Abstract
Despite representing the majority of bee species, non-eusocial bees (e.g. solitary, subsocial, semisocial, and quasisocial species) are comparatively understudied in learning, memory, and cognitive-like behaviour compared to eusocial bees, such as honeybees and bumblebees. Ecologically relevant colour discrimination tasks are well-studied in eusocial bees, and research has shown that a few non-eusocial bee species are also capable of colour learning and long-term memory retention. Australia hosts over 2000 native bee species, most of which are non-eusocial, yet evidence of cognitive-like behaviour and learning abilities under controlled testing conditions is lacking. In the current study, I examine the learning ability of a non-eusocial Australian bee, Lasioglossum (Chilalictus) lanarium, using aversive differential conditioning during a colour discrimination task. L. lanarium learnt to discriminate between salient blue- and yellow-coloured stimuli following training with simulated predation events. This study acts as a bridge between cognitive studies on eusocial and non-social bees and introduces a framework for testing non-eusocial wild bees on elemental visual learning tasks using aversive conditioning. Non-eusocial bee species are far more numerous than eusocial species and contribute to agriculture, economics, and ecosystem services in Australia and across the globe. Thus, it is important to study their capacity to learn flower traits allowing for successful foraging and pollination events, thereby permitting us a better understanding of their role in plant-pollinator interactions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A large amount of research has focused on the learning abilities of eusocial bees. Honeybees (Apis mellifera) and bumblebees (Bombus terrestris) are among the most-studied bee species in terms of learning, memory, and cognition. For example, honeybees demonstrate evidence of both elemental (simple) and non-elemental (complex/relational) learning. Honeybees can discriminate between colours (Dyer and Arikawa 2014; Dyer and Neumeyer 2005; Dyer et al. 2008) and patterns (Dyer and Griffiths 2012; Efler and Ronacher 2000; Giurfa et al. 1999; Horridge 1997; Zhang and Srinivasan 1994), tasks which are generally considered elemental learning facilitated by associative mechanisms. Honeybees can perform more complex tasks including learning relational rules such as above vs. below (Avarguès-Weber et al. 2011), same vs. different (Giurfa et al. 2001), greater vs. lesser (Bortot et al. 2019a; Howard et al. 2018a), and smaller vs. larger (Avarguès-Weber et al. 2014; Howard et al. 2017a, b). They also demonstrate the acquisition of complex concepts such as maze navigation (Collett et al. 1993; Zhang et al. 1996, 2000), facial recognition (Avargues-Weber et al. 2018; Avarguès-Weber et al. 2010b; Chittka and Dyer 2012; Dyer et al. 2005), abstract character use (Howard et al. 2019d; Zhang et al. 1999), counting (Chittka and Geiger 1995; Dacke and Srinivasan 2008), arithmetic (Howard et al. 2019a, b), and quantity discrimination (Howard et al. 2018a, 2019c, 2020).
While learning and memory tasks are comparatively understudied in non-eusocial bees, previous work shows evidence of learning abilities in a number of solitary bee species. Osmia lignaria (Amaya-Marquez and Wells 2008), Xylocopa virginica (Dukas and Real 1991), Heriades truncorum (Chittka et al. 1992), and Osmia rufa (Menzel et al. 1988) can learn important colour discrimination tasks. Eucera sp. appears to learn an ecologically relevant counting task (Bar-Shai et al. 2011), where flower nectaries are counted as bees feed to avoid return visits to empty nectaries. Osmia caerulescens and Osmia leaiana (Loukola et al. 2020) learn to avoid or visit nests based on abstract characters representing parasitized or non-parasitised nesting sites. Xylocopa micans demonstrates learning of flower colour, showing more visits to rewarding flowers (higher in nectar concentration or volume) in less than six trials (Perez and Waddington 1996). X. micans also demonstrates the use of colour and location learning when foraging on vertical inflorescences of three artificial flowers, where just one contained nectar. Although they preferred to use colour cues, in the absence of colour information, X. micans could use spatial information (Orth and Waddington 1997). Colour and spatial information of rewarding flowers were learnt within five foraging bouts by X. micans, suggesting rapid visual and spatial learning (Orth and Waddington 1997; Somanathan et al. 2019). Additionally, other ecologically relevant learning tasks have been demonstrated in the nocturnal Xylocopa tranquebarica which can visually learn coloured landmarks for nest recognition (Somanathan et al. 2008).
Bees are a model species for testing learning, cognition, and memory in invertebrates (Avarguès-Weber and Giurfa 2013; Chittka 2017; Dyer 2012; Srinivasan 2010). Research has shown that the method of conditioning impacts the learning ability of bees. The major types of differential conditioning, where two or more stimuli are presented to the bee simultaneously, are appetitive differential conditioning, aversive differential conditioning, and appetitive-aversive differential conditioning. Appetitive differential conditioning involves rewarding a correct choice of stimulus while providing no outcome for an incorrect choice. Aversive conditioning involves a punishment for an incorrect stimulus choice and no outcome for a correct choice. Finally, appetitive-aversive differential conditioning involves providing a reward for a correct choice and a punishment for an incorrect choice. Using appetitive-aversive differential conditioning is known to improve learning in bees during visually and conceptually difficult tasks such as fine colour discrimination (Avarguès-Weber et al. 2010a; Chittka et al. 2003) and quantity discrimination (Howard et al. 2019c). While appetitive-aversive and appetitive differential conditioning can be used in free-flying bee experiments [for examples, see Avarguès-Weber et al. (2014); Avarguès-Weber et al. (2015); Avarguès-Weber et al. 2011; Bortot et al. (2019a, b); Chittka and Geiger (1995); Dyer and Chittka (2004); Dyer et al. (2005); Garcia et al. (2018); Giurfa et al. (2001); Howard et al. (2019c, d); Perry and Barron (2013)], aversive learning is used in experiments where bees are constrained in harnesses or closed arenas (Marchal, 2019; Nouvian and Galizia 2019; Vergoz et al. 2007). The type of aversive outcome/punishment can include mild electric shocks (Marchal et al. 2019; Nouvian and Galizia 2019; Vergoz et al. 2007) or simulated predation events (Jones and Dornhaus 2011; Zhang and Nieh 2015). In the current study, I employ the latter. Past work has shown that simulated crab-spider predation events (Fig. 1a) are successful as an aversive outcome in learning tasks related to flower choice and colour discrimination in eusocial bee species (Ings et al. 2012; Ings and Chittka 2008, 2009; Jones and Dornhaus 2011; Wang et al. 2013; Zhang and Nieh 2015).
In the current study, I tested a widespread Australian native bee, Lasioglossum (Chilalictus) lanarium, for its ability to learn a simple colour discrimination task. I presented bees with two colours, known to be easily discriminated by honeybee vision in a previous study (Howard et al. 2019b). Bees were trained using aversive differential conditioning, with a simulated predation event acting as the aversive outcome to an incorrect choice. The current study aims to aid in bridging the gap between learning and cognitive studies on eusocial and non-eusocial bees and demonstrates how non-eusocial bees may learn important plant-pollinator signals such as flower colour to find rewarding flowers and avoid predators.
Methods
Study species and collection
The native Australian bee, L. lanarium (family Halictidae), was used in this study. Halictids are short-tongued bees with most species being ground-nesting and a few burrowing into rotten branches, logs, or stumps (Houston 2018). Lasioglossum bees are found across Australia (Danforth and Ji 2001). Specifically, L. lanarium is a widespread (Atlas of Living Australia website), ground-nesting bee species which forages on multiple flowering plant species. L. lanarium nest in communal aggregations of females (Houston 2018).
Male and female L. lanarium were collected in southeast Melbourne, Australia (near the latitudes and longitudes of − 37.858, 145.0952 and − 37.871, 145.187 in residential gardens) on the days of experiments during November 2020. The sex of bees was not determined for all individuals; thus these data were not included in the analysis. Bees were captured in small transparent plastic vials with air holes, transported to a testing arena in a dark bag, and behavioural assays were conducted within 24 h of capture during daylight hours.
The transport of the bees to the experimental area while in the vial in a dark opaque bag appeared to prevent escape behaviour enough so that when released from the vial, they did not exhibit an escape response but still moved around the arena with motivation to approach the stimuli (Online Resource 1). Different individuals exhibited different levels of activity and escape behaviour following capture but did not try to escape at the beginning of the experiments.
Apparatus
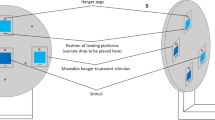
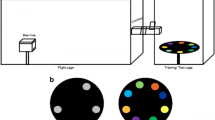
The circular arena was constructed of white plastic 16 cm in diameter and 4.5 cm in height (Fig. 1b). The bee was released at one edge of the arena when presented with two stimuli at the other edge. Bees were individually tested. Experiments were conducted under natural diffuse daylight conditions.
Stimuli
Salient blue and yellow circles on a background of neutral grey were used as the stimuli (Fig. 1b, c). Stimuli were 6 × 6 cm (36 cm2) laminated cards presenting the coloured circle of 10 cm2 in surface area. The colours are easily discriminable by bees, which is demonstrated in a previous study on honeybees (Howard et al. 2019b) and based on current knowledge of bee colour vision (Briscoe and Chittka 2001; Chittka 1992; Dyer and Arikawa 2014). The reflectance spectrum of each colour (grey background and target colours) was measured with a spectrophotometer fitted with quartz optics and a PX-2 pulsed xenon UV–visible radiation source (USB 2000 + , Ocean Optics, Dunedin, FL, USA) that closely matches the spectral profile of typical daylight illumination. The spectrophotometer was attached to a computer running SpectraSuite software 2011. The results of these measurements are plotted in Fig. 1c.
Training and testing procedure
Individual bees were placed 13 cm away from the two stimuli presented. Bees were transported in and out of the arena and manipulated around the experimental area on a toothpick. Once bees were placed into the arena for the first time, a choice was recorded for either the yellow or blue stimulus. This first choice acted as preference test or ‘pre-test’; bees would then be trained on the opposite colour to what they had first chosen. For example, if a bee preferred blue on the first choice, they would then be trained to avoid the blue stimulus, similar to previous work with honeybees (Buatois et al. 2018). During the preference test, n = 21 bees chose the yellow stimulus and thus were trained to avoid the yellow stimulus, and n = 19 bees chose the blue stimulus and thus were trained to avoid the blue stimulus. The pre-test was necessary as recent evidence has shown L. lanarium bees have colour preferences (Howard et al. 2021). To demonstrate the motivation of bees and the attraction to stimuli, a video of a pre-test is included in Online Resource 1.
Bees underwent ten aversive training trials. Then ten unreinforced test choices (no outcomes for a correct or incorrect choice) were conducted to determine whether they had learnt to avoid the incorrect stimulus, similar to unreinforced tests in other bee behaviour studies (Howard et al. 2019b, c, d; Howard et al. 2020, 2021). During aversive differential conditioning, bees were placed at the starting point and then walked towards a stimulus and made a choice (usually within 1 min of being placed into the arena). If bees chose incorrectly, they would receive an aversive outcome — a simulated predation event. If bees chose correctly, there was no outcome.
The simulated predation event involved gently squeezing the bee’s abdomen with forceps when it climbed onto the incorrect stimulus. This procedure simulates the attack of a crab spider, a predator of L. lanarium (personal observation; Fig. 1a). This method or similar has been successfully used previously with forceps (Jones and Dornhaus 2011; Zhang and Nieh 2015) and robotic crab spiders (Ings et al. 2012; Ings and Chittka 2008, 2009; Wang et al. 2013). Bees were provided breaks of 10–30 s between trials.
If bees exhibited escape behaviour following the aversive outcome for an incorrect choice, polyethylene film was placed over the arena which prevented escape but allowed both visible light and UV light to pass through. The cover still allowed the aversive outcome to be applied due to the plastic film’s flexibility.
Statistical analysis
To determine if bees demonstrated significant learning during training, data from the ten aversive differential conditioning trials were analysed with a generalized linear mixed-effects model (GLMM) with a binomial distribution using the ‘glmer’ package within the R environment for statistical analysis. I first fitted a full model with choice as the categorial response variable with two levels (correct; incorrect), individual trial number as a continuous predictor (1–10), colour as a categorical predictor with two levels (yellow; blue), and an interaction between trial and colour as a predictor. Subject (bee ID) was included as a random factor to account for repeated choices of individual bees.
To determine whether bees learnt to discriminate between yellow vs. blue stimuli, I analysed the test data by employing a GLMM with a binomial distribution including categorial response variable with two levels (correct; incorrect), individual test choice number as a continuous predictor (1–10), colour as a categorical predictor with two levels (yellow; blue), and an interaction between trial and colour as a predictor. Subject was included as a random factor to account for repeated choices of individual bees. The proportion of choices for the correct colour (MPCC) recorded from the tests was used as the response variable in the model. The Wald statistic (z) tested if the mean proportion of correct choices recorded from the test, represented by the coefficient of the intercept term, was significantly different from chance expectation, i.e. H0: MPCC = 0.5.
The models were estimated using the routine ‘glmer’ available as part of the ‘lme4’ package written for the R statistical language, run in R version 4.0.3 (Bates et al. 2014; R Core Team 2020).
Results
Training
The analysis from the full model including trial (continuous), colour (categorical with two levels: blue; yellow), and an interaction between the two predictors showed no effect of the interaction (z = − 0.846, P = 0.398); thus the interaction term was removed. The subsequent reduced model showed no effect of colour (z = 1.744, P = 0.0812), but an effect of trial (z = 3.277, P = 0.001). The simplified model excluding colour showed a significant effect of trial on bee performance (z = 3.265; P = 0.001). See Fig. 2a.
Results of the training and testing phases. Left panel (a) shows the performance of bees during the learning phase. The solid black line represents a function describing the learning phase of n = 40 bees as modelled by a generalized linear mixed-effect model (GLMM). Shaded area indicates the ± 95% confidence intervals (CIs) of correct choices for the bees (turquoise). The increase in learning performance was significant. Dashed line at 0.5 indicates chance level performance. Black circles show the mean performance of all bees (n = 40) for each trial. Right panel (b) shows the mean proportion of correct choices [mean ± 95% confidence intervals (CIs)] by L. lanarium during all ten choices during the unreinforced test. Broken black line shows chance expectation at 0.5. Blue circles indicate the raw data from each individual bee (n = 40) as a bee-swarm plot. Significance from chance level performance is indicated by *** ≥ 0.001
As the P value for the effect of colour was < 0.100 but > 0.050, I compared the AIC values of the model including and excluding colour (both models excluding the interaction effect). The model including colour had an AIC value of 543.700. The simplified model excluding the effect of colour had an AIC value of 544.700; thus the model of best fit included colour and showed that bees learnt the task over the ten aversive conditioning trials with an overdispersion (OV) value of 1.349 showing that the training data was slightly overdispersed but within a reasonable range to conduct the analysis.
Testing
Each bee underwent ten unreinforced choices during a learning test to determine if they had learnt the task of yellow vs. blue (Fig. 2b). There was no significant difference between bees trained to avoid the yellow or blue stimulus (z = 0.988; P = 0.323); therefore the data were pooled for analysis.
Bees discriminated between blue and yellow stimuli during the learning test following the ten training choices at a level of 62.75% (confidence intervals [CIs] = 0.557, 0.690; z = 5.043; P < 0.001; n = 40; Fig. 2b). The data collected was slightly overdispersed (OV = 1.32) but still within a reasonable range to conduct the analysis.
When examining only the first choice of bees during the test (thus without a subject as a random term as there were no repeated measures in the model), bees chose the correct stimulus at a level of 75.00% (CIs = 0.595, 0.860; z = 3.009; P < 0.003; n = 40), which is consistent with what was found when bees underwent all ten unreinforced choices in the test.
Discussion
The current study presents evidence that a wild non-eusocial bee can learn the elemental task of colour discrimination using aversive differential conditioning when colours are saliently different. Colour discrimination is a potentially useful capacity in L. lanarium’s natural environment. Bees are known to use different signals or cues from flowers to choose which flowers to visit. These signals and cues include scent (Raguso 2008), colour (Giurfa et al. 1995), shape (Howard et al. 2018b; Lehrer et al. 1995), size (Martin 2004), quantity of flowers (Howard et al. 2020), and/or symmetry (Giurfa et al. 1996); thus bees must learn which flowers to visit based on these traits. Colour signalling by flowers appears to be the main plant-pollinator communication channel in studies from around the world (Barth 1985; Mitchell et al. 2009; Sargent and Ackerly 2008), and so colour discrimination learning is an important behaviour to examine in understudied non-eusocial bees. The ability of L. lanarium to discriminate between two colours suggests that in a natural foraging situation, this species may learn to visit rewarding flower colours, avoid non-rewarding flower colours, or even learn to avoid flower colours which host crab-spiders (Fig. 1a), similar to findings in past studies (for example, Jones and Dornhaus 2011).
A few studies have directly compared the learning abilities of eusocial and solitary bees. These studies suggest that while there are differences between learning in eusocial and non-eusocial bees, fundamental aspects of learning are shared between the two groups. In a colour discrimination experiment, A. mellifera (eusocial) learnt more quickly than O. lignaria (solitary) in the case of discriminating between flower colour morphs (Amaya-Marquez and Wells 2008). The eusocial bumblebee, B. bimaculatus, and the solitary carpenter bee, X. virginica, were compared for learning of flower colours. While B. bimaculatus demonstrated higher rates of learning, both species had similar levels of overnight memory retention (Dukas and Real 1991). In the field, it appears that B. terrestris (eusocial) may have a more exact counting strategy for flower nectaries than Eucera sp. (solitary) (Bar-Shai et al. 2011). Thus, in these few comparisons, eusocial bees appear to learn more quickly (Amaya-Marquez and Wells 2008) or demonstrate higher rates of learning then non-eusocial solitary bees (Dukas and Real 1991), but more research is needed to determine whether this trend is widespread or restricted to a few species comparisons.
Non-eusocial wild bees are important pollinators in terms of agriculture, economics, and ecosystem services in Australia and across the globe (Bell et al. 2006; Bray 1973; Heard 1999; Hogendoorn et al. 2010, 2007, 2006, 2000; Hogendoorn and Keller 2012; Kleijn 2015); however they are threatened by a number of anthropogenic activities such as land-clearing, habitat loss and fragmentation, urbanisation, pesticide and insecticide use, climate change, disease, fire, and invasive species introductions (Batley and Hogendoorn 2009; Brown and Paxton 2009; Goulson et al. 2015; Herrera 2020; Mallinger et al. 2017; Owens et al. 2020; Potts et al. 2010; Santos et al. 2020). The biggest threat to Australian native bees remains a lack of information, knowledge, and expertise (Batley and Hogendoorn 2009; Sands 2018; Taylor et al. 2018). Thus, the current study and further research should work towards filling this gap in knowledge and developing robust frameworks to study non-eusocial wild bees. Future work should aim to determine effective ways to test learning and memory in non-eusocial bees using both aversive and appetitive conditioning and develop testing of more complex learning tasks for comparison with eusocial bees. As non-eusocial bees are important but comparatively understudied pollinators, efforts to examine their behaviour and foraging decisions are imperative.
Data availability
All data generated or analysed during this study are included in this published article [and its supplementary information files].
Code availability
Available upon request.
References
Amaya-Marquez M, Wells H (2008) Social complexity and learning foraging tasks in bees. Caldasia 30:469–477 Atlas of Living Australia website. Species page: https://bie.ala.org.au/species/urn:lsid:biodiversity.org.au:afd.taxon:8634ccfb-2dea-48d4-8853-6f91018440f2. Accessed 16th February 2021
Avarguès-Weber A, de Brito Sanchez MG, Giurfa M, Dyer AG (2010a) Aversive reinforcement improves visual discrimination learning in free-flying honeybees. PLoS ONE 5:e15370–e15370
Avarguès-Weber A, Portelli G, Benard J, Dyer A, Giurfa M (2010b) Configural processing enables discrimination and categorization of face-like stimuli in honeybees. J Exp Biol 213:593–601
Avarguès-Weber A, Dyer AG, Giurfa M (2011) Conceptualization of above and below relationships by an insect. Proc Biol Sci 278:898–905
Avarguès-Weber A, Giurfa M (2013) Conceptual learning by miniature brains. Proc Biol Sci 280:20131907
Avarguès-Weber A, d’Amaro D, Metzler M, Dyer AG (2014) Conceptualization of relative size by honeybees. Front Behav Neurosci 8:1–8
Avarguès-Weber A, Dyer AG, Ferrah N, Giurfa M (2015) The forest or the trees: preference for global over local image processing is reversed by prior experience in honeybees. Proc Biol Sci 282:20142384
Avargues-Weber A, d’Amaro D, Metzler M, Finke V, Baracchi D, Dyer AG (2018) Does holistic processing require a large brain? Insights from honeybees and wasps in fine visual recognition tasks. Front Psychol 9:1313
Bar-Shai N, Keasar T, Shmida A (2011) How do solitary bees forage in patches with a fixed number of food items? Anim Behav 82:1367–1372
Barth FG (1985) Insects and flowers. The biology of a partnership. Princeton University Press USA, Princeton
Bates D, Mächler M, Bolker B, Walker S (2014) Fitting linear mixed-effects models using lme4. arXiv preprint arXiv:14065823:1-51
Batley M, Hogendoorn K (2009) Diversity and conservation status of native Australian bees. Apidologie 40:347–354
Bell M, Spooner-Hart R, Haigh AM (2006) Pollination of greenhouse tomatoes by the Australian bluebanded bee Amegilla (Zonamegilla) holmesi (Hymenoptera: Apidae). J Econ Entomol 99:437–442
Bortot M, Agrillo C, Avarguès-Weber A, Bisazza A, Miletto Petrazzini ME, Giurfa M (2019a) Honeybees use absolute rather than relative numerosity in number discrimination. Biol Let 15:20190138
Bortot M, Stancher G, Vallortigara G (2019b) Transfer from number to size reveals abstract coding of magnitude in honeybees. IScience 23:101122
Bray R (1973) Characteristics of some bees of the family Megachilidae in Southeast Queensland and their potential as lucerne pollinators. Aust J Entomol 12:99–102
Briscoe AD, Chittka L (2001) The evolution of color vision in insects. Annu Rev Entomol 46:471–510
Brown MJ, Paxton RJ (2009) The conservation of bees: a global perspective. Apidologie 40:410–416
Buatois A, Flumian C, Schultheiss P, Avarguès-Weber A, Giurfa M (2018) Transfer of visual learning between a virtual and a real environment in honey bees: the role of active vision. Front Behav Neurosci 12:139
Chittka L (1992) The colour hexagon: a chromaticity diagram based on photoreceptor excitations as a generalized representation of colour opponency. J Comp Physiol A 170:533–543
Chittka L (2017) Bee cognition. Curr Biol 27:R1049–R1053
Chittka L, Geiger K (1995) Can honey bees count landmarks? Anim Behav 49:159–164
Chittka L, Dyer A (2012) Cognition: Your face looks familiar. Nature 481:154–155
Chittka L, Beier W, Hertel H, Steinmann E, Menzel R (1992) Opponent colour coding is a universal strategy to evaluate the photoreceptor inputs in Hymenoptera. J Comp Physiol A 170:545–563
Chittka L, Dyer AG, Bock F, Dornhaus A (2003) Psychophysics: bees trade off foraging speed for accuracy. Nature 424:388–388
Collett T, Fry S, Wehner R (1993) Sequence learning by honeybees. J Comp Physiol A 172:693–706
Dacke M, Srinivasan MV (2008) Evidence for counting in insects. Anim Cogn 11:683–689
Danforth BN, Ji S (2001) Australian Lasioglossum + Homalictus form a monophyletic group: resolving the “Australian enigma”. Syst Biol 50:268–283
Dukas R, Real LA (1991) Learning foraging tasks by bees: a comparison between social and solitary species. Anim Behav 42:269–276
Dyer AG (2012) The mysterious cognitive abilities of bees: why models of visual processing need to consider experience and individual differences in animal performance. J Exp Biol 215:387–395
Dyer AG, Chittka L (2004) Bumblebees (Bombus terrestris) sacrifice foraging speed to solve difficult colour discrimination tasks. J Comp Physiol A 190:759–763
Dyer AG, Neumeyer C (2005) Simultaneous and successive colour discrimination in the honeybee (Apis mellifera). J Comp Physiol A 191:547–557
Dyer AG, Griffiths DW (2012) Seeing near and seeing far; behavioural evidence for dual mechanisms of pattern vision in the honeybee (Apis mellifera). J Exp Biol 215:397–404
Dyer AG, Arikawa K (2014) A hundred years of color studies in insects: with thanks to Karl von Frisch and the workers he inspired. J Comp Physiol A 200:409–410
Dyer AG, Neumeyer C, Chittka L (2005) Honeybee (Apis mellifera) vision can discriminate between and recognise images of human faces. J Exp Biol 208:4709–4714
Dyer AG, Spaethe J, Prack S (2008) Comparative psychophysics of bumblebee and honeybee colour discrimination and object detection. J Comp Physiol A 194:617–627
Efler D, Ronacher B (2000) Evidence against a retinotopic-template matching in honeybees’ pattern recognition. Vision Res 40:3391–3403
Garcia JE, Shrestha M, Howard SR, Petersen P, Dyer AG (2018) Signal or cue: the role of structural colors in flower pollination. Curr Zool 65:467–481
Giurfa M, Hammer M, Stach S, Stollhoff N, Müller-Deisig N, Mizyrycki C (1999) Pattern learning by honeybees: conditioning procedure and recognition strategy. Anim Behav 57:315–324
Giurfa M, Nunez J, Chittka L, Menzel R (1995) Colour preferences of flower-naive honeybees. J Comp Physiol A 177:247–259
Giurfa M, Eichmann B, Menzel R (1996) Symmetry perception in an insect. Nature 382:458–461
Giurfa M, Zhang S, Jenett A, Menzel R, Srinivasan MV (2001) The concepts of ‘sameness’ and ‘difference’ in an insect. Nature 410:930–933
Goulson D, Nicholls E, Botías C, Rotheray EL (2015) Bee declines driven by combined stress from parasites, pesticides, and lack of flowers. Science 347:1255957
Heard TA (1999) The role of stingless bees in crop pollination. Annu Rev Entomol 44:183–206
Herrera CM (2020) Gradual replacement of wild bees by honeybees in flowers of the Mediterranean Basin over the last 50 years. Proc R Soc B 287:20192657
Hogendoorn K, Keller M (2012) Native Australian bees as potential pollinators of lucerne (Publication No 12/048; Project No. PRJ-005657). Rural Industries Research and Development Corporation. Canberra, Australia
Hogendoorn K, Steen Z, Schwarz MP (2000) Native Australian carpenter bees as a potential alternative to introducing bumble bees for tomato pollination in greenhouses. J Apic Res 39:67–74
Hogendoorn K, Gross CL, Sedgley M, Keller MA (2006) Increased tomato yield through pollination by native Australian Amegilla chlorocyanea (Hymenoptera: Anthophoridae). J Econ Entomol 99:828–833
Hogendoorn K, Bartholomaeus F, Keller MA (2010) Chemical and sensory comparison of tomatoes pollinated by bees and by a pollination wand. J Econ Entomol 103:1286–1292
Hogendoorn K, Coventry S, Keller MA (2007) Foraging behaviour of a blue banded bee, Amegilla chlorocyanea in greenhouses: implications for use as tomato pollinators. Apidologie 38:86–92
Horridge GA (1997) Pattern discrimination by the honeybee: disruption as a cue. J Comp Physiol A 181:267–277
Houston T (2018) A guide to native bees of Australia. CSIRO Publishing, Australia
Howard SR, Garcia JE, Dyer AG (2021) Comparative psychophysics of colour preferences in two species of non-eusocial Australian native halictid bees. J Comp Physiol A
Howard SR, Avarguès-Weber A, Garcia J, Dyer AG (2017a) Free-flying honeybees extrapolate relational size rules to sort successively visited artificial flowers in a realistic foraging situation. Anim Cogn 20:627–638
Howard SR, Avarguès-Weber A, Garcia JE, Stuart-Fox D, Dyer AG (2017b) Perception of contextual size illusions by honeybees in restricted and unrestricted viewing conditions. Proc Biological Sci 284:20172278
Howard SR, Avarguès-Weber A, Garcia JE, Greentree AD, Dyer AG (2018a) Numerical ordering of zero in honey bees. Science 360:1124–1126
Howard SR, Shrestha M, Schramme J, Garcia JE, Avarguès-Weber A, Greentree AD, Dyer AG (2018b) Honeybees prefer novel insect-pollinated flower shapes over bird-pollinated flower shapes. Curr Zool 65:457–465
Howard SR, Avarguès-Weber A, Garcia JE, Greentree AD, Dyer AG (2019a) Achieving arithmetic learning in honeybees and examining how individuals learn. Commun Integr Biol 12:166–170
Howard SR, Avarguès-Weber A, Garcia JE, Greentree AD, Dyer AG (2019b) Numerical cognition in honeybees enables addition and subtraction. Sci Adv 5:easv0961
Howard SR, Avarguès-Weber A, Garcia JE, Greentree AD, Dyer AG (2019c) Surpassing the subitizing threshold: appetitive–aversive conditioning improves discrimination of numerosities in honeybees. J Exp Biol 222:jeb205658
Howard SR, Avarguès-Weber A, Garcia JE, Greentree AD, Dyer AG (2019d) Symbolic representation of numerosity by honeybees (Apis mellifera): Matching characters to small quantities. Proc Biological Sci 286:20190238
Howard SR, Schramme J, Garcia JE, Ng L, Avarguès-Weber A, Greentree AD, Dyer AG (2020) Spontaneous quantity discrimination of artificial flowers by foraging honeybees. J Exp Biol 223:jeb223610
Ings T, Wang M-Y, Chittka L (2012) Colour-independent shape recognition of cryptic predators by bumblebees. Behav Ecol Sociobiol 66:487–496
Ings TC, Chittka L (2009) Predator crypsis enhances behaviourally mediated indirect effects on plants by altering bumblebee foraging preferences. Proc Biol Sci 276:2031–2036
Ings TC, Chittka L (2008) Speed-accuracy tradeoffs and false alarms in bee responses to cryptic predators. Curr Biol 18:1520–1524
Jones EI, Dornhaus A (2011) Predation risk makes bees reject rewarding flowers and reduce foraging activity. Behav Ecol Sociobiol 65:1505–1511
Kleijn D et al (2015) Delivery of crop pollination services is an insufficient argument for wild pollinator conservation. Nat Commun 6:7414
Lehrer M, Horridge G, Zhang S, Gadagkar R (1995) Shape vision in bees: innate preference for flower-like patterns. Series B: Biological Sciences 347:123–137
Loukola OJ, Gatto E, Híjar-Islas AC, Chittka L (2020) Selective interspecific information use in the nest choice of solitary bees. Anim Biol 70:215–225
Mallinger RE, Gaines-Day HR, Gratton C (2017) Do managed bees have negative effects on wild bees?: a systematic review of the literature. PloS One 12:e0189268
Marchal P et al (2019) Inhibitory learning of phototaxis by honeybees in a passive-avoidance task. Learn Mem 26:412–423
Martin NH (2004) Flower size preferences of the honeybee (Apis mellifera) foraging on Mimulus guttatus (Scrophulariaceae). Evol Ecol Res 6:777–782
Menzel R, Steinmann E, De Souza J, Backhaus W (1988) Spectral sensitivity of photoreceptors and colour vision in the solitary bee, Osmia rufa. J Exp Biol 136:35–52
Mitchell RJ, Irwin RE, Flanagan RJ, Karron JD (2009) Ecology and evolution of plant–pollinator interactions. Ann Bot 103:1355–1363
Nouvian M, Galizia CG (2019) Aversive training of honey bees in an automated Y-maze. Front Physiol 10:678
Orth AI, Waddington KD (1997) Hierarchical use of information by nectar-foraging carpenter bees on vertical inflorescences: floral color and spatial position. Israel J Plant Sci 45:213–221
Owens A, Cochard P, Durrant J, Perkin E, Seymoure B (2020) Light pollution is a driver of insect declines. Biol Conserv 241:108259
Perez SM, Waddington KD (1996) Carpenter bee (Xylocopa micans) risk indifference and a review of nectarivore risk-sensitivity studies. Am Zool 36:435–446
Perry CJ, Barron AB (2013) Honey bees selectively avoid difficult choices. Proc Natl Acad Sci 110:19155–19159
Potts SG, Biesmeijer JC, Kremen C, Neumann P, Schweiger O, Kunin WE (2010) Global pollinator declines: trends, impacts and drivers. Trends Ecol Evol 25:345–353
R Core Team (2020) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/
Raguso RA (2008) Wake up and smell the roses: the ecology and evolution of floral scent. Annu Rev Ecol Evol Syst 39:549–569
Sands DP (2018) Important issues facing insect conservation in Australia: now and into the future. Austral Entomol 57:150–172
Santos AA, Leijs R, Picanço MC, Glatz R, Hogendoorn K (2020) Modelling the climate suitability of green carpenter bee (Xylocopa aerata) and its nesting hosts under current and future scenarios to guide conservation efforts. Austral Ecol 45:271–282
Sargent RD, Ackerly DD (2008) Plant–pollinator interactions and the assembly of plant communities. Trends Ecol Evol 23:123–130
Somanathan H, Borges RM, Warrant EJ, Kelber A (2008) Nocturnal bees learn landmark colours in starlight. Curr Biol 18:R996–R997
Somanathan H, Saryan P, Balamurali G (2019) Foraging strategies and physiological adaptations in large carpenter bees. J Comp Physiol A 205:387–398
Srinivasan MV (2010) Honey bees as a model for vision, perception, and cognition. Annu Rev Entomol 55:267–284
Taylor GS, Braby MF, Moir ML, Harvey MS, Sands DP, New TR, Kitching RL, McQuillan PB, Hogendoorn K, Glatz RV (2018) Strategic national approach for improving the conservation management of insects and allied invertebrates in Australia. Austral Entomol 57:124–149
Vergoz V, Roussel E, Sandoz J-C, Giurfa M (2007) Aversive learning in honeybees revealed by the olfactory conditioning of the sting extension reflex. PloS One 2:e288
Wang M-Y, Ings TC, Proulx MJ, Chittka L (2013) Can bees simultaneously engage in adaptive foraging behaviour and attend to cryptic predators? Anim Behav 86:859–866
Zhang E, Nieh JC (2015) The neonicotinoid imidacloprid impairs honey bee aversive learning of simulated predation. J Exp Biol 218:3199–3205
Zhang S, Srinivasan M (1994) Prior experience enhances pattern discrimination in insect vision. Nature 368:330–332
Zhang S, Bartsch K, Srinivasan M (1996) Maze learning by honeybees. Neurobiol Learn Mem 66:267–282
Zhang SW, Lehrer M, Srinivasan MV (1999) Honeybee memory: navigation by associative grouping and recall of visual stimuli. Neurobiol Learn Mem 72:180–201
Zhang S, Mizutani A, Srinivasan MV (2000) Maze navigation by honeybees: learning path regularity. Learn Mem 7:363–374
Acknowledgements
SRH thanks Associate Professor Matthew Symonds for his comments on the study and the editors and two anonymous reviewers for their constructive and helpful suggestions. SRH acknowledges funding from Deakin University.
Funding
SRH acknowledges funding from the Alfred Deakin Postdoctoral Research Fellowship.
Author information
Authors and Affiliations
Contributions
SRH performed data collection, analysis, and wrote the manuscript.
Corresponding author
Ethics declarations
Ethics approval
All animal care was in accordance with institutional guidelines. Formal ethics approval was not required for invertebrate behavioural testing.
Conflicts of interest
The author declares no competing interests.
Additional information
Communicated by: Sean O’Donnell
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Online Resource 1 (ESM 1): Video showing the pre-test of a L. lanarium bee. (MP4 9295 KB)
Rights and permissions
About this article
Cite this article
Howard, S.R. Wild non-eusocial bees learn a colour discrimination task in response to simulated predation events. Sci Nat 108, 28 (2021). https://doi.org/10.1007/s00114-021-01739-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00114-021-01739-9