Abstract
Along with afferent information, centrally generated motor command signals may play a role in joint position sense. Isometric muscle contractions can produce a perception of joint displacement in the same direction as the joint would move if unrestrained. Contradictory findings of perceived joint displacement in the opposite direction have been reported. As this only occurs if muscle spindle discharge in the contracting muscle is initially low, it may reflect increased muscle spindle firing from fusimotor activation, rather than central motor command signals. Methodological differences including the muscle contraction task and use of muscle conditioning could underlie the opposing findings. Hence, we tested perceived joint position during two contraction tasks (‘hold force’ and ‘hold position’) at the same joint (wrist) and controlled muscle spindle discharge with thixotropic muscle conditioning. We expected that prior conditioning of the contracting muscle would eliminate any effect of increased fusimotor activation, but not of central motor commands. Muscle conditioning altered perceived wrist position as expected. Further, during muscle contractions, subjects reported wrist positions displaced ~12° in the direction of contraction, despite no change in wrist position. This was similar for ‘hold force’ and ‘hold position’ tasks and occurred despite prior conditioning of the agonist muscle. However, conditioning of the antagonist muscle did reduce the effect of voluntary contraction on position sense. The errors in position sense cannot be explained by fusimotor activation. We propose that central signals combine with afferent signals to determine limb position and that multiple sources of information are weighted according to their reliability.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The sense of limb position is vital to our ability to accurately control the movement of our limbs in space. The traditional view is that the sense of limb position is derived from afferent information in which muscle spindle endings are attributed the major role (McCloskey 1978; Gandevia 1996; Proske 2006) with skin stretch receptors and slowly adapting joint receptors providing supplementary information (Ferrell et al. 1987; Collins et al. 2005; Weerakkody et al. 2007). Evidence for muscle spindles as limb position sensors includes the presence of position and movement illusions induced by muscle vibration (Goodwin et al. 1972; Roll and Vedel 1982), which is known to be a strong stimulus for muscle spindles (Brown et al. 1967; Burke et al. 1976; Gregory et al. 1988; Cordo et al. 1993; Bergenheim et al. 2000).
Two groups of recent studies have shown that voluntary contractions affect position sense. Gandevia et al. (2006) reported that when the arm was paralysed and anaesthetised by ischaemia, a steady effort about the wrist created the perception of displacement in the direction of the attempted contraction. That is, when subjects pushed into flexion they perceived their paralysed wrist to be more flexed than during rest, and this effect was large (~15°). The same effect occurs with isometric contraction of the wrist muscles with the motor and afferent pathways intact, although this effect is smaller in magnitude (Smith et al. 2009). These results suggest a role for motor command signals in the perception of limb position. In contrast, Ansems et al. (2006) exploited the thixotropic properties of muscle (Proske et al. 1993) to set an initially high spindle firing rate in either the elbow flexor or extensor muscles. They reported that perception of joint position is only affected by muscle contraction if the muscle spindles in the agonist muscle have low firing rates before the contraction. Moreover, when judgements of limb position were altered, perceived displacement was in the opposite direction to the contraction, so that when subjects contracted the elbow flexors they perceived the elbow to be more extended (Walsh et al. 2004; Allen and Proske 2006; Walsh et al. 2006). Ansems and colleagues’ results are consistent with the effect of the muscle contraction on position sense being due to an increase in muscle spindle firing rates produced by co-activation of fusimotor drive with descending drive to the alpha motoneurones.
Thus, two sets of studies show that muscle contraction has an effect on limb position sense. However, one set supports the role of central motor command signals in position sense during muscle contraction, while the other set suggests an effect due to changes in muscle spindle firing caused by the fusimotor activation that accompanies muscle contractions. We postulated that these different findings were due to the different loading tasks used. One task was a ‘hold force’ task in which subjects pushed isometrically against a fixed object and were given force feedback to maintain the level of contraction (Gandevia et al. 2006; Smith et al. 2009). The other was a ‘hold position’ task in which subjects were instructed to maintain the position of their arm and the contraction level was controlled because they supported a weight (Ansems et al. 2006). Altered reflex responses and motor unit firing suggest that muscle spindle inputs contribute to motor output more during hold position than hold force tasks (Akazawa et al. 1983; Maluf et al. 2007; Baudry et al. 2009). As H-reflexes and long-latency stretch reflexes are altered in these tasks, muscle spindle input is likely to be processed differently at both the spinal and cortical levels (Doemges and Rack 1992; Maluf et al. 2007). However, it is not known whether the altered motor actions of muscle spindles are associated with changes in the perception of limb position. In addition, if motor cortical neurones or motoneurones are more facilitated by muscle spindle input, then less voluntary descending drive should be required to produce the same motor output. We hypothesised that, in the hold position task, the balance between the higher spindle discharge caused by fusimotor activation and voluntary motor output was altered when compared with the hold force task so that the spindle discharge was perceived as signalling that the contracting muscle was lengthened. We set out to test this hypothesis by having subjects do both a hold force and hold position task at the same joint in the same experiment under controlled conditions.
Due to the thixotropic properties of intrafusal muscle fibres, the contraction history of the muscle can alter the effect of subsequent contraction on perceived limb position (Ansems et al. 2006). Therefore, in the current study, we used prior conditioning contractions to control the contraction history of the muscle. This allowed us to set the firing rates of the muscle spindles in either the wrist flexor or wrist extensor muscles to a high firing rate, which meant we would then be able to predict changes in muscle spindle firing with muscle contraction. The study was designed to test whether voluntary contraction about the same joint in the same experiment resulted in different effects on perceived limb position in hold position and hold force tasks. We expected that prior contraction of the muscle at a short length would eliminate any effect that increased muscle spindle firing induced by fusimotor co-activation had on position sense. Effects due to a central motor command signal would be unchanged.
Some of these findings have been presented as an abstract (Walsh et al. 2008).
Methods
Eight subjects (5 male) aged 23–37 participated in this experiment. All subjects gave informed consent and the experimental procedures were carried out in accordance with the Declaration of Helsinki and with the approval of the University of New South Wales Human Research Ethics Committee. Subjects were unaware of the experimental hypotheses.
Subjects had their right forearm strapped down and their hand fixed in a manipulandum with the fingers extended and the dorsal surface of the hand aligned with the vertical plane. This manipulandum was connected to a rotating platform via a load cell (XTran 250N, Applied Measurements, Australia) and this arrangement restricted movement of the wrist to flexion and extension only (Fig. 1). The signal from the load cell was amplified and low-pass filtered with a 1 kHz cut-off. The manipulandum was marked with graduations in degrees so that wrist angle could be accurately set, and reproduced, by the experimenter. An angle of 0° was defined as when the hand and fingers were collinear with the forearm, with angles into flexion defined as negative and into extension as positive. A pointer placed above the wrist and with its axis collinear with the flexion-extension axis of the wrist, allowed subjects to signal the perceived angle of the wrist using their left hand. The scale was graduated in degrees and could only be seen by the experimenter. The subject’s arm was covered below the elbow to block the subject’s vision. Prior to experimental trials, the subjects performed maximal voluntary contractions (MVCs) into flexion and extension with the wrist clamped at 0°. Subjects then performed a series of trials in which they signalled the angle of their wrist under one of the three conditions detailed below. EMG was monitored using surface electrodes over the flexor and extensor carpi radialis muscles (band filtered at 16–1,000 Hz; CED 1902 amplifiers) in order to ensure that the muscle conditioning was applied. It was important in these experiments that we knew that subjects were contracting the correct muscles and following instructions to stay relaxed. EMG, torque at the wrist and wrist angle were sampled at 100 Hz and recorded using a CED 1401 data acquisition system (Cambridge Electronic Design, Cambridge, UK).
A diagram of the experimental setup. Left panel shows the subject’s right hand held in the manipulandum on the rotating platform. The right panel shows how the subject’s arm was covered with the dial used to indicate wrist angle. The axis of the dial was co-linear with the axis of the wrist and rotating platform. The wrist flexors or extensors were conditioned with a muscle contraction then the wrist was moved to the test angle and loaded into flexion or extension. Subjects either supported a weight (hold position) or made an isometric contraction (hold force) with the platform clamped into position. At the same time, they made a judgement of their wrist angle and indicated it with the pointer using their left hand
Conditioning contractions of either the wrist flexor or wrist extensor muscles were used at the start of each trial. We did this to manipulate the ongoing firing rate of muscle spindle endings (Proske et al. 1993). For flexion conditioning, the wrist was fixed into 20° of flexion, the subject then performed a contraction that was 4 s long with the wrist flexors, at 30% of the force measured during their MVC. Visual feedback of the force was provided on an oscilloscope. Extension conditioning was done in the same way, with the wrist fixed at 20° of extension. One of these muscle conditioning procedures was used before every trial throughout the experiment.
After each conditioning contraction, the experimenter moved the wrist into a test position while wrist extensor and wrist flexor EMG were monitored to ensure that these muscle groups were relaxed. Occasional trials had to be repeated due to poor relaxation, typically only 2–3 per subject. Based on pilot studies, the following three experimental conditions were combined and studied in one testing session.
Condition one: position signalled at rest
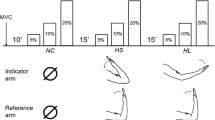
The purpose of this condition was to confirm that the muscle conditioning was working as expected and to provide a baseline for the comparison of the loaded conditions. After muscle conditioning, the manipulandum was moved and then clamped at either 10° wrist flexion or 10° wrist extension (0° is a straight wrist) (Fig. 2a). While the right arm remained relaxed, subjects used the pointer to indicate their perceived wrist angle. The instruction was “show me where your wrist is.” This task was performed three times at both 10° wrist flexion and 10° wrist extension, after both types of muscle conditioning. This gave a total of 12 trials for this condition.
Stylised examples of trials for the three experimental conditions. The two boxes mark the conditioning phase (left in each panel) and the judgement phase (right in each panel). The arrows indicate the direction of wrist flexion. a An example of a ‘rest’ trial. The wrist extensor muscle was conditioned but there was no contraction while wrist angle was judged. b An example of a hold force trial. In this task the subject contracted during the judgement of wrist angle, but wrist position was fixed by clamping the manipulandum. This panel is an example of a trial when the same muscle was conditioned and loaded (i.e. agonist conditioning). c In the hold position task the subject also contracted during the judgement phase. Here both the position and force were unsteady, as the subject had to control the load and hold it in position. This is an example of a trial in which the muscles that were conditioned and the muscles that were loaded were different (i.e. antagonist conditioning)
Condition two: position signalled during a ‘hold force’ contraction
After the same muscle conditioning as above, the wrist was moved passively and clamped into either 10° flexion or 10° extension. Using visual feedback of force, subjects were required to produce either a wrist flexion force or a wrist extension force using isometric contractions of 30% MVC for the relevant muscle group. While maintaining this contraction, the subject indicated the perceived wrist angle using the pointer (Fig. 2b). Subjects usually took 2–3 s to stabilise the contraction before they signalled wrist position. If subjects took more than 5 s the trial was aborted and restarted. The condition involved a total of 24 trials. Three trials were performed at both test angles after both muscle conditioning types, and during both wrist flexion and wrist extension (4 different tasks at two test angles). With this hold force task, we expected to confirm that perceived wrist position was displaced in the direction of effort (Smith et al. 2009) and that this was not affected by manipulation of muscle spindle firing rates with muscle conditioning.
Condition three: position signalled during a ‘hold position’ contraction
After muscle conditioning, the wrist was moved into the test position (10° wrist flexion or wrist extension) but was not clamped (Fig. 2c). Instead, the subject was given visual feedback of wrist angle on an oscilloscope and was instructed to maintain their wrist angle. The wrist was then loaded in the direction of wrist flexion or wrist extension by applying a weight. The weight was chosen to produce a force equal to that in the isometric task (30% MVC). Subjects were required to maintain an isotonic contraction and, at the same time, indicate perceived wrist angle with the pointer. Again the instruction was “show me where your wrist is.” As for condition two, subjects performed three trials at each of the eight different task combinations resulting in another 24 trials. Visual feedback of wrist angle was only available once the weight was applied to the wrist and provided no information on absolute wrist angle. This was achieved by applying a DC offset to the feedback signal so that it always had the same baseline when the subject saw it. Trials in which subjects did not maintain wrist angle accurately were aborted and repeated. With this hold position task, we expected that, as previously shown at the elbow, perceived wrist position would be displaced in the direction opposite to the effort and that this would be abolished by prior conditioning of the contracting muscle.
The combined trials for each of the three conditions (total of 60) were presented in a random order. They were presented in blocks of 20 with 5 min rests between blocks to avoid loss of attention or muscle fatigue. At no time was the subject removed from the apparatus or allowed to see their arm.
Data and statistical analysis
The errors made by subjects were calculated as the difference between their actual wrist angle and the angle they indicated with the pointer. Data from the two test angles were pooled after a paired t test showed that there was not a significant difference between these two groups of data. Within each type of muscle conditioning the mean position error during the rest task was subtracted from the mean position error for each loading task. This is the subtraction of a large number from a small one, and so has the visual effect of reversing the direction of the muscle conditioning effect in Fig. 4. This reversal indicates the reduced effect of the prior conditioning during muscle contraction. The statistical analysis for the hold force and hold position data consisted of three-way repeated measures ANOVAs testing the factors of task (hold force/hold position), direction of loading (flexor muscles/extensor muscles) and type of muscle conditioning (flexion conditioning/extension conditioning) as well as all of the interactions. Analysis of the rest condition was carried out with a paired t test. The EMG was compared across tasks (hold force/hold position) with a t test and there was no significant difference. For all tests the threshold for statistical significance was set at p < 0.05 and all data are presented as the mean ± SEM.
Results
The perceived position of the wrist was compared under three conditions: during muscle relaxation, in a ‘hold force’ contraction and ‘hold position’ contraction. The subjects were able to maintain the target force during the hold force task and did so to within 20% of the target force (±6% MVC). During the hold position task, subjects were typically good at maintaining wrist angle given visual feedback. They were able to do this within ≈1°.
After conditioning of wrist extensor muscles with a voluntary contraction at short length, subjects at rest perceived the wrist to be more flexed than it actually was. With conditioning of the wrist flexor muscles, there was a trend towards perception of a more extended wrist angle. Pooled data for eight subjects is shown in Fig. 3. Perceived wrist angle was significantly different after extension and flexion conditioning (13.3° ± 1.5° into flexion and 1.8° ± 1.5° into extension, respectively, t 7 = 10.00, p < 0.0005). This difference is consistent with a contribution from the muscle spindles in the conditioned muscle to the perception of joint position. During the hold force trials, the difference between extension and flexion muscle conditioning was on average 5.9° ± 6.5° (F 1,7 = 27.95, p < 0.005), which was smaller than during rest (15.1° ± 3.0°).
Errors in indication of wrist angle during rest and during hold force contraction after muscle conditioning. The means (±SEM) of eight subjects for indicated wrist position after muscle conditioning during rest and during hold force contractions. Either the subject’s wrist flexor muscles (closed circles) or wrist extensor muscles (open circles) were conditioned with a prior contraction. The wrist was then moved to the test position and wrist position was signalled with a pointer, either at rest, or during a hold force contraction into flexion or extension. The effect of the muscle conditioning was consistent with increase firing rates in the conditioned muscles. The effect of the muscle conditioning was less during a contraction than during rest. Asterisk indicates a statistically significant difference (p < 0.05) and the arrows indicate the direction of the errors
Figure 4 displays pooled data for both tasks (hold force and hold position). Each data point is the mean position error for that type of muscle conditioning and loading task, minus the mean error for that type of muscle conditioning during rest. Thus Fig. 4 displays the component of the position error that is associated with the muscle contraction. These results show an effect of the direction of the load (F 1,7 = 6.24, p < 0.05), such that when subjects contracted the wrist flexors and judged position they perceived the wrist to be more flexed than when they contracted the wrist extensors. This difference between a contraction of the wrist flexors and a contraction of the wrist extensor muscles was ~12°. There was no difference in the size and duration of this effect between hold force and hold position contractions.
Errors in indication of wrist angle for the group of subjects. Means (±SEM) of eight subjects for the two loading conditions shown as differences from the resting conditions. Trials with extension conditioning are shown as the difference from rest trials with extension conditioning. Trials with flexion conditioning are shown as the difference from the rest trials with flexion conditioning. The hold force task required subjects to indicate wrist angle while they pushed into flexion or extension (left) against an immoveable surface and the hold position task required subjects to indicate wrist angle during contraction into flexion or extension (right) to support a weight. When subjects made either hold force or hold position contractions into flexion, they perceived their wrist to be more flexed than when they made extension contractions. Open circles indicate data obtained when the wrist extensor muscles were conditioned with a brief contraction at a short muscle length before the trial and closed circles indicate data when the wrist flexor muscles were conditioned with a brief contraction at short length. Asterisk indicates a statistically significant difference (p < 0.05) and the arrows indicate the direction of the errors
In Fig. 5, instead of grouping trials depending upon whether the flexors or extensors were conditioned, data are grouped depending on whether the conditioned muscle was the agonist or antagonist compared to the muscle used in the hold force and hold position tasks. Figure 5 shows that when the agonist muscle was conditioned, subjects perceived a change of the position of the wrist in the direction of the muscle contraction. There was no difference in this effect between flexion or extension conditioning. However, when the antagonist muscle was conditioned there was little or no change in perceived wrist angle associated with muscle contraction. The difference in perceived wrist angle between prior conditioning of the muscle contracting in the task (agonist) or the antagonist muscle was ~9°, which was significant (F 1,7 = 27.95, p < 0.005).
Error in indication of wrist angle for the group of subjects when the data are separated into trials in which there was conditioning of the agonist or antagonist muscles. The mean (±SEM) of eight subjects for the two loading conditions of hold force (left) and hold position (right). Errors have been plotted in reference to the agonist or antagonist direction for both the direction of the errors and the muscle conditioning. Open circles indicate data obtained when the antagonist muscles were conditioned with a brief contraction at short muscle length before each trial and the closed circles indicate data obtained when the agonist muscle was conditioned at short length. Asterisk indicates a significant difference (p < 0.05) between agonist and antagonist conditioning
Discussion
When subjects performed a contraction with the wrist muscles they perceived the angle of the wrist to be displaced in the same direction as the contraction although the actual wrist angle did not change. Voluntary flexor or extensor contractions were made prior to trials to control the contraction history of the muscle. Thixotropic muscle conditioning manipulates muscle spindle firing rates (Morgan et al. 1984; Wilson et al. 1995) and also produced illusions of altered wrist position.
We hypothesised that for the ‘hold position’ task higher spindle discharge rates with contraction would be perceived as signalling that the contracting muscle was lengthened. This is not what occurred. We did not find any differences in subjects’ perception of wrist position between the two loading tasks ‘hold force’ and ‘hold position’ (Fig. 4). However, there was a novel interaction of the muscle conditioning and the muscle contraction (Fig. 5), but not in the way we expected. If the interaction were due to the effects of fusimotor drive on spindle firing then we would expect that the muscle contraction would have no effect on perceived wrist position when the agonist muscle was conditioned, as found by Ansems et al. (2006). However, the interaction we observed was that conditioning the antagonist muscles reduced or removed the effect of the muscle contraction on position sense, whereas after conditioning the agonist muscle, muscle contraction resulted in perceived displacement of the wrist in the same direction as the contraction.
One interpretation of our results, which could be suggested by the data in Fig. 3, is that contraction of the muscle to perform the task removes any effect of the thixotropic conditioning of the muscle when the intrafusal muscle fibres are activated by fusimotor drive. This would imply that an accurate perception of wrist position is signalled by muscle spindles in the contracting muscle and that some central process subtracts the increases in spindle firing rates resulting from fusimotor drive. However, if cancellation of thixotropic conditioning was the sole mechanism for perceived wrist displacement associated with muscle contraction, then we would see no effect when an ‘unconditioned muscle’ was contracted. Although it is not possible to test a muscle with no contraction history (i.e. unconditioned), it is possible to produce neutral conditions where wrist position is perceived as midway between those produced by wrist flexor or wrist extensor conditioning. In Smith et al. (2009) prior to each test “the wrist was rapidly moved backwards and forwards over a range of angles from 30 to 90°” to minimise thixotropy. After this neutral muscle conditioning, with both wrist flexor muscle spindles and wrist extensor muscle spindles in a state of low firing, muscle contraction resulted in perceived displacement of the wrist in the direction of contraction. Therefore, muscle contraction influences perceived limb position independently of changes in muscle spindle firing produced by fusimotor drive.
Some proprioceptive judgements require a combination of afferent and efferent signals (Gandevia and McCloskey 1978). Another possible explanation for the differential effect on perceived wrist position of antagonist or agonist conditioning combined with contraction is that the brain is weighting the signals available to it. In order to determine the position of a joint, the brain has access to muscles spindles on both (or more) sides of the joint, motor commands from any muscles that are contracting as well as information from skin and joint receptors. None of these signals is a perfect indicator of position and all can be affected by outside sources of noise. In some conditions, multisensory cues are weighted according to their reliability so that the integrated perception is statistically optimal (Ernst and Banks 2002; Alais and Burr 2004; Helbig and Ernst 2007). Here, the brain may give less weight to signals that it knows are contaminated, for example muscle spindles in a contracting muscle, when it combines all of the sensory information available into a perception of limb position.
In summary, this study has shown that despite any mechanistic difference in motor control behaviour between hold force and hold position loading tasks (Mottram et al. 2005; Rudroff et al. 2005; Maluf et al. 2007), when subjects indicated their joint angles the perception was the same for both types of task. Our hypothesis that the different loading tasks would cause different effects of muscle contraction on perceived limb position was disproved and the reason for discrepancies between the findings of previous studies remains unclear. One possible reason is the different methods used to indicate joint angle (a bilateral match, e.g. Ansems et al. 2006; an indication with a pointer Gandevia et al. 2006; Smith et al. 2009). The sense of limb position is complex, with many sources of information interacting to produce perception. An interaction between the perception of the position of both arms and the internal body image could result in the suppression or removal of the influence of motor commands when a matching task is performed, as opposed to the indicating task we have performed here. The question needs further investigation. In addition to the demonstration that perception of arm position does not depend on the type of loading, we have also shown a novel interaction between thixotropic muscle conditioning and muscle contraction during perception of limb position. We interpret our findings as consistent with a role of motor command signals in position sense and with the brain weighting the multiple sources of information available to it to create a perception.
References
Akazawa K, Milner TE, Stein RB (1983) Modulation of reflex EMG and stiffness in response to stretch of human finger muscle. J Neurophysiol 49:16–27
Alais D, Burr D (2004) The ventriloquist effect results from near-optimal bimodal integration. Curr Biol 14:257–262
Allen TJ, Proske U (2006) Effect of muscle fatigue on the sense of limb position and movement. Exp Brain Res 170:30–38
Ansems GE, Allen TJ, Proske U (2006) Position sense at the human forearm in the horizontal plane during loading and vibration of elbow muscles. J Physiol (Lond) 576:445–455
Baudry S, Jordan K, Enoka RM (2009) Heteronymous reflex responses in a hand muscle when maintaining constant finger force or position at different contraction intensities. Clin Neurophysiol 120:210–217
Bergenheim M, Ribot-Ciscar E, Roll JP (2000) Proprioceptive population coding of two-dimensional limb movements in humans: I. muscle spindle feedback during spatially oriented movements. Exp Brain Res 134:301–310
Brown MC, Engberg I, Matthews PBC (1967) The relative sensitivity to vibration of muscle receptors of the cat. J Physiol 192:773–800
Burke D, Hagbarth KE, Lofstedt L, Wallin BG (1976) The responses of human muscle spindle endings to vibration of non-contracting muscles. J Physiol 261:673–693
Collins DF, Refshauge KM, Todd G, Gandevia SC (2005) Cutaneous receptors contribute to kinesthesia at the index finger, elbow, and knee. J Neurophysiol 94:1699–1706
Cordo P, Gandevia SC, Hales JP, Burke D, Laird G (1993) Force and displacement-controlled tendon vibration in humans. Electroencephalogr Clin Neurophysiol 89:45–53
Doemges F, Rack PM (1992) Task-dependent changes in the response of human wrist joints to mechanical disturbance. J Physiol 447:575–585
Ernst MO, Banks MS (2002) Humans integrate visual and haptic information in a statistically optimal fashion. Nature 415:429–433
Ferrell WR, Gandevia SC, McCloskey DI (1987) The role of joint receptors in human kinaesthesia when intramuscular receptors cannot contribute. J Physiol 386:63–71
Gandevia SC (1996) Kinesthesia: roles for afferent signals and motor command. In: Rowell L, Shepherd J (eds) Handbook on integration of motor, circulatory, respiratory and metabolic control during exercise. American Physiological Society, Bethesda, pp 128–172
Gandevia SC, McCloskey DI (1978) Interpretation of perceived motor command by reference to afferent signals. J Physiol 283:493–499
Gandevia SC, Smith JL, Crawford M, Proske U, Taylor JL (2006) Motor commands contribute to human position sense. J Physiol 571:703–710
Goodwin GM, McCloskey DI, Matthews PBC (1972) The contribution of muscle afferents to kinaesthesia shown by vibration induced illusion of movement and by the effects of paralysing joint afferents. Brain 95:705–748
Gregory JE, Morgan DL, Proske U (1988) Aftereffects in the responses of cat muscle spindles and errors of limb position sense in man. J Neurophysiol 59:1220–1230
Helbig H, Ernst M (2007) Optimal integration of shape information from vision and touch. Exp Brain Res 179:595–606
Maluf KS, Barry BK, Riley ZA, Enoka RM (2007) Reflex responsiveness of a human hand muscle when controlling isometric force and joint position. Clin Neurophysiol 118:2063–2071
McCloskey DI (1978) Kinaesthetic sensibility. Physiol Rev 58:763–820
Morgan DL, Prochazka A, Proske U (1984) The after-effects of stretch and fusimotor stimulation on the responses of primary endings of cat muscle spindles. J Physiol 356:465–477
Mottram CJ, Christou EA, Meyer FG, Enoka RM (2005) Frequency modulation of motor unit discharge has task-dependent effects on fluctuations in motor output. J Neurophysiol 94:2878–2887
Proske U (2006) Kinesthesia: the role of muscle receptors. Muscle Nerve 34:545–558
Proske U, Morgan DL, Gregory JE (1993) Thixotropy in skeletal muscle and in muscle spindles: a review. Progr Neurobiol 41:705–721, 0301-0082
Roll JP, Vedel JP (1982) Kinaesthetic role of muscle afferents in man, studied by tendon vibration and microneurography. Exp Brain Res 47:177–190
Rudroff T, Poston B, Shin I-S, Bojsen-Moller J, Enoka RM (2005) Net excitation of the motor unit pool varies with load type during fatiguing contractions. Muscle Nerve 31:78–87
Smith JL, Crawford M, Proske U, Taylor JL, Gandevia SC (2009) Signals of motor command bias joint position sense in the presence of feedback from proprioceptors. J Appl Physiol 106:950–958
Walsh LD, Hesse CW, Morgan DL, Proske U (2004) Human forearm position sense after fatigue of elbow flexor muscles. J Physiol 558:705–715
Walsh LD, Allen TJ, Gandevia SC, Proske U (2006) The effect of eccentric exercise on position sense at the human forearm in different postures. J Appl Physiol 100:1109–1116
Walsh LD, Gandevia SC, Taylor JL, Smith JL (2008) The role of motor command signals in human position sense is preserved in different loading conditions. In: Proceedings of Australian Neuroscience Society 28th Annual Meeting
Weerakkody NS, Mahns DA, Taylor JL, Gandevia SC (2007) Impairment of human proprioception by high-frequency cutaneous vibration. J Physiol 581:971–980
Wilson LR, Gandevia SC, Burke D (1995) Increased resting discharge of human spindle afferents following voluntary contractions. J Physiol 488:833–840
Acknowledgments
This work was supported by the National Health and Medical Research Council. We are grateful to Dr. Hiske van Duinen for comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Walsh, L.D., Smith, J.L., Gandevia, S.C. et al. The combined effect of muscle contraction history and motor commands on human position sense. Exp Brain Res 195, 603–610 (2009). https://doi.org/10.1007/s00221-009-1832-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-009-1832-3