Abstract
The dissipation and residues of bromothalonil in apple and soil under field condition were analyzed by QuEChERS (quick, easy, cheap, effective, rugged and safe) combined with gas chromatography–mass spectrometer method. The recoveries were ranged from 80.8 % to 106 % with coefficient variation for repeatability ranged from 3.08 % to 7.09 % at fortification levels of 0.02, 0.04 and 0.2 mg/kg in apple and soil. The limit of quantification of the method was 0.31 μg/kg. The dissipation rates of bromothalonil followed the first-order kinetics and the half-lives were from 3.61 to 3.98 days in apple and from 4.65 to 9.29 days in soil. In apple, the terminal residues of bromothalonil were below the China maximum residue limit (0.2 mg/kg) after 7 days of application. This work contributed to provide the basic information for a safe usage of bromothalonil in apple orchard and preventing health problem from consumers in China.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
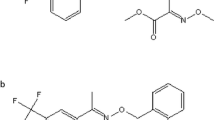
Apple is one of the most important fruits in China. The annual production of apple in 2011 in China is more than 35,000 thousands tons which exceeds 50 % of the world’s total output. Apple anthracnose is a common destructive disease of apples which can infect and destroy the fruits and stems of apple (VON Schrenk and Spaulding 1902). Bromothalonil (2-bromo-2-(bromomethyl) pentanedinitrile (Fig. 1), CAS No. (35691-65-7) is widely used in apple to control anthracnose, citrus anthracnose in China and the MRL for bromothalonil in apple is 0.2 mg/kg in China (PRC 2012). The ADI is 0.001 mg/kg bw.
Liquid chromatography coupled with electrochemical detector (Weyland et al. 1994), ultra performance liquid chromatography (UPLC) coupled to inductively coupled plasma mass spectrometry (ICP-MS) (Bendahl et al. 2006) and GC–MS (Pellegrini et al. 2011) were used to analysis bromothalonil in cosmetic products. Quick, Easy, Cheap, Effective, Rugged and Safe (QuEChERS) is a method first introduced by Anastassiades et al. (2003) for the monitoring of pesticide multi-residues in fruits and vegetables. This method is particularly popular for pesticide residues determination in various kind of food matrices dueing to its low time-consuming and reduced glassware and organic solvent usage (Abd-Alrahman and Almaz 2012; Urvashi et al. 2012). Now the QuEChERS method has been the Official Method of AOAC International (Lehotay 2007). In this present study, we set up and validated a QuEChERS combined with GC–MS/MS method for analyzing and quantifying bromothalonil in apple and soil matrices. The residue levels and degradation dynamics of bromothalonil apple and soil after application were also determined.
Materials and Methods
Twenty-five percentage Bromothalonil wettable power was provided by Beijing Organic Agriculture Technology Development Center. Bromothalonil standard material (≥97.1 % purity) was purchased from Beijing Helishun Technology Co., Ltd. Ethyl acetate was HPLC grade (Dikma Technologies Inc.) and sodium chloride was analytic grade (Beijing Reagent Company). Water was distilled and primary secondary amine (PSA) was bought from Agela Technologies Inc. The stock standard solution of 100 mg/L bromothalonil was prepared in ethyl acetate. Working standard solutions were prepared by serial dilution with ethyl acetate and all standards solutions were stored in amber bottles (50 mL) at 4°C.
Field Trials
The field trials including the dissipation and final residue study were conducted in apple orchards in Anhui, Beijing and Shanxi in China in 2012. Every treatment was repeated three times and each experiment plot was two apple trees. A 15-m distance buffer area was retained for separating each plot.
Bromothalonil 25 % wettable powder (wp) was sprayed one time on apple and soil with the dosage of 750 mg/kg. The control plot sprayed water simultaneously. Sample 1 kg was collected randomly from each plot at 0, 1, 3, 5, 7, 10, 14, 21 and 30 days after application. All samples were putted into plastic bags and transported to laboratory, where the soil samples were sieved and the apple samples were chopped. Each individual sample was mixed thoroughly then placed 250 g into polyethylene bags, labeled and stored frozen at −20°C until analysis.
Bromothalonil 25 % wp was sprayed three or four times with the dosage of 500 or 750 mg/kg. The interval of each application was 7 days. Spray water on individual plot as control. Sample 1 kg was collected randomly from each plot at 7, 14, 21 and 28 days after the last treatment. The collection, transportation, processing and storage were as the same as the degradation dynamic experiment.
Extraction Procedure
10.0 g previously homogenized apple or soil samples were weighed in 50 mL centrifuge tube. Then 5 mL distilled water and 10 mL ethyl acetate were added into the tube following ultrasonic extraction for 30 min. Add 5 mL NaCl saturation solution into the sample tube and vortex for 1 min. Afterwards, the extract was centrifuged at 3,000 rpm for 5 min. Transfer 1.5 mL aliquot of upper ethyl acetate layer to a 2-mL centrifuge tube containing 30 mg sorbent PSA and 500 mg MgSO4. Shake the tube vigorously for 1 min then stand for 5 min. The supernatant was filtrated through a 0.45 μm nylon filter thereafter analyzed by Thermo scientific GC–MS/MS.
Apparatus and Chromatography
The determination of bromothalonil was done by Thermo scientific TSQ 8000 triple quadrupole gas chromatography–mass spectrometer (GC–MS/MS) equipped with silica capillary RTX-35 MS column (30 m × 0.25 mm × 0.25 μm). Helium (purity ≥99.99 %) was used as carrier gas with a flow rate of 1 mL/min. The injector temperature was 250°C and the oven temperature was programmed as follows: 100°C increased to 180°C at 15°C/min (hold 1 min), raised to 280°C at 45°C/min and held 5 min. The injection volume was 1 μL in split less mode. The samples were ionized using positive electron ionization mode. Argon (purity ≥99.99 %) was used as collision gas with a flow rate of 50 mL/min. Bromothalonil was identified and quantified in selected reaction monitoring mode at m/z 106/66 and 106/39 under the following conditions: interface temperatures at 280°C, ion source at 230°C, and collision energy 10 eV, respectively.
Results and Discussion
Calibration curve was calculated by plotting peak area versus concentration (mg/L) of five gradient standard solutions. A good linearity (y = 2.97 × 106x + 10,837) was achieved between 0.01 and 1.00 mg/L with a correlation coefficient of 1.0000. The instrument limits of detection (LOD) and quantitation (LOQ) were calculated as 3 and 10 times the signal-to-noise ratio (S/N) which were 0.09 and 0.31 μg/kg. Recoveries of bromothalonil at 0.02, 0.04 and 0.2 mg/kg in apple and soil were determined in five replicates to evaluate the accuracy and precision of the method. The obtained recoveries are given in Table 1 which range from 80.8 % to 106.0 %. The coefficient variation of the methods (CV %) for repeatability ranged from 3.08 % to 7.09 %. Both the recoveries and coefficient variation were within acceptable range (EC 2009).
The dissipation curves of bromothalonil in apple and soil under natural conditions were shown in Fig. 2. The initial deposits of bromothalonil were 0.412, 0.372 and 0.229 mg/kg in apples and 0.436, 0.315 and 0.243 mg/kg in soils in Anhui, Beijing and Shanxi respectively. Determine the degradation kinetics of bromothalonil in apple and soil by plotting residue concentration against time after treatment. The decrease of bromothalonil in apple and soil followed the first order kinetics pattern. The half-life values of bromothalonil calculated as Hoskins (1966) in apple were 3.61, 3.98 and 3.84 days and 4.65, 9.29 and 8.80 days in soil respectively.
Bromothalonil contained two bromine moieties (Fig. 1) which were extremely labile and readily eliminated. It was readily debrominated and degraded into 2-methyleneglutaronitrile (Bao et al. 1998; Sauer et al. 1998). Many factors such as the local soil characteristics and climate might affect the dissipation of bromothalonil (Brouwer et al. 1997; Katagi 2004). The dissipation of bromothalonil in apple was faster than in soil under field conditions. The growth dilution factor might have played a significant role (Tewary et al. 2005). Comparison of the degradation rates revealed that bromothalonil degraded most fastly in Anhui and slowly in Beijing. It might due to the climate of Anhui is rainy and the average temperature of Beijing is lower than the other two.
The final residues of bromothalonil in apple and soil under field condition are shown in Table 2. When bromothalonil (25 % wp) was applied three or four times at low dosage (500 mg/kg) in apple, the residues of bromothalonil in apple and soil were <0.02–0.071 and <0.02 mg/kg. When it was applied three or four times at high dosage (750 mg/kg), the residues of bromothalonil in apple and soil were 0.043–0.169 and <0.02–0.044 mg/kg respectively. The above findings indicated that higher dosage and rate of application of bromothalonil results in higher residues and the residues declined with time.
The final residues of bromothalonil in apple were 0.025–0.169 mg/kg at PHI (Pre-Harvest Interval) of 7 days and <0.02–0.124 mg/kg at PHI of 14 days under field condition. The MRL of bromothalonil in apple established by the Peoples’ Republic of China was 0.2 mg/kg. It could be recommended on the basis of the above results that 25 % bromothalonil wettable powder could be applied on apple at dosage of 500–700 mg/kg, 3–4 times and PHI of 7 day. This work established the safe and proper use of bromothalonil on apple in China.
In this study, ethyl acetate was selected as the extraction solvent, PSA was used as sorbent for removal of matrices and the samples were detected by GC–MS/MS and no impurity interfered. Extraction and purification used limited organic solvent and avoided time-consuming. GC–MS/MS used to analysis was more sensitive than LC-ED (Weyland et al. 1994), UPLC-ICP-MS (Bendahl et al. 2006) and GC–MS (Pellegrini et al. 2011) which had lower LOD and LOQ. The method of QuEChERS extraction combined with GC–MS/MS analysis to determine bromothalonil residues in apple and soil achieved good validation results. The results indicated that the proposed method of QuEChERS extraction coupled with GC–MS/MS analysis was simple, quick, cheap, and reliable for the determination of bromothalonil in apple and soil.
References
Abd-Alrahman SH, Almaz MM (2012) Degradation of propamocarb-hydrochloride in tomatoes, potatoes and cucumber using HPLC-DAD and QuEChERS methodology. Bull Environ Contam Toxicol 89:302–305
Anastassiades M, Lehotay SJ, Stajnbaher D, Schenck FJ (2003) Fast and easy multiresidue method employing acetonitrile extraction/partitioning and “dispersive solid phase extraction” for the determination of pesticides resi-dues in produce. J AOAC Int 86:412–431
Bao J, Sauer JM, Smith RL, Kuester RK, Kattnig MJ, Sipes IG (1998) Sulfhydryl-dependent biotransformation and macromolecular binding of 1,2-dibromo-2,4-dicyanobutane in blood. Drug Metab Dispos 26:1001–1007
Bendahl L, Hansen SH, Gammelgaard B, Sturup S, Nielsen C (2006) Hyphenation of ultra- performance liquid chromatography (UPLC) with inductively coupled plasma mass spectrometry (ICP-MS) for fast analysis of bromine containing preservatives. J Pharm Biomed Anal 40:648–652
Brouwer DH, de Haan M, Leenheers LH, de Vreede SA, van Hemmen JJ (1997) Half-lives of pesticides on greenhouse crops. Bull Environ Contam Toxicol 58:976–984
EC (European Commission) (2009) Method validation and quality control procedures for pesticide residues analysis in food and feed. SANCO/10684/2009 (01 Jan, 2010)
Hoskins WM (1966) Mathematical treatment of the rate of loss of pesticide residues. FAO Plant Protect Bull 9:163–168
Katagi T (2004) Photodegradation of pesticides on plant and soil surfaces. Rev Environ Contam Toxicol 182:1–189
Lehotay SJ (2007) Determination of pesticide residues in foods by acetonitrile extraction and partitioning with magnesium sulfate: collaborative study. J AOAC Int 90:485–520
Pellegrini M, Bossù E, Rotolo MC, Pacifici R, Pichini S (2011) Simple and rapid analysis of methyldibromo glutaronitrile in cosmetic products by gas chromatography mass spectrometry. J Pharm Biomed Anal 56:1112–1116
PRC (People’s Republic of China) (2012) GB 2763-2012. National food safety standard maximum residue limits for pesticides in food. Beijing, p 115
Sauer JM, Smith RL, Bao J, Kattnig MJ, Kuester RK, Sipes IG (1998) Metabolic and dispositional fate of 1, 2-dibromo-2, 4-dicyanobutane in the male fischer 344 rat. Drug Metab Dispos 26:1001–1007
Tewary DK, Vipin K, Ravindranath SD, Adarsh S (2005) Dissipation behavior of bifenthrin residues in tea and its brew. Food Control 16:231–237
Urvashi Jyot G, Sahoo SK, Kaur S, Battu RS, Singh B (2012) Estimation of indoxacarb residues by QuEChERS technique and its degradation pattern in cabbage. Bull Environ Contam Toxicol 88:372–376
VON Schrenk H, Spaulding P (1902) The bitter rot disease of apples. Science 408:669–670
Weyland JW, Stern A, Rooselaar J (1994) Determination of bronopol, bronidox, and methyldibromo glutaronitrile in cosmetics by liquid chromatography with electrochemical detection. J AOAC Int 77:1132–1136
Acknowledgments
This research work was supported by the National High Technology Research and Development Program (863) of China (No. 2012AA06A302) and National Natural Science Foundation of China (21277163).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, H., Guo, B., Wang, H. et al. Determination of Bromothalonil Residues and Degradation in Apple and Soil by QuEChERS and GC–MS/MS. Bull Environ Contam Toxicol 92, 451–454 (2014). https://doi.org/10.1007/s00128-014-1224-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-014-1224-7