Abstract
Tryptophanyl residues of A niger glucoamylase G2 (EC 3.2.1.3) involved in substrate and inhibitor binding have been identified following N-bromosuccinimide (NBS) treatment in the presence and absence of protective ligands. Appropriate proteolytic cleavages of the glucoamylase derivatives enabled isolation of individual peptide fragments containing the 15 thytophan positions and the extent of tryptophan oxidation was measured employing normal and 2nd derivative UV-spectrophotometry.
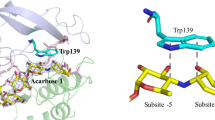
Trp-52,-141,-156,-228,-317, and-335 remained unoxidized while compflete oxidation to Trp-6,-28, and-466 and partial oxidation of Trp-170 and-178 was observed after NBS-treatment whether ligands were added or not. Trp-212,-417, and-437 were partially converted in uncomplexed glucoamylase while the presence of either the substrate maltose or the inhibitor acarbose prevented the oxidation of these residues. Trp-120, required for maintaining the active catalytic site, was protected by acarbose only, and its oxidation did not prevent ligand binding. The functional roles of Trp-212,-417,-437, and-120 are discussed.
NBS-treatment of the acarbose-protected large form of glucoamylase, G1, destroyed its unique capacity to adsorb onto starch granules while catalytic activity was preserved towards soluble substrates. This effect could be correlated with the oxidation of Trp-590 and-615 located near the COOH-terminus.
The reactivities, probably reflecting the degree of solvent exposure, were also assessed for individual tyrosyl residues in G2.
The thermal stability of oxidized, catalytically active G1 and G2 was remarkably low as compared to the unmodified forms.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- G1 and G2:
-
designate the larger and the smaller forms of glucoamylase from A. niger (29,43)
- CM:
-
S-carboxymethyl
- NBS:
-
N-bromosuccinimide
- nWox :
-
number (n) of oxidized tryptophanyl residues per enzyme molecule
- 2-pe:
-
2-pyridylethyl
- RP-HPLC:
-
reversed-phase high performance liquid chromatography
- Tris:
-
2-amino-2(hydroxymethyl)-1,3-propandiol
References
Adler, A. J., N. J. Greenfield &G. D. Fasman: Circular dichroism and optical rotatory dispersion of proteins and polypeptides. Meth. Enzymol. 27, 675–735 (1973)
Baldwin, R. L.: Intermediates in protein folding reactions and the mechanism of protein folding. Ann. Rev. Biochem. 44, 453–475 (1975)
Beddell, C. R., C. C. F. Blake &S. J. Oatley: An X-ray study of the structure and binding properties of iodine-inactivated lysozyme. J. Mol. Biol. 97, 643–654 (1975)
Blake, C. C. F., R. Cassels, C. M. Dobson, F. M. Poulsen, R. J. P. Williams &K. S. Wilson: Structure and binding properties of hen lysozyme modified at tryptophan 62, J. Mol. Biol. 147, 73–95 (1981)
Boel, E., I., Hjort, B. Svensson, F. Norris, K. E. Norris &N. P. Fill: Glucoamylases G1 and G2 from Aspergillus niger are synthesized from two different but closely related mRNAs EMBO J. 3, 1097–1102 (1984)
Burley, S. K. &G. A. Petsko: Aromatic-aromatic interaction: A mechanism of protein structure-stabilization. Science 229, 23–28 (1985)
Clarke, A. J. &B. Svensson: The role of tryptophanyl residues in the function of Aspergillus niger glucoamylase G1 and G2. Carlsberg Res. Commun. 49, 111–122 (1984)
Clarke, A. J. &B. Svensson: Identification of an essential tryptophanyl residue in the primary structure of glucoamylase G2 from Aspergillus niger. Carlsberg Res. Commun. 49, 559–566 (1984)
Fauchere, J. L.: How hydrophobic is tryptophan? Trends Biochem. Sci. 10, 268 (1985)
Friedman, M., J. C. Zahnley &J. R. Wagner: Estimation of the disulfide content of trypsin inhibitors as S-β-(2-pyridylethyl)-L-cysteine. Anal. Biochem. 106, 27–34 (1980)
Hartdegen, F. J. &J. A. Rupley: Oxidation of lysozyme by iodine: Identification of oxindolealanine 108. J. Mol. Biol. 80, 649–656 (1973)
Hayashida, S. &E. Yoshino: Formation of active derivatives of glucoamylase I during the digestion with fungal acid protease and α-mannosidase. Agric. Biol. Chem. 42, 927–933 (1978)
Hiromi, K., M. Ohnishi &A. Tanaka: Subsite structure and ligand binding mechanism of glucoamylase. Mol. Cell. Biochem. 51, 79–95 (1983)
Hirs, C. H. W.: Reduction and S-carboxymethylation of proteins. Meth. Enzymol. 25, 199–203 (1972)
Johansen, J. T., C. Overballe-Petersen, B. Martin, V. Hasemann &I. Svendsen: The complete amino acid sequence of copper, zinc superoxide dismutase from Saccharomyces cerevisiae. Carlsberg Res. Commun. 44, 201–217 (1979)
Jolley, M. E. &C. J. Gray: Tryptophanyl and carboxylic acid residues in the active center of glucoamylase I from Aspergillus niger. Carbohydr. Res. 49, 361–370 (1976)
Joly, M.: A physicochemical approach to the denaturation of proteins. Academic Press, New York (1965)
Klapper, D. G., C. E. Wilde III &J. D. Capra: Automated amino acid sequence of small peptides utilizing Polybrene. Anal. Biochem. 85, 126–131 (1978)
Kobayashi, S., T. Tazawa, M. Sasaki, S. Kiryu &M. Sugiura: Chemical modification of tryptophan and histidine residues in semi-alkaline proteinase from Aspergillus melleus. Chem. Pharm. Bull. 32, 3111–3117 (1984)
Lineback, D. R. &L. A. Aira: Structural characterization of the two forms of glucoamylase from Aspergillus niger. Cereal Chem. 49, 283–296 (1972)
MacGregor, E. A. &A. W. MacGregor: The action of cereal α-amylases on solubilized starch and cereal starch granules. In: New Approaches to Research on Cereal Carbohydrates (ed., R. D. Hill & L. Munck, Elsevier, Amsterdam, 1985) 149–160
Mahoney, W. C. &M. A. Hermodson: Separation of large denatured peptides by reverse phase high performance liquid chromatography. Trifluoroacetic acid as a peptide solvent. J. Biol. Chem. 255, 11199–11203 (1980)
Matsuura, Y., M. Kusunoki, W. Harada &M. Kakudo: Structure and possible catalytic residues of Taka-amylase A. J. Biochem. 95, 697–702 (1984)
Nunberg, J.-H., J. H. Meade, G. Cole, F. C. Lawyer, P. McCage, V. Schweickart, R. Tal, V. P. Wittman, J. E. Flatgaard &M. A. Innis: Molecular cloning and characterization of the glucoamylase gene of Aspergillus awamori. Mol. Cell. Biol. 4, 2306–2315 (1984)
Ohnishi M., T. Kawagishi, T. Abe &K. Hiromi: Stopped-flow studies on the chemical modification with N-bromosuccinimide of model compounds of tryptophan residues. J. Biochem. 87, 273–279 (1980)
Ohnishi, M., M. Taniguchi &K. Hiromi: Kinetic discrimination of tryptophan residues of glucoamylase from Rhizopus niveus by fast chemical modification with N-bromosuccinimide. Biochim. Biophys. Acta 744, 64–70 (1983)
Ohnishi, M., H. Kegai &K. Hiromi: Studies on the subsite structure of amylases I. Interaction of glucoamylase with substrate and analogues studied by difference spectrophotometry. J. Biochem. 77, 695–703 (1975)
Payan, F., R. Haser, M. Pierrot, M. Frey &J. P. Astier: The three-dimensional structure of α-amylase from porcine pancreas at 5Å resolution.—The active site location. Acta Cryst. B 36, 416–421 (1980)
Pazur, J. H. &T. Ando: The action of an amyloglucosidase of Aspergillus niger on starch and malto-oligosaccharides. J. Biol. Chem. 234, 1966–1970 (1959)
Pazur, J. H., H. R. Knull &A. Cepure: Glycoenzymes: structure and properties of the two forms of glucoamylase from Aspergillus niger. Carbohydr. Res. 20, 83–96 (1971)
Ponnuswamy, P. K., R. Muthusamy &P. Monavalan: Amino acid composition and thermal stability of proteins. Int. J. Biological Macromolecules 4, 186–190 (1982)
Reilly, P. J.: Starch hydrolysis with soluble and immobilized glucoamylase. Appl. Biochem. Bioeng. 2, 185–206 (1979)
Saha, B. C. &S. Ueda: Raw starch adsorption. elution and digestion behaviour of glucoamylase ofRhizopus niveus. J. Ferment. Technol. 61, 67–72 (1983)
Saha, B. C. &S. Ueda: Production and characteristics of inhibitory factor of raw starch digestion from Aspergillus niger. Appl. Microbiol. Biotechnol. 19, 341–346 (1984)
Savel'ev, A. N. &L. M. Firsov: Effect of modification of some amino acid radicals on enzymatic activity of glucoamylase from Aspergillus awamori. Biokhimiya 48, 1311–1318 (1983)
Savel'ev, A. N., V. R. Sergeev &L. M. Firsov: Study of the active site of glucoamylase from Aspergillus awamori. Biokhimiya 47, 390–397 (1982)
Servillo, L., G., Colonna, C. Balestrieri, R. Ragone &G. Irace: Simultaneous determination of tyrosine and tryptophan residues in proteins by second-derivative spectroscopy. Anal. Biochem. 126, 251–257 (1982)
Spande, T. F. &Witkop, B.: Determination of the tryptophan content of proteins with N-bromosuccinimide. Meth. Enzymol. XI, 498–506 (1967)
Svendsen, I., B. Martin &I. Jonassen: Characteristics of hiproly barley III. Amino acid sequences of two lysine-rich proteins. Carlsberg Res. Commun. 45, 79–85 (1980)
Svensson, B., K. Larsen &A. Gunnarsson: Characterization of a glucoamylase G2 from Aspergillus niger. Eur. J. Biochem. 154, 497–502 (1985)
Svensson, B., K. Larsen &I. Svendsen: Amino acid sequence of tryptic fragments of glucoamylase G1 from Aspergillus niger. Carlsberg Res. Commun. 48, 517–527 (1983)
Svensson, B., K. Larsen, I. Svendsen &E. Boel: The complete amino acid sequence of the glycoprotein, glucoamylase G1 from Aspergillus niger. Carlsberg Res. Commun. 48, 529–544 (1983)
Svensson, B., T. G. Pedersen, I. Svendsen, T. Sakai &M. Ottesen: Characterization of two forms of glucoamylase from Aspergillus niger. Carlsberg Res. Commun. 47, 55–69 (1982)
Truscheit, E., W. Frommer, B. Junge, L. Müller, D. D. Schmidt &W. Wingender: Chemie und Biochemie mikrobieller α-Glucosidase-ninhibitoren. Angew. Chem. 93, 738–755 (1981)
Ueda, S.: Fungal glucoamylases and raw starch digestion. Trends Biochem. Sci. 6, 89–90 (1981)
Yamashita, I., K. Suzuki &S. Fukui: Nucleotide sequence of the extracellular glucoamylase gene STA1 in the yeast Saccharomyces diastaticus. J. Bacteriol. 161, 567–573 (1985)
Author information
Authors and Affiliations
Additional information
Accepted byH. Klenow
Rights and permissions
About this article
Cite this article
Svensson, B., Clarke, A.J. & Svendsen, I. Influence of acarbose and maltose on the reactivity of individual tryptophanyl residues in glucoamylase from aspergillus niger. Carlsberg Res. Commun. 51, 61 (1986). https://doi.org/10.1007/BF02907996
DOI: https://doi.org/10.1007/BF02907996