Abstract
Purpose
Rupture of the anterior cruciate ligament is common and may necessitate surgical reconstruction. Surgical reconstruction aims to restore normal kinematics and biology within the knee. The acute phase response after surgical reconstruction remains poorly defined but may influence graft integration through modulation of host tissue remodelling.
Methods
The very early host production of key cytokines after surgery was studied. A consecutive series of 14 patients undergoing reconstructive surgery were studied per-operatively, 1 and 6 h after surgery, examining the hypothesis that the acute phase response would be non-specific but consistent between individuals, demonstrating increases of pro-inflammatory cytokines.
Results
A consistent increased release of monocyte-driven, non-specific, IL-1 and IL-6 release but not T cell-derived IL-2 was found. Perhaps, more interestingly, very early high concentrations of secondary growth factors PDGF and TGF-β suggestive of an anabolic response were found.
Conclusion
These data support the contention that an anabolic response starts earlier than previously thought within the surgically reconstructed knee.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The anterior cruciate ligament (ACL) is one of the most frequently injured structures in the knee [16, 19, 22]. Surgical reconstruction involves transplantation of autograft tissue, integration of the graft within host peri-articular subchondral bone and remodelling of the host-tissue construct. The host response to surgical insult and the generation of a haemarthrotic response is associated with cellular infiltration and the release of high concentrations of reactive mediators [16]. There remains limited understanding of the very early host-generated response, catabolic pro-inflammatory and anabolic growth factor mediators, to iatrogenic surgical intervention yet it is considered crucial to successful restoration of knee biology and graft integration. However, failure of such incorporation with resultant recurrent laxity and late chondral damage remains a significant clinical problem [4, 5, 43]. Such failure may occur in up to 15% of patients within 2 years from date of such surgery [32, 41].
ACL graft incorporation is believed to be different for patellar tendon autografts [6, 23, 35, 44] and hamstring autografts [28]), but the histological stages are believed to follow a standard pathway of inflammation, necrosis, revascularisation and remodelling [16, 30, 40] that may become disordered [11, 16]. These cellular changes are mediated through cytokine release. Demirag et al. [17] showed that administration of α2-macroglobulin to block MMP activity in a rabbit ACL reconstruction model leads to production of denser connective tissue in-growth in the bone tunnels. This late phase of successful graft integration and remodelling is dependent upon coordinated cytokine release, fibroblast ingrowth and stromal maturation. It has been postulated that a disordered cytokine pattern of release may herald failure of successful integration with an imbalance between anabolic and catabolic mediators acting locally [17].
The primary hypothesis was that the acute phase response to such surgery would follow an identifiable course in the majority of patients and secondly that the characterisation of this normal response could ultimately allow for early identification of patients in whom the response is disordered.
Materials and methods
Patients listed for ACL reconstruction under the care of the senior author over a four-month period and who were between the ages of 16 and 50 were eligible for inclusion in the study provided that their knee was otherwise quiescent, had a full range of movement and was clinically in the chronic, steady state, phase of recovery after their ACL rupture. Patients undergoing multiple ligament reconstruction were excluded. Sequential patients were approached and asked to give appropriately informed consent to the retention of biological material that would otherwise have been discarded. Approval for this project was granted by the (blinded for submission) Local Research Ethics Committee (LREC).
The senior author performed all ACL reconstructions, using an arthroscopically assisted technique. All operations took place between 0830 and 1130 to ensure that all samples from each patient were taken and processed the same day and to minimise any potential error from diurnal variation. Each patient received per-operative intravenous antibiotics (co-amoxyclav 1.2 g) before induction and maintenance of general anaesthesia. The gracilis and semitendinosus tendons were identified and harvested, via a short oblique incision at the pes anserinus, and then prepared. The joint was inspected. The femoral and tibial tunnels were drilled. The graft was inserted and fixed on the femoral side using an endobutton and at the tibial side by an interference screw (Acufex, Smith and Nephew, Memphis). After closure of the harvest site, an intra-articular drain was inserted and clamped. Twenty millilitres of 0.5% marcain (bupivacaine) and 10 mg of morphine sulphate were injected into the joint. The drain was unclamped forty minutes post-operatively, removing all joint fluid and residual local anaesthetic solution, and removed after 6 h.
Samples
The first author prepared and processed all samples.
-
1.
In preparation of the ACL graft, small sections of hamstring tendon in excess of requirement are generally discarded: these were retained and cultured. These samples represent the new tissue being brought into the knee, and its potential contribution to the acute phase response (APR).
-
2.
Before insertion of the arthroscope, a sample of synovial fluid was aspirated from the knee and retained. As the majority of synovial fluid samples were very viscous, they were diluted 1 + 4 by volume with sterile distilled water prior to storage. These samples represent the steady state of the knee prior to the start of the operation.
-
3.
During preparation of the notch, the native ACL stump is generally removed: samples of this were taken with an arthroscopic instrument and retained and cultured. These samples represent contribution of the soft tissue within the knee to the APR.
-
4.
Fluid in the drain was collected at one and 6 h after surgery and stored separately. These samples represent the post-surgical APR.
Tissue samples were placed into sterile containers with phosphate-buffered saline (PBS) and kept on ice until transfer to the laboratory for preparation, within 4 h. In the laboratory, the samples were cut up into 1–3-mm pieces and cultured at 37°C in a 5% CO2 incubator for 24 h in Petri dishes with Dulbecco’s Modified Eagle’s Medium (DMEM) (Invitrogen Ltd, Paisley, UK), containing 4 mM glutamine (Sigma–Aldrich Company Ltd, Dorset, UK) and 50 IU penicillin and 50 μg ml−1 streptomycin (Invitrogen Ltd, Paisley, UK). Following culture, the medium was removed and transferred into 15-ml sterile tubes to be micro-centrifuged at 4,000 rpm for 4 min to allow removal of tissue debris before storage at −80°C until required for cytokine and growth factor assay.
All knees were quiescent—exhibited minimal effusion, had active full movement and the patient could demonstrate normal gait pattern. This was consistent with the absence of a synovial response found at arthroscopic inspection at the time of surgical intervention.
After skin preparation with a dilute betadine solution and under tourniquet control, an aspirate of joint fluid (<5 ml) was obtained from each patient and this aliquot transferred into sterile containers. The drain fluid was emptied directly into similar tubes. All samples were kept on ice, before being transported back to the laboratory for preparation within 4 h. Fluid samples were centrifuged at 300g for 30 min to remove any cellular debris, and the supernatant was removed and transferred to new 15-ml sterile tubes before being stored at −80°C until required for cytokine and growth factor assay.
Assays
A Human 12-Plex Custom Build Multiplex Assay (a solid phase sandwich immunoassay) and a Multi Species TGF-β1 Assay (Invitrogen (formally BioSource) (LCP0012 and LHG0121)) were used to measure the levels, relative to volume, of the following cytokines and growth substances in the samples: IFN-γ, IL-1β, IL-1 RA, IL-2, IL-4, IL-6, IL-6 R, EGF, FGF-basic, PDGF, TGF-β, TNF-α and VEGF.
Cytokines/growth factors
The principal actions of the studied interleukins (IL-) and growth factors are given in Table 1.
Statistical analysis
SigmaPlot (Version 10.0) and SigmaStat (Version 3.1) were used for all data analysis and plotting. Results are presented as mean ± standard deviation where normally distributed or as median and interquartile range [25, 75%] where the distribution was not normal. Paired t-tests were used to assess differences between the sample means where the data were normally distributed. The Mann–Whitney test was used for unpaired data and the Wilcoxon signed rank test for repeated measures of non-normally distributed data. Values of P < 0.05 were considered to be statistically significant and if >0.05 are reported as not significant (n.s.).
Results
Fourteen sequential patients were approached to participate in the study, and none declined. Samples were obtained from all fourteen patients. The subjects were all men, aged 17–45 (median: 28 years), with symptomatic ACL deficiency 4–22 months after injury and no other significant medical conditions. All knees were quiescent with no effusion and all patients at pre-assessment had normal serum biochemical and haematological markers. No patients had greater than grade 1 Outerbridge grade localised (<1 cm) chondral injury. Eight of the fourteen patients were found to have minor meniscal partial tears necessitating trimming but not formal resection or suture. No patient underwent arthrotomy.
Cytokines
IFN-γ was found in all samples from each patient and showed significant increases over time in the drain samples (median 4 pg/ml (25% 3; 75% 6), rising to 6 pg/ml (25% 4; 75% 7) (P = 0.023). Production by graft and native ACL tissue was similar, and median concentration was 3 pg/ml.
The presence of IL-1β was detected in all per-operative synovial fluid samples (Fig. 1), whereas IL-1 Ra was only confirmed in six of the twelve samples analysed. IL-1β and IL-1 Ra were found in all the drain fluid samples collected, increasing over time (P = 0.008 and P = 0.025, for IL-1β and IL-1 Ra, respectively), with patient 12 being an outlier (Fig. 1). IL-1β was present in all of the culture media samples. IL-1 Ra was only present in three cases.
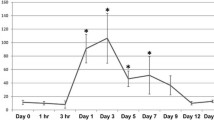
Mean Concentration (horizontal line), box plot and 95% CI of the studied cytokine/growth factor for the following time points with the number of studied samples given below. a Per-operative synovial fluid samples (n = 12). b 1-h drain fluid (n = 12). c 6-h drain fluid (n = 14). d ACL stump tissue culture (n = 14). e Graft tissue culture (n = 14)
IL-2 was identified in all per-operative synovial fluid samples, culture medium samples and increased with time in drain fluid samples (Fig. 1).
IL-4 was only confirmed at low concentrations in one of the per-operative fluid samples, four of the six-hour drain samples and in only one of the culture medium samples.
IL-6 was found in low concentration in the per-operative synovial fluid samples and in the 1-h drain samples (median concentration 2 pg/ml). The soluble receptor was found in all per-operative synovial fluid samples and 1-h drain samples. The increases in concentration over time for IL-6 and sIL-6r were both shown to be statistically significant (P = 0.025 and P = 0.010, respectively) (Fig. 1). IL6 was found in all but two culture media samples, in several cases above the working range of the assay (>1,480 pg/ml) as was the soluble receptor.
TNF-α was found in all per-operative synovial fluid samples at a median concentration of 19 pg/ml. Levels increased in all drain samples between one and six hours (9 pg/ml and 11 pg/ml, P = 0.005) (Fig. 1). The median concentration in culture medium samples was 5 pg/ml.
Growth factors
EGF was found in the per-operative synovial fluid samples of only nine patients with a median of 131 pg/ml (25% 71, 75% 260).
FGF-β was not found in any of the per-operative synovial fluid samples and in only five of the 1-h drain samples [median 153 pg/ml, (25% 109, 75% 242)] and nine of the 6-h drain samples [median 244 pg/ml (25% 174, 75% 311), n.s.] (Fig. 1).
PDGF was confirmed in all per-operative synovial fluid samples (median 11 pg/ml (25% 3, 75% 54). It was found in 1-h and 6-h drain samples (314 pg/ml (25% 203, 75% 547) and 765 pg/ml (25% 628, 75% 1,024), respectively, P = 0.013) (Fig. 1) but only at low levels in culture medium samples (ACL stump median 1 pg/ml, Graft 2 pg/ml).
TGF-β was confirmed in all the per-operative synovial fluid samples collected, in the range of 65–575 pg/ml for ten of these patients. It was present in all drain fluid samples and increased from a median of 1,410 pg/ml (25% 920, 75% 1,606) at 1 h to 2,594 pg/ml (25% 1,964, 75% 3,043) at 6 h (P < 0.001).
VEGF was shown to increase with time in drain samples at 1 h 23 pg/ml (25% 12, 75% 52), at 6 h 251 (25% 228, 75% 275) pg/ml (P = 0.008).
Discussion
The most important finding of the present study is the consistent release in the acute inflammatory post-surgical phase of anabolic growth factors within the knee. This represents a unique attempt to understand the acute phase response in a human model of surgical autograft transplantation within the contained environment of the knee joint. Through distinct time point sampling a host cytokine response with increases in IL-1, sIL-6r and TNF-α, coupled with an increase in the immunosuppressive B-cell cytokine Il-4, was found. Furthermore, tissue substrate culture has demonstrated differential ability of uninjured autograft tendon when compared with native ACL stump to produce both PDGF and FGF (which is not normally seen at such high levels so early in the APR). Sequential analysis of cytokine and growth factor concentration in a series of patients undergoing reconstructive surgery for ACL deficiency and per patient comparison of pro- and anti-inflammatory mediator levels is key to understanding the host response to such surgery and ultimately to modulating the integrative response by the host to this procedure. These key mediators have short half-lives, and the acute phase response shifts to a chronic response after 48 h, so close early monitoring is most likely to identify important changes [8].
Previous work has examined at a single time frame local concentrations of specific mediators from synovial fluid aspirated in patients with a variety of meniscal/chondral and ligamentous injured knees; this study has examined the temporal changes in such cytokine levels in a distinct and controlled group of patients [26]. It is crucial to examine both the early and the late phase of cytokine release within the knee joint so as to successfully unravel the pattern of such mediated cellular response and graft integration. This may help to explain possibly why these workers failed with sampling at day 7 after surgery to identify any particular pattern or relationship between radiographic tunnel widening and concentrations of pro-inflammatory mediators [45]. One of the key hypotheses underscoring this preliminary work is the view that sustained growth factor release will direct appropriate graft healing. This study has given an insight for the first time into the active agents released at the initiation of the host response to surgical insult. Previous work has failed to identify the correct time frame for study of the biochemical processes underscoring the healing response and in our opinion this has lead to poor interpretation of the subsequent single time frame cytokine concentrations. It is clear from this study that two major markers PDGF and TGF are active within 6 h of surgical insult. Longer time frame study and analysis of mRNA are clearly required. Furthermore, study of greater numbers of patients and correlation with these key growth factor concentrations may help with our understanding of graft failure.
The biomarkers targeted were based on previous relevant work and the availability of multiplex assay plates. The growth substances targeted were ones most documented and those with established functions. Our work has identified some molecules of interest, while perhaps reducing interest in others and this information can guide further targeted research. Future work may also want to look at additional cytokines and growth factors such as IL-8, 10 and 17, that are also well documented, for their pro- and anti-inflammatory roles.
In the per-operative synovial fluid samples, all the biomarkers studied provided a baseline for assessment [38]. IL-4 acts predominantly as an immune down regulator and would not be expected to be raised in a quiescent knee. Previous work has found, in single time frame sampling, higher baseline concentrations of the pro-inflammatory cytokines (IL-1β, TNF-α and IL-6) in synovial fluid of ACL deficient knees [25]. Patients with chronic anterior cruciate ligament deficiency and recurrent chondral injury were previously found to have had a sustained inflammatory response predisposing the patient to an increased risk of osteoarthritis [9, 10, 29]. Interpretation of local peak concentrations of key mediators at single time points is confounded by the lack of a clear pattern of change and must be placed in the context of a defined acute clinical picture.
Sample intervals for drain fluid samples revealed an evolving inflammatory response between one and 6 h after surgery. For all sample types, there were some patients with samples that appeared to be outliers, the importance of which remains to be clarified.
There are limitations to the interpretation of this work. The study group was small and were all men, a chance occurrence, as sequential eligible patients were recruited. Hormonal differences in women have been suggested as the reason that they have higher injury rates (per hour of specific sport played) than men [24], so this information may not be generalisable to women. It is accepted that the instillation of bupivicaine, known to be chondrocytoxic in animal studies [12] and morphine to the knee may, to some degree, alter the acute phase response, but it was felt to be unethical to withhold what has proven to be a useful analgesic approach for these patients [37]. Further, the unclamping of the drain tube would be expected to remove the local non-bound anaesthetic, reducing the potential effect on chondrocytes. This preliminary study was not intended to provide any correlation between acute phase response and eventual graft incorporation, as the small numbers would invalidate any firm clinical conclusions. Rather this dataset can be viewed as a first stage of characterisation of the acute phase response in the human knee after ACL reconstructive surgery.
The underlying mechanism of failure of ACL graft integration remains relatively unknown. This first observational study in a human model has identified sequential production of key cytokines and growth factors in the knee in a consecutive series of controlled patients undergoing a standard surgical reconstruction procedure. Such analysis has been shown to be of value in the evaluation of acute knee pain [14] and acute ACL ruptures [15] and may lead to a better understanding of the complex interactions involved in successful graft incorporation.
Conclusion
This work has found rapid local increase in the concentrations of IL-1, IL-6 but not IL-2. This suggests a non-specific monocyte-driven early response. However, the novel finding of rapid intra-articular elution of key growth factors within 6 h of iatrogenic injury, in particular TGF-β and PDGF, would argue for a more controlled and directed early anabolic response. More focused work on these key growth factors may help us unravel the signalling pathways which underlie the normal homeostatic response following surgical reconstruction at the knee.
References
Assoian RK, Sporn MB (1986) Type beta transforming growth factor in human platelets: release during platelet degranulation and action on vascular smooth muscle cells. J Cell Biol 102:1217–1223
Bennett NT, Schultz GS (1993) Growth factors and wound healing: part II. role in normal and chronic wound healing. Am J Surg 166:74–81
Bennett NT, Schultz GS (1993) Growth factors and wound healing: biochemical properties of growth factors and their receptors. Am J Surg 165:728–737
Beynnon BD, Fleming BC, Labovitch R, Parsons B (2002) Chronic anterior cruciate ligament deficiency is associated with increased anterior translation of the tibia during the transition from non-weightbearing to weightbearing. J Orthop Res 20:332–337
Beynnon BD, Johnson RJ, Fleming BC, Kannus P, Kaplan M, Samani J, Renstrom P (2002) Anterior cruciate ligament replacement: comparison of bone-patellar tendon-bone grafts with two-strand hamstring grafts. A prospective, randomized study. J Bone Joint Surg [Am] 84:1503–1513
Blickenstaff KR, Grana WA, Egle D (1997) Analysis of a semitendinosus autograft in a rabbit model. Am J Sports Med 25:554–559
Border WA, Ruoslahti E (1992) Transforming growth factor-beta in disease: the dark side of tissue repair. J Clin Invest 90:1–7
Fitzgerald KA, O’Neill AJ, Gearing AJH, Callard RF (2001) The cytokine factsbook. 2nd Edn, Academic Press, London Chapter 2, pp 12–19
Cameron ML, Fu FH, Paessler HH, Schneider M, Evans CH (1994) Synovial fluid cytokine concentrations as possible prognostic indicators in the ACL-deficient knee. Knee Surg Sports Traumatol Arthrosc 2:38–44
Cameron M, Buchgraber A, Paessler H, Vogt M, Thonar E, Fu F, Evans C (1997) The natural history of the anterior cruciate ligament-deficient knee. changes in synovial fluid cytokine and keratan sulfate concentrations. Am J Sports Med 25:751–754
Cawston TE, Wilson AJ (2006) Understanding the role of tissue degrading enzymes and their inhibitors in development and disease. Best Pract Res Clin Rheumatol 20:983–1002
Chu CR, Coyle CH, Chu CT, Szczodry M, Seshadri V, Karpie JC, Cieslak KM, Pringle EK (2010) In vivo effects of single intra-articular injection of 0.5% bupivacaine on articular cartilage. J Bone Joint Surg [Am] 92:599–608
Cohen S, Carpenter G (1975) Human epidermal growth factor: isolation and chemical and biological properties. Proc Natl Acad Sci 72:1317–1321
Cuellar JM, Scuderi GJ, Cuellar VG, Golish SR, Yeomans DC (2009) Diagnostic utility of cytokine biomarkers in the evaluation of acute knee pain. J Bone Joint Surg [Am] 91:2313–2320
Cuellar JM, Golish SR, Yeomans DC, Scuderi GJ (2010) Cytokine profiling in acute anterior cruciate ligament injury. Arthroscopy 26:1296–1301
Deehan DJ, Cawston TE (2005) The biology of integration of the anterior cruciate ligament. J Bone Joint Surg [Br] 87:889–895
Demirag B, Sarisozen B, Ozer O, Kaplan T, Ozturk C (2005) Enhancement of tendon-bone healing of anterior cruciate ligament grafts by blockage of matrix metalloproteinases. J Bone Joint Surg [Am] 87:2401–2410
Dinarello CA (1994) The biological properties of interleukin-1. Eur Cytokine Netw 5:517–531
Duthon VB, Barea C, Abrassart S, Fasel JH, Fritschy D, Menetrey J (2006) Anatomy of the anterior cruciate ligament. Knee Surg Sports Traumatol Arthrosc 14:204–213
Fibbe WE, Schaafsma MR, Falkenburg JH, Willemze R (1989) The biological activities of interleukin-1. Blut 59:147–156
Gearing AJ, Beckett P, Christodoulou M et al (1994) Processing of tumour necrosis factor-alpha precursor by metalloproteinases. Nature 370:555–557
Gianotti SM, Marshall SW, Hume PA, Bunt L (2009) Incidence of anterior cruciate ligament injury and other knee ligament injuries: a national population-based study. J Sci Med Sport 12:622–627
Grana WA, Egle DM, Mahnken R, Goodhart CW (1994) An analysis of autograft fixation after anterior cruciate ligament reconstruction in a rabbit model. Am J Sports Med 22:344–351
Griffin LY, Agel J, Albohm MJ et al (2000) Noncontact anterior cruciate ligament injuries: risk factors and prevention strategies. J Am Acad Orthop Surg 8:141–150
Higuchi H, Shirakura K, Kimura M, Terauchi M, Shinozaki T, Watanabe H, Takagishi K (2006) Changes in biochemical parameters after anterior cruciate ligament injury. Int Orthop 30:43–47
Irie K, Uchiyama E, Iwaso H (2003) Intraarticular inflammatory cytokines in acute anterior cruciate ligament injured knee. Knee 10:93–96
Jones SA, Horiuchi S, Topley N, Yamamoto N, Fuller GM (2001) The soluble interleukin 6 receptor: mechanisms of production and implications in disease. FASEB J 15:43–58
Kapoor B, Clement DJ, Kirkley A, Maffulli N (2004) Current practice in the management of anterior cruciate ligament injuries in the United Kingdom. Br J Sports Med 38:542–544
Marks PH, Donaldson MLC (2005) Inflammatory cytokine profiles associated with chondral damage in the anterior cruciate Ligament–Deficient knee. Arthrosc J Arthrosc Relat Surg 21:1342–1347
McFarland EG (1993) The biology of anterior cruciate ligament reconstructions. Orthopedics 16:403–410
McGeehan GM, Becherer JD, Bast RC Jr et al (1994) Regulation of tumour necrosis factor-alpha processing by a metalloproteinase inhibitor. Nature 370:558–561
Menetrey J, Duthon VB, Laumonier T, Fritschy D (2008) “Biological failure” of the anterior cruciate ligament graft. Knee Surg Sports Traumatol Arthrosc 16:224–231
Neufeld G, Cohen T, Gengrinovitch S, Poltorak Z (1999) Vascular endothelial growth factor (VEGF) and its receptors. FASEB J 13:9–22
Nugent MA, Iozzo RV (2000) Fibroblast growth factor-2. Int J Biochem Cell Biol 32:115–120
Rodeo SA, Arnoczky SP, Torzilli PA, Hidaka C, Warren RF (1993) Tendon-healing in a bone tunnel. A biomechanical and histological study in the dog. J Bone Joint Surg [Am] 75:1795–1803
Davies PJ, Martin SJ, Burton DR, Roitt IM (2006) Chapter 5, The primary interaction with antigen, in Essential Immunology, Eleventh Edition, Wiley-Blackman, London, pp 86–110
Senthilkumaran S, Tate R, Read J, Sutherland AG (2010) The use of intra-articular morphine in addition to local anaesthetic for post-operative analgesia in anterior cruciate ligament reconstruction: a prospective randomised controlled trial. Knee Surg Sports Traumatol Arthrosc 18:731–735
Steed DL (1998) Modifying the wound healing response with exogenous growth factors. Clin Plast Surg 25:397–405
Okada H, Banchereau J, Lotze MT (2003). Interleukin-4. In: Thomson AW, Lotze MT (eds) The cytokine handbook, 4th edn. Academic Press, London, pp 227–264
Tohyama H, Yasuda K (2005) Anterior cruciate ligament (ACL) healing: ACL graft biology. Sports Med Arthrosc Rev 13:156–160
Tyler TF, McHugh MP, Gleim GW, Nicholas SJ (1999) Association of KT-1000 measurements with clinical tests of knee stability 1 year following anterior cruciate ligament reconstruction. J Orthop Sports Phys Ther 29:540–545
Werner S, Grose R (2003) Regulation of wound healing by growth factors and cytokines. Physiol Rev 83:835–870
Woo SL, Abramowitch SD, Kilger R, Liang R (2006) Biomechanics of knee ligaments: Injury, healing, and repair. J Biomech 39:1–20
Yoshiya S, Nagano M, Kurosaka M, Muratsu H, Mizuno K (2000) Graft healing in the bone tunnel in anterior cruciate ligament reconstruction. Clin Orthop Relat Res 376:278–286
Zysk SP, Fraunberger P, Veihelmann A et al (2004) Tunnel enlargement and changes in synovial fluid cytokine profile following anterior cruciate ligament reconstruction with patellar tendon and hamstring tendon autografts. Knee Surg Sports Traumatol Arthrosc 12:98–103
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hayward, A.L., Deehan, D.J., Aspden, R.M. et al. Analysis of sequential cytokine release after ACL reconstruction. Knee Surg Sports Traumatol Arthrosc 19, 1709–1715 (2011). https://doi.org/10.1007/s00167-011-1486-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-011-1486-0