Abstract
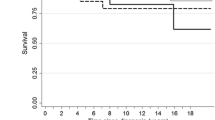
12 patients: (7 males and 5 females) with recurrent brainstem gliomas were treated with the oral topoisomerase inhibitor VP-16 (Etoposide). Patients ranged in age from 3 to 49 years with a median age of 7 years. All patients had been previously treated with radiation therapy (conventional fractionation: 4; hyperfractionation: 8) and 5 had received prior nitrosourea-based chemotherapy at time of tumor recurrence. Tumor recurrence was documented by radiographic tumor enlargement utilizing brain MRI with gadolinium enhancement (12) and clinical neurologic deterioration (9). Two patients underwent biopsy pathologically documenting tumor recurrence. Each cycle of therapy consisted of 21 days of VP-16 (50 mg/m2/day) followed by a 14 day rest followed by an additional 21 days of VP-16 (50mg/m2 day). Complete blood counts were followed bi-weekly and a neurologic examination and brain MRI scan with contrast were performed prior to initiation of each cycle of therapy. Treatment related complications included: partial alopecia (5); diarrhea (5); weight loss (4); neutropenia (2); and thrombocytopenia (4). No patient required transfusion or antibiotic treatment of neutropenic fever. There were no treatment related deaths. 12 patients were evaluable of whom 6 demonstrated a radiographic response (1 complete; 3 partial; 2 stable disease) with a median duration of response of 8 months. In summary; oral VP-16 is a well tolerated and relatively non-toxic chemotherapeutic agent with apparent activity in this small cohort of patients with recurrent brainstem gliomas.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Edwards MSB, Prados M: Current management of brain stem gliomas. Pediat Neurosci 13: 309–315, 1987
Stroink AR, Hoffman HJ, Hendrick EB, Humphreys RP: Diagnosis and management of pediatric brain-stem gliomas. J Neurosurg 65: 745–750, 1986
Mantravadi RVP, Phatak R, Bellur S, Liebner EJ, Haas R: Brain stem gliomas: An autopsy study of 25 cases. Cancer 49: 1294–1296, 1982
Berger MS, Edwards MSB, LaMasters D, Davis RL, Wilson CB: Pediatric brain stem tumors: Radiographic, pathological, and clinical correlations. Neurosurgery 12: 298–302, 1983
Epstein F, McCleary L: Intrinsic brain-stem tumors of childhood: surgical indications. J Neurosurg 64:11–15, 1986
Abernathey CD, Camacho A, Kelly PJ: Sterotaxic suboccipital transcerebellar biopsy of pontine mass lesions. J Neurosurg 70: 195–200, 1989
Epstein F, Wisoff JH: Intrinsic brainstem tumors in childhood: Surgical indications. J Neuro-Oncol 6: 309–317, 1988
Freeman CR, Suissa S: Brain stem tumors in children: Results of a survey of 62 patients treated with radiotherapy. J Rad Oncol Biol Phys 12: 1823–1828, 1986
Packer RJ, Zimmerman RA, Luerssen TG, Sutton LN, Bilaniuk LT, Bruce DA, Schut L: Brainstem gliomas of childhood: Magnetic resonance imaging. Neurology 35: 397–401, 1985
Packer RJ, Littman PA, Sposto RM, D'Angio GD, Priest JR, Heideman RL, Bruce DA, Nelson DF: Results of pilot study of hyperfractionated radiation therapy for children with brain stem gliomas. Int J Rad Oncol Biol Phys 13: 1647–1651, 1987
Freeman CR, Krischer J, Sanford RA, Burger PC, Cohen M, Norris D: Hyperfractionated radiotherapy in brain stem tumors: Results of a pediatric oncology group study. Int J Rad Oncol Biol Phys 15: 311–318, 1988
Edwards MSB, Wara WM, Urtasun RC, Prados M, Levin VA, Fulton D, Wilson CB, Hannigan J, Silver P: Hyperfractionated radiation therapyn for brain-stem glioma: a Phase I–II trial. J Neurosurg 70: 691–700, 1989
Jenkin RDT, Jenkin MB, Boesel C, Ertel I, Evans A, Hittle R, Ortega J, Sposto R, Wara W, Wilson C, Anderson J, Leikin S, Hammond GD: Brain-stem tumors in childhood: a prospective randomized trial of irradiation with and without adjuvant CCNU, VCR, and prednisone. J Neurosurg 66: 227–213, 1987
Fulton DS, Levin VA, Wara WM, Edwards MS, Wilson CB: Chemotherapy of Pediatric brain-stem tumors. J Neurosurg 54: 721–725, 1981
Mundiger F, Baus DF, Krauss JK, Birg W: Long-term outcome of 89 low-grade brain-stem gliomas after interstitial radiation therapy. Mundinger F, Braus DF, Krauss JK, Birg W. Long-term outcome of 89 low-grade brain-stem gliomas after interstitial radiation therapy. J Neurosurg 75: 740–746, 1991
Rodrieguez LA, Prados M, Fulton D, Edwards MSB, Silver P, Levin V: Treatment of recurrent brain stem gliomas and other central nervous system tumors with 5-fluorouracil, CCNU, Hydroxyurea, and 6-mercaptopurine. Neurosurgy 22: 691–693, 1988
Finlay JL, Coins SC: Brain tumors in children; III. Advances in chemotherapy. A J Ped Hem/Oncol 9(3): 264–271, 1987
Pendergrass TW, Milstein JM, Geyer JR, Mulne AF, Kosnik EJ, Morris JD, Heideman RL, Ruymann FB, Stuntz JT, Bleyer WA. Eight drugs in one day chemotherapy for brain tumors: Experience in 107 children and rationale for preradiation chemotherapy. J Clin Oncol 5(8): 1221–1231, 1987
Friedman HS, Oakes WJ: The chemotherapy of posterior Fossa tumors in childhood. J Neuro-Oncol 5:217–229, 1987
Allen JC, Helson L: High-dose cyclophosphamide chemotherapy for recurrent CNS tumors in children. J Neurosurg 55: 749–756, 1981
van Eys J, Cangir A, Coody D, Smith B: MOPP regimen as primary chemotherapy for brain tumors in infants. J Neuro-Oncol 3: 237–243, 1985
Bertolone SJ, Baum ES, Krivit W, Hammond GD: A phase II study of cisplatin therapy in recurrent childhood brain tumors. J Neuro-Oncol 7: 5–11, 1989
Fleming RA, Miller AA, Stewart CF: Etoposide: An update. Clin Pharm 8: 274–293, 1989
Clark PI, Slevin ML: The clinical pharmacology of etoposide and teniposide. Clin Pharm 12: 223–252, 1987
O'Dwyer PJ, Leyland-Jones B, Alonso MT, Marsoni S, Wittes RE: Drug Therapy: Etoposide (VP-16-213) Current status of an active anticancer drug. N Engl J Med 312: 692–700, 1985
Greco FA: Future directions for etoposide therapy. Cancer 67: 315–318, 1991
Slevin ML: The clinical pharmacology of etoposide. Cancer 67: 319–329, 1991
O'Dwyer MB, Leyland-Jones B, Alonso MT, Marsoni S, Wites RE: Etoposide (VP-16-213): Current status of an active anticancer drug. N Engl J Med 312: 692–700, 1985
Seiler RW, Zimmerman A, Markwalder H: Adjuvant chemotherapy with VM-26 and CCNU after operation and radiotherapy of high-grade supratentorial astrocytomas. Surg Neurol 13: 65–68, 1980
Slansky BD, Mann-Kaplan RS, Reynolds AF,et al.: 4′-demethylepiopdophyllotoxin beta-D-thenylodene-glucoside (PTG) in the treatment of malignant intracranial neoplasm. Cancer 33: 460–467, 1974
Sweet DL, Hendler FJ, Hanlon K,et al.: Treatment of grade III and IV astrocytomas with BCNU alone and in combination with VM-26 following surgery and radiation therapy. Cancer Treat Rep 63: 1707–1711, 1979
Tirelli U, D'lncalci M, Canetta R, Tumolo S, Franchin G, Veronesi A, Galligioni E, Trovo MG, Rossi C, Grigoletto E: Etoposide (VP-16-213) in malignant brain tumors: A phase II study. J Clin Oncol 2: 432–436, 1984
Corden BJ, Strauss LC, Killmond T, Carson BS, Wharam MD, Kumar AJ, Piantodosi S, Robb PA, Phillips PC: Cisplatin, cytosine arabinoside and etoposide in the treatment of recurrent childhood brain tumors. Pediatr Neurosci 14: 163, 1988 (Abstract)
Donfrancesco A, Cozza R, Deb G, De Laurentis C, Fidani P, Castello M, Clerico A, Dominici C, Jenkher A: Etoposide and high-dose carboplatin is an active regimen in childhood brain tumors (BT). Pediatr Neurosci 14: 163, 1988 (Abstract)
EORTC Brain Tumor Group: Evaluation of CCNU, VM-26 plus CCNU, and procarbazine in supratentorial brain gliomas. Final evaluation of a randomized study. J Neurosurg 55: 27–31, 1981
Garbino CE, Gordon-Firing S: Adjuvant chemotherapy with VM-26 in glioblastoma multiforme. Proc Ann Meet Am Soc Clin Oncol 3: C-997, 1984 (Abstract)
Gerosa MA, DiStefano E, Olivi A: A VM-26 monochemotherapy trial in the treatment of recurrent supratentorial gliomas: Preliminary report. Surg Neurol 15:128–134, 1981
Hayes FA, Green A, Thompson E, Wilimas J, Etcubanas E, Pratt C: Phase II trial of VP16-213 in Pediatric solid tumors. Proc Am Assoc Cancer Res 2: 66, 1983 (Abstract)
Kessinger A, Lemon HM, Foley JF: VM-26 as a second drug in treatment of brain gliomas. Cancer Treat Rep 63: 511–512, 1979
Creaven PJ: The clinical pharmacology of VM-26 and VP-16-213: A brief overview. Cancer Chemother Pharmacol 7: 133–140, 1982
D'Incalci M, Farina P, Sessa C, Mangioni C, Conter V, Maera G, Rocchetti M, Brambilla Pisoni M, Piazza E, Beer M, Cavelli F: Pharmacokinetics of VP-16-213 given by different administration methods. Cancer Chemother Pharmacol 7: 141–145, 1982
Nissen NI, Pajak TF, Leone LA,et al.: Clinical trial of VP-16(NSC-141540)I.V. twice weekly in advanced neoplastic disease. Cancer 45: 232–235, 1980
Smyth RD, Pfeffer M, Scalzo A, Comis RL: Bioavailability and pharmacokinetics of etoposide (VP-16). Semin Oncol XII(I, Suppl 2): 48–51, 1985
Greco FA, Johnson DH, Hainsworth JD: Chronic oral etoposide. Cancer 67: 303–309, 1991
Finlay J, August C, Packer R, Kamani N, Bayever E, Sutton L, Fried A, Zimmerman R, Nachman J, Turski P, Steeves R, Longo W, Rozenthal J, Levin A: High-dose chemotherapy with autologous marrow ‘Rescue’ in children with recurrent brain tumors. Proc ASCO 8(347): 89, 1989
Giannone L, Wolff SW: Phase II treatment of central nervous system gliomas with high-dose etoposide and autologous bone marrow transplantation. Cancer Treat Rep 71 (7-8): 759–761, 1987
Kovnar E, Kun L, Horowitz M, Douglass E, Kellie S, Sanford R, Langston J, Jenkins J, Fairclough D: Preirradiation cisplatin and VP-16 (CDDP/VP) for high risk medulloblastoma and other primitive neuroectodermal tumors of the central nervous system (CNS). Pediatr Neurosci 14: 164, 1988 (Abstract)
Pons M, Finolay JL, Walker RW, McElwain M, Packer R: Chemotherapy with vincristine and etoposide in children with low-grade astrocytoma. Ann Neurol 28(3): 419, 1990
Author information
Authors and Affiliations
Additional information
Reported in part at the Child Neurology Society Meetings, October 1991 in Portland, Oregon
Rights and permissions
About this article
Cite this article
Chamberlain, M.C. Recurrent brainstem gliomas treated with oral VP-16. J Neuro-Oncol 15, 133–139 (1993). https://doi.org/10.1007/BF01053934
Issue Date:
DOI: https://doi.org/10.1007/BF01053934