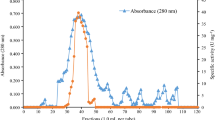
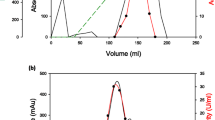
Summary
A thermophilic amyloglucosidase (EC 3.2.1.3) from a strain of Aspergillus niger was purified and separated from contaminating α-amylase (EC 3.2.1.1) and transglucosidase (EC 2.4.1.24) by ammonium sulphate fractionation, acetone precipitation and CM-BIO-GEL A chromatography. A 61-fold purification was achieved. The enzyme had highest affinity for starch (100), maltotriose (68) and maltose (31) and Km values of 0.025% and 1.42 mM with starch and maltose, respectively. It had a molecular weight of 63,000. The enzyme operated most efficiently on starch and maltose at pH 4.5 and surprisingly at the high temperature of 70°C. It possessed considerable pH stability, with 79% and 50% activity retained at pH 2.0 and pH 11.0, respectively, after 30 min at 40°C. The enzyme was 100% stable up to 50°C and 90% stable at 60°C for 30 min; above this latter temperature activity was rapidly destroyed. The presence of starch or glycerol improved the thermal stability of the enzyme. Polyvalent anions stimulated activity while the cations Cu2+ and Ag+ and to a lesser extent Ni2+ and Co2+ caused notable inhibitory effects. When incubated with high concentrations of glucose the enzyme formed small amounts of isomaltose as a reversion product.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Alazard D, Baldensperger JF (1982) Amylolytic enzymes from Aspergillus hennebergi (A. niger group): purification and characterization of amylases from solid and liquid cultures. Carbohydr Res 107:231–241
Alazard D, Raimbault M (1981) Comparitive study of amylolytic enzyme production by Aspergillus niger in liquid and solid-state cultivation. Eur J Appl Microbiol Biotechnol 12:113–117
Andrews P (1964) Estimation of molecular weight of proteins by Sephadex gel-filtration. Biochem J 91:222–233
Aunstrup K (1979) Production, isolation and economics of extracellular enzymes. In: Wingard L, Katchalski-Katzir E, Goldstein L (eds) Applied biochemistry and bioengineering, vol 2 Academic Press, London, pp 27–69
Banks GT, Binns F, Cutcliffe RL (1967) Recent developments in the production and industrial applications of amylolytic enzymes derived from filamentous fungi. In: Hockenhull DJD (ed.) Progress in industrial microbiology. Haywood Books, London, pp 95–137
Benson CP, Fogarty WM (1979) A rapid, quantitative assay procedure for transglucosidase. FEMS: NWEMG: SGM Meeting, Dublin. p 9
Benson CP, Kelly CT, Fogarty WM (1982) Production and quantification of transglucosidase from Aspergillus niger. J Chem Technol Biotechnol 32:790–798
Bernfeld P (1955) Amylases α and β methods. Enzymol 1:149–158
Campbell LL (1955) Purification and properties of an α-amylase from facultative thermophilic bacteria. Arch Biochem Biophys 54:154–161
Cramer F, Kampe W (1962) Katylse der Decarboxylierung durch Cyclodextrine. Eine Modellreaktion, die Wirkungsweise der Enzyme. Tetrahed Letters 353–356
Day DF (1978) A thermophilic glucoamylase from Cephalosporium eichhorniae. Curr Microbiol 1:181–184
Fleming ID (1968) Amyloglucosidase. In: Radley JA (ed) Starch and its derivatives, 4th edn. Chapman and Hall, London, pp 498–508
Fleming ID, Stone BA (1965) Fractionation of Aspergillus niger amyloglucosidase. Biochem J P 97: 13 p
Fogarty WM (1983) Microbial amylases In: Fogarty WM (ed) Microbial enzymes and biotechnology. Applied Science Publs Ltd, London, pp 1–92
Fogarty WM, Kelly CT (1979) Starch-degrading enzymes of microbial origin In: Bull MJ (ed) Progress in industrial microbiology. Elsevier Scientific Publ. Co, Amsterdam, 15:87–150
Fogarty WM, Kelly CT (1980) Amylases, amyloglucosidases and related glucanases. In: Rose AH (ed) Microbial enzymes and bioconversions. Academic Press, New York, 5:115–170
Freedberg IM, Levin Y, Kay C, McCubbin W, Katchalski-Katzir E (1975) Purification and characterization of Aspergillus niger exo-1,4-glucosidase. Biochim Biophys Acta 391:361–381
Green AA, Hughes WL (1955) Protein fractionation on the basis of solubility in aqueous solutions of salts and organic solvents. Methods Enzymol 1:67–90
Hansen SA (1975) Thin-layer chromatographic methods for identification of oligosaccharides in starch hydrolysates. J Chromatagr 105:388–390
Hugget SA, Nixon DA (1957) Enzymic determination of blood glucose. Biochem J P 66: 12 p
Kaji A, Sato M, Kobayashi M, Murao T (1976) Acid-stable glucoamylase produced in medium containing sucrose by Corticium rolfsii. J Agric Chem Soc Jpn 50:509–517
Kobrehel D (1979) The determination of glucotransferase activity in amyloglucosidase preparations. Biotechnol Letters 1:251–254
Krzechowska M, Urbanek H (1975) Isolation and some properties of glucoamylase from Cephalosporium charticola Lindau. Appl Microbiol 30:163–166
Lineback DR, Baumann WE (1970) Properties of a glucoamylase from Aspergillus phoenicis. Carbohydr Res 14:341–353
Lineback DR, Russell IJ, Rasmussen CR (1969) Two forms of the glucoamylase of Aspergillus niger. Arch Biochem Biophys 134:539–553
Lineback DR, Aira LA, Horner RL (1972) Structural characterisation of the two forms of glucoamylase from Aspergillus niger. Cereal Chem 49:293–298
LKB-Producter AB Annedalsvagen 33–39, 5–161 26 Bromma, Sweden
Maher GG (1968) Inactivation of transglucosidase in enzyme preparation from Aspergillus niger. Die Starke 20:228–232
Miah MN, Ueda S (1977) Multiplicity of glucoamylase of Aspergillus oryzae. Die Starke 29:191–235
Okada G (1977) Glucoamylase from Trichoderma viride J Jap Soc St Sci 24: 120–127
Pazur J, Ando T (1959) The action of amyloglucosidase of Aspergillus niger on starch and malto-oligosaccharides. J Biol Chem 234:1966–1970
Pazur JH, Kleppe K (1962) The hydrolysis of α-d-glucosides by amyloglucosidase from Aspergillus niger. J Biol Chem 237:1002–1006
Pazur J, Okada S (1967) Properties of the glucoamylase from Rhizopus delemar. Carbohydr Res 4: 371–378
Pazur J, Knull HR, Cepure A (1971) Glycoenzymes: structure and properties of the two forms of glucoamylase from Aspergillus niger. Carbohydr Res 20:83–96
Ramasesh N, Sreekantiah KR, and Murthy VS (1982) Studies on the two forms of amyloglucosidase of Aspergillus niger van Tieghem Starke 34:346–351
Rao BV, Maheshwari R, Sastry NV, Rao PV (1979) A thermostable glucoamylase from the thermophilic fungus Thermomyces lanuginous. Curr Sci 48:113–115
Smiley KL, Hensley DE, Smiley JM, Gasdorf HS (1971) Kinetic patterns of glucoamylase isoenzymes isolated from Aspergillus sp. Arch Biochem Biophys 144: 694–699
Taylor PM, Napier EJ, Fleming ID (1978) Some properties of a glucoamylase produced by the thermophilic fungus Humicola lanuginosa. Carbohydr Res 61:301–308
Tsuboi A, Yamasaki Y, Suzuki Y (1974) Two forms of glucoamylase from Mucor rouxianus. 1. Purification and crystallization. Agric Biol Chem 38:543–550
Underkofler LA (1968) Development of a commercial enzyme process: Glucoamylase. Adv Chem Series 5:343–358
Underkofler L, Denault J, Hou EF (1965) Enzymyes in the starch industry. Die Starke 17:179–184
Yamasaki Y, Suzuki Y, Czawa J (1977) Three forms of α-glucosidase and a glucoamylase from Aspergillus awamori. Agric Biol Chem 41:2149–2161
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fogarty, W.M., Benson, C.P. Purification and properties of a thermophilic amyloglucosidase from Aspergillus niger . European J. Appl. Microbiol. Biotechnol. 18, 271–278 (1983). https://doi.org/10.1007/BF00500491
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00500491