabetes status. To calculate the average difference in T-score between T2DM and non-DM women with the same risk of incident fractures, we used logistic models to estimate the associations of LS T-score and T2DM with the outcomes of incident VFs, and used Cox proportional hazards models to estimate the associations of FN T-score and T2DM with the outcomes of incident non-VFs, with adjustment for age, BMI, YSM and any previous fractures. The difference in T-score for those with and without T2DM but with the same incident fracture risk was calculated as the ratio of the regression coefficient for T2DM to the coefficient for T-score as previously published [40], with a 95% CI obtained by the delta method. p values of less than 0.05 were considered to be significant.
Results
Difference in the Risk of Incident Fractures Between T2DM and Non-DM Women
Baseline characteristics of all of the participants according to diabetes status are summarized in Table 1. 43 (5.0%) incident VFs and 59 (6.0%) incident clinical non-VFs were reported during the follow-up, with 8 (0.9%) VFs and 18 (1.8%) clinical non-VFs associated with type 2 diabetic women (Table 1). Type 2 diabetic women had mildly lower risk for VFs than women without diabetes (OR 0.95, 95% CI 0.43–2.09). However, this difference did not reach statistical significance in any analyzed model (Table 3), and 95% CIs for these estimates were wide and the power to detect modest associations was limited. The incidence and hazard ratio for clinical non-VFs among women with T2DM were nearly twice those among women without diabetes (incidence: 9.7% vs. 5.2%; HR 1.95, 95% CI 1.11–3.35). This hazard ratio became even higher after adjusting for age, YSM, BMI, BMD, and previous fractures (Table 2). When fractures by anatomic site were compared (data not shown), only the difference in incident foot fractures remained statistically significant (HR 3.56, 95% CI 1.24–10.22), but the limited number of fractures at other sites did not allow us to make a definite conclusion.
The Modifying Effect of BMI on the Association Between T2DM and Incident Fractures
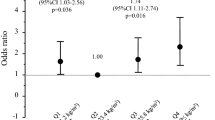
When stratifying the population by BMI status, we performed the same regression models in individuals with BMI ≥ 24 kg/m2 and in individuals with BMI < 24 kg/m2 (Table 3). The risk of VFs remained similar between diabetics and non-diabetics despite their BMI status. However, the increased risk of non-VFs was only associated with T2DM in women BMI ≥ 24 kg/m2 (HR 2.80, 95% CI 1.41–5.57, p for interaction = 0.002).
The Association Between BMD T-Scores and Incident Fractures in Either T2DM Women or Non-DM Women
Higher BMD T-scores at the lumbar spine were similarly and negatively associated with incident VFs both in women with T2DM (OR 0.34, 95% CI 0.12–0.88) and in women without diabetes (OR 0.60, 95%CI 0.44–0.82, p for interaction = 0.430). However, no significant associations were found between LS T-scores and incident non-VFs in either T2DM women or non-DM women (Table 4). Therefore, the LS T-score predicts VF risks similarly in T2DM and non-DM, but does not predict non-VF in either group. Although BMD T-scores at the femoral neck were found to be negatively associated with incident non-VFs (HR 0.55, 95% CI 0.37–0.81) and incident VFs (OR 0.54, 95% CI 0.34–0.84) only in non-DM women, no evidence of interaction by diabetes status indicated that the FN T-score predicts fracture risks similarly in T2DM as in non-DM (Table 4). Neither BTM was found to be significantly associated with incident fractures (Table 4).
Considering the challenge of interpreting BMD T-scores in type 2 diabetic patients, we analyzed mean differences in T-scores, comparing women with and without diabetes at a similar fracture risk. The difference in LS T-score comparing T2DM and non-DM women with similar VF risk was − 0.36 (95% CI − 1.77 to 1.04). The difference in FN T-score comparing T2DM and non-DM women with similar non-VF risk was 1.61 (95% CI − 0.11 to 3.34). It seemed that LS T-score would slightly overestimate the risk of VFs in T2DM, while the FN T-score would possibly underestimate the risk of non-VFs in T2DM. However, none of the results were statistically significant.
Discussion
In this longitudinal cohort study based on data from PK-VF, we first demonstrated that the odds ratio for osteoporotic fracture in patients with type 2 diabetes is about 1.5, which is same as reported by Ferrari et al. [29] Nevertheless, no significant association existed between incident VFs and T2DM, even after BMDs and previous fracture history were taken into account. This finding is consistent with some studies [9, 13, 23, 24] while inconsistent with others [5, 9, 11, 12]. Most studies reporting an increased risk of incident VFs used clinical VFs as the outcome, not morphometric VFs [5, 11]. Our study and the MrOS study [21] respectively showed that only 11.6% and 13.5% of morphometric VFs could be recognized as clinical VFs, most of which had a SQ score change ≥ 2, indicating more severe VFs. Other studies [12, 13, 23, 24] based on spine radiographs, including our previous study [6], mainly investigated prevalent VFs and yielded conflicting results. In agreement with our data, the CaMos study also found an insignificantly lower prevalence of VFs in men with T2DM (OR 0.77, 95% CI 0.49–1.22) [23]. However, slightly higher risks of incident VFs were detected in the MrOS study (OR 1.28, 95% CI 0.81–2.00) [24] and SOF study (OR 1.06–1.09, 95% CI 0.33–3.55) [13], but none of those differences were statistically significant. Meanwhile, the risk of incident non-VFs was substantially increased in women with T2DM compared to women without diabetes. This difference was mainly caused by foot fractures in our study. These results are compatible with some previous research also reporting an increased risk of non-VFs in patients with T2DM [4, 5, 7, 9, 13, 14], and among these studies, those of Luetters et al. [4] and Schwartz [13] respectively described positive associations between T2DM and prevalent foot fractures (OR 1.45, 95% CI 1.07–1.83) and incident foot fractures (HR 2.68, 95% CI 1.18–6.06).
Taken together, our results suggest that the risk of incident fractures associated with T2DM differed according to fracture site. The difference in the cortical to trabecular bone ratio between vertebral and non-vertebral bones may be a reasonable explanation for this discrepancy [41]. Increased cortical porosity in populations with T2DM has been proven to be related to elevated fracture risk at the hip and other non-vertebral sites [30, 42,43,44] that have a higher proportion of cortical bone. Another possible explanation for the increased incidence of non-VFs, especially of foot fractures, is that diabetic individuals have an elevated risk of falling, which is probably due to some disease-related chronic complications (peripheral neuropathy and retinopathy) [30, 45]. Previous studies have described an association of peripheral neuropathy with metatarsal fracture [46] and calcaneal fracture [47].
High BMI is a known protective factor for fractures, most evidence for which comes from studies of hip fractures [9, 17, 34]. However, our study found that the modifying effect of BMI on diabetes-related fractures also differed according to the site. Compared to non-diabetic women, women with T2DM always had a similar risk of incident VFs despite the different BMI status, but had a significantly higher risk of incident non-VFs only in individuals with BMI ≥ 24 kg/m2. These results suggest that higher BMI increased the association between T2DM and risk of non-VFs, but did not modify the effect of T2DM on the risk of VFs. In agreement with our findings, Holmberg et al. [9] reported that high BMI increases the risk of ankle fractures; while decreasing the risk of hip fractures and maintaining the risk of vertebral fractures. One hypothesis for this discrepancy is that different risk factors predominate in different types of fractures, such as poor bone quality in hip fractures and a tendency to fall in foot fractures [9].
Finally, we characterized the relationship between BTMs, BMDs, and incident fractures in T2DM and non-DM women. Unlike previous studies [25, 31,32,33], we did not find any significant association between BTMs and fractures either in T2DM women or in non-DM women, indicating that BTMs might not be reliable predictors for incident fractures. In line with the results for VFs in T2DM men reported by Napoli et al. [24] our results also described a similar and negative association between LS T-score and incident VFs both in T2DM women and non-DM women, suggesting that a lower LS T-score is a possible risk factor for VFs in T2DM women. Similar with the results of Schwartz et al. [40] the lack of interaction between FN T-score and diabetes status indicates that the FN T-score predicts the risks of VF and non-VF in those with T2DM just as it does in those without diabetes. There might be a difference in the association between FN T-score and fracture risk by diabetes status, but only a larger study would be able to determine if there is a real difference or if this is just a chance finding. In order to address how well the T-score predicts incident fractures in T2DM and non-DM women, we estimated the reduction in T-score equivalent to having T2DM. For incident non-VFs, we found an insignificant trend suggesting that having T2DM equates to having a FN T-score 1.61 units lower as compared with not having DM, indicating that the FN T-score would possibly underestimate the incident non-VF risk, although the degree of underestimation is difficult to determine with the small number of fractures in T2DM. Although the point estimate (1.61) reported by our study appears quite different from the value reported by Schwartz et al. (0.59) [40], there is no good evidence that they are really different, given the wide 95% CI (− 0.11 to 3.34), which also includes the point estimate for the study by Schwartz et al. Similarly, for LS T-score and incident VFs, comparing these results with those of Napoli et al. [24] which reported a difference in LS T-score of 0.64 (95% CI − 0.43 to 1.71) with a similar VF risk, the point estimates are in opposite directions, the 95% CI values are wide for both studies, and the point estimate for our study is within the 95% CI for that of Napoli et al. Thus, it is not clear whether LS T-score would under- or over-estimates VF risk in T2DM.
To our knowledge, this is the first and largest longitudinal cohort study investigating the risk of incident fractures associated with T2DM in a mainland Chinese population. We analyzed a well-characterized cohort of postmenopausal women with a long follow-up, for whom both self-reported fractures and morphometric VFs were available. We also examined the modifying effect of BMI on the diabetes-fracture association. For the first time in a Chinese population, we analyzed the value of BMD T-scores in predicting incident fracture risk and calculated the point estimate and 95% CI for their relationship.
However, our study has some inevitable limitations. The sample size was not large enough and the incidence of VFs or hip fractures in the T2DM group was relatively low as aforementioned, both of which limited the power to make definitive conclusions. Selection bias existed in our study due to the high ratio of loss to follow-up (46, 86%) and relatively low response rate (53.14%). Study participants were volunteers, community dwelling, and ambulatory, and therefore women with or without diabetes in PK-VF may be healthier than the general population, which limited the application of our results to the broader population of older women and women with T2DM. Since the original study was not designed to investigate diabetes, essential data needed to diagnose diabetes or assess diabetic severity were lacking, such as HbA1c, advanced glycation end-products (AGEs), and diabetic complications, and information on the specific anti-diabetic medications used by each participant was incomplete. Thus, it is possible that some undiagnosed diabetics existed in our study, but the adoption of FPG to ascertain diabetes almost eliminated this possibility. Similarly, since we did not confirm non-VFs by radiologic methods, there may be under- or over-reporting of non-VFs. Therefore, a large-scale prospective study is required in the future, with a higher incidence of radiologically confirmed fractures in diabetics, and considering falls, diabetic complications, and anti-diabetic drugs as confounders, and while measuring the levels of HbAlc and AGEs.
In conclusion, in this analysis of PK-VF, not only the association between T2DM and incident fractures, but also the modifying effect of BMI on this association differed according to the fracture site. Type 2 diabetic women had a similar risk of VFs, but a significantly higher risk of incident non-VF, compared to non-diabetic women, even after adjustment for age, BMI, BMD, and previous fractures. Higher BMI increased the association between diabetes and the risk of non-VFs, but it did not modify the effect of diabetes on risk of VFs. LS BMD T-score was similarly and negatively associated with VF risk in T2DM and non-DM women, and appears to be useful for clinical evaluation of VF risk. However, FN BMD T-score seemed to underestimate the non-VF risk in T2DM women despite the increased non-VF risk in this population.
References
Meyer HE, Tverdal A, Falch JA (1993) Risk factors for hip fracture in middle-aged Norwegian women and men. Am J Epidemiol 137(11):1203–1211
Forsén L, Meyer HE, Midthjell K, Edna TH (1999) Diabetes mellitus and the incidence of hip fracture: results from the Nord-Trøndelag Health Survey. Diabetologia 42(8):920–925
Ivers RQ, Cumming RG, Mitchell P, Peduto AJ (2001) Diabetes and risk of fracture: The Blue Mountains Eye Study. Diabetes Care 24(7):1198–1203
Luetters CM, Keegan TH, Sidney S et al (2004) Risk factors for foot fracture among individuals aged 45 years and older. Osteoporos Int 15(12):957–963
Bonds DE, Larson JC, Schwartz AV et al (2006) Risk of fracture in women with type 2 diabetes: the Women’s Health Initiative Observational Study. J Clin Endocrinol Metab 91(9):3404–3410
Jiajue R, Jiang Y, Wang O et al (2014) Suppressed bone turnover was associated with increased osteoporotic fracture risks in non-obese postmenopausal Chinese women with type 2 diabetes mellitus. Osteoporos Int 25(8):1999–2005
Liao CC, Lin CS, Shih CC et al (2014) Increased risk of fracture and postfracture adverse events in patients with diabetes: two nationwide population-based retrospective cohort studies. Diabetes Care 37(8):2246–2252
Koh WP, Wang R, Ang LW, Heng D, Yuan JM, Yu MC (2010) Diabetes and risk of hip fracture in the Singapore Chinese Health Study. Diabetes Care 33(8):1766–1770
Holmberg AH, Johnell O, Nilsson PM, Nilsson J, Berglund G, Akesson K (2006) Risk factors for fragility fracture in middle age. A prospective population-based study of 33,000 men and women. Osteoporos Int 17(7):1065–1077
Janghorbani M, Feskanich D, Willett WC, Hu F (2006) Prospective study of diabetes and risk of hip fracture: the Nurses’ Health Study. Diabetes Care 29(7):1573–1578
Kanazawa I, Yamaguchi T, Yamamoto M, Yamauchi M, Yano S, Sugimoto T (2008) Combination of obesity with hyperglycemia is a risk factor for the presence of vertebral fractures in type 2 diabetic men. Calcif Tissue Int 83(5):324–331
Yamamoto M, Yamaguchi T, Yamauchi M, Kaji H, Sugimoto T (2009) Diabetic patients have an increased risk of vertebral fractures independent of BMD or diabetic complications. J Bone Miner Res 24(4):702–709
Schwartz AV, Sellmeyer DE, Ensrud KE et al (2001) Older women with diabetes have an increased risk of fracture: a prospective study. J Clin Endocrinol Metab 86(1):32–38
Napoli N, Strotmeyer ES, Ensrud KE et al (2014) Fracture risk in diabetic elderly men: the MrOS study. Diabetologia 57(10):2057–2065
Tseng VL, Yu F, Lum F, Coleman AL (2012) Risk of fractures following cataract surgery in Medicare beneficiaries. JAMA 308(5):493–501
Holm JP, Jensen T, Hyldstrup L, Jensen JB (2018) Fracture risk in women with type II diabetes. Results from a historical cohort with fracture follow-up. Endocrine 60(1):151–158
Vestergaard P (2007) Discrepancies in bone mineral density and fracture risk in patients with type 1 and type 2 diabetes–a meta-analysis. Osteoporos Int 18(4):427–444
Ford ES, Giles WH, Dietz WH (2002) Prevalence of the metabolic syndrome among US adults: findings from the third National Health and Nutrition Examination Survey. JAMA 287(3):356–359
Torréns JI, Skurnick J, Davidow AL et al (2004) Ethnic differences in insulin sensitivity and beta-cell function in premenopausal or early perimenopausal women without diabetes: the Study of Women’s Health Across the Nation (SWAN). Diabetes Care 27(2):354–361
Gu D, Reynolds K, Wu X et al (2005) Prevalence of the metabolic syndrome and overweight among adults in China. Lancet 365(9468):1398–1405
Ensrud KE, Blackwell TL, Fink HA et al (2016) What proportion of incident radiographic vertebral fractures in older men is clinically diagnosed and vice versa: a prospective study. J Bone Miner Res 31(8):1500–1503
Cooper C, Atkinson EJ, O’Fallon WM, Melton LJ (1992) Incidence of clinically diagnosed vertebral fractures: a population-based study in Rochester, Minnesota, 1985-1989. J Bone Miner Res 7(2):221–227
Hanley DA, Brown JP, Tenenhouse A et al (2003) Associations among disease conditions, bone mineral density, and prevalent vertebral deformities in men and women 50 years of age and older: cross-sectional results from the Canadian Multicentre Osteoporosis Study. J Bone Miner Res 18(4):784–790
Napoli N, Schwartz AV, Schafer AL, Vittinghoff E, Cawthon PM, Parimi N, Orwoll E, Strotmeyer ES, Hoffman AR, Barrett-Connor E, Black DM (2018) Vertebral fracture risk in diabetic elderly men: the MrOS study. J Bone Miner Res 33:63–69
Yamamoto M, Yamaguchi T, Nawata K, Yamauchi M, Sugimoto T (2012) Decreased PTH levels accompanied by low bone formation are associated with vertebral fractures in postmenopausal women with type 2 diabetes. J Clin Endocrinol Metab 97:1277–1284
Tuominen JT, Impivaara O, Puukka P, Rönnemaa T (1999) Bone mineral density in patients with type 1 and type 2 diabetes. Diabetes Care 22:1196–1200
Sahin G, Bağis S, Cimen OB, Ozişik S, Güler H, Erdoğan C (2001) Lumbar and femoral bone mineral density in type 2 Turkish diabetic patients. Acta Medica (Hradec Kralove) 44:141–143
Compston J (2018) Type 2 diabetes mellitus and bone. J Intern Med 283:140–153
Ferrari SL, Abrahamsen BD, Napoli N et al (2018) Diagnosis and management of bone fragility in diabetes: an emerging challenge. Osteoporos Int 29(12):2585–2596
Napoli N, Chandran M, Pierroz DD, Abrahamsen B, Schwartz AV, Ferrari SL (2017) Mechanisms of diabetes mellitus-induced bone fragility. Nat Rev Endocrinol 13(4):208–219
Starup-Linde J, Eriksen SA, Lykkeboe S, Handberg A, Vestergaard P (2014) Biochemical markers of bone turnover in diabetes patients—a meta-analysis, and a methodological study on the effects of glucose on bone markers. Osteoporos Int 25:1697–1708
Starup-Linde J, Vestergaard P (2016) Biochemical bone turnover markers in diabetes mellitus—a systematic review. Bone 82:69–78
Yamamoto M, Yamauchi M, Sugimoto T (2013) Elevated sclerostin levels are associated with vertebral fractures in patients with type 2 diabetes mellitus. J Clin Endocrinol Metab 98:4030–4037
De Laet C, Kanis JA, Odén A et al (2005) Body mass index as a predictor of fracture risk: a meta-analysis. Osteoporos Int 16(11):1330–1338
Department of Disease Control Ministry of Health, PR China (2006) Guidelines for prevention and control of overweight and obesity in Chinese adults. People’s Medical Publishing House, Beijing
Zhao J, Xia W, Nie M et al (2011) The levels of bone turnover markers in Chinese postmenopausal women: peking Vertebral Fracture study. Menopause 18(11):1237–1243
Zhao J, Xia W, Nie M et al (2012) A haplotype of MATN3 is associated with vertebral fracture in Chinese postmenopausal women: peking Vertebral Fracture (PK-VF) study. Bone 50(4):917–924
Zhang ZHSJX, Shen JX (2005) Liu ZH (2005) Retrospective study on standardization of BMD machines in China. Chin J Osteoporos 11(2):133–145
Genant HK, Wu CY, van Kuijk C, Nevitt MC (1993) Vertebral fracture assessment using a semiquantitative technique. J Bone Miner Res 8(9):1137–1148
Schwartz AV, Vittinghoff E, Bauer DC, Hillier TA, Strotmeyer ES, Ensrud KE, Donaldson MG, Cauley JA, Harris TB, Koster A, Womack CR, Palermo L, Black DM (2011) Association of BMD and FRAX score with risk of fracture in older adults with type 2 diabetes. JAMA 305:2184–2192
Christiansen BA, Kopperdahl DL, Kiel DP, Keaveny TM, Bouxsein ML (2011) Mechanical contributions of the cortical and trabecular compartments contribute to differences in age-related changes in vertebral body strength in men and women assessed by QCT-based finite element analysis. J Bone Miner Res 26(5):974–983
Burghardt AJ, Issever AS, Schwartz AV et al (2010) High-resolution peripheral quantitative computed tomographic imaging of cortical and trabecular bone microarchitecture in patients with type 2 diabetes mellitus. J Clin Endocrinol Metab 95(11):5045–5055
Oei L, Zillikens MC, Dehghan A et al (2013) High bone mineral density and fracture risk in type 2 diabetes as skeletal complications of inadequate glucose control: the Rotterdam Study. Diabetes Care 36(6):1619–1628
Patsch JM, Burghardt AJ, Yap SP et al (2013) Increased cortical porosity in type 2 diabetic postmenopausal women with fragility fractures. J Bone Miner Res 28(2):313–324
Kurra S, Siris E (2011) Diabetes and bone health: the relationship between diabetes and osteoporosis-associated fractures. Diabetes Metab Res Rev 27(5):430–435
Cundy TF, Edmonds ME, Watkins PJ (1985) Osteopenia and metatarsal fractures in diabetic neuropathy. Diabetes Med 2(6):461–464
Kathol MH, el-Khoury GY, Moore TE, Marsh JL (1991) Calcaneal insufficiency avulsion fractures in patients with diabetes mellitus. Radiology 180(3):725–729
Acknowledgements
Our deepest gratitude to all the study participants and to the participating centers of the original parent study: Department of Endocrinology, China Rehabilitation Research Center; Department of Endocrinology, Beijing Liangxiang Hospital; Department of Cadre Unit, General Hospital of the Second Artillery Force; Department of Endocrinology, Peking University Shougang Hospital; Department of Endocrinology, Beijing Chaoyang Hospital, Capital University of Medical Science; Department of Endocrinology, Beijing Haidian Hospital; Department of Geriatric Endocrinology, Chinese People’s Liberation Army, General Hospital. We would also like to thank Ms. Yingying Hu for her valuable work in testing biomarker. The Peking Vertebral Fracture Study (PK-VF) was funded by the National Natural Science Foundation of China (No.81070687 and 81170805), National Science and Technology Pillar Program (2006BAI02B03), National Science and Technology Major Projects for “Major New Drugs Innovation and Development” (Grant2008ZX09312-016), Beijing Natural Science Foundation (No. 7121012), Scientific Research Foundation of Beijing Medical Development (No. 2007-3029), and National Key Program of Clinical Science (WBYZ2011-873).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Ruizhi Jiajue, Xuan Qi, Yan Jiang, Qiuping Wang, Wenbo Wang, Yu Pei, Xiran Wang, Wei Huang, Xin Zheng, Zhiwei Ning, Ou Wang, Mei Li, Xiaoping Xing, Wei Yu, Ling Xu, and Weibo Xia declare that they don’t have any conflicts of interest in this work.
Human and Animal Rights and Informed Consent
All of the subjects agreed to participate in this study and signed informed consent forms.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jiajue, R., Qi, X., Jiang, Y. et al. Incident Fracture Risk in Type 2 Diabetic Postmenopausal Women in Mainland China: Peking Vertebral Fracture Study. Calcif Tissue Int 105, 466–475 (2019). https://doi.org/10.1007/s00223-019-00598-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-019-00598-x