Abstract
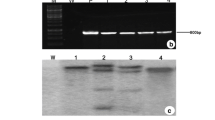
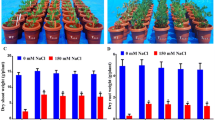
OsNHX1 gene (Na+/H+ antiporter gene ofOryza sativa L.) was introduced into Poplar 84K withAgrobacterium tumefaciens- mediated transformation. PCR, Southern and Northern blot analysis showed thatOsNHX1 gene was incorporated successfully into the genome of Poplar 84K and expressed in these transgenic plants. Salt tolerance test showed that three lines of transgenic plants grew normally in the presence of 200 mmol/L NaCl, while the Na+ content in the leaves of the transgenic plants grown at 200 mmol/L NaCl was significantly higher than that in plants grown at 0 mmol/L NaCl. The osmotic potential in the transgenic plants with high salinity treatment was lower than that of control plants. Our results demonstrate the potential use of these transgenic plants for agricultural use in saline soils.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Yokoi, S., Bressan, R. A., Hasegawa, P. M., Salt stress tolerance of plant, JIRCAS Working Rep, 2002, 25–33.
Blumwald, E., Aharon, G. S., Apse, M. P., Sodium transport in plant cells, Biochem. Biophys. Acta, 2000, 1465: 140–151.
Apse, M. P., Aharon, G. S., Snedden, W. A. et al., Salt tolerance conferred by overexpression of a vacuolar Na+/H+ antiport inArabidopsis, Science, 1999, 285: 1256–1258.
Zhang, H. X., Blumwald, E., Transgenic salt-tolerent tomato plants accumulate salt in foliage but not in fruit, Nat. Biotechnol., 2001, 19: 765–768.
Zhang, H. X., Hodson, J. N., Williams, J. P. et al., Engineering salt-tolerant Brassica plants: Characterization of yield and seed oil quality in transgenic plants with increased vacuolar sodium accumulation, Proc. Natl. Acad. Sci. USA, 2001, 98: 12832–12836.
Fukuda, A., Nakamura, A., Tanaka, Y., Molecular cloning and expression of the Na+/H+ exchanger gene inOryza sativa, Biochim. Biophys. Acta., 1999, 1446: 149–155.
Fukuda, A., Nakamura, A., Tagiri, A., Function, intracellular localization and the importance in salt tolerance of a vacuolar Na+/H+ antiporter from rice, Plant Cell Physiol., 2004, 45(2): 146–159.
Horsch, R. B., Fry, T. E., Hoffmann, N. L. et al., A simple and general method for transferring genes into plants, Science, 1995, 227: 1227–1231.
Murray, M. G., Thompson, W. F., Rapid isolation of high-molecular-weight plant DNA, Nucl. Acids. Res., 1980, 8: 4321–4325.
Sambrook, J., Fritsch, E., Maninatis, T., Molecular Cloning: A Laboratory Manual, 2rd ed., New York: Cold Spring Harbor Laboratory, 1989.
Fan, J. F., Li, L., Han, Y. F. et al., Establishment of leaf-explant regeneration system of Poplar 84K, J Northwest Forestry University, 2002, 17(2): 33–36.
Chun, Y. W., Morphogenic potential of leaf, internode, and root explants fromPopulus alba × P. grandidentata plantlets, Forest and Humanity, 1990, 3: 31–44.
Fan, G. Q., Zhai, X. Q., Zhai, C. J. et al., Callus induction from leaves of differentPaulownia species and its plantlet regeneration Journal of Forestry Research, 2001, 12(4): 209–214.
Brich, R. G., Plant transformation: Problems and strategies for practical application, Annu. Rev. Plant Physiol. Plant Mol. Biol., 1997, 48: 297–326.
Denney, B. K., Petersen, W. L., Ford-Santino, C. et al., Comparison of selective agents for use with the selectable marker gene bar in maize transformation, Plant Cell, Tissue Organ Culture, 1994, 36: 1–7.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Wang, S., Chen, Q., Wang, W. et al. Salt tolerance conferred by over-expression ofOsNHX1 gene in Poplar 84K. Chin.Sci.Bull. 50, 225–229 (2005). https://doi.org/10.1007/BF02897531
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02897531