Abstract
Purpose
Anterior cranial fossa (ACF) dural arteriovenous fistulae (DAVF) are rare, unique, and ominous. While surgical disconnection is considered as the favored management option, endovascular treatment has lately gained importance. We present a single institution experience of seven cases.
Method
A retrospective analysis was performed on the institutional patient database. Features analyzed were demographic details, symptoms, angioarchitecture, treatment course, angiographic results, procedural complications, and follow-up.
Results
This study included seven patients. The age at presentation ranged from 5–67 years. Clinical symptomatology was as intracranial hemorrhage in 4 patients and headache, chemosis and seizures in one patient each. The fistulae were paramedian at the ACF base. All DAVFs were Cognard type 4. The arterial feeders were from the anterior ethmoidal branches of the ophthalmic artery in all cases (bilateral in n = 5), frontal branches of the middle meningeal artery (MMA) (n = 6), and multiple ECA branches. The arterial route was the choice for access. Complete fistula obliteration was achieved in all but one patient. A traversed vein underwent rupture in one patient. One patient suffered postsurgical hemorrhage. No clinical or angiographic recurrence was noted.
Conclusion
The DAVFs of the ACF are inherently high-grade lesions. Transorbital ECA-ICA branch anastomoses may be recruited as feeders. They may be best managed by multidisciplinary means personalized on an angioarchitectural basis. Endovascular embolization is safe and efficacious when performed through a navigable feeder from the frontal division of the MMA, which according to our interpretation is in anastomosis with the anterior falcine branch of the anterior ethmoidal artery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Dural arteriovenous fistulae (DAVF) amount to ~10–15% of intracranial arteriovenous malformations [1]. The annual risk of parenchymal hemorrhage exemplifies the high-risk characteristics of these lesions in the background of cortical venous drainage, up to 7.6%, which exceeds that of most arteriovenous malformations [2]. The cumulative annual neurological event rates in patients with DAVF can be as high as 15%, with mortality rates of up to 10% [3]. Notably, a small subset of these lesions located in the anterior cranial fossa (ACF) has a sinister course, with bleeding rates scaling up to 91% [4]. The propensity for direct cortical venous drainage [5], partially explains the high-risk traits. Notwithstanding the nature of the clinical symptomatology, this disorder warrants urgent management [6].
Management options of ACF DAVF are manifold, including surgical disconnection, endovascular obliteration, and radiosurgery [7, 8]. The current implicit consensus is towards surgical exclusion in light of its efficiency in achieving a complete cure [5, 6]. The limitations with surgical resection reside in the inherent risks of the craniotomy, possibility of paranasal sinus extension of the defect, cerebrospinal fluid leak, pachymeningeal infection, and retraction damage to the frontal lobe and olfactory nerves [9]. Radiosurgery has a waiting period before occlusion of the fistula [7].
The option of endovascular management was hitherto not preferred, given the usually challenging arterial access to these lesions. The literature on endovascular management of ACF DAVF has been sparse and sporadic, with approximately 48 cases published so far [4]. Endovascular treatment for ACF DAVF has recently gained popularity with technology-driven advances in access catheters and embolisates [4, 5, 10, 11]. With favorable access, either a transarterial or a transvenous route may be considered [9, 12]. We introduce a single institution experience of seven consecutive cases managed at our facility and review the literature on this entity.
Method
A retrospective analysis was performed on the institutional and departmental patient database to identify patients diagnosed with ACF DAVF from 2010 to October 2019. Features derived and analyzed were demographic details, presenting symptoms, angioarchitecture (including arterial feeders, venous drainage scheme, the existence of venous ectasia, Cognard classification), treatment course (surgical and/or endovascular), angiographic results, procedural complications, and follow-up results. Follow-up data were gathered from the clinical evaluation and angiography at scheduled intervals or telephonic inquiry.
Results
A total of seven patients who were managed at our facility were included in the analysis. The clinicodemographic profile, imaging phenotype, interventional approach, outcome, and follow-up are summarized in Table 1.
Clinical and Demographic Profile
The age at clinical presentation ranged from 5 to 67 years (mean age 51.5 years) and 5 patients were male (male:female 5:2). Clinical symptomatology was predominantly in the form of intracranial hemorrhage in 4 patients (57.2% %). Headache and tinnitus were reported by one patient and one patient presented with primary ophthalmological complaints, such as, insidious onset and progressive proptosis, chemosis of the right eye with diminution of vision. Cross-sectional imaging revealed evidence of anterior cranial fossa base fistula at the right lesser wing of the sphenoid with a dilated superior ophthalmic vein (SOV). The youngest patient in the series, presented with generalized tonic-clonic seizures (GTCS) whose imaging evaluation showed dual vascular lesions.
Imaging and Angioarchitecture
Cross-sectional imaging (CT/MRI) was available for 6 patients. The fistula locations were inferable at the plane of the anterior cranial fossa base, mainly in a paramedian location (n = 6). Evidence of a remote parenchymal hematoma was at the level of the orbitofrontal gyrus, gyrus rectus region (n = 3). One patient had evidence of subdural and subarachnoid hemorrhage with extensive near-holohemispheric ectatic draining veins. Imaging evaluation of the youngest patient demonstrated dual pathology, an unruptured left perisylvian arteriovenous malformation (Spetzler Martin grade 3 AVM; with feeders from the M2 and M3 segments of the MCA) and anterior cranial fossa base DAVF. Classified under the Cognard scheme, all DAVFs were type 4 (type 3 as per Borden classification). The arterial feeders were from the anterior ethmoidal branches of the ophthalmic artery in all cases (bilateral in n = 5), frontal branches of the middle meningeal artery (n = 6), the angular branch of the facial artery in one case, infraorbital branches of the internal maxillary artery in two cases, the septal branch of the sphenopalatine artery in one patient and the greater palatine branch of the internal maxillary artery (IMA) in one patient. Transosseous branches of the right occipital artery in one case and anastomoses between the zygomatico-orbital branches of the superficial temporal artery, supratrochlear and supraorbital branches of the ophthalmic artery were notable feeders in one case each. Facial artery anastomosis with sphenopalatine artery was evident, possibly through the presumed superior labial artery, another alternative anastomosis was the anastomosis through the jugal branches. Another interesting anastomosis was evident between the sphenopalatine and the infraorbital artery in two cases. Anastomosis between the ophthalmic artery and the infraorbital artery was also an alternative pathway to the anterior ethmoidal artery (AEA) feeder (Fig. 1). Flow-associated feeder artery aneurysms were remarkably absent. No pial feeders were encountered. Venous drainage was into a cortical vein (frontal, namely, frontopolar and orbitofrontal) and thence into the anterior aspect of the superior sagittal sinus in three of the patients. One patient had venous outflow into the basal vein of Rosenthal. Posteriorly directed venous drainage into the cavernous sinus, along with cortical venous reflux, was evident in one case. Anterior outflow into the ophthalmic-facial venous system was observed in another patient who presented with chemosis. Diffuse ectatic cortical venous drainage with no prompt opacification of the superior sagittal sinus was observed in two of the patients. Venous sacs were evident in the early phase of the angiogram in 5 patients. It was noteworthy that all patients who had the clinical presentation suggestive of hemorrhage indeed had demonstrable venous sacs.
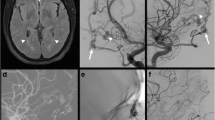
Varied anastomoses observed in anterior skull base fistulae. a Lateral and b frontal projection of the left ECA run in case 4 reveals a prominent facial artery (arrow) with its alar and angular branch involved in anastomosis with the nasal branches of the infraorbital artery (double arrow). The superior labial artery (asterisk) is prominent. Curved arrows in a and b denote the anterior temporal, zygomatic branches of the superficial temporal artery involved as feeders in the fistula. c Lateral and d frontal projection of the right ECA run in case 4. The septal branches of the sphenopalatine artery are visualized as feeders to the ACF base fistula (single arrow) as are the branches of the infraorbital artery (double arrow). e Microcatheter injection in feeders to the ACF base fistula via a left frontal branch of the MMA in case 4 shows reflux opacification of the facial artery (double asterisk) via the angular branch and the superficial temporal artery (single asterisk) via the anterior temporal and zygomatic branches. f Lateral and g frontal projection of the right ECA run in case 3 reveals transosseous occipitocarotid anastomoses (arrowhead) between the occipital artery with a midline extending up to the anterior skull base. The anterior temporal branches (double asterisk) and zygomatic branches (single asterisk) of the right superficial temporal artery are depicted
Interventional Management
The interventional procedures were done with the patient under general anesthesia. Diagnostic cerebral angiogram of all six vessels was performed before the interventional phase for all patients. The institutional anticoagulation protocol for endovascular treatment of DAVFs was an intravenous loading dose bolus of 80 U/kg of unfractionated heparin (UFH) at the start of the procedure. It was supported by heparin administration at an equivalent of 18 U/kg/h throughout the procedure. Posttreatment, the patient was maintained on 80 U/kg of UFH for 3 days (to prevent exaggerated cortical venous thrombosis), after which the heparinization was stopped. The arterial route was the primary choice for access (n = 5). The arteries accessed were: falcine branch of the frontal division of the middle meningeal artery in 5 cases (Fig. 2); ophthalmic artery in 1 case (Fig. 3). A triaxial access system was used across a femoral arterial route (6–8F short sheath). The long access systems used were 0.088″ NeuronMax (Penumbra Inc, Alameda, CA, USA) and 7F Vista Brite Tip sheath (Cordis, Miami, FL, USA). The intermediate catheters used were 5F Navien (ev3 Neurovascular; ev3/Covidien, Irvine, CA, USA), AXS Catalyst 5 (Stryker Neurovascular, Mountain View, CA, USA), and distal access catheter (DAC, Stryker Neurovascular, Freemont, CA, USA). Marathon (ev3 Neurovascular; ev3/Covidien; n = 5), and Echelon 10 microcatheter (ev3 Neurovascular; ev3/Covidien; n = 1), over Traxcess (MicroVention, Tustin, CA, USA) microwire was used for the distal access. Coil embolization of the fistulous sac with six coils was performed in one patient. The liquid embolic agents used were Onyx 34, Onyx 18 (ev3 Neurovascular; ev3/Covidien), and Squid 12 (Balt, Montmorency, France). Complete fistula obliteration was observed in all but one patient (mild residual filling in the patient in case 1, who was managed with coiling of the feeder from MMA close to the fistula site).
Case 3: a axial Non-Contrast Computed Tomography (NCCT) shows serpiginous extra-axial hyperdensities in the right frontal region, a few with mural calcification suggestive of ectatic venous channels. b Coronal T2 weighted image demonstrates flow voids corresponding to the cortical venous reflux in the right paramedian anterior cranial fossa base region. c Frontal projection of right ICA injection and d, e lateral projection of bilateral ICA injections reveal Cognard type 4 DAVF at the anterior cranial fossa base with numerous minute feeders from the anterior ethmoidal branches of the bilateral ophthalmic arteries and extensive cortical venous reflux. f, g Frontal and lateral projections of left ECA injection reveal a dominant feeder from the frontal branch of the left MMA converging on the fistula, which was chosen as the access to the fistula. h, i Frontal and lateral projection of microcatheter injection after reaching the fistulous site. j, k, l Postembolization lateral and bilateral frontal projections of bilateral ICA injections reveal no residual fistula
Case 2: a axial NCCT reveals gyrus rectus and right orbitofrontal hematoma, b T2 weighted axial section reveals hemorrhagic residue and a few flow voids in this location. c Frontal and d lateral projection of right ICA injection reveals Cognard type 4 DAVF at the anterior cranial fossa base with feeders from the anterior ethmoidal branches of the right ophthalmic artery and extensive cortical venous reflux. e Lateral projections of bilateral ECA injections reveals no distinct navigable feeder from the MMA. f, g, h Progressive navigation (top row—lateral; bottom row—frontal projection) of the ophthalmic artery with the support of a triaxial system reaching the desired position close to the fistula. i Frontal projection of right ICA injection and j lateral projection of the left ICA injection postembolization reveal complete exclusion of fistula
A primary venous approach was adopted in one case where the drainage was into the basal vein of Rosenthal. The straight sinus was accessed with a Neuron 6F (Penumbra Inc) from the right internal jugular vein. Headway Duo (MicroVention)/Traxcess (MicroVention) co-axial system was employed for cannulation of the fistulous segment via basal vein of Rosenthal. Three coils were deployed, and because of incomplete exclusion of the fistula, the procedure was subsequently completed successfully via an arterial route from the left MMA. In one case (case 7), a secondary venous approach across the superior sagittal sinus (after difficulty with arterial access via the falcine branch of the middle meningeal artery) was adopted using a NeuronMAX-Navien-Marathon co-axial system via the left internal jugular vein. Difficulty was encountered in negotiating the draining frontopolar vein to reach the site of the fistula despite multiple attempts. Check arteriograms revealed contrast extravasation across the cannulated vein. Suboptimal percolation of the liquid embolic agent was achieved. Postprocedural CT revealed right frontoparietal convexity subdural hemorrhage (22 mm thick) with mass effect in the form of subfalcine and uncal herniation causing a midline shift of ~15 mm. An emergent decompressive hinge craniotomy was performed with ligation of the vein and evacuation of the hematoma. Following a decompressive craniotomy, the patient had fixed dilated pupils, with a Glasgow Coma Scale (GCS) score of E1VTM1. Follow-up CT revealed multiple progressive hypodensities in the bilateral cerebral hemispheres, suggestive of infarcts due to vascular compression with ongoing mass effect. The patient died following a sudden cardiac arrest 5 days later.
Surgical management was undertaken in one patient who could not afford the neurointerventional procedure. The patient underwent a bifrontal craniotomy with coagulation and removal of coagulated draining veins. Bilateral frontal durotomy and disconnection of superior sagittal sinus disconnected from ACF base attachment with dura closure with pericranial graft were done.
Follow-up
The follow-up angiography was performed at 6 months. The follow-up clinical examination at the neurosurgery outpatient department of the institute was performed at 6‑month intervals from the time point of treatment.
The patient (case 5; 5‑year-old boy; with an associated arteriovenous malformation) who underwent surgical exclusion of the lesion had three episodes of GTCS in the immediate postoperative period. The CT revealed hemorrhage in the left frontotemporal region. Subsequent management was conservative with a single antiepileptic drug with no deficits at discharge. He reported no seizures at follow-up. One patient (case 2; who was managed with a transophthalmic arterial route) had transient visual obscuration, was initiated on pulse methylprednisolone for 5 days, and reported a complete reversal of symptoms. Prompt resolution of chemosis/proptosis was observed in the patient who had anterior drainage (case 1). Although the patient had a small remnant filling in the immediate postintervention phase angiogram, he had prompt resolution of symptoms. He was heparinized for 3 days postprocedure. The follow-up angiogram performed at 6 months revealed obliteration of the remnant and no recurrence of the fistula. Two patients (cases 2 and 4) who reported sudden-onset headache at admission, mentioned no recurrence of the symptoms posttreatment and at clinical follow-ups. The patient in case 3 presented with headache and left hemiparesis for 15 days pretreatment, both of which responded to treatment, with no recurrence at follow-up. No patient reported a recurrence of symptoms. Angiographic follow-up data were available (6 months) for all patients with no evidence of recurrence.
Discussion
First described by Lepoire et al. in 1963, anterior cranial fossa DAVFs, for the most of neurosurgical and neurointerventional literature, has been a rarity [13]. The frequency of ACF base lesions is estimated at approximately 4–10% of all intracranial DAVFs [14, 15]. Lasjaunias et al. in a review of neurological manifestations of cranial dural arteriovenous fistulae, underscored the perilous natural course of these rare lesions referring to a rate of hemorrhage at the presentation of up to 84% [14]. Stratified for location and Borden type, a comprehensive review by Gross et al. observed that the anterior cranial fossa DAVF are invariably Borden type III reiterating their propensity for cortical venous drainage [16]. Cortical venous drainage and ectasia unequivocally dictate the hemorrhage risk in patients with DAVFs, explaining the innate high-risk nature of these lesions [17, 18].
Our patient population followed the generally reported trend of male predilection [4, 5]. Hemorrhage is the most frequent primary presentation, but the clinical presentation in our group has been diverse [19, 20]. A referral bias driven representation may be possible in a tertiary center such as ours, where the proportion of lesions with a non-hemorrhagic presentation may be higher [5].
The localization of the fistulous site (usually solitary) is at the plane of the cribriform plate, in an off-midline/midline location at the lateral epidural compartment overlying the lamina cribrosa and roof of the orbits [9]. Bilateral fistulous points are rare [21]. The ophthalmological pattern of presentation in one patient may be adduced by the transosseous exit of the draining vein across the thin roof of the orbit converging into the superior ophthalmic vein. Two case reports exist of similar departure from the usual patterns (hemorrhage) of clinical presentation [22, 23].
In keeping with the literature, our patient database was characterized by fistulae with arterial feeders principally from ethmoidal branches of the ophthalmic arteries with bilateral feeders in all of the cases [4, 9]. The frontal branch of the middle meningeal artery (involvement in approximately 20% of cases) and the septal branch of the sphenopalatine artery and its anastomoses with the anterior and posterior ethmoidal artery (to the tune of 20–60%) have been implicated as next most likely feeders as was the case in our study [5, 7, 9, 15]. The frontal (anterior) division of the middle meningeal artery has a dural territorial supply over the frontal convexity, superior sagittal sinus walls, and the falx and is therefore in a reciprocal relationship with the anterior falcine artery territory (branch of the ophthalmic artery in the third segment), thus potentially explaining its hypertrophy and contribution in these fistulous states [24, 25]. The contribution from the superficial temporal artery may be explained by its participation in the transorbital anastomoses. The superficial temporal artery’s frontal branch and the zygomatico-orbital branch are engaged in collateralization with supraorbital and supratrochlear branches of the ophthalmic artery at the anterior orbit, offering an insight into their induction as feeders to these lesions [26]. Occipitocarotid anastomoses have not been described earlier as candidate feeders for the ACF DAVF.
Cortical venous outflow, the well-established angiographic signature of these lesions rendering them a higher Cognard grading, was almost invariant in our series. Additional venous ectasia was present in 3 cases. Furthermore, the absence of a discrete collecting dural venous sinus in the anterior cranial fossa base leaves these lesions with no alternative drainage pathways [20]. Intracranial DAVFs are hypothesized to arise from minute, intramural, pre-existent shunts on the venous sinuses that drain into the sinus lumen in the background of sinovenous thrombosis. The floor of the ACF, the usual location of anterior DAVFs has no dural sinus, thereby questioning the genesis of fistulae at these locations [10]. The fact that one of the patients in the current analysis was very young (the youngest reported) with a co-existent temporal AVM also prompts to seek alternative explanations for the pathogenesis. The hemodynamic stress on the fragile vein is the driver for ultrastructural changes in the wall leading to venous ectasia, pouches, and aneurysms [27]. Posterior drainage into the cavernous sinus and the basal vein of Rosenthal have been described earlier at lower frequencies [8, 28]. Very few case reports exist of anterior venous drainage of an ethmoidal fistula with a similar clinical presentation of chemosis, proptosis, and diminution of vision [22, 28].
The management of ACF DAVF with Onyx/Squid via an MMA feeder as a safe and efficacious option has been explored earlier [12, 29]. The first line choice of the frontal branch of the middle meningeal artery to access the fistula in our series is driven by the strategical and anatomic advantages that it confers. Microcatheter access is less treacherous and uncomplicated except in extremely tortuous anatomy. Angiographic outcomes are good irrespective of the size of the MMA feeders considering the lesser likelihood of rupture while retracting the microcatheter, and allowance for reflux. Unlike the ophthalmic artery route, the margin of error is higher when embolisate reflux is a concern [7, 12]. It seems plausible that when the tolerance to reflux is higher, the chances of a forward percolation of the embolisate is higher, and thus, the possibility of incomplete embolization would be correspondingly lower (this incidentally happens to be the argument for a transvenous approach to the lesions). No case of liquid embolic agent delivery via MMA in our series had incomplete embolization. The benefits of the anterior division of MMA as access are strikingly similar to its posterior counterpart in the management of the torcular/transverse-sigmoid DAVF [30]. Approaches through the septal branch of the internal maxillary artery or balloon-assisted management via ophthalmic artery exist, albeit seemingly hazardous [10]. As demonstrated in our case of ophthalmic artery access, if catheter location and stability is a concern, it may be achieved using a supple co-axial (triaxial) system that supports microcatheter navigation. Coils as the primary embolic agent have been used earlier along the venous route but have largely been superseded by liquid embolic agents (Onyx/Squid), which have now become the norm [9]. Transvenous embolization of ACF-DAVF has been described earlier in at least 15 instances [9, 31,32,33,34,35,36]. The prior reports have all demonstrated a complete cure with no recurrence of symptoms. No description of the intraprocedural rupture of the venous outflow has been described earlier in the treatment of ACF-DAVF. Defreyne et al. prescribe a phlebography of the accessed vein and ophthalmic arteriography to ascertain the location of the fistula [9]. In the context of ACF DAVF, the transvenous retrograde catheterization is fraught with the long, tortuous, and tenuous course of the traversed vessels prompting a recommendation for jugular access [9, 37]. The venous route of access spares the operator the risk of compromising the central retinal artery and of inadvertent embolization to the brain parenchyma; however, the draining veins of the DAVF in the anterior cranial fossa may be drawn out, tortuous, and fragile, with a concomitant risk of intraoperative perforation during microcatheter navigation [9]. The possibility of the veins draining normal parenchyma cannot be altogether excluded [4]. The solitary case (case 7) of our series that was approached via a venous route was complicated by perforation and a downhill clinical course. On reflection, the extreme tortuosity of the draining frontopolar vein factored in the difficulty with traversing it despite robust co-axial access for supporting navigation. The cannulated fragile venous pathway eventually ruptured culminating in subdural hemorrhage and its sequelae. It is to be noted that excessive tortuous anatomy is considered a relative contraindication to the venous approach.
Patient and access selection based on angioarchitecture remains the cornerstone of angiographic outcomes and success. The reported complete occlusion rate via arterial means ranges between 12.5% and 63%, while a venous route has met with an occlusion rate of 63–91% [5, 6]. Recently, Piergallini et al. reported on the ophthalmic artery as the choice of access for embolization of these fistulae [38]. Notably, their study demonstrates that the ophthalmic artery offers access for safe and complete obliteration of the ACF-DAVFs using liquid embolic agents. The patient demographics, angiographic grades of the lesions in our series were similar to theirs. They opine that the ophthalmic access confers the advantage of the highest flow, and pressure enables percolation of liquid embolic agents to the fistulous site, thereby enabling the likelihood of complete cure. None of the cases in their study was complicated by visual deterioration. They propose that identification of the anatomical landmark of the take-off of the posterior ciliary arteries (consequently that of the central retinal artery) is of value. When the reflux of the embolisate is not allowed proximal to the distal third of the ophthalmic artery, visual complications seldom occur. Cannulation of a hypertrophic ophthalmic artery in an ACF DAVF is at least theoretically easier than in other routine contexts.
We acknowledge the limitations of the study in its retrospective nature, small patient population, and non-availability of all modalities of cross-sectional imaging in all patients.
The ACF DAVFs may be best managed by multidisciplinary means with the therapeutic endpoint being exclusion/arteriovenous disconnection of the lesions. The evaluation and management principles should be highly personalized based on the angioarchitecture of the fistula. Given a favorable access endovascular management of ACF DAVF compared to surgical exclusion is less invasive and more cost-effective. We emphasize that the endovascular approach should be considered as a management option for ACF DAVF, particularly in the presence of an amenable MMA feeder. This also happens to reflect the evolution of treatment strategy at our institution. The risk of perforation/rupture of the traversed vein, especially when tortuous and ectatic, should be borne in mind when employing the transvenous route for embolization of ACF DAVF.
Abbreviations
- ACF:
-
Anterior cranial fossa
- AEA:
-
Anterior ethmoidal artery
- AVM:
-
Arteriovenous malformation
- BVR:
-
Basal vein of Rosenthal
- CT:
-
Computed tomography
- DAVF:
-
Dural arteriovenous fistulae
- ECA:
-
External carotid artery
- ICA:
-
Internal carotid artery
- IMA:
-
Internal maxillary artery
- MMA:
-
Middle Meningeal Artery
- MRI:
-
Magnetic resonance imaging
- SOV:
-
Superior ophthalmic vein
References
Kwon BJ, Han MH, Kang HS, Chang KH. MR imaging findings of intracranial dural arteriovenous fistulas: relations with venous drainage patterns. AJNR Am J Neuroradiol. 2005;26:2500–7.
Zipfel GJ, Shah MN, Refai D, Dacey RG, Derdeyn CP. Cranial dural arteriovenous fistulas: modification of angiographic classification scales based on new natural history data. Neurosurg Focus. 2009;26:E14.
van Dijk JMC, terBrugge KG, Willinsky RA, Wallace MC. Clinical course of cranial dural arteriovenous fistulas with long-term persistent cortical venous reflux. Stroke. 2002;33:1233–6.
Xu K, Ji T, Li C, Yu J. Current status of endovascular treatment for dural arteriovenous fistulae in the anterior cranial fossa: a systematic literature review. Int J Med Sci. 2019;16:203–11.
Gross BA, Moon K, Kalani MY, Albuquerque FC, McDougall CG, Nakaji P, Zabramski JM, Spetzler RF. Clinical and anatomic insights from a series of ethmoidal dural arteriovenous fistulas at Barrow Neurological Institute. World Neurosurg. 2016;93:94–9.
Meneghelli P, Pasqualin A, Lanterna LA, Bernucci C, Spinelli R, Dorelli G, Zampieri P. Surgical treatment of anterior cranial fossa dural arterio-venous fistulas (DAVFs): a two-centre experience. Acta Neurochir (Wien). 2017;159:823–30.
Agid R, Terbrugge K, Rodesch G, Andersson T, Söderman M. Management strategies for anterior cranial fossa (ethmoidal) dural arteriovenous fistulas with an emphasis on endovascular treatment. J Neurosurg. 2009;110:79–84.
Kobayashi H, Hayashi M, Noguchi Y, Tsuji T, Handa Y, Caner HH. Dural arteriovenous malformations in the anterior cranial fossa. Surg Neurol. 1988;30:396–401.
Defreyne L, Vanlangenhove P, Vandekerckhove T, Deschrijver I, Sieben G, Klaes R, Kunnen M. Transvenous embolization of a dural arteriovenous fistula of the anterior cranial fossa: preliminary results. AJNR Am J Neuroradiol. 2000;21:761–5.
Li Q, Fang YB, Huang QH, Zhang Q, Hong B, Zhao WY, Liu JM, Xu Y. Transarterial embolization of dural arteriovenous fistulas of the anterior cranial fossa with Onyx. J Clin Neurosci. 2013;20:287–91.
Lee B, Mehta VA, Amar AP, Tenser MS, Mack WJ. Transarterial embolization of an anterior fossa cranial base dural arteriovenous fistula. Neurosurg Focus. 2014;37(1 Suppl):1.
Deng JP, Li J, Zhang T, Yu J, Zhao ZW, Gao GD. Embolization of dural arteriovenous fistula of the anterior cranial fossa through the middle meningeal artery with Onyx. Clin Neurol Neurosurg. 2014;117:1–5.
Lepoire J, Montaut J, Bouchot M, Laxenaire M. Intrafrontal arteriovenous aneurysms vascularized by the anterior ethmoidal artery. Apropos of 3 cases. Neurochirurgie. 1963;9:159–66.
Lasjaunias P, Chiu M, ter Brugge K, Tolia A, Hurth M, Bernstein M. Neurological manifestations of intracranial dural arteriovenous malformations. J Neurosurg. 1986;64:724–30.
Başkaya MK, Suzuki Y, Seki Y, Negoro M, Ahmed M, Sugita K. Dural arteriovenous malformations in the anterior cranial fossa. Acta Neurochir. 1994;129:146–51.
Gross BA, Du R. The natural history of cerebral dural arteriovenous fistulae. Neurosurgery. 2012;71:594–602. discussion 602–3.
Reynolds MR, Lanzino G, Zipfel GJ. Intracranial dural arteriovenous fistulae. Stroke. 2017;48:1424–31.
Bulters DO, Mathad N, Culliford D, Millar J, Sparrow OC. The natural history of cranial dural arteriovenous fistulae with cortical venous reflux—the significance of venous ectasia. Neurosurgery. 2012;70:312–9.
Martin NA, King WA, Wilson CB, Nutik S, Carter LP, Spetzler RF. Management of dural arteriovenous malformations of the anterior cranial fossa. J Neurosurg. 1990;72:692–7.
Abrahams JM, Bagley LJ, Flamm ES, Hurst RW, Sinson GP. Alternative management considerations for ethmoidal dural arteriovenous fistulas. Surg Neurol. 2002;58:410–6.
Deshmukh VR, Chang S, Albuquerque FC, McDougall CG, Spetzler RF. Bilateral ethmoidal dural arteriovenous fistulae: a previously unreported entity: case report. Neurosurgery. 2005;57:E809.
Inoue A, Tagawa M, Kumon Y, Watanabe H, Shoda D, Sugiu K, Ohnishi T. Ethmoidal dural arteriovenous fistula with unusual drainage route treated by transarterial embolization. BMJ Case Rep. 2014;2014:bcr2013011098.
Shumway CL, Motlagh M, Wade M. Anatomy, Head and Neck, Orbit Bones. 2019 Jun 20. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020.
Geibprasert S, Pongpech S, Armstrong D, Krings T. Dangerous extracranial-intracranial anastomoses and supply to the cranial nerves: vessels the neurointerventionalist needs to know. AJNR Am J Neuroradiol. 2009;30:1459–68.
Martins C, Yasuda A, Campero A, Ulm AJ, Tanriover N, Rhoton A Jr. Microsurgical anatomy of the dural arteries. Neurosurgery. 2005;56(2 Suppl):211-51; discussion 211–51.
Bracco S, Venturi C, Leonini S, Romano DG, Cioni S, Vallone IM, Gennari P, Hadjistilianou T, De Francesco S, Bertelli E. Transorbital anastomotic pathways between the external and internal carotid systems in children affected by intraocular retinoblastoma. Surg Radiol Anat. 2016;38:79–87.
Ogawa T, Okudera T, Miyauchi T, Inugami A, Uemura K, Yasui N. Anterior cranial fossa dural arteriovenous fistula with a varix mimicking an anterior communicating artery aneurysm. Neuroradiology. 1996;38:252–3.
Halbach VV, Higashida RT, Hieshima GB, Wilson CB, Barnwell SL, Dowd CF. Dural arteriovenous fistulas supplied by ethmoidal arteries. Neurosurgery. 1990;26:816–23.
Tahon F, Salkine F, Amsalem Y, Aguettaz P, Lamy B, Turjman F. Dural arteriovenous fistula of the anterior fossa treated with the onyx liquid embolic system and the sonic microcatheter. Neuroradiology. 2008;50:429–32.
Jiang C, Lv X, Li Y, Wu Z. Transarterial Onyx packing of the transverse-sigmoid sinus for dural arteriovenous fistulas. Eur J Radiol. 2011;80:767–70.
Cannizzaro D, Peschillo S, Cenzato M, Pero G, Resta MC, Guidetti G, Burdi N, Piccirilli M, Santoro A, Lanzino G. Endovascular and surgical approaches of ethmoidal dural fistulas: a multicenter experience and a literature review. Neurosurg Rev. 2018;41:391–8.
Limbucci N, Leone G, Nappini S, Rosi A, Renieri L, Consoli A, Pedicelli A, Mangiafico S. Transvenous Embolization of Ethmoidal Dural Arteriovenous Fistulas: Case Series and Review of the Literature. World Neurosurg. 2018;110:e786–93.
Albuquerque FC, Ducruet AF, Crowley RW, Bristol RE, Ahmed A, McDougall CG. Transvenous to arterial Onyx embolization. J Neurointerv Surg. 2014;6:281–5.
Spiotta AM, Hawk H, Kellogg RT, Turner RD, Chaudry MI, Turk AS. Transfemoral venous approach for Onyx embolization of anterior fossa dural arteriovenous fistulae. J Neurointerv Surg. 2014;6:195–9.
Lv X, Li Y, Wu Z. Endovascular treatment of anterior cranial fossa dural arteriovenous fistula. Neuroradiology. 2008;50:433–7.
Roa JA, Dabus G, Dandapat S, Hasan D, Samaniego EA. Ethmoidal dural arteriovenous fistulas: endovascular transvenous embolization technique. J Neurointerv Surg. 2020;12:610.
Mendes GA, Caire F, Saleme S, Ponomarjova S, Mounayer C. Retrograde leptomeningeal venous approach for dural arteriovenous fistulas at foramen magnum. Interv Neuroradiol. 2015;21:244–8.
Piergallini L, Tardieu M, Cagnazzo F, Gascou G, Dargazanli C, Derraz I, Costalat V, Bonafé A, Lefevre PH. Anterior cranial fossa dural arteriovenous fistula: Transarterial embolization from the ophthalmic artery as first-line treatment. J Neuroradiol. 2019. https://doi.org/10.1016/j.neurad.2019.05.009. Epub ahead of print.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
K. Kulanthaivelu, H. Pendharkar, C. Prasad, A. Kumar Gupta, A. Hanumanthapura Ramalingaiah, J. Saini and D. Srinivas declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Kulanthaivelu, K., Pendharkar, H., Prasad, C. et al. Anterior Cranial Fossa Dural Arteriovenous Fistulae – Angioarchitecture and Intervention. Clin Neuroradiol 31, 661–669 (2021). https://doi.org/10.1007/s00062-020-00932-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00062-020-00932-z