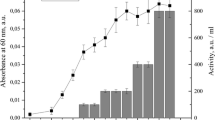
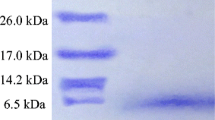
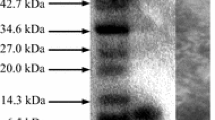
Abstract
A strain ofEnterococcus faecium isolated from Bulgarian yellow cheese “kashkaval” produced a bacteriocin-like substance named enterococcin A 2000. The antibacterial substance had a low molar mass (<2 kDa), was relatively stable toward heat but was sensitive to selected proteolytic enzymes. It was active against Gram-positive bacteria including enterococci, such asListeria, Bacillus andStreptococcus, and also against Gram-negativeE. coli. Production of enterococcin A 2000 has a maximum near the end of the exponential phase of producer growth. The peptide was purified by ammonium sulfate precipitation, butanol extraction, followed by cation-exchange chromatography and reversed-phase chromatography. A partial sequence of purified enterococcin A 2000 indicated that this substance does not belong to the class IIa of bacteriocins presenting the consensus anti-Listeria motif YGNGV.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aymerich T., Holo H., Havarstein L.S., Hugas M., Garriga M., Nes I.F.: Biochemical and genetic characterization of enterocin A fromEnterococcus faecium, a new antilisterial bacteriocin in the pediocin family of bacteriocins.Appl. Environ. Microbiol. 62, 1676–1682 (1996).
Belicová A., Krajčovič J., Dobias J., Ebringer L.: Antimutagenicity of milk fermented byEnterococcus faecium.Folia Microbiol. 44, 513–518 (1999).
Bidlingmeyer B.A., Cohen S.A., Tarvin T.L.: Rapid analysis of amino acids using precolum derivatization.J. Chromatogr. 336, 93–104 (1984).
Casaus P., Nilsen T., Cintas L.M., Nes I.F., Hernandez P.E., Holo H.: Enterocin B, a new bacteriocin fromEnterococcus faecium T136 which can act synergistically with enterocin A.Microbiology 143, 2287–2294 (1997).
Cintas L.M., Casaus P., Havarstein L.S., Hernandez P.E., Nes I.F.: Biochemical and genetic characterization of enterocin P, a novel sec-dependent bacteriocin fromEnterococcus faecium P13 with a broad antimicrobial spectrum.Appl. Environ. Microbiol. 63, 4321–4330 (1997).
Cintas L.M., Casaus P., Holo H., Hernandez P.E., Nes I.F., Havarstein L.S.: Enterocins L50A and L50B, two novel bacteriocins fromEnterococcus faecium L50, are related to staphylococcal hemolysins.J. Bacteriol. 180, 1988–1994 (1998).
Cintas L.M., Casaus P., Herranz C., Havarstein L.S., Holo H., Hernandez P.E., Nes I.F.: Biochemical and genetic evidence thatEnterococcus faecium L50 produces enterocins L50A and L50B, the sec-dependent enterocin P, and a novel bacteriocin secreted without an N-terminal extension termed enterocin Q.J. Bacteriol. 23, 6806–6814 (2000).
Devriese L.A., Pot B., Collins M.D.: Phenotypic identification of the genusEnterococcus and differentiation of phylogenetically distinct enterococcal species and species groups.J. Appl. Bacteriol. 75, 399–408 (1993).
Farýas M.E., Farýas R.N., de Ruýz Holgado A.P., Sesma F.: Purification and N-terminal amino acid sequence of enterocin CRL35, a ‘pediocin-like’ bacteriocin produced byEnterococcus faecium CRL35.Lett. Appl. Microbiol. 22, 412–419 (1996).
Franz C.M., Schillinger U., Holzapfel W.H.: Production and characterization of enterocin 900, a bacteriocin produced byEnterococcus faecium BFE 900 from black olives.Internat. J. Food Microbiol. 29, 255–270 (1996).
Giraffa G.: Enterococcal bacteriocins: their potential as anti-Listeria factors in dairy technology.Food Microbiol. 12, 291–299 (1995).
Groves M.L., Dower H.J., Farell H.M.J.: Re-examination of the polymeric distributions of κ-casein isolated from bovine milk.J. Prot. Chem. 11, 21–28 (1992).
Gürtler V., Stanisich V.A.: New approaches to typing and identification of bacteria using the 16S–23S rDNA spacer region.Microbiology 142, 3–16 (1996).
Holo H., Casaus P., Nilsen T., Cintas L.M., Nes I.F., Hernandez P.E.: Enterocin B, a new bacteriocin fromEnterococcus faecium T136 which can act synergistically with enterocin A.Microbiology 143, 2287–2294 (1997).
Klaenhammer T.R.: Genetics of bacteriocins produced by lactic acid bacteria.FEMS Microbiol. Rev. 12, 39–85 (1993).
Lauková A., Czikková S., Burdová O.: Anti-staphylococcal effect of enterocin in Sunar® and yogurt.Folia Microbiol. 44, 707–712 (1999).
Lauková A., Mareková M.: Production of bacteriocins by different enterococcal isolates.Folia Microbiol. 46, 49–52 (2001).
Lauková A., Vlaemynck G., Czikková S.: Effect of enterocin CCM 4231 onListeria monocytogenes in Saint-Paulin cheese.Folia Microbiol. 46, 157–160 (2001).
Maisnier-Patin S., Forni E., Richard J.: Purification, partial characterization and mode of action of enterococcin EFS2, an antilisterial bacteriocin produced by a strain ofEnterococcus faecalis isolated from cheese.Internat. J. Food Microbiol. 30, 255–270 (1996).
McKay L.L., Baldwin K.A.: Applications for biotechnology: present and future improvements in lactic acid bacteria.FEMS Microbiol. Rev. 7, 3–14 (1990).
Meisel H.: Overview on milk protein-derived peptides.Internat. Dairy J. 8, 363–373 (1998).
Migliore-Samour D., Floch F., Jollès P.: Biologically active casein peptides implicated in immunomodulation.J. Dairy Res. 56, 357–362 (1989).
Morovský M., Pristaš P., Javorský P.: Bacteriocins of ruminal bacteria.Folia Microbiol. 46, 61–62 (2001).
Moschetti G., Blaiotta G., Aponte M., Catzeddu P., Villani F., Deiana P., Coppola S.: Random amplified polymorphic DNA and amplified ribosomal DNA spacer polymorphism: powerful methods to differentiateStreptococcus thermophilus strains.J. Appl. Microbiol. 85, 25–36 (1998).
Moschetti G., Blaiotta G., Villani F., Coppola S., Parente E.: Comparison of statistical methods for identification ofStreptococcus thermophilus, Enterococcus faecalis, andEnterococcus faecium from randomly amplified polymorphic DNA patterns.Appl. Environ. Microbiol. 67, 2156–2166 (2001).
Naimi A., Beck G., Branlant C.: Primary and secondary structures of rRNA spacer regions in enterococci.Microbiology 143, 823–834 (1997).
Nes I.F., Diep D.B., Havarstein L.S., Brurberg M.B., Eijsink V., Holo H.: Biosynthesis of bacteriocins in lactic acid bacteria.Antonie van Leeuwenhoek 70, 113–128 (1996).
Nesterenko M.V., Tilley M., Upton S.J.: A simple modification of Blum’s silver stain method allows for 30-min detection of proteins in polyacrylamide gels.J. Biochem. Biophys. Meth. 28, 239–242 (1994).
Neviani E., Giraffa G., Carminati D.: Enterococci isolated from dairy products: a review of risks and potential technological use.J. Food Prot. 60, 732–738 (1997).
Nunez M., Rodriguez J.L., Garcia E., Gaya P., Medina M.: Inhibition ofListeria monocytogenes by enterocin 4 during manufacture and ripening of Manchego cheese.J. Appl. Microbiol. 83, 671–677 (1997).
Olasupo N.A., Schillinger U., Franz C.M.A.P., Holzapfel W.H.: Bacteriocin production byEnterococcus faecium NA01 from ‘Wara’—a fermented skimmed cow milk product from West Africa.Lett. Appl. Microbiol. 19, 438–441 (1994).
Pantev A., Valcheva R., Danova S., Ivanova I., Minkov I., Haertlé T., Chobert J.-M.: Effect of enterococcin A 2000 on biological and synthetic phospholipid membranes.Internat. J. Food Microbiol., in press (2002).
Piard J.C., Kuipers O.P., Rollema H.S., Desmazeaud M.J., de Vos W.M.: Structure, organization, and expression of thelct gene for lacticin 481, a novel lantibiotic produced byLactococcus lactis.J. Biol. Chem. 22, 16361–16368 (1993).
Rekhif N., Atrih A., Lefevre G.: Activity of plantaricin SA6, a bacteriocin produced byLactobacillus plantarum SA6 isolated from fermented sausage.J. Appl. Bacteriol. 78, 349–358 (1995).
Saruta K., Matsunaga T., Hoshina S., Kono M., Kitahara S., Kanaemo S., Sakai O., Machida K.: Rapid identification ofStreptococcus pneumoniae by PCR amplification of ribosomal DNA spacer region.FEMS Lett. 132, 165–170 (1995).
Schagger H., von Jagow G.: Tricine-sodium dodecyl sulfate-polyacrylamide gel electrophoresis for the separation of proteins in the range from 1 to 100 kDa.Anal. Biochem. 166, 368–379 (1987).
Schillinger U., Lucke F.K.: Antibacterial activity ofLactobacillus sakei isolated from meat.J. Appl. Bacteriol. 70, 134–142 (1989).
Smacchi E., Gobetti M.: Bioactive peptides in dairy products: synthesis and interaction with proteolytic enzymes.Food Microbiol. 17, 129–141 (2000).
Smid E.J., Bennik M.H., Vanloo B., Brasseur R., Gorris L.G.: A novel bacteriocin with a YGNGV motif from vegetable-associatedEnterococcus mundtii: full characterization and interaction with target organisms.Biochim. Biophys. Acta 1373, 47–58 (1998).
Sperber W.H., Swan J.: Hot-loop test for the determination of carbon dioxide production from glucose by lactic acid bacteria.Appl. Environ. Microbiol. 31, 990–991 (1976).
Torri Tarelli G., Carminati D., Giraffa G.: Production of bacteriocins active againstListeria monocytogenes andListeria innocua from dairy enterococci.Food Microbiol. 11, 243–252 (1994).
Villani F., Salzano G., Sorrentino E., Pepe O., Marino P., Coppola S.: Enterocin 226NWC, a bacteriocin produced byEnterococcus faecalis 226, active againstListeria monocytogenes.J. Appl. Bacteriol. 74, 380–387 (1993).
Vlaemynck G., Herman L., Coudijzer K.: Isolation and characterization of two bacteriocins produced byEnterococcus faecium strains inhibitory toListeria monocytogenes.Internat. J. Food Microbiol. 24, 211–225 (1994).
Worobo R.W., Henkel T., Sailer M., Roy K.L., Vederas J.C., Stiles M.E.: Characteristics and genetic determinant of a hydrophobic peptide bacteriocin, carnobacteriocin A, produced byCarnobacterium piscicola LV17A.Microbiology 140, 517–526 (1994).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pantev, A., Kabadjova, P., Dalgalarrondo, M. et al. Isolation and partial characterization of an antibacterial substance produced byEnterococcus faecium . Folia Microbiol 47, 391–400 (2002). https://doi.org/10.1007/BF02818696
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02818696