Abstract
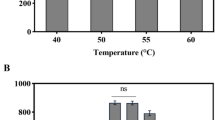
A study of the kinetics and performance of solvent-yielding batch fermentation of individual sugars and their mixture derived from enzymic hydrolysis of sago starch byClostridium acetobutylicum showed that the use of 30 g/L gelatinized sago starch as the sole carbon source produced 11.2 g/L total solvent,i.e. 1.5–2 times more than with pure maltose or glucose used as carbon sources. Enzymic pretreatment of gelatinized sago starch yielding maltose and glucose hydrolyzates prior to the fermentation did not improve solvent production as compared to direct fermentation of gelatinized sago starch. The solvent yield of direct gelatinized sago starch fermentation depended on the activity and stability of amylolytic enzymes produced during the fermentation. The pH optima for α-amylase and glucoamylase were found to be at 5.3 and 4.0–4.4, respectively. α-Amylase showed a broad pH stability profile, retaining more than 80% of its maximum activity at pH 3.0–8.0 after a 1-d incubation at 37°C. SinceC. acetobutylicum α-amylase has a high activity and stability at low pH, this strain can potentially be employed in a one-step direct solvent-yielding fermentation of sago starch. However, theC. acetobutylicum glucoamylase was only stable at pH 4–5, maintaining more than 90% of its maximum activity after a 1-d incubation at 37°C.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Annous B.A., Blaschek H.P.: Regulation and localization of amylolytic enzymes inClostridium acetobutylicum ATCC 824.Appl. Environ. Microbiol. 56, 2559–2561 (1990).
Ariff A.B., Webb C.: Influence of different fermentor configurations and modes of operation on glucoamylase production byAspergillus awamori.Asia Pacific J. Mol. Biol. Biotechnol. 4, 183–195 (1996).
Barton L.L., Georgi C.E., Lineback D.R.: Effect of maltose on glucoamylase formation byAspergillus niger.J. Bacteriol. 111, 771–777 (1972).
Bhella S.R., Altosaar I.: Purification and some properties of the extracellular α-amylase fromAspergillus awamori.Can. J. Microbiol. 28, 1340–1346 (1984).
Buonocore V., Caporale C., De Rosa M., Gambacorta A.: Stable, inducible thermoacidophilic α-amylase fromBacillus acidocaldarius.J. Bacteriol. 128, 515–520 (1976).
Chojecki A., Blanschek H.P.: Effect of carbohydrate source on α-amylase and glucoamylase formation byClostridium acetobutylicum SA-1.Ind. Microbiol. 1, 63–67 (1986).
Ennis B.M., Maddox I.S.: Use ofClostridium acetobutylicum P262 for production of solvents from whey permeate.Biotechnol. Lett. 7, 601–606 (1985).
Ennis B.M., Gutierrez N.A., Maddox I.S.: The acetone-butanol-ethanol fermentation: a current assessment.Proc. Biochem. 21, 131–146 (1986).
Ensley B., Mchugh J.J., Barton L.L.: Effect of carbon sources on the formation of α-amylase and glucoamylase byClostridium acetobutylicum.J. Gen. Appl. Microbiol. 21, 51–59 (1975).
Fond O., Matta-Ammouri G., Petitdemange H., Engasser J.M.: The role of acids in the production of acetone and butanol byClostridium acetobutylicum.Appl. Microbiol. Biotechnol. 22, 195–200 (1985).
Fond O., Engasser J.E., El-Amouri G.M., Petitdemange H.: The acetone-butanol fermentation on glucose and xylose. 1. Regulation and kinetics in batch cultures.Biotechnol. Bioeng. 28, 160–166 (1986).
Forgaty W.M., Benson C.P.: Purification and properties of thermophilic amyloglucosidase fromAspergillus niger.Appl. Microbiol. Biotechnol. 18, 271–278 (1983).
Gutierrez N.A., Maddox I.S., Schuster K.C., Swobida H., Gapes J.R.: Strain comparison and medium preparation for the acetone-butanol-ethanol (ABE) fermentation process using substrate of potato.Biores. Technol. 66, 263–265 (1998).
Hyun H.H., Zeikus J.G.: Simultaneous and enhanced production of thermostable amylases and ethanol from starch by cocultures ofClostridium thermosulfurogenes andClostridium thermohydrosulfuricum.Appl. Environ. Microbiol. 49, 1174–1181 (1985).
Jones T.J., Woods D.R.: Acetone-butanol fermentation revisited.Microbiol. Rev. 20, 484–524 (1986).
Linden I.C., Moreira A.R., Lenz T.G.: Acetone and butanol comprehensive biotechnology. The principles, application and regulations of biotechnology in industry, agriculture and medicine, pp. 915–929 in M.Y. Murray, W.B. Harvey, S. Drew, D.I.C. Wang (Eds.):The Practice of Biotechnology, Current Commodity Product, Vol. 3. Pergamon Press, London 1985.
Long S., Jones D.T., Woods D.R.: The relationship between sporulation and solvent production inClostridium acetobutylicum P262.Biotechnol. Lett. 6, 529–534 (1984).
Maher G.G.: Inactivation of transglucosidase in enzyme preparation formAspergillus niger.Starke 20, 228–232 (1968).
Miller T.L., Wolin M.J.: A serum bottle modification of the Hungate technique for culturing obligate anaerobes.Appl. Microbiol. 27, 985–987 (1974).
Ounine K., Petitdemange H., Raval G., Gay R.: Regulation and butanol inhibition ofd-xylose andd-glucose uptake inClostridium acetobutylicum.Appl. Environ. Microbiol. 49, 874–878 (1985).
Paquet V., Croux C., Goma G., Soucaille P.: Purification and characterization of the extracellular α-amylase fromClostridium acetobutylicum ATCC 824.Appl. Environ. Microbiol. 57, 212–218 (1991).
Rosfarizan M., Ariff A.B., Hassan M.A., Karim M.I.A.: Kojic acid production byAspergillus flavus using gelatinized and hydrolyzed sago starch as carbon sources.Folia Microbiol. 43, 459–464 (1998).
Schoutens G.H., Groot W.J.: Economic feasibility of the production ofiso-propanol-butanol-ethanol fuels from whey permeate.Proc. Biochem. 20, 117–121 (1985).
Scott D., Hedrick L.R.: The amylase ofClostridium acetobutylicum.J. Bacteriol. 63, 795–803 (1958).
Soni B.K., Soucaille P., Goma G.: Continuous acetone butanol fermentation: a global approach for the improvement in the solvent productivity in synthetic medium.Appl. Microbiol. Biotechnol. 25, 317–321 (1987).
Soni B.K., Kapp, Goma G., Soucaille P.: Solvent production from starch: effect of pH on α-amylase and glucoamylase localization and synthesis in synthetic medium.Appl. Microbiol. Biotechnol. 37, 539–543 (1992).
Srivasta R.A.K., Mathur S.N.: Regulation of amylase biosynthesis in growing and nongrowing cells ofBacillus stearothermophilus.J. Appl. Bacteriol. 57, 147–151 (1984).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Madihah, M.S., Ariff, A.B., Khalil, M.S. et al. Anaerobic fermentation of gelatinized sago starch-derived sugars to acetone—1-butanol—Ethanol solvent byClostridium acetobutylicum . Folia Microbiol 46, 197–204 (2001). https://doi.org/10.1007/BF02818533
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02818533