Abstract
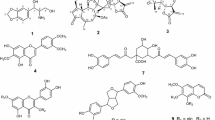
Phytochemical investigation of the hexane and CH2Cl2 extracts ofErythrina stricta roots andE. subumbrans stems led to the isolation of six pterocarpans, one flavanone, one isoflavone, two alkaloids, five triterpenes, six steroids and alkyl trans-ferulates. The structures of all known compounds were determined on the basis of spectroscopic evidence. Sophoradiol (15), a mixture of stigmast-4-en-3-one (19) and stigmasta-4,22-dien-3-one (20), lupeol (21), cycloeucalenol (22), a mixture of 3β-hydroxystigmast-5-en-7-one (23) and 3β-hydroxystigmast-5,22-dien-7-one (24) and melilotigenin C (25) were first isolated from the genusErythrina. The isolated compounds were evaluated for antiplasmodial activity, antimycobacterial activity and cytotoxicity. Among the tested compounds, 5-hydroxysophoranone (8) exhibited the highest antiplasmodial activity againstPlasmodium falciparum (IC50 2.5 μg/mL). Compound8, erystagallin A (5), erycristagallin (7) and erysubin F (10) showed the same level of antimycobacterial activity againstMycobacterium tuberculosis (MIC 12.5 μg/mL). For cytotoxicity, erybraedin A (2) showed the highest activity against the NCI-H187 and BC cells (IC50 2.1 and 2.9 μg/mL, respectively), whereas 10 exhibited the highest activity against the KB cells (IC50 4.5 μg/mL).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Achenbach, H. and Schwinn, A., Aporphinoid alkaloids and terpenoids fromPiptostigma fugax.Phytochemistry, 38, 1037–1048 (1995).
Andayi, A. W., Yenesew, A., Derese, S., Midiwo, J. O., Gitu, P. M., Jondiko, O. J. I., Akala, H., Liyala, P., Wangui, J., Waters, N. C., Heydenreich, M., and Peter, M. G., Anti-plasmodial flavonoids fromErythrina sacleuxii.Planta Med., 72, 187–189 (2006).
Barton, D. H. R., James, R., Kirby, G. W., Turner, D. W., and Widdowson, D. A., Phenol oxidation and biosynthesis. XVIII. The structure and biosynthesis ofErythrina alkaloids.J. Chem. Soc. C., 12, 1529–1537 (1986).
Baruah, P., Barua, N. C., Sharma, R. P., Baruah, J. N., Kulanthaivel, P., and Herz, W., Flavonoids fromMillettia pulchra.Phytochemistry, 23, 443–447 (1984).
Chopra, R. N., Nayar, S. L., and Chopra, I. C., Glossary of Indian Medicinal Plants, Council of Scientific & Industrial Research, New Delhi, pp.111 (1956).
Collins, L. and Franzblau, S. G., Microplate alamar blue assay versus BACTEC 460 system for high-throughput screening of compounds againstMycobacterium tuberculosis andMycobacterium avium.Antimicrob. Agents Chemother., 4, 1004–1009 (1997).
Dagne, E. and Steglich, W., 8-Oxoerythrinine: an alkaloid fromErythrina brucei.Phytochemistry, 23, 449–451 (1984).
Desjardins, R. E., Canfield, C. J., Haynes, J. D., and Chulay, J. D., Quantitative assessment of antimalarial activityin vitro by a semiautomated microdilution technique.Antimicrob. Agents and Chemother., 16, 710–718 (1979).
El-Masry, S., Amer, M. E., Abdel-Kader, M. S., and Zaatout, H. H., Prenylated flavonoids ofErythrina lysistemon grown in Egypt.Phytochemistry, 60, 783–787 (2002).
Folkers, K., and Koniuszy, F., Erythrina alkaloids. III. Isolation and characterization of a new alkaloid, erythramine.J. Am. Chem. Soc., 61, 1232–1235 (1939).
Folkers, K., Shavel, J. Jr., and Koniuszy, F., Erythrina alkaloids. X. Isolation and characterization of erysonine and other liberated alkaloids.J. Am. Chem. Soc., 63, 1544–1549 (1941).
Fomum, Z. T., Ayafor, J. F, Wandji, J., Fomban, W. G., and Nkengfack, A. E., Erythrinasinate, an ester from threeErythrina species.Phytochemistry, 25, 757–759 (1986).
Games, D. E., Jackson, A. H., Khan, N. A., and Millington, D. S., Alkaloids of some African, Asian, Polynesian and Australian species ofErythrina.Lloydia, 37, 581–588 (1974).
Greca, M. D., Monaco, P., and Previtera, L., Stigmasterols fromTypha latifolia.J. Nat. Prod., 53, 1430–1435 (1990).
Hussain, S. S., A new alkaloid from flowers ofErythrina stricta.J. Sciences, Islamic Republic of Iran, 13, 35–38, (2002).
Khaomek, P., Ruangrunsi, N., Saifah, E., Sriubolmas, N., Ichino, C., Kiyohara, H., and Yamada, H., A new pterocarpan fromErythrina fusca.Heterocycles, 63, 879–884 (2004).
Khuong-Huu, F., Sangare, M., Chari, V. M., Bekaert, A., Devys, M., Barbier, M., and Lukacs, G., Carbon-13 nuclear magnetic resonance spectral analysis of cycloartanol and related compounds.Tetrahedron Lett., 1787–1790 (1975).
Kinjo, J., Miyamoto, I., Miurakami, K., Kida, K., Tomimatsu, T., Yamasaki, M., and Nohara, T., Oleanene-sapogenols fromPuerariae radix.Chem. Pharm. Bull., 33, 1293–1296 (1985).
Kitagawa, I., Yoshikawa, M., and Yosioka, I., Revised structures of soyasapogenols A, B, and E.Chem. Pharm. Bull., 24, 121- 129 (1976).
Kocor, M., and Pyrek, J. St., Cyclotrichosantol, a new C31 31- nor triterpene.J. Org. Chem., 38, 3688–3690 (1973).
Macias, F. A., Simonet, A. M., Galindo, J. C. G., Pacheco, P. C., and Sanchez, J. A., Bioactive polar triterpenoids fromMelilotus messanensis.Phytochemistry, 49, 709–717, (1998).
Mahato, S. B. and Banerjee, S., Microbiological transformations of β-sitosterol and stigmasterol by a soil pseudomonad.Experientia, 36, 515–516 (1980).
Mahato, S. B. and Kundu, A. P.,13C NMR spectra of pentacyclic triterpenoids-a compilation and some salient features.Phytochemistry, 37, 1517–1575 (1994).
Matsuura, N., Nakai, R., Iinuma, M., Tanaka, T., and Inoue, K., A prenylated flavanone from roots ofMaackia amurensis subsp.Buergeri. Phytochemistry, 36, 255–256 (1994).
Maximo, P. and Lourenco, A., A pterocarpan fromUlex parviflorus.Phytochemistry, 48, 359–362 (1998).
Mitscher, L. A., Ward, J. A., Drake, S., and Rao, G S., Antimicrobial agents from higher plants. Erycristagallin, a new pterocarpene from the roots of the bolivian coral tree,Erythrina crista-galli.Heterocycles, 22, 1673–1675 (1984).
Mitscher, L. A., Drake, S., Gollapudi, S. R., and Okwute, S. K., A modern look at folkloric use of anti-infective agents.J. Nat. Prod., 50, 1025–1040 (1987).
Mitscher, L. A., Gollapudi, S. R., Geriach, D. C., Drake, S., Veliz, E. A., and Ward, J. A., Erycristin, a new antimicrobial pterocarpan fromErythrina crista-galli.Phytochemistry, 27, 381–385 (1988a).
Mitscher, L. A., Okwute, S. K., Gollapudi, S. R., Drake, S., and Avona, E., Antimicrobial Ptercarpans of NigerianErythrina mildbraedii.Phytochemistry, 27, 3449–3452 (1988b).
Nkengfack, A. E., Azebaze, A. G. B., Waffo, A. K., Fomum, Z. T., Meyer, M., and Van Heerden, F. R., Cytotoxic isoflavones fromErythrina indica.Phytochemistry, 58, 1113–1120 (2001).
Notaro, G., Piccialli, V., and Sica, D., New steroidal hydroxy-ketones and closely related diols from the marine spongeCliona copiosa.J. Nat. Prod., 55, 1588–1594 (1992).
Pouchert, C. J. and Behnke, J., The Aldrich Library of13C and1H FT NMR Spectra. 3rd ed., Aldrich Chemical Company, Inc., U.S.A., 3, pp. 569A (1993).
Reynolds, W. F., McLean, S., Poplawski, J., Enriquez, R. G., Escobar, L. I., and Leon, I., Total assignment of13C and1H spectra of three isomeric triterpenol derivatives by 2D NMR: an investigation of the potential utility of1H chemical shifts in structural investigations of complex natural products.Tetrahedron, 42, 3419–3428 (1986).
Rukachaisirikul, T., Intaraudom, J., Chawanasak, S., and Suksamram, A., Phenylpropanoids fromCinnamomum parthenoxylon.ScienceAsia, 26, 159–161 (2000).
Rukachaisirikul, T., Innok, P., Aroonrerk, N., Boonamnuaylap, W., Limrangsun, S., Boonyon, C., Woonjina, U., and Suksamrarn, A., Antibacterial pterocarpans fromErythrina subumbrans.J. Ethnopharmacol., 110, 171–175 (2007).
Singh, H., Chawla, A. S., Kapoor V. K., and Kumar, N., Investigation ofErythrina spp. IX. Chemical constituents ofErythrina stricta bark.J. Nat. Prod., 44, 526–529, (1981).
Skehan, P., Storeng, R., Scudiero, D., Monks, A., McMahon, J., Vistica, D., Warren, J. T., Bokesch, H., Kenny, S., and Boyd, M. R., New colorimetric cytotoxicity assay for anticancer-drug screening.J. Nat. Cancer Inst., 82, 1107–1112 (1990).
Tanaka, H., Tanaka, T., and Etoh, H., Three pterocarpans fromErythrina crista-galli.Phytochemistry, 45, 835–838 (1997).
Tanaka, H., Tanaka, T., and Etoh, H., Two pterocarpans fromErythrina orientalis.Phytochemistry, 47, 475–477 (1998).
Tanaka, H., Etoh, H., Watanabe, N., Shimizu, H., Ahmad, M., and Rizwani, G. H., Erysubins C-F, four isoflavonoids fromErythrina suberosa var.glabrescences. Phytochemistry, 56, 769–773, (2001).
Tandon, S. and Rastogi, R. P., Studies on the chemical constituents ofSpondias pinnata.Planta Med., 29, 190–192, (1976).
Trager, W. and Jensen, J. B., Human malaria parasites in continuous culture.Science, 193, 673–675 (1976).
Wen, L, Weiming, C., Zhi, X., and Xaotain, L., New triterpenoids ofPholidota chinensis.Planta Med., 52, 4–6 (1986).
Yenesew, A., Derese, S., Irungu, B., Midiwo, J. O., Waters, N. C., Liyala, P., Akala, H., Heydenreich, M., and Peter, M. G., Flavonoids and isoflavonoids with antiplasmodial activities from the root bark ofErythrina abyssinica.Planta Med., 69, 658–661 (2003).
Yenesew, A., Induli, M., Derese, S., Irung, B., Midiwo, J. O., Heydenreich, M., Peter, M. G., Akala, H., Wangui, J., Liyala, P., and Waters, N. C., Anti-plasmodial flavonoids from the stem bark ofErythrina abyssinica.Phytochemistry, 65, 3029–3032 (2004).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rukachaisirikul, T., Saekee, A., Tharibun, C. et al. Biological Activities of the Chemical Constituents ofErythrina stricta andErythrina subumbrans . Arch Pharm Res 30, 1398–1403 (2007). https://doi.org/10.1007/BF02977363
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02977363