Abstract
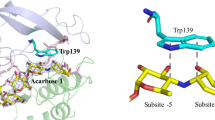
Enzymatically active, N-bromosuccinimide oxidized glucoamylase G2 (EC 3.2 1.3) was prepared in the presence of the inhibitor nearbose and inactive, oxidized G2 with capacity to bind substrate was prepared in the presence of maltose. Four out of 15 tryptophanyl residues were oxidized in the first derivative and five in the latter. In order to identify this fifth and essential residue, tryptic fragments of the two G2 derivatives were isolated. The peptide fragment from the inactive G2 derivative containing the additional oxindolealanine residue was located in HPLC chromatograms by UV-absorption measurements. Normal and second derivative UV-spectra, amino acid composition and NH2-terminal sequence of the isolated fragment led to identification of Trp(120) as a residue essential for activity. A short homologous amino acid sequence precedes both this Trp(120) and the Trp(83) which is involved in substrate binding in Taka-amylase A (J. Biochem. 95. 697–702 (1984)).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- AH:
-
aminohexyl
- G1 and G2:
-
the larger and the smaller of the two forms of glucoamylase from A, niger (31)
- HPLC:
-
high pressure liquid chromatography
- NBS:
-
N-bromosuccinimide
- TFA:
-
trifluoroactic acid
- Tris:
-
2-amino-2(hydroxymethyl)-1,3-propandiol
References
Bachmeyer, H.: Effect of tryptophan modification on the activity of bacterial and viral neuraminidase. FEBS Lett. 23, 217–219 (1972)
Barker, S.A., C.J. Gray &M.E. Jolley: Photo-oxidation of glucoamylase I from Aspergillus niger. Biochem. Biophys. Res. Comm. 45, 654–661 (1971)
Beddell, C.R., C.C.F. Blake &S.J. Oatley: An X-ray study of the structure and binding proporties of iodine-inactivated lysozyme. J. Mol. Biol. 97, 643–654 (1975)
Blake, C.C.F., R. Cassels, C.M. Dobson, F.M. Poulsen, R.J.P. Williams &K.S. Wilson: Structure and binding properties of heb lysozyme modified at tryptophan 62. J. Mol. Biol. 147, 73–95 (1981)
Boel, E., I. Hiort, B. Svensson, F. Norris, K.E. Norris &N.P. Fin: Glucoamylases G1 and G2 from Aspergillus niger are synthesized from two different but closely related mRNAs. EMBO J. 3, 1097–1102 (1984)
Boel, E., M. Trier Hansen, I. Hiort, I. Høegh &N.P. Ful: Two different types of intervening sequences in the glucoamylase gene from Aspergillus niger. EMBO J. 3, 1581–1585 (1984)
Buisson, G., E. Duee, R. Haser & F. Payan: Crystallographic structure of pig pancreas α-amylase (2.9 Á resolution), 8th International Biophysic Congress. Bristol (1984), abstract 112, p. 60.
Chou, P.Y. &G.D. Fasman: Prediction of protein conformation. Biochemistry 13, 222–244 (1974)
Clarke, A.J. &B. Svensson: The role of tryptophanyl residues in the function of A spergillus niger glucoamylase G1 and G2. Carlsberg Res. Commun. 49, 111–122 (1984)
Hagenbuchle, O., R. Bovey &R.A. Young: Tissue-specific expression of mouse α-amylase genes: nucleotide sequence of isoenzyme mRNAs from pancreas and salivary gland. Cell 21, 179–187 (1980)
Hiromi, K., M. Ohnishi &A. Tanaka: Subsite structure and ligand binding mechanism of glucoamylase. Mol. Cell. Biochem. 51, 79–95 (1983)
Imoto, T., L.N. Johnson, A.C.T. North, D.C. Phillips & J.A. Rupley: Vertebrate lysozymes Enzymes 3rd ed. 7, 665–868 (1972)
Inokuchi, N., T. Takahashi, A. Yoshimoto &M. Irie: N-Bromosuccinimide oxidation of a glucoamylase from Aspergillus saitol. J. Biochem. 91, 1661–1668 (1982)
Johansen, J.T., C. Overballe-Petersen, B. Martin, V. Hasemann &I. Svendsen: The complete amino acid sequence of copper, zinc superoxide dismutase from Saccharomyces cerevisiae. Carlsberg Res. Commun. 44, 201–217 (1979)
Jolley, M.E. &C.J. Gray: Tryptophanyl and carboxylic acid tesidues in the active center of glucoamylase I from Aspergillus niger. Carbohyd. Res. 49, 361–370 (1976)
Kita, Y., M. Fukazawa, Y. Nitta &T. Watanabe: Kinetic study on chemical modification of Taka-amylase A. L. Location and role of tryptophan residues. J. Biochem. 92, 653–659 (1982)
Klapper, D.G., C.E. Wilde III &J.D. Capra: Automated amino acid sequence of small peptides utilizing Polybrene. Anal. Biochem. 85, 126–131 (1978)
Klub, J.: Amino acid sequence of hog pancreatic α-amylase isoenzyme I. FEBS Lett. 136, 231–234 (1981)
Maboney, W.C. &M.A. Hermodson: Separation of large denatured peptides by reverse phase high performance liquid chromatography. Trifluoroacetic acid as a peptide solvent. J. Biol. Chem. 255, 11199–11203 (1980)
Matsuura, Y., M. Kusunoki, W. Harada &M. Kakudo: Structure and possible catalytic residues of Taka-amylase A. J. Biochem. 95, 697–702 (1984)
Muirhead, H.: Triose phosphate isomerase, pyruvate kinase and other α/β-barrel enzymes. Trends Bioch. Sci. 8, 326–330 (1983)
Nitta, Y., T. Konikata &T. Watanabe: Difference spectroscopic study of the interaction between soybean β-amylase and substrate or substrate analogues. J. Biochem. 93, 1195–1201 (1983)
Ohnish, M., M. Taniguchi &K. Hiromi: Kinetic discrimination of tryptophan residues of glucoamylase from Rhizopus niveus by fast chemical modification with N-bromosuccinimide. Biochim. Biophys. Acta 744, 64–70 (1983)
Rogers, J.C. &C. Milliman: Isolation and sequence analysis of a barley α-amylase cDNA clone. J. Biol. Chem. 258, 8169–8174 (1983)
Savelev, A.N. &L.M. Firsov: Effect of modification of some amino acid radicals on enzymatic activity of glucoamylase from Aspergillus awamori. Biokhimiya 48, 1311–1318 (1983)
Servillo, L., G. Colonna, C. Balestrieri, R. Ragone &G. Irace: Simultaneous determination of tyrosine and tryptophan residues in proteins by second-derivative spectroscopy. Anal. Biochem. 126, 251–257 (1982)
Spande, T.F. &B. Witkop: Determination of the tryptophan content of proteins with N-bromosuccinimide. Meth. Enzymol. XI, 498–506 (1967)
Svendsen, I., B. Martin &I. Jonassen: Characteristics of Hiproly barley III. Amino acid sequences of two lysine-rich proteins. Carlsberg Res. Commun. 45, 79–85 (1980)
Svensson, B., K. Larsen &I. Svendsen: Amino acid sequence of tryptic fragments of glucoamylase G1 from Aspergillus niger. Carlsberg Res. Commun. 48, 517–527 (1983)
Svensson, B., K. Larsen, I. Svendsen &E. Boel: The complete amino acid sequence of the glycoprotein, glucoamylase G3 from Aspergillus niger. Carlsberg Res. Commun. 48, 529–544 (1983)
Svensson, B., T.G. Pedersen, I. Svendsen, T. Sakai &M. Ottesen: Characterization of two forms of glucoamylase from Aspergillus niger. Carlsberg Res. Commun. 47, 55–69 (1982)
Takkinen, K., R.F. Pettersson, N. Kalkinen, I. Palva, H. Soderlund &L. Kääidänen: Amino acid sequence of α-amylase from Bacillus amyloliquefaciens deduced from the nucleotide sequence of the cloned gene. J. Biol. Chem. 258, 1007–1013 (1983)
Toda, H., K. Kondo & K. Narita: The complete amino acid sequence of Taka-amylase A. Proc. Japan Acad. 58, Ser. B. 208–212 (1982)
Truscheyt, E., W. Frommer, B. Junge, L. Müller, D.D. Schmidt &W. Wingender: Chemie und Biochemie mikrobieller α-Glucosidaseninhibitoren. Angew. Chem. 93, 738–755 (1981)
Author information
Authors and Affiliations
Additional information
Accepted by:H. Klenow, E. Lund and S.O. Andersen
Rights and permissions
About this article
Cite this article
Clarke, A.J., Svensson, B. Identification of an essential tryptophanyl residue in the primary structure of glucoamylase G2 from aspergillus niger. Carlsberg Res. Commun. 49, 559 (1984). https://doi.org/10.1007/BF02908684
DOI: https://doi.org/10.1007/BF02908684