Abstract
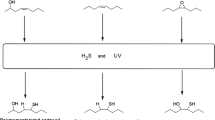
Glycol diesters and mixtures of mono- and diesters have been prepared from methyl esters of partially hydrogenated soybean oil fatty acids and diethylene, dipropylene, neopentyl and triethylene glycols. The catalyst used in these reactions was a mixture of calcium acetate/barium acetate (3∶1, w/w). The reactions were carried out under nitrogen with 0.5% catalyst at temperatures in the range of 190–275°C. Borated esters of mixed mono- and diesters were prepared with 0.33 equivalent of boric acid per 1.0 equivalent hydroxyl group on the ester. Refractive indices, viscosities, and flash and fire points were determined for diesters, mixed mono- and diesters, and mixed diesters and borated esters. The viscosities, flash points and fire points indicate that these esters can be used as a component of lubricating oils. Wear-prevention characteristics of mixed diesters and borated esters indicated that they can be used as antifriction additives in lubricating oils.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Naegley, P.C., Presented at the 83rd American Oil Chemists' Society Annual Meeting & Expo joint with 20th International Society of Fat World Congress, Toronto, Canada, May 10–14, 1992.
Bell, E.W., L.E. Gast, F.L. Thomas and R.E. Koos,J. Am. Oil. Chem. Soc. 54:239 (1977).
Zipf, R.L. (Werner G. Smith, Inc.), U.S. Patent 3,850,827 (1974).
Bell, E.W., J.C. Cowan and L.E. Gast,J. Am. Oil Chem. Soc. 49:552 (1972).
Bell, E.W. (U.S. Secretary of Agriculture), U.S. Patent 4,031,019 (1977).
Messina, J.F. (U.S. Secretary of the Army), U.S. Patent 3,493,509 (1970).
Friedrich, J.P. (U.S. Secretary of Agriculture), U.S. Patent 3,373,176 (1968).
Friedrich, J.P., inFatty Acids, edited by E.H. Pryde, American Oil Chemists' Society, Champaign, 1979, pp. 596–598.
Luck, K.M., and G.C. Gainer (Westinghouse Electric Corporation), U.S. Patent 3,878,112 (1975).
Sonntaga, N.O.V., inBailey's Industrial Oil and Fat Products, edited by D. Swern, 4th edn., Vol. 2, John Wiley & Sons, New York, 1982, pp. 131–132.
Annual Book of ASTM Standards, edited by F. Paulac, D. Fisher, E.L. Gutman, C.T. Hasia, S.L. Kauffman, J. Kramer, M. Lane, C.M. Leinweber, V.A. Mayer and P.A. McGee, Vol. 05.02, 1992, pp. 114–116, Method D2270-91.
Ibid. Annual Book of ASTM Standards, edited by F. Paulac, D. Fisher, E.L. Gutman, C.T. Hasia, S.L. Kauffman, J.Kramer, M. Lane, C.M. Leinweber, V.A. Mayer and P.A. McGee, Vol. 05.02, 1992, pp. 54–55, Method D2161-87
Lee, Jr., R.E., and E.R. Booser, inEncyclopedia of Chemical Technology, 2nd revised edn., Interscience Publishers, a Division of John Wiley & Sons, Inc., Vol. 12, 1967, p. 572.
Annual Book of ASTM Standards, edited by F. Paulac, D. Fisher, E.L. Gutman, C.T. Hasia, S.L. Kauffman, J. Kramer, M. Lane, C.M. Leinweber, V.A. Mayer and P.A. McGee, Vol. 05.01, 1992, pp. 23–26, Method D92-90.
Official Methods and Recommended Practices of the American Oil Chemists' Society, 4th edn, edited by D. Firestone, American Oil Chemists' Society, Champaign, 1990, Method Ce 9A-48, Reapproved 1989.
, Method Cd 13-60, Reapproved 1989, Method Cd 1-25, Revised 1990.
Annual Book of ASTM Standards, edited by F. Paulac, D. Fisher, E.L. Gutman, C.T. Hasia, S.L. Kauffman, J. Kramer, M. Lane, C.M. Leinweber, V.A. Mayer and P.A. McGee, Vol. 05.03, 1992, pp. 266–269, Method D4172.
Bell, E.W. (U.S. Secretary of Agriculture), U.S. Patent 4,031,019 (1977).
Watanabe, S., T. Fujita and M. Sakamoto,J. Am. Oil Chem. Soc. 65:1479 (1988).
Bailey, A.V., G.J. Boudreaux and G. Sumrell,53:632 (1976).
Small, V.R. (Chevron Research Co.), U.S. Patent 4,394,277 (1983).
Liston, T.V. (Chevron Research Co.), U.S. Patent 4,455,243 (1984); Japanese Patent 90051959 (1990).
Author information
Authors and Affiliations
Additional information
An erratum to this article is available at http://dx.doi.org/10.1007/BF02638910.
About this article
Cite this article
Basu, H.N., Robley, E.M. & Norris, M.E. Preparation of glycol derivatives of partially hydrogenated soybean oil fatty acids and their potential as lubricants. J Am Oil Chem Soc 71, 1227–1230 (1994). https://doi.org/10.1007/BF02540542
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02540542