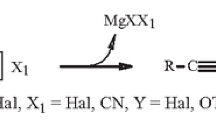Abstract
Diunsaturated C18 cyclic fatty acid methyl esters of known structure and configuration were synthesized as model derivatives of cyclic fatty acids formed in heat-abused vegetable oils for characterization and further biological evaluation. The Wittig reaction was used to prepare 5 pure methyl esters: (a) 12-(3-cyclohexenyl)-11-dodecenoate, (b) 11-(6-methyl-3-cyclohexenyl)-10-undecenoate, (c) 10-(6-ethyl-3-cyclohexenyl)-9-decenoate, (d) 9-(6-propyl-3-cyclohexenyl)-8-nonenoate and (e) 8-(6-butyl-3-cyclohexenyl)-7-octenoate. Diels-Alder cycloaddition reactions between 1,3-butadiene and appropriate (E)-2-alkenals produced 3-cyclohexenal intermediates. The appropriate methyl ω-bromoesters and their triphenylphosphonium bromides were made and converted to their respective ylids with NaOCH3 in DMF. The appropriate 3-cyclohexenals and phospho-ylids were reacted, and the desired cyclic ester products were isolated in crude yields of 30–83% as liquids and fractionally distilled. The crude cyclic esters were purified either by preparative TLC or by saponification-esterification. Double bonds in purified cyclic esters weretrans-isomerized and hydrogenated. Each derivative was characterized by IR,1H-NMR,13C-NMR, capillary GLC and GC-MS. On the basis of these analyses, no positional isomers were detected, Z-unsaturated isomers were produced in better than 90% purity, and the alkyl and ester ring substituents were predominantlytrans to each other.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Artman, N.R. (1969) Adv. Lipid Res. 7, 245–329.
Perkins, E.G. (1976) Rev. Fr. Corps Gras 23, 257–262 and 313–322.
Potteau, B., Dubois, P., and Rigaud, J. (1978) Ann. Technol. Agri. 27, 655–679.
Combe, N., Constantin, M.J., and Entressangles, B. (1981) Lipids 16, 8–14.
McInnes, A.G., Cooper, F.P., and MacDonald, J.A. (1961) Can. J. Chem. 39, 1906–1914.
Hutchinson, R.B., and Alexander, J.C. (1963) J. Org. Chem. 28, 2522–2526.
Michael, W.R. (1966) Lipids 1, 365–368.
Graille, J., Bonfand, A., Perfetti, P., and Naudet, M. (1980) Chem. Phys. Lipids 27, 23–41.
Holmes, H.L. (1948) in Organic Reactions (Adams, R., ed.), Vol. 4, pp. 60–173, John Wiley and Sons, New York, NY.
Gosney, I., and Rowley, A.G. (1980) in Organophosphorous Reagents in Organic Synthesis (Cadogan, J.I.G., ed.), pp. 17–53, Academic Press, Inc. New York, NY.
Bergelson, L.D., Vaver, V.A., YuKovtun, V., Senyavina, L.B., and Shemyakin, M.M. (1962) Zh. Obsshch. Khim. 32, 1802–1807; C.A. (1963) 58, 4415g.
Diaper, D.G.M., and Mitchell, D.L. (1960) Can. J. Chem. 38, 1976–1982.
Schaefer, J.P., Higgins, J.G., and Shenov, P.K. (1968) Org. Synth. 48, 51–53.
Siclari, F. (1974) U.S. Patent 3,856,833.
Siclari, F. (1980) U.S. Patent 4,192,813.
Ackman, R.G., Retson, M.E., Gallay, L.R., and Vandenheuvel, F.A. (1961) Can. J. Chem. 39, 1956–1963.
Bergelson, L.D., Vaver, V.A., Bezzubov, A.A., and Shemyakin, M.M. (1962) Zh. Obshch. Khim. 32, 1807–1811;C.A. (1963) 58, 4416c.
Gibson, T.W., and Strassburger, P. (1976) J. Org. Chem. 41, 791–793.
Scholfield, C.R. (1981) J. Am. Oil Chem. Soc. 58, 662–663.
MacDonald, J.A. (1956) J. Am. Oil Chem. Soc. 33, 394–496.
Eliel, E.L. (1962) Stereochemistry of Carbon Compounds, McGraw-Hill Book Company, Inc., New York, NY.
Kugatova-Shemyakina, G.P., Nikolaev, G.M., and Andreev, V.M. (1967) Tetrahedron 23, 2721–2733.
Kugatova-Shemyakina, G.P., and Nikolaev, G.M. (1967) Tetrahedron 23, 2987–2995.
Zefirov, N.S., Sergeev, N.M., and Gurvich, L.G. (1970) Dokl. Akad. Nauk SSSR 190, 345–347.
Johnson, L.F., and Jankowski, W.C. (1972) Carbon-13 NMR Spectra, Spectrum No. 272, John Wiley & Sons, Inc., New York, NY; and Sadtler Standard Carbon-13 NMR Indexes (1977), Vol. 5, Spectrum No. 896, Sadtler Research Laboratories, Inc., Philadelphia, PA.
Nakagawa, K., Sawai, M., Ishii, Y., and Ogawa, M. (1977) Bull. Chem. Soc. Jpn. 50, 2487–2488.
Kugatova-Shemyakina, G.P., and Nikolaev, G.M. (1967) Zh. Org. Khim. 3, 1448–1455.
Miyajima, S., and Inukai, T. (1972) Bull. Chem. Soc. Jpn. 45, 1553–1554.
Bus, J., Sies, I., and Lie Ken Jie, M.S.F. (1976) Chem. Phys. Lipids 17, 501–518.
Friedrich, J.P. (1967) J. Am. Oil Chem. Soc. 44, 244–248.
Oshima, M., and Ariga, T. (1975) J. Biol. Chem. 250, 6963–6968.
Silverstein, R.M., Bassler, G.C., and Morrill, T.C. (1974) Spectrometric Identification of Organic Compounds, 3rd edn., pp. 5–71, John Wiley & Sons, Inc., New York, NY.
Diels, O., and Alder, K. (1929) Justus Liebigs Ann. Chem. 470, 62–103.
Sobecki, W. (1910) Chem. Ber. 43, 1038–1041.
Winter, M., and Goutschi, F. (1962) Helv. Chim. Acta 45, 2567–2575.
Prevost, C. (1944) Bull. Soc. Chim. Fr. 11, 218–225.
Delaby, R., and Guillot-Allegre, S. (1931) Compt. Rend. 192, 1467–1469.
Lycan, W.H., and Adams, R. (1929) J. Am. Chem. Soc. 51, 625–629.
Pryde, E.H., Theirfelder, C.M., and Cowan, J.C. (1976) J. Am. Oil Chem. Soc. 53, 90–93.
Chuit, P., and Hausser, J. (1929) Helv. Chim. Acta 12, 463–492.
Hunsdiecker, H., and Hunsdiecker, C. (1942) Chem. Ber. 75, 291–297.
Author information
Authors and Affiliations
About this article
Cite this article
Awl, R.A., Frankel, E.N. Cyclic fatty esters: Synthesis and characterization of methyl ω-(6-alkyl-3-cyclohexenyl) alkenoates. Lipids 17, 414–426 (1982). https://doi.org/10.1007/BF02535220
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02535220